Biofuel Production and Phosphorus Recovery through an Integrated Treatment of Agro-Industrial Waste
Abstract
1. Introduction
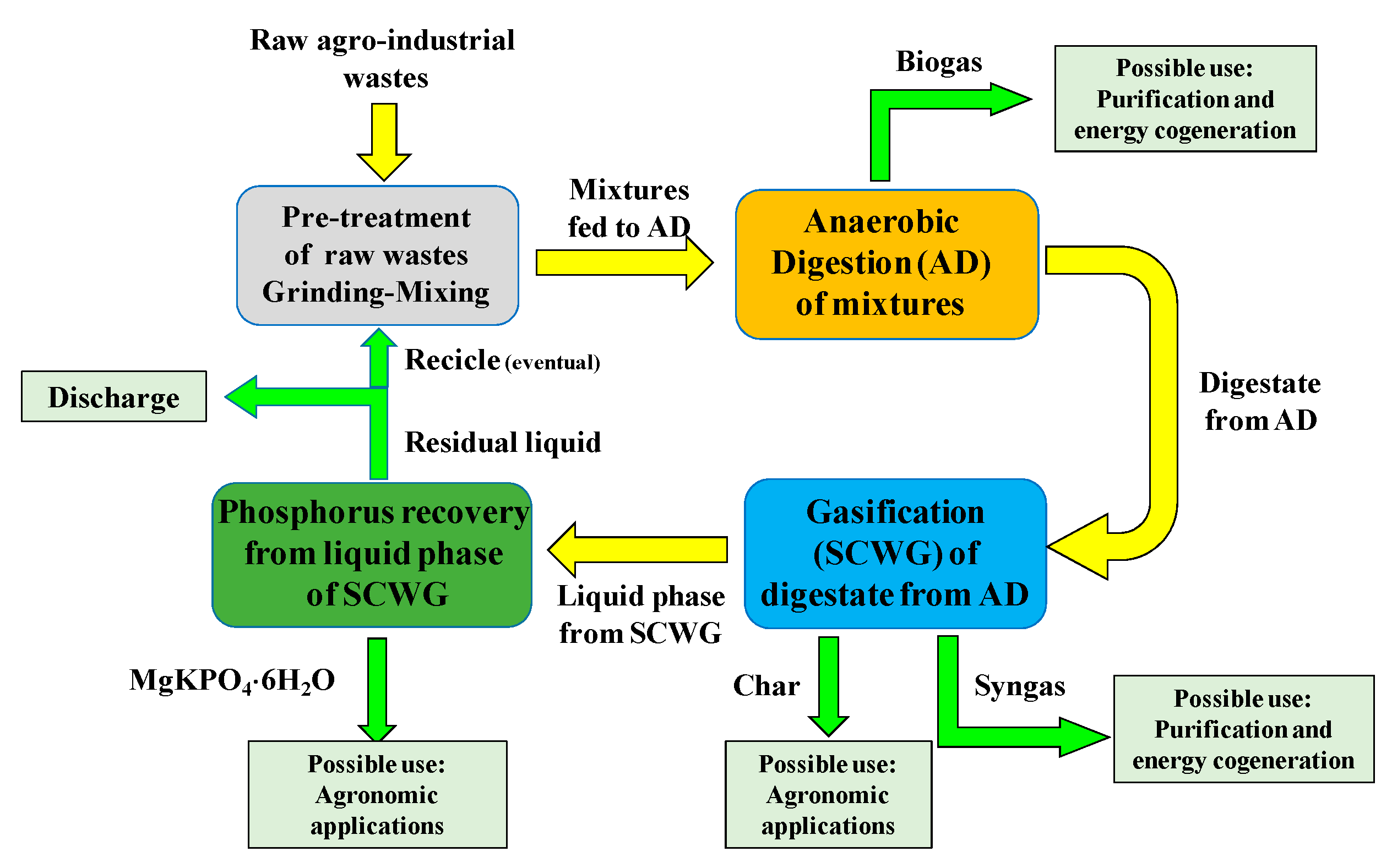
2. Materials and Methods
2.1. Co-Digestion Tests
2.2. SCWG Reactor and Procedure for SCWG Tests
2.3. Precipitation Tests for Phosphorus Recovery
2.4. Analytical Methods
3. Results and Discussion
3.1. Characterization of Agro-industrial Waste and Activated Sludge
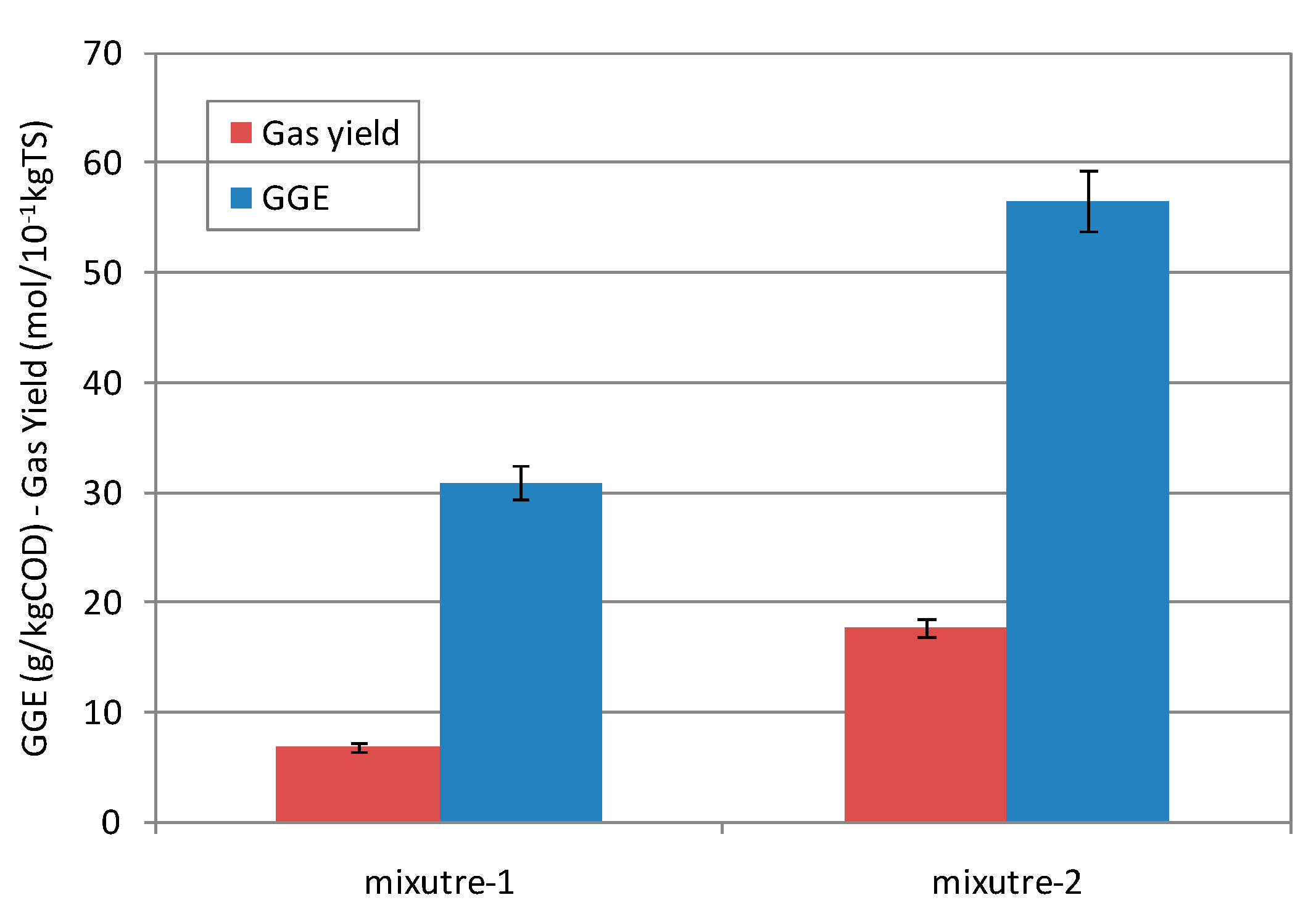
3.2. Co-Digestion Tests
3.2.1 Characterization of Mixtures Prepared for Co-Digestion
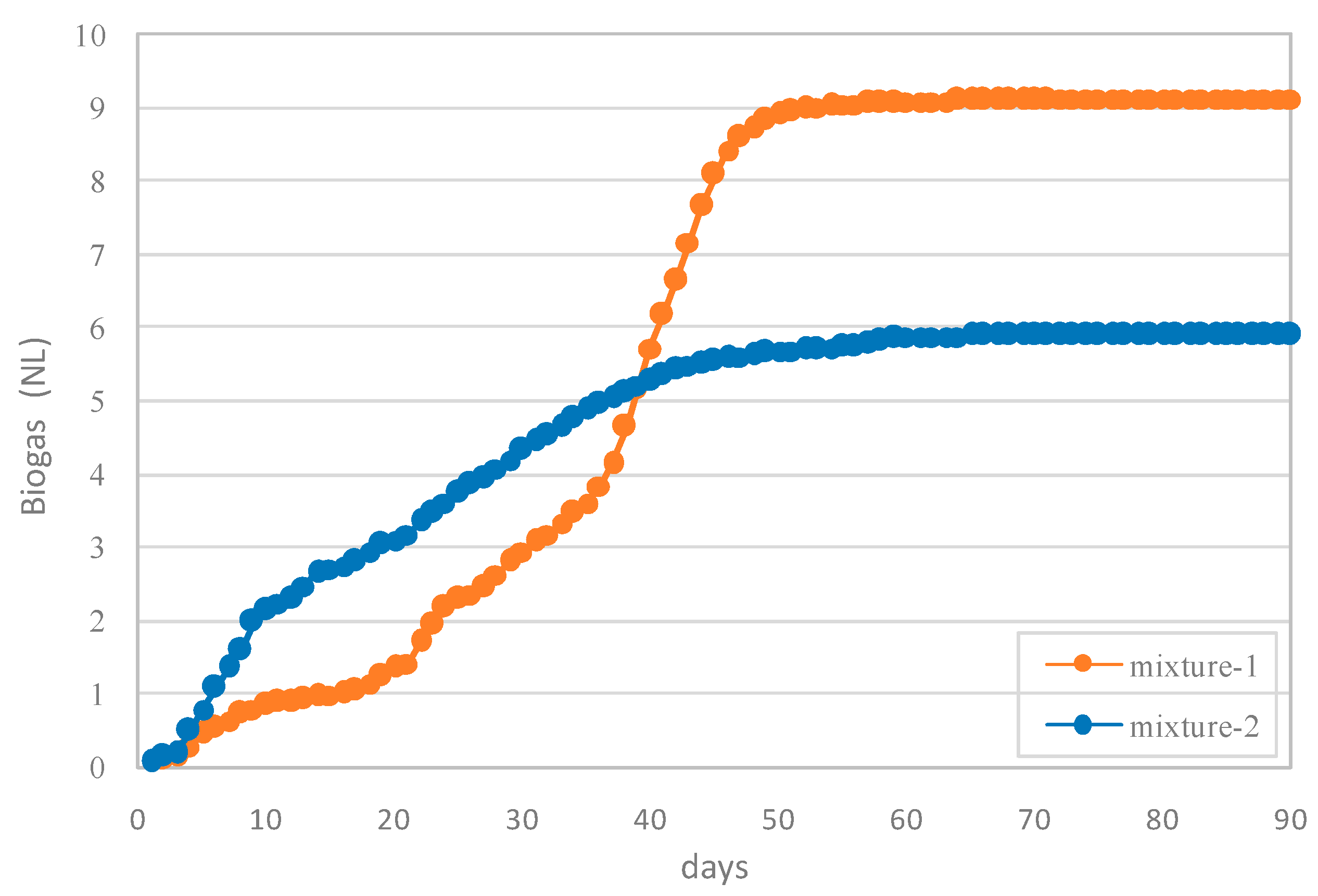
3.2.2 Performance of the Co-Digestion Process
3.3. SCWG Tests
3.3.1 Characteristics of Digestate Fed to SCWG
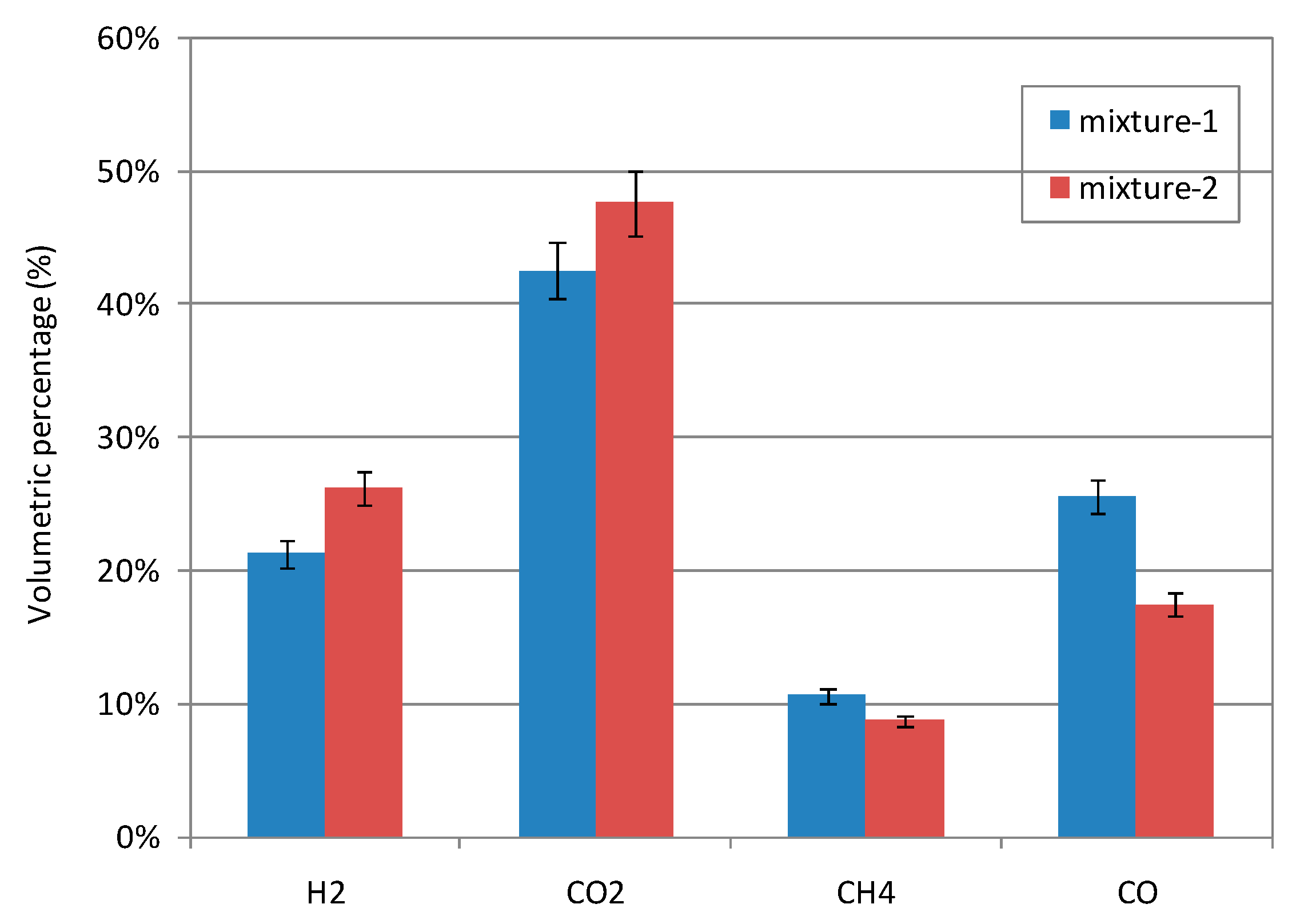
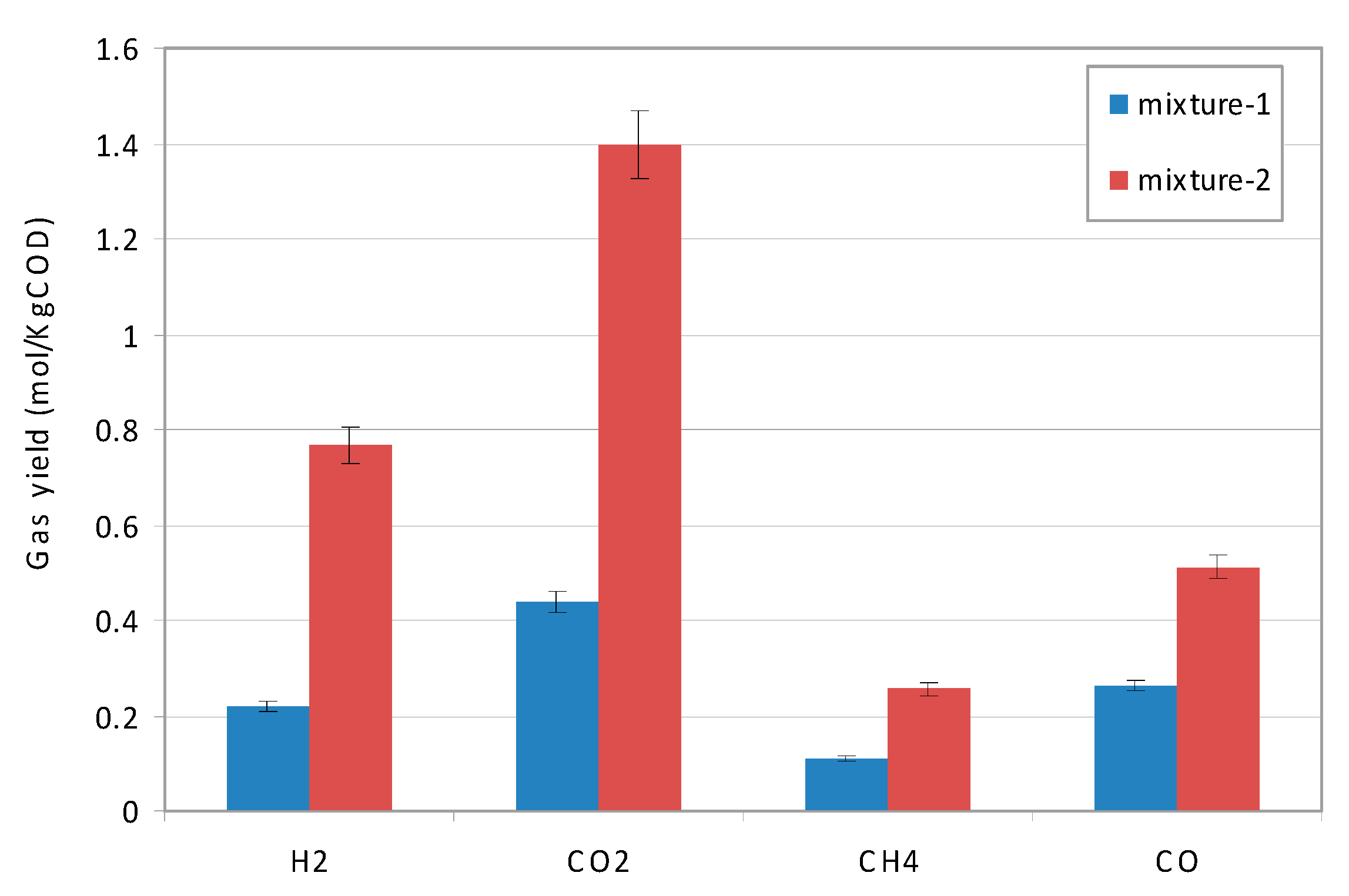
3.3.2 Performance of SCWG
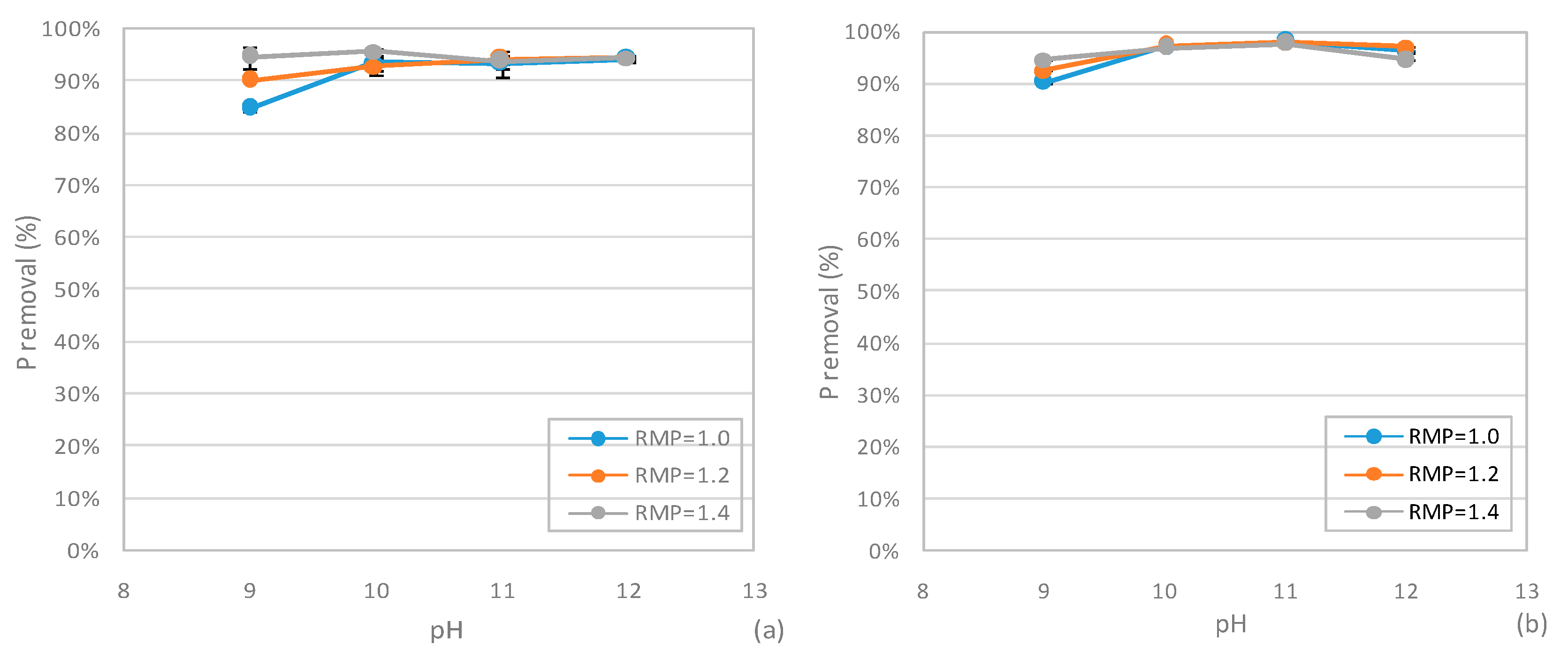
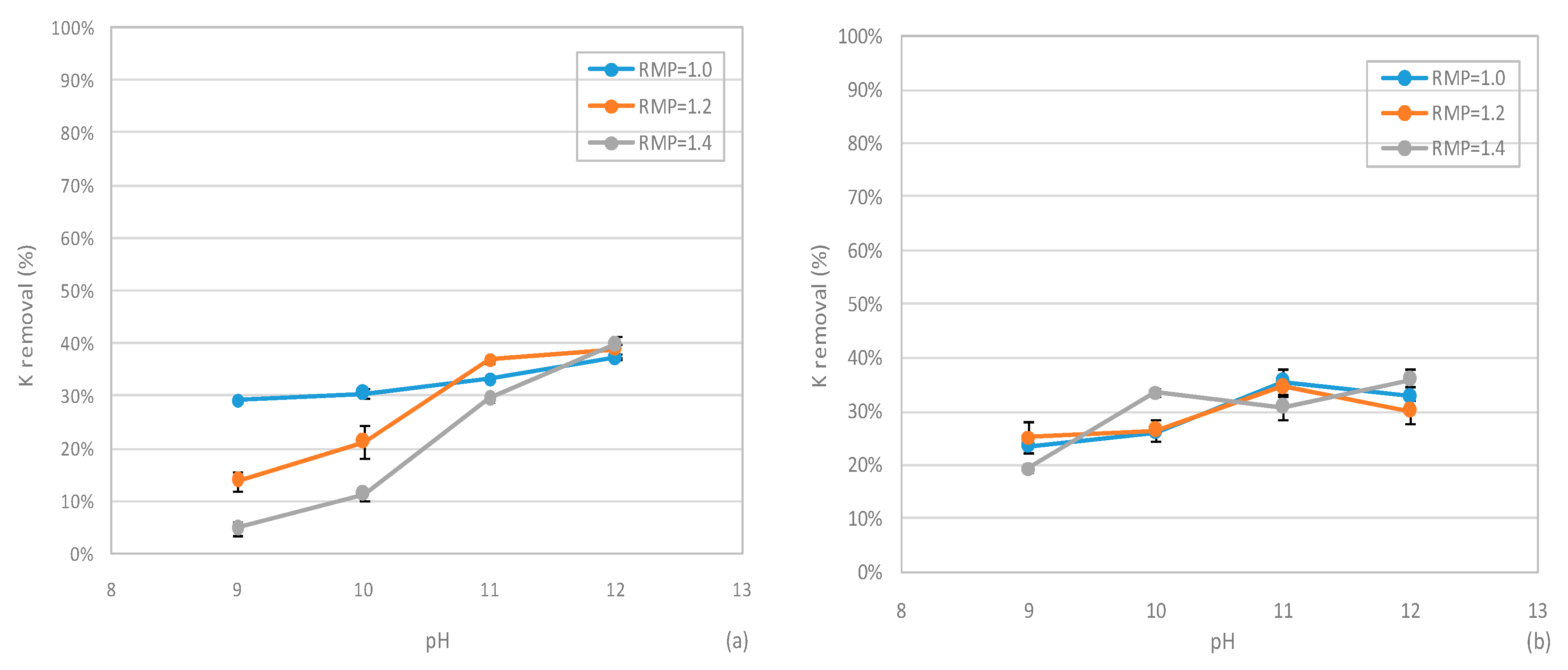
3.4. Phosphorus Recovery from SCWG Liquid Phase
3.4.1 Characteristics of Samples Subjected to Phosphorus Recovery
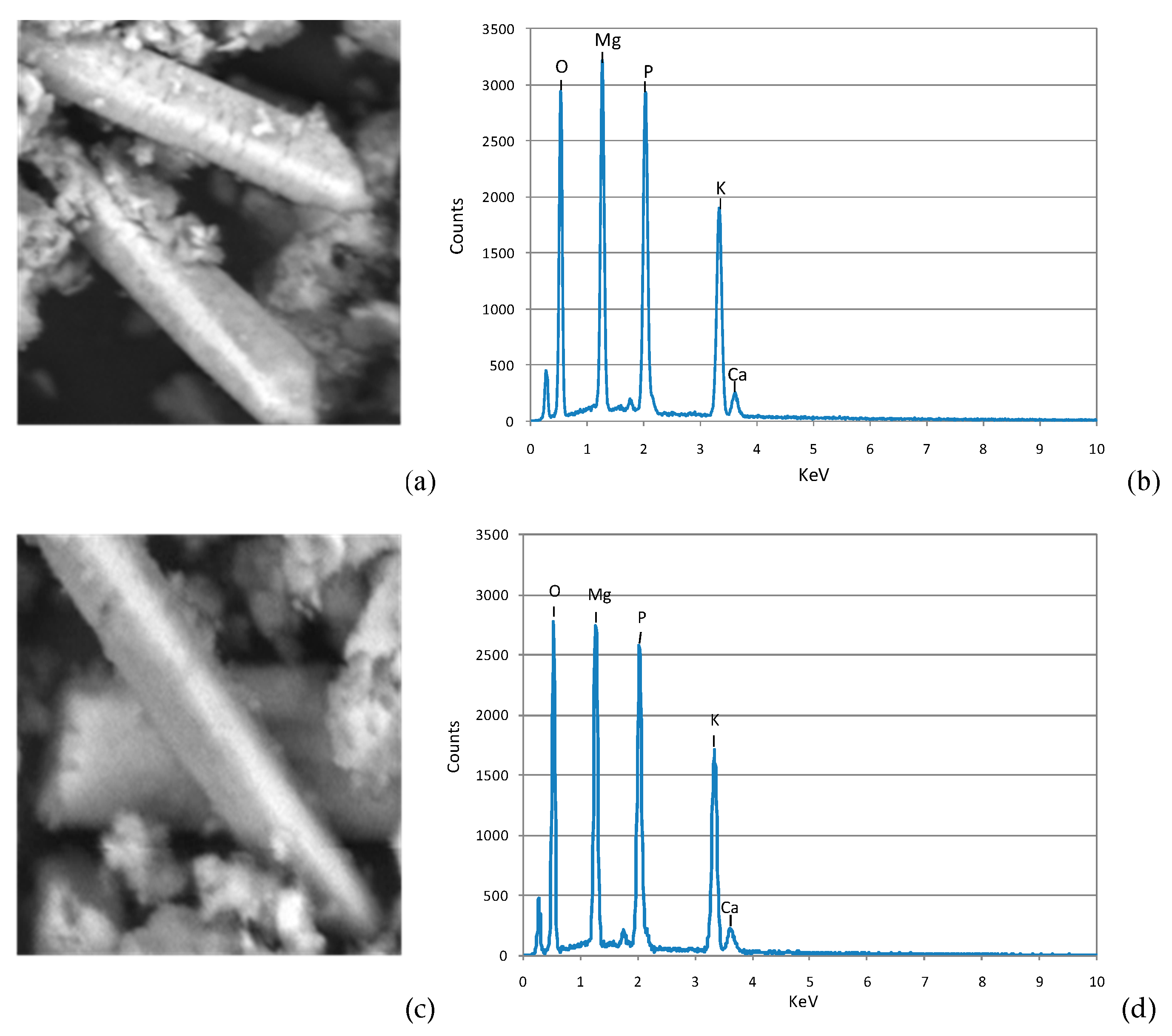
3.4.2 Performances of MPP Precipitation Tests
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Gontard, N.; Sonesson, U.; Birkved, M.; Majone, M.; Bolzonella, D.; Celli, A.; Angellier-Coussy, H.; Jang, G.-W.; Verniquet, A.; Broeze, J.; et al. A research challenge vision regarding management of agricultural waste in a circular bio-based economy. Crit. Rev. Environ. Sci. Technol. 2018, 1–41. [Google Scholar] [CrossRef]
- Carlini, M.; Mosconi, E.M.; Castellucci, S.; Villarini, M.; Colantoni, A. An economical evaluation of anaerobic digestion plants fed with organic agro-industrial wastes. Energies 2017, 10, 1165. [Google Scholar] [CrossRef]
- Hernández, D.; Riaño, B.; Coca, M.; García-González, M.C. Treatment of agro-industrial wastewater using microalgae–bacteria consortium combined with anaerobic digestion of the produced biomass. Bioresour. Technol. 2013, 135, 598–603. [Google Scholar] [CrossRef] [PubMed]
- Siciliano, A.; Stillitano, M.A.; De Rosa, S. Increase of the anaerobic biodegradability of olive mill wastewaters through a pre-treatment with hydrogen peroxide in alkaline conditions. Desal. Water. Treat. 2014, 55, 1735–1746. [Google Scholar] [CrossRef]
- Calabrò, P.S.; Fòlino, A.; Tamburino, V.; Zappia, G.; Zema, D.A. Increasing the tolerance of polyphenols of the anaerobic digestion of olive wastewater through microbial adaptation. Biosyst. Eng. 2018, 172, 19–28. [Google Scholar] [CrossRef]
- Fezzani, B.; Ben Cheickh, R. Two-phase anaerobic co-digestion of olive mill wastes in semi-continuous digesters at mesophilic temperature. Bioresour. Technol. 2010, 101, 1628–1634. [Google Scholar] [CrossRef] [PubMed]
- Siciliano, A.; Stillitano, M.A.; De Rosa, S. Biogas production from wet olive mill wastes pretreated with hydrogen peroxide in alkaline conditions. Renew. Energy 2016, 85, 903–916. [Google Scholar] [CrossRef]
- Juneja, A.; Ceballos, R.M.; Murthy, G.S. Effects of Environmental Factors and Nutrient Availability on the Biochemical Composition of Algae for Biofuels Production: A Review. Energies 2013, 6, 4607–4638. [Google Scholar] [CrossRef]
- Siciliano, A.; Stillitano, M.A.; Limonti, C. Energetic valorization of wet olive mill wastes through a suitable integrated treatment: H2O2 with lime and anaerobic digestion. Sustainability 2016, 8, 1150. [Google Scholar] [CrossRef]
- Zema, D.A.; Fòlino, A.; Zappia, G.; Calabrò, P.S.; Tamburino, V.; Zimbone, S.M. Anaerobic digestion of orange peel in a semi-continuous pilot plant: An environmentally sound way of citrus waste management in agro-ecosystems. Sci. Total Environ. 2018, 630, 401–408. [Google Scholar] [CrossRef]
- Khanal, S. Anaerobic Biotechnology for Bioenergy Production: Principles and Applications; Wiley-Blackwell: Ames, IA, USA, 2008. [Google Scholar]
- Roubík, H.; Mazancová, J.; Le Dinh, P.; Van Dinh, D.; Banout, J. Biogas quality across small-scale biogas plants: A case of central Vietnam. Energies 2018, 11, 1794. [Google Scholar] [CrossRef]
- Mehariya, S.; Patel, A.K.; Obulisamy, P.K.; Punniyakotti, E.; Wong, J.W.C. Co-digestion of food waste and sewage sludge for methane production: Current status and perspective. Bioresour. Technol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Álvarez, J.A.; Otero, L.; Lema, J.M. A methodology for optimising feed composition for anaerobic co-digestion of agro-industrial wastes. Bioresour. Technol. 2010, 101, 1153–1158. [Google Scholar] [CrossRef] [PubMed]
- Zahan, Z.; Othman, M.Z.; Muster, T.H. Anaerobic digestion/co-digestion kinetic potentials of different agro-industrial wastes: A comparative batch study for C/N optimisation. Waste Manag. 2018, 71, 663–674. [Google Scholar] [CrossRef] [PubMed]
- Keucken, A.; Habagil, M.; Batstone, D.; Jeppsson, U.; Arnell, M. Anaerobic Co-Digestion of Sludge and Organic Food Waste—Performance, Inhibition, and Impact on the Microbial Community. Energies 2018, 11, 2325. [Google Scholar] [CrossRef]
- Goberna, M.; Podmirseg, S.M.; Waldhuber, S.; Knapp, B.A.; García, C.; Insam, H. Pathogenic bacteria and mineral N in soils following the land spreading of biogas digestates and fresh manure. Appl. Soil Ecol. 2011, 49, 18–25. [Google Scholar] [CrossRef]
- Codignole Luz, F.; Volpe, M.; Fiori, L.; Manni, A.; Cordiner, S.; Mulone, V.; Rocco, V. Spent coffee enhanced biomethane potential via an integrated hydrothermal carbonization-anaerobic digestion process. Bioresour. Technol. 2018, 256, 102–109. [Google Scholar] [CrossRef]
- Basu, P.; Mettanant, V. Biomass Gasification in Supercritical Water—A Review. Int. J. Chem. React. Eng. 2009, 7. [Google Scholar] [CrossRef]
- Nanda, S.; Isen, J.; Dalai, A.K.; Kozinski, J.A. Gasification of fruit wastes and agro-food residues in supercritical water. Energy Convers. Manag. 2016, 110, 296–306. [Google Scholar] [CrossRef]
- Amrullah, A.; Matsumura, Y. Supercritical water gasification of sewage sludge in continuous reactor. Bioresour. Technol. 2018, 249, 276–283. [Google Scholar] [CrossRef]
- Molino, A.; Larocca, V.; Chianese, S.; Musmarra, D. Biofuels Production by Biomass Gasification: A Review. Energies 2018, 11, 811. [Google Scholar] [CrossRef]
- Kruse, A. Supercritical water gasification. Biofuels Bioprod. Biorefining 2008, 2, 415–437. [Google Scholar] [CrossRef]
- Yakaboylu, O.; Harinck, J.; Smit, K.G.; de Jong, W. Supercritical Water Gasification of Biomass: A Literature and Technology Overview. Energies 2015, 8, 859–894. [Google Scholar] [CrossRef]
- Matsumura, Y.; Minowa, T. Fundamental design of a continuous biomass gasification process using a supercritical water fluidized bed. Int. J. Hydrogen Energy 2004, 29, 701–707. [Google Scholar] [CrossRef]
- Molino, A.; Nanna, F.; Villone, A.; Iovane, P.; Tarquini, P.; Migliori, M.; Giordano, G.; Braccio, G. Pressure and time effect over semi-continuous gasification of zootechnical sludge near critical condition of water for green chemicals production. Fuel 2014, 136, 172–176. [Google Scholar] [CrossRef]
- Molino, A.; Migliori, M.; Blasi, A.; Davoli, M.; Marino, T.; Chianese, S.; Catizone, E.; Giordano, G. Municipal waste leachate conversion via catalytic supercritical water gasification process. Fuel 2017, 206, 155–161. [Google Scholar] [CrossRef]
- Chianese, S.; Loipersböck, J.; Malits, M.; Rauch, R.; Hofbauer, H.; Molino, A.; Musmarra, D. Hydrogen from the high temperature water gas shift reaction with an industrial Fe/Cr catalyst using biomass gasification tar rich synthesis gas. Fuel Process. Technol. 2015, 132, 39–48. [Google Scholar] [CrossRef]
- Molino, A.; Larocca, V.; Valerio, V.; Martino, M.; Marino, T.; Rimauro, J.; Casella, P. Biofuels and Bio-based Production via Supercritical Water Gasification of Peach Scraps. Energy Fuels 2016, 30, 10443–10447. [Google Scholar] [CrossRef]
- Jahirul, M.I.; Rasul, M.G.; Chowdhury, A.A.; Ashwath, N. Biofuels Production through Biomass Pyrolysis —A Technological Review. Energies 2012, 5, 4952–5001. [Google Scholar] [CrossRef]
- Panepinto, D.; Fiore, S.; Genon, G.; Acri, M. Thermal valorization of sewer sludge: Perspectives for large wastewater treatment plants. J. Clean. Prod. 2016, 137, 1323–1329. [Google Scholar] [CrossRef]
- Molino, A.; Giordano, G.; Migliori, M.; Lauro, V.; Santarcangelo, G.; Marino, T.; Larocca, V.; Tarquini, P. Process Innovation Via Supercritical Water Gasification to Improve the Conventional Plants Performance in Treating Highly Humid Biomass. Waste Biomass Valorization 2016, 7, 1289–1295. [Google Scholar] [CrossRef]
- Gasafi, E.; Reinecke, M.-Y.; Kruse, A.; Schebek, L. Economic analysis of sewage sludge gasification in supercritical water for hydrogen production. Biomass Bioenergy 2008, 32, 1085–1096. [Google Scholar] [CrossRef]
- Gong, M.; Zhu, W.; Xu, Z.R.; Zhang, H.W.; Yang, H.P. Influence of sludge properties on the direct gasification of dewatered sewage sludge in supercritical water. Renew. Energy 2014, 66, 605–611. [Google Scholar] [CrossRef]
- Gorazda, K.; Tarko, B.; Werle, S.; Wozrek, Z. Sewage sludge as a fuel and raw material for phosphorus recovery: Combined process of gasification and P extraction. Waste Manag. 2018, 73, 404–415. [Google Scholar] [CrossRef] [PubMed]
- Acelas, N.Y.; López, D.P.; Brilman, D.W.F.; Kersten, S.R.A.; Kootstra, A.M.J. Supercritical water gasification of sewage sludge: Gas production and phosphorus recovery. Bioresour. Technol. 2014, 174, 167–175. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Wang, C.; Liu, H.; Qian, Y. Simultaneous removal of phosphorus and potassium from synthetic urine through the precipitation of magnesium potassium phosphate hexahydrate. Chemosphere 2011, 84, 207–212. [Google Scholar] [CrossRef]
- Wilsenach, J.A.; Schuurbiers, C.A.H.; van Loosdrecht, M.C.M. Phosphate and potassium recovery from source separated urine through struvite precipitation. Water Res. 2007, 41, 458–466. [Google Scholar] [CrossRef]
- Siciliano, A.; De Rosa, S. Recovery of ammonia in digestates of calf manure through a struvite precipitation process using unconventional reagents. Environ. Technol. 2014, 35, 841–850. [Google Scholar] [CrossRef]
- Karabegovic, L.; Uldal, M.; Werker, A.; Morgan-Sagastume, F. Phosphorus recovery potential from a waste stream with high organic and nutrient contents via struvite precipitation. Environ. Technol. 2013, 34, 871–883. [Google Scholar] [CrossRef]
- Kabdaşli, I.; Tünay, O.; Özcan, P. Application of struvite precipitation coupled with biological treatment to slaughterhouse wastewaters. Environ. Technol. 2009, 30, 1095–1101. [Google Scholar] [CrossRef]
- Battistoni, P.; Boccadoro, R.; Fatone, F.; Pavan, P. Auto-Nucleation and Crystal Growth of Struvite in a Demonstrative Fluidized Bed Reactor (FBR). Environ. Technol. 2010, 26, 975–982. [Google Scholar] [CrossRef] [PubMed]
- Siciliano, A. Assessment of fertilizer potential of the struvite produced from the treatment of methanogenic landfill leachate using low-cost reagents. Environ. Sci. Pollut. Res. 2016, 23, 5949–5959. [Google Scholar] [CrossRef] [PubMed]
- Korchef, A.; Saidou, H.; Ben Amor, M. Phosphate recovery through struvite precipitation by CO2 removal: Effect of magnesium, phosphate and ammonium concentrations. J. Hazard. Mater. 2011, 186, 602–613. [Google Scholar] [CrossRef] [PubMed]
- Siciliano, A.; Stillitano, M.A.; Limonti, C.; Marchio, F. Ammonium removal from landfill leachate by means of multiple recycling of struvite residues obtained through acid decomposition. Appl. Sci. 2016, 6, 345. [Google Scholar] [CrossRef]
- Quintana, M.; Colmenarejo, M.F.; Barrera, J.; García, G.; García, E.; Bustos, A. Use of a byproduct of magnesium oxide production to precipitate phosphorus and nitrogen as struvite from wastewater treatment liquors. J. Agric. Food Chem. 2004, 52, 294–299. [Google Scholar] [CrossRef] [PubMed]
- Siciliano, A.; Ruggiero, C.; De Rosa, S. A new integrated treatment for the reduction of organic and nitrogen loads in methanogenic landfill leachates. Process Saf. Environ. Prot. 2013, 91, 311–320. [Google Scholar] [CrossRef]
- Gunay, A.; Karadag, D.; Tosun, I.; Ozturk, M. Use of magnesit as a magnesium source for ammonium removal from leachate. J. Hazard. Mater. 2008, 156, 619–623. [Google Scholar] [CrossRef]
- APHA. Standard Methods for the Examination of Water and Wastewater, 20th ed.; APHA: Washington, DC, USA, 1998. [Google Scholar]
- Metcalf & Eddy. Wastewater Engineering: Treatment and Reuse, 4th ed.; McGraw-Hill: New York, NY, USA, 2003. [Google Scholar]
- Siciliano, A.; De Rosa, S. Experimental formulation of a kinetic model describing the nitrification process in biological aerated filters filled with plastic elements. Environ. Technol. 2015, 36, 293–301. [Google Scholar] [CrossRef]
- Siciliano, A.; De Rosa, S. An experimental model of COD abatement in MBBR based on biofilm growth dynamic and on substrates’ removal kinetics. Environ. Technol. 2016, 37, 2058–2071. [Google Scholar] [CrossRef]
- Le Corre, K.S.; Valsami-Jones, E.; Hobbs, P.; Parsons, S.A. Impact of calcium on struvite crystal size, shape and purity. J. Cryst. Growth. 2005, 283, 514–522. [Google Scholar] [CrossRef]

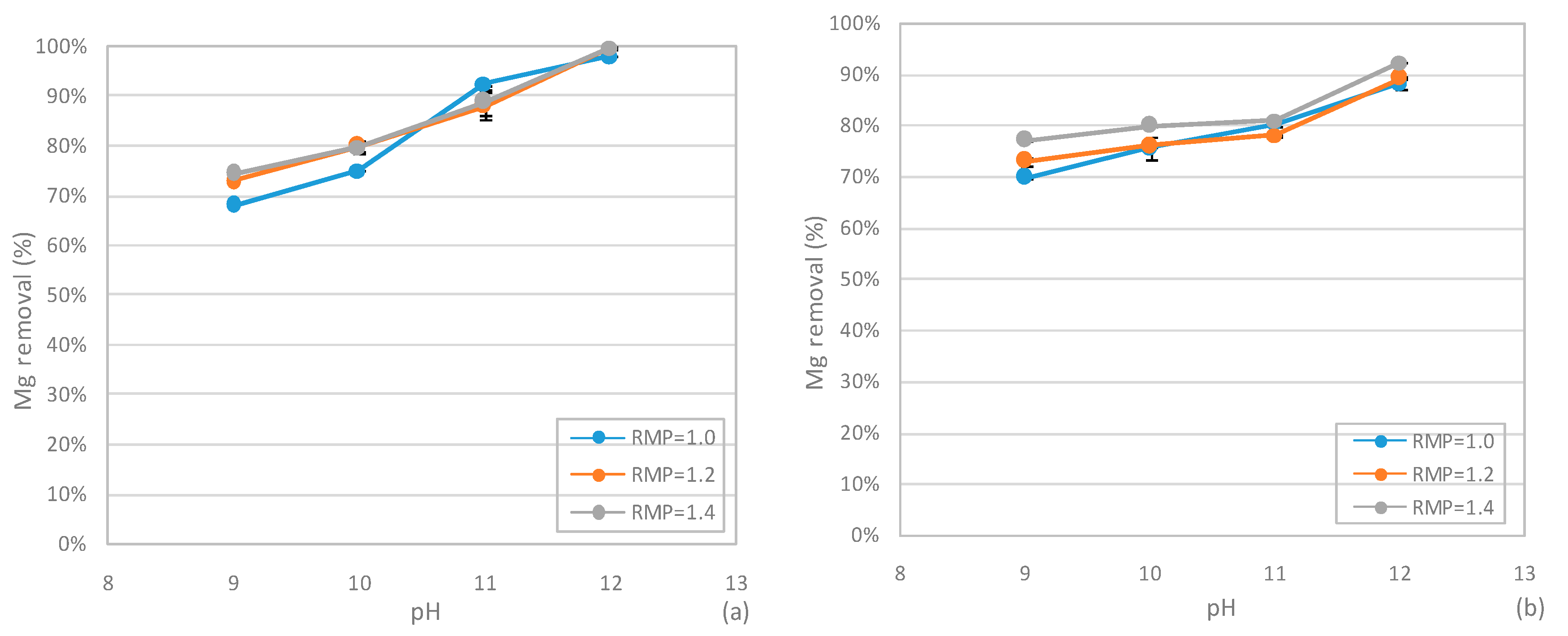
| Parameter | Type and Characteristics of Agro-Industrial Wastes | |||||
|---|---|---|---|---|---|---|
| Tomato Waste | Kiwifruit Waste | Asparagus Waste | Potato Waste | Cattle Manure | Activated Sludge | |
| pH | 4.4 | 3.2 | 6.4 | 6.0 | 7.2 | 6.7 |
| Conductivity [mS/cm] | 8.4 | 14.4 | 17.8 | 13.8 | 34.1 | 2.2 |
| COD [g/L] | 88.0 | 236.9 | 105.9 | 208.9 | 104.8 | 115.4 |
| TS [g/L] | 87.5 | 222.9 | 81.4 | 180.8 | 93.1 | 98.9 |
| VS [g/L] | 78.0 | 204.5 | 74.3 | 170.1 | 73.9 | 81.3 |
| TS [%TQ] | 8.75 | 22.3 | 8.1 | 18.1 | 9.3 | 9.9 |
| VS [%TQ] | 7.8 | 20.4 | 7.4 | 17.0 | 7.4 | 8.1 |
| VS [%TS] | 89.1 | 91.7 | 91.3 | 94 | 79.3 | 82.2 |
| TKN [g/L] | 1.67 | 3.10 | 4.51 | 3.54 | 4.92 | 1.63 |
| N-NH4+ [g/L] | 0.342 | 0.360 | 0.424 | 0.588 | 1.259 | 0.693 |
| P-PO43− [g/L] | 0.574 | 0.697 | 1.174 | 0.380 | 0.908 | 0.702 |
| SO42− [g/L] | 0.480 | 0.317 | 1.575 | 0.254 | 3.135 | 1.207 |
| Alkalinity [gCaCO3/L] | 6.205 | 5.160 | 9.292 | 14.777 | 28.055 | 1.506 |
| VFA [g CH3COOH/L] | 4.055 | 4.049 | 7.442 | 16.806 | 17.814 | 2.962 |
| VFA/Alkalinity | 0.65 | 0.78 | 0.8 | 1.13 | 0.0.63 | 1.97 |
| COD/N | 52.7 | 76.4 | 23.5 | 59.0 | 21.3 | 70.67 |
| N/P | 2.9 | 4.4 | 3.8 | 9.3 | 5.4 | 2.3 |
| Parameter | Mixture 1 | Mixture 2 | ||||
|---|---|---|---|---|---|---|
| Mixture fed to AD | Digestate from AD | Liquid from SCWG | Mixture fed to AD | Digestate from AD | Liquid from SCWG | |
| pH | 7.2 | 8.2 | 11.9 | 7.5 | 5.0 | 11.88 |
| Conductivity [mS/cm] | 11.5 | 16.3 | 31.7 | 12.4 | 18.2 | 34.5 |
| COD [g/L] | 100.8 | 30.4 | 0.150 | 170.1 | 82.5 | 0.181 |
| TS [g/L] | 89.7 | 45.9 | 28.5 | 151.5 | 95.1 | 30.7 |
| VS [g/L] | 81.4 | 20.5 | 0.14 | 135.1 | 57.3 | 0.17 |
| TS [%TQ] | 8.9 | 4.6 | 2.85 | 15.15 | 9.5 | 3.07 |
| VS [%TQ] | 8.14 | 2.0 | 0.014 | 13.5 | 5.7 | 0.017 |
| VS [%TS] | 90.7 | 44.5 | 0.5 | 89.2 | 60.25 | 0.55 |
| TKN [g/L] | 2.54 | 2.25 | - | 2.876 | 2.68 | - |
| N-NH4+ [g/L] | 0.58 | 1.468 | - | 0.576 | 1.196 | - |
| P-PO43− [g/L] | 0.826 | 1.128 | 0.462 | 0.604 | 0.988 | 0.503 |
| SO42− [g/L] | 1.373 | 1.310 | 0.592 | 0.767 | 0.602 | 0.534 |
| K+ [g/L] | 1.64 | 1.71 | 1.81 | 2.58 | 2.61 | 2.82 |
| Mg2+ [g/L] | 0.39 | 0.32 | 0.036 | 0.38 | 0.41 | 0.028 |
| Ca2+ [g/L] | 0.44 | 0.37 | 0.07 | 0.49 | 0.55 | 0.08 |
| Alkalinity [gCaCO3/L] | 20.171 | 17.693 | 18.48 | 14.843 | 15.130 | 19.33 |
| VFA [gCH3COOH/L] | 6.144 | 3.489 | - | 8.260 | 61.461 | - |
| VFA/ALK | 0.30 | 0.19 | - | 0.55 | 4.06 | - |
| COD/N | 39.87 | 13.51 | - | 59.1 | 30.8 | - |
| N/P | 3.07 | 2.0 | - | 4.76 | 2.71 | - |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Siciliano, A.; Limonti, C.; Mehariya, S.; Molino, A.; Calabrò, V. Biofuel Production and Phosphorus Recovery through an Integrated Treatment of Agro-Industrial Waste. Sustainability 2019, 11, 52. https://doi.org/10.3390/su11010052
Siciliano A, Limonti C, Mehariya S, Molino A, Calabrò V. Biofuel Production and Phosphorus Recovery through an Integrated Treatment of Agro-Industrial Waste. Sustainability. 2019; 11(1):52. https://doi.org/10.3390/su11010052
Chicago/Turabian StyleSiciliano, Alessio, Carlo Limonti, Sanjeet Mehariya, Antonio Molino, and Vincenza Calabrò. 2019. "Biofuel Production and Phosphorus Recovery through an Integrated Treatment of Agro-Industrial Waste" Sustainability 11, no. 1: 52. https://doi.org/10.3390/su11010052
APA StyleSiciliano, A., Limonti, C., Mehariya, S., Molino, A., & Calabrò, V. (2019). Biofuel Production and Phosphorus Recovery through an Integrated Treatment of Agro-Industrial Waste. Sustainability, 11(1), 52. https://doi.org/10.3390/su11010052









