A New Approach to Assess the Effect of Various Humic Compounds on the Metabolic Activity of Cells Participating in Methanogenesis
Abstract
1. Introduction
2. Materials and Methods
2.1. Characteristics of the Consortia and HCs
2.2. Methanogenic Tests
2.3. Analytical Methods
3. Results
3.1. Influence of HCs on the ATP Level of Cells in the Natural Anaerobic Consortia Catalyzing Methanogenesis
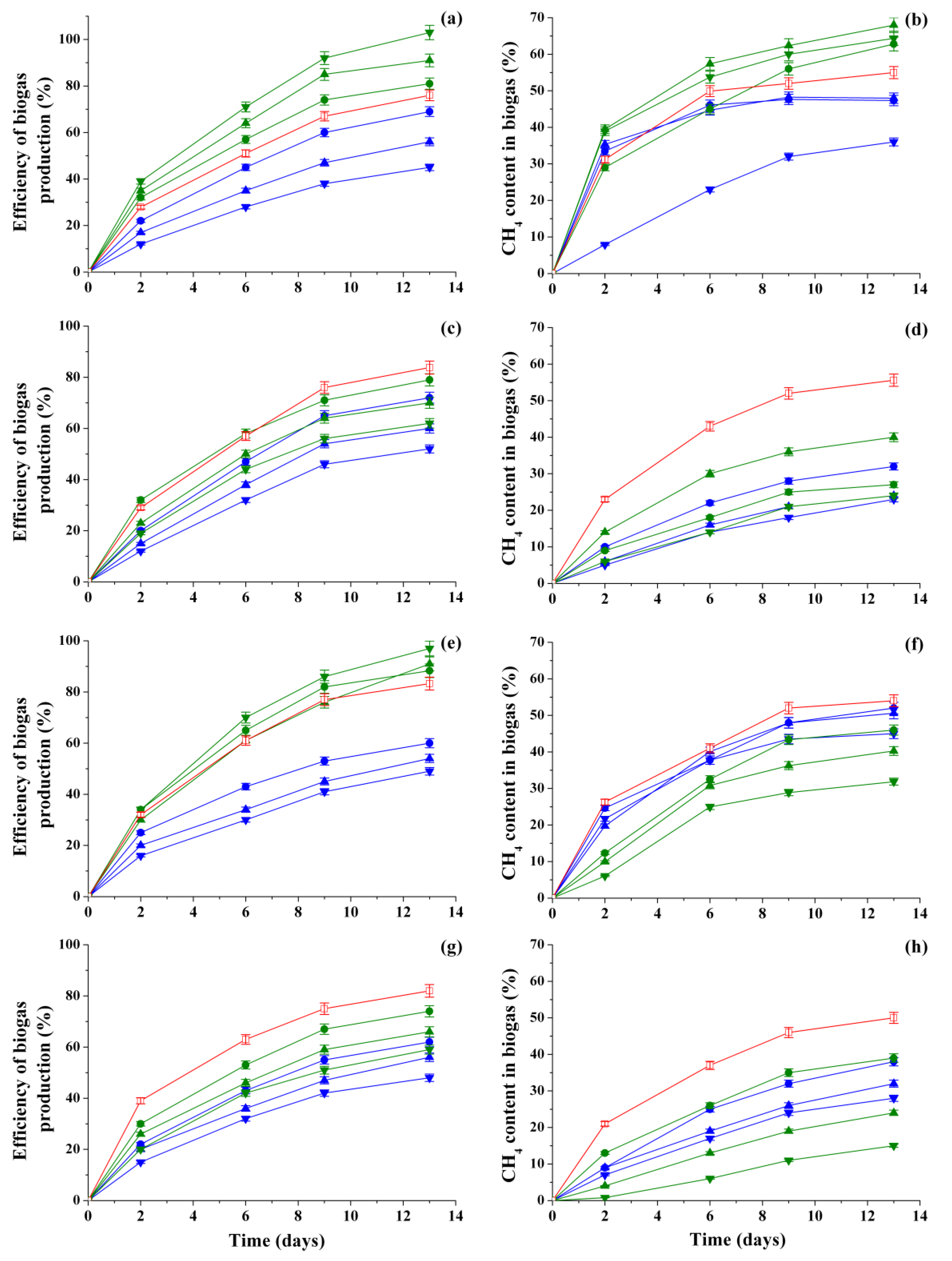
3.2. Analysis of the Biogas Production Efficiency and CH4 Content in Biogas Accumulated by Methanogenic Consortia in the Presence of Different Concentrations of HCs
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Awe, O.W.; Zhao, Y.; Nzihou, A.; Minh, D.P.; Lyczko, N.A. Review of biogas utilisation, purification and upgrading technologies. Waste Biomass Valoriz. 2017, 8, 267–283. [Google Scholar] [CrossRef]
- Lee, U.; Han, J.; Wang, M. Evaluation of landfill gas emissions from municipal solid waste landfills for the life-cycle analysis of waste-to-energy pathways. J. Clean. Prod. 2017, 166, 335–342. [Google Scholar] [CrossRef]
- Moqbel, S.; Reinhart, D.; Chen, R.H. Factors influencing spontaneous combustion of solid waste. Waste Manag. 2010, 30, 1600–1607. [Google Scholar] [CrossRef] [PubMed]
- Chiemchaisri, C.; Chiemchaisri, W.; Kumar, S.; Wicramarachchi, P.N. Reduction of methane emission from landfill through microbial activities in cover soil: A brief review. Crit. Rev. Environ. Sci. Technol. 2012, 42, 412–434. [Google Scholar] [CrossRef]
- Sadasivam, B.Y.; Reddy, K.R. Landfill methane oxidation in soil and bio-based cover systems: A review. Rev. Environ. Sci. Biotechnol. 2014, 13, 79–107. [Google Scholar] [CrossRef]
- Majdinasab, A.; Yuan, Q. Performance of the biotic systems for reducing methane emissions from landfill sites: A review. Ecol. Eng. 2017, 104, 116–130. [Google Scholar] [CrossRef]
- Terent’eva, I.E.; Sabrekov, A.F.; Glagolev, M.V.; Kotsyurbenko, O.R. Methane emission from municipal solid waste landfills. Russ. Meteorol. Hydrol. 2017, 42, 327–334. [Google Scholar] [CrossRef]
- Pyzik, A.; Ciezkowska, M.; Krawczyk, P.S.; Sobczak, A.; Drewniak, L.; Dziembowski, A.; Lipinski, L. Comparative analysis of deep sequenced methanogenic communities: Identification of microorganisms responsible for methane production. Microb. Cell Fact. 2018, 17, 197. [Google Scholar] [CrossRef]
- Chen, Y.; Cheng, J.J.; Creamer, K.S. Inhibition of anaerobic digestion process: A review. Bioresour. Technol. 2008, 99, 4044–4064. [Google Scholar] [CrossRef]
- Mudhoo, A.; Kumar, S. Effects of heavy metals as stress factors on anaerobic digestion processes and biogas production from biomass. Int. J. Environ. Sci. Technol. 2013, 10, 1383–1398. [Google Scholar] [CrossRef]
- de Melo, B.A.G.; Motta, F.L.; Santana, M.H.A. Humic acids: Structural properties and multiple functionalities for novel technological developments. Mater. Sci. Eng. C 2016, 62, 967–974. [Google Scholar] [CrossRef] [PubMed]
- Khadem, A.F.; Azman, S.; Plugge, C.M.; Zeeman, G.; van Lier, J.B.; Stams, A.J. Effect of humic acids on the activity of pure and mixed methanogenic cultures. Biomass Bioenergy 2017, 99, 21–30. [Google Scholar] [CrossRef]
- Guo, M.; Xian, P.; Yang, L.; Liu, X.; Zhan, L.; Bu, G. Effect of humic acid in leachate on specific methanogenic activity of anaerobic granular sludge. Environ Technol. 2015, 36, 2740–2745. [Google Scholar] [CrossRef] [PubMed]
- Yap, S.D.; Astals, S.; Lu, Y.; Peces, M.; Jensen, P.D.; Batstone, D.J.; Tait, S. Humic acid inhibition of hydrolysis and methanogenesis with different anaerobic inocula. Waste Manag. 2018, 80, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, T.V.; van Lier, J.B.; Zeeman, G. Humic acid-like and fulvic acid-like inhibition on the hydrolysis of cellulose and tributyrin. BioEnergy Res. 2015, 8, 821–831. [Google Scholar] [CrossRef]
- Lomakina, G.Y.; Modestova, Y.A.; Ugarova, N.N. Bioluminescence assay for cell viability. Biochemistry 2015, 80, 701–713. [Google Scholar] [CrossRef] [PubMed]
- Ismayilov, I.T.; Stepanov, N.A.; Efremenko, E.N.; Abbasov, V.M. Evaluation of biocidal properties of vegetable oil-based corrosion inhibitors using bioluminescent enzymatic method. Moscow Univ. Chem. Bull. 2015, 70, 197–201. [Google Scholar] [CrossRef]
- Mempin, R.; Tran, H.; Chen, C.; Gong, H.; Ho, K.K.; Lu, S. Release of extracellular ATP by bacteria during growth. BMC Microbiol. 2013, 13, 301. [Google Scholar] [CrossRef] [PubMed]
- Efremenko, E.N.; Azizov, R.E.; Makhlis, T.A.; Abbasov, V.M.; Varfolomeev, S.D. Determination of minimal concentrations of biocorrosion inhibitors by a bioluminescence method. Prikl Biokhim Mikrobiol. 2005, 41, 377–381. [Google Scholar] [CrossRef]
- Efremenko, E.N.; Azizov, R.E.; Raeva, A.A.; Abbasov, V.M.; Varfolomeyev, S.D. An approach to the rapid control of oil spill bioremediation by bioluminescent method of intracellular ATP determination. Int. Biodeterior. Biodegrad. 2005, 56, 94–100. [Google Scholar] [CrossRef]
- Efremenko, E.N.; Tatarinova, N.Y. The effect of long-term preservation of bacterial cells immobilized in poly(vinyl alcohol) cryogel on their viability and biosynthesis of target metabolites. Microbiology 2007, 76, 336–341. [Google Scholar] [CrossRef]
- Senko, O.; Maslova, O.; Gladchenko, M.; Gaydamaka, S.; Akopyan, A.; Lysenko, S.; Karakhanov, E.; Efremenko, E. Prospective approach to the anaerobic bioconversion of benzo- and dibenzothiophene sulfones to sulfide. Molecules 2019, 24, 1736. [Google Scholar] [CrossRef] [PubMed]
- Kononikhin, A.S.; Zherebker, A.Y.; Kazachkov, M.A.; Grigiriev, A.S.; Kostyukevich, Y.I.; Pekov, S.I.; Bocharov, K.V.; Popov, I.A.; Perminova, I.V.; Nikolaev, E.N. The study of the molecular composition of humic substances of coal and peat by high resolution mass spectrometry with optimal electrospray conditions. Proc. Russ. Acad. Sci. Power Eng. J. 2017, 1, 107–114. [Google Scholar]
- Dubber, D.; Gray, N.F. Replacement of chemical oxygen demand (COD) with total organic carbon (TOC) for monitoring wastewater treatment performance to minimize disposal of toxic analytical waste. J. Environ. Sci. Health A Tox. Hazard. Subst. Environ. Eng. 2010, 45, 1595–1600. [Google Scholar] [CrossRef] [PubMed]
- Angelidaki, I.; Alves, M.; Bolzonella, D.; Borzacconi, L.; Campos, J.L.; Guwy, A.J.; Kalyuzhnyi, S.; Jenicek, P.; Van Lier, J.B. Defining the biomethane potential (BMP) of solid organic wastes and energy crops: A proposed protocol for batch assays. Water Sci. Technol. 2009, 59, 927–934. [Google Scholar] [CrossRef] [PubMed]
- Stepanov, N.; Efremenko, E. Immobilised cells of Pachysolen tannophilus yeast for ethanol production from crude glycerol. New Biotechnol. 2017, 34, 54–58. [Google Scholar] [CrossRef] [PubMed]
- Senko, O.; Gladchenko, M.; Maslova, O.; Efremenko, E. Long-term storage and use of artificially immobilized anaerobic sludge as a powerful biocatalyst for conversion of various wastes including those containing xenobiotics to biogas. Catalysts 2019, 9, 326. [Google Scholar] [CrossRef]
- Liu, K.; Chen, Y.; Xiao, N.; Zheng, X.; Li, M. Effect of humic acids with different characteristics on fermentative short-chain fatty acids production from waste activated sludge. Environ. Sci. Technol. 2015, 49, 4929–4936. [Google Scholar] [CrossRef]
- Azman, S.; Khadem, A.F.; Plugge, C.M.; Stams, A.J.; Bec, S.; Zeeman, G. Effect of humic acid on anaerobic digestion of cellulose and xylan in completely stirred tank reactors: Inhibitory effect, mitigation of the inhibition and the dynamics of the microbial communities. Appl. Microbiol. Biotechnol. 2017, 101, 889–901. [Google Scholar] [CrossRef]
- Li, H.; Li, Y.; Li, C. Evolution of humic substance during anaerobic sludge digestion. Environ. Eng. Manag. J. 2017, 16, 1577–1582. [Google Scholar] [CrossRef]
- Yan, Z.; Joshi, P.; Gorski, C.A.; Ferry, J.G. A biochemical framework for anaerobic oxidation of methane driven by Fe (III)-dependent respiration. Nat. Commun. 2018, 9, 1642. [Google Scholar] [CrossRef] [PubMed]
- Holmes, D.; Ueki, T.; Tang, H.Y.; Zhou, J.; Smith, J.; Chaput, G.; Lovley, D. A membrane-bound cytochrome enables Methanosarcina acetivorans to conserve energy to support growth from extracellular electron transfer. bioRxiv 2019, 590380. [Google Scholar] [CrossRef]

| Consortium | Dry Matter (g/L) | Ash (%) | Biomass VSS (g/L) | * Activity (mg COD/g VSS/day) | Initial Concentration of ATP, ×10−12 mol/mL | |
|---|---|---|---|---|---|---|
| Acidogenic | Methanogenic | |||||
| I | 64.3 ± 3.1 | 42.4 ± 0.9 | 35.3 ± 1.1 | 2210 ± 30 | 315 ± 5 | 15.7 ± 0.6 |
| II | 57.1 ± 2.3 | 37.8 ± 1.6 | 35.4 ± 1.2 | 1550 ± 30 | 86 ± 1 | 26.4 ± 0.8 |
| III | 56.6 ± 1.6 | 38.5 ± 1.5 | 34.8 ± 1.1 | 2180 ± 30 | 195 ± 10 | 4.4 ± 0.3 |
| IV | 58.2 ± 2.2 | 42.7 ± 1.7 | 33.0 ± 1.1 | 1970 ± 30 | 157 ± 1 | 15.1 ± 0.6 |
| HС, g/L | Consortium | |||
|---|---|---|---|---|
| I | II | III | IV | |
| 0 (control) | 14.5 ± 0.5 | 6.6 ± 0.3 | 6.3 ± 0.3 | 10.2 ± 0.4 |
| PH | ||||
| 1 | 12.7 ± 0.4 | 5.2 ± 0.2 | 0.7 ± 0.1 | 1.5 ± 0.2 |
| 5 | 4.5 ± 0.3 | 1.7 ± 0.2 | 0.3 ± 0.1 | 0.9 ± 0.1 |
| 10 | 0.5 ± 0.1 | 0.9 ± 0.1 | 0.2 ± 0.1 | 0.3 ± 0.1 |
| FA | ||||
| 1 | 20.1 ± 0.6 | 5.9 ± 0.3 | 25.2 ± 0.6 | 2.3 ± 0.2 |
| 5 | 24.9 ± 0.7 | 3.5 ± 0.2 | 17.6 ± 0.5 | 1.5 ± 0.2 |
| 10 | 32.9 ± 0.9 | 2.1 ± 0.2 | 15.4 ± 0.4 | 0.6 ± 0.1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stepanov, N.; Senko, O.; Perminova, I.; Efremenko, E. A New Approach to Assess the Effect of Various Humic Compounds on the Metabolic Activity of Cells Participating in Methanogenesis. Sustainability 2019, 11, 3158. https://doi.org/10.3390/su11113158
Stepanov N, Senko O, Perminova I, Efremenko E. A New Approach to Assess the Effect of Various Humic Compounds on the Metabolic Activity of Cells Participating in Methanogenesis. Sustainability. 2019; 11(11):3158. https://doi.org/10.3390/su11113158
Chicago/Turabian StyleStepanov, Nikolay, Olga Senko, Irina Perminova, and Elena Efremenko. 2019. "A New Approach to Assess the Effect of Various Humic Compounds on the Metabolic Activity of Cells Participating in Methanogenesis" Sustainability 11, no. 11: 3158. https://doi.org/10.3390/su11113158
APA StyleStepanov, N., Senko, O., Perminova, I., & Efremenko, E. (2019). A New Approach to Assess the Effect of Various Humic Compounds on the Metabolic Activity of Cells Participating in Methanogenesis. Sustainability, 11(11), 3158. https://doi.org/10.3390/su11113158





