The Social-Ecological Keystone Concept: A Quantifiable Metaphor for Understanding the Structure, Function, and Resilience of a Biocultural System
Abstract
1. Introduction
1.1. Social-Ecological Systems and the Application of Ecological Terminology
1.2. Quantifying Biocultural Elements within Social-Ecological Systems
- exploring the concept of functional groups within social-ecological systems,
- quantitatively classifying particular elements as either keystone components or redundant components of social-ecological systems,
- quantitatively relating loss of keystone components to loss of social-ecological system structure and function,
- quantitatively classifying loss of redundant components to diminished resilience in social-ecological systems,
- identifying alternative regime states within a single social-ecological system,
- quantifying regime shifts between social-ecological systems.
1.3. The Hawaiian Social-Ecological System
2. Theoretical Foundations
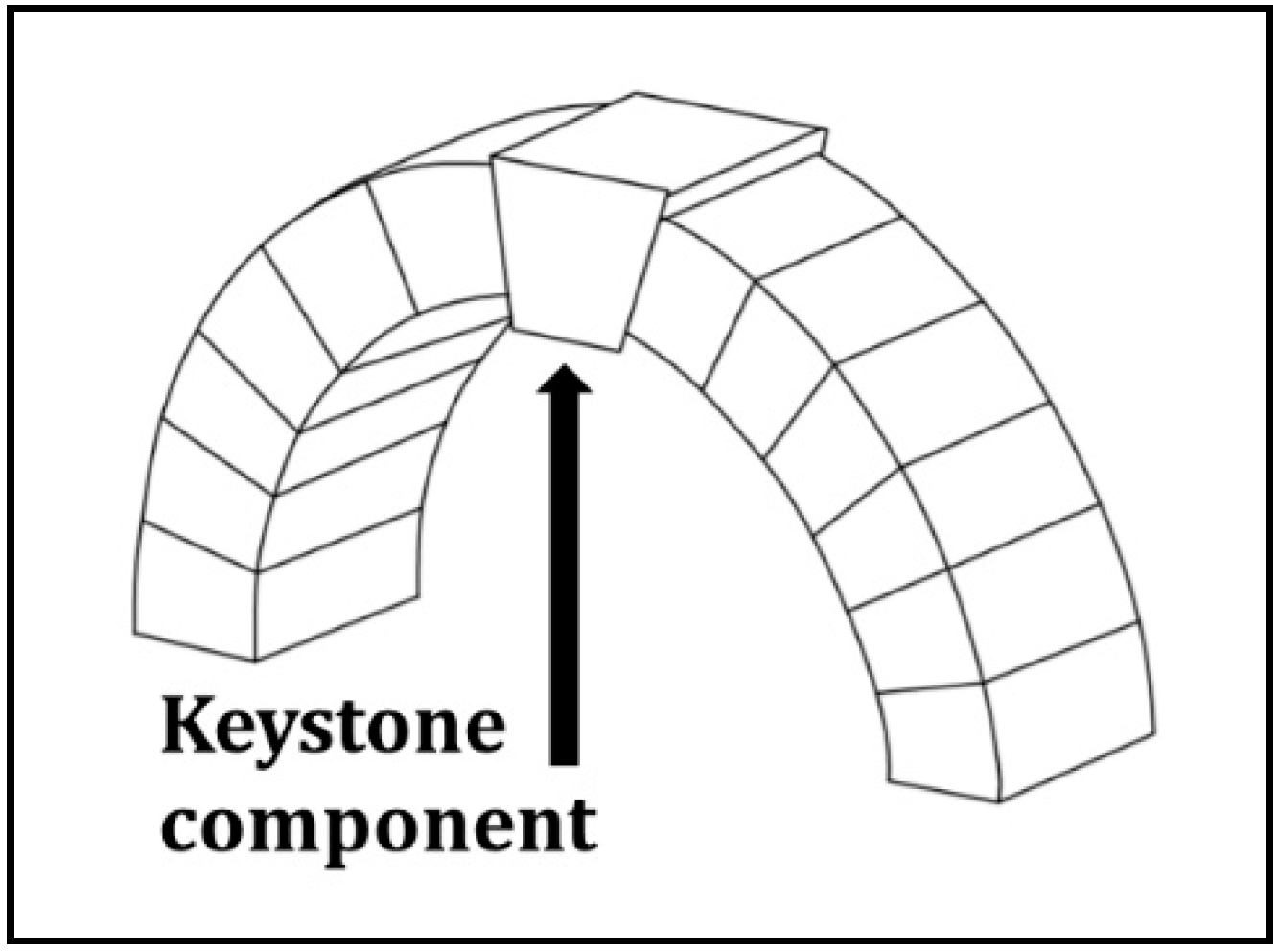
2.1. The Keystone Concept as Relates to System Structure and Function
2.2. The Social-Ecological Keystone Concept
2.3. The Influence of Crop Diversity and Cropping Systems on the Structure of the Hawaiian Social-Ecological System
2.4. Social-Ecological System Resilience and the Role of Redundant Components
2.5. Theoretical Assumptions
- Keystone function of a system can be viewed in terms of a functional group.
- Keystone components of functional groups are dominants within that functional group.
- Dominant components of a functional group (i.e., keystone components of a system) are not necessarily dominant components within the overall system.
- Shifting dominance within a keystone functional group replaces the keystone component of the system, thus influencing the structure of that system.
3. Testing the Keystone Theory in Social-Ecological Systems
The Hawaiian Social-Ecological System as a Model
4. Methodology
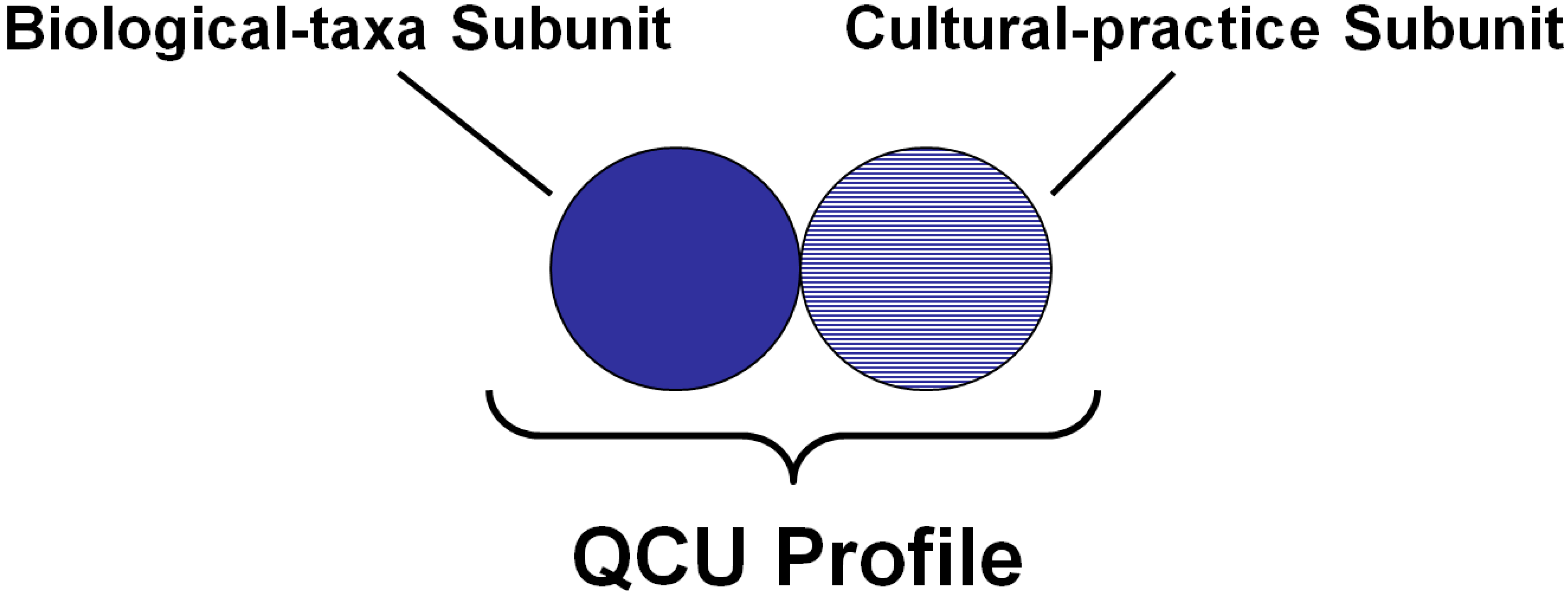
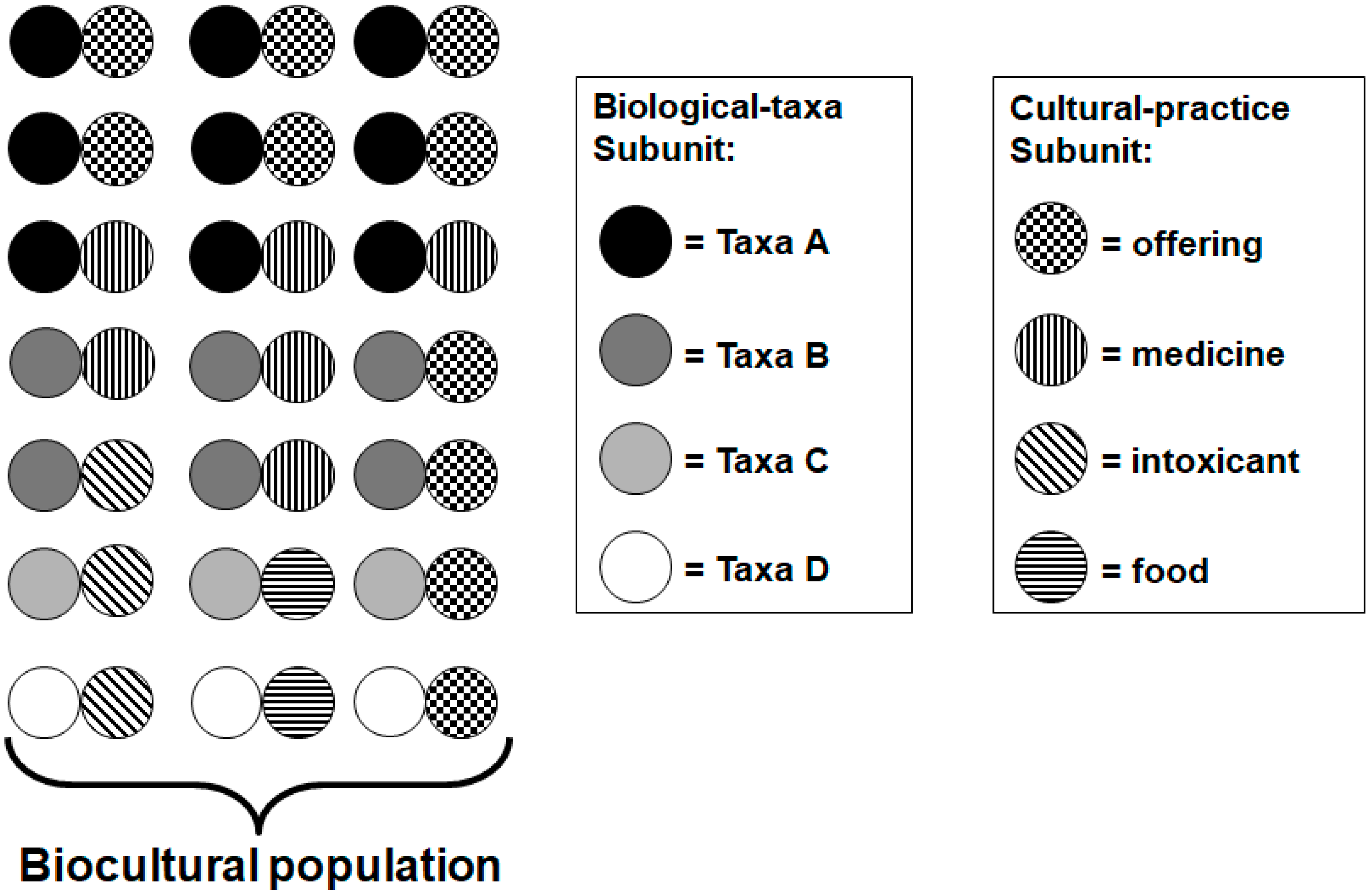
4.1. Quantification of Biocultural Diversity
4.2. Assessing Biocultural Functional Groups in Social-Ecological Systems
4.3. Quantitatively Classifying Keystone and Redundant Components
5. Results
6. Analysis
7. Discussion
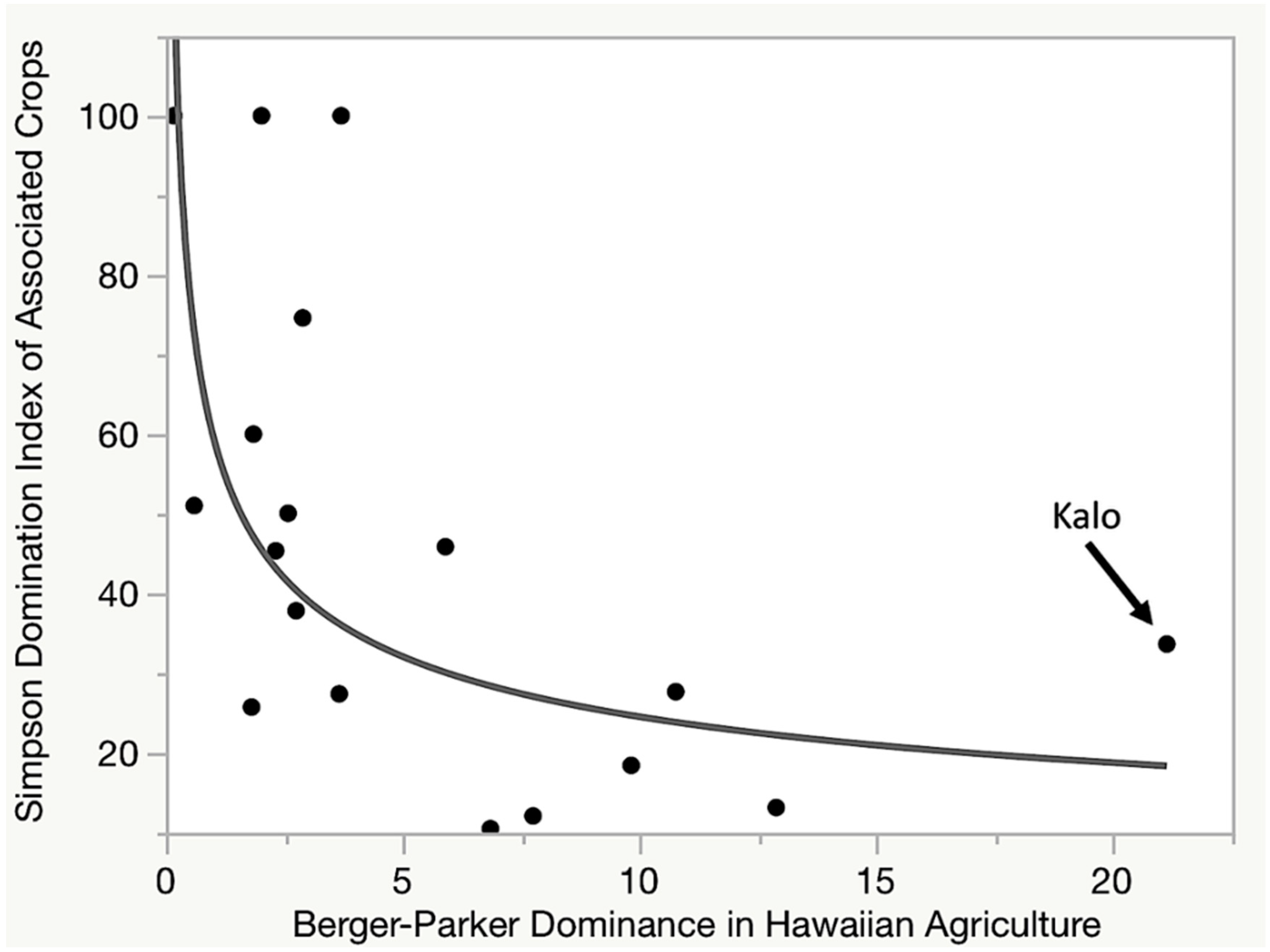
7.1. Is Kalo Cultivation a Keystone Component of the Hawaiian Social-Ecological System?
7.2. Biocultural Relationships between Kalo and Hawaiian Culture
7.3. Kalo Is a Dominant Component in Hawaiian Cropping Systems
7.4. Kalo Is a Dominant in a Key Biocultural Functional Group
7.5. Cropping Systems Associated with Kalo Influenced the Structure of the Hawaiian Social-Ecological System
- This conversion induced localized regime shifts in large areas of land (valley floors and alluvial plains) from forest biome to riparian ecotone. This, in essence, expanded and stabilized riparian habitat—a highly productive ecotone—from a relatively limited to a very broad area. Archaeological evidence suggests that such localized regime shifts have occurred [67], and likely extended the range of native water (i.e., riparian) fowl allowing for increases in their populations [68].
- This conversion theoretically increased the capacity of aquifers (i.e., the islands’ ability to retain water). The expansive flooded-field system slowed the flow rate of water on its journey towards the sea, and increased surface area of land covered by water. This cumulatively increased the potential for aquifer recharge. Increasing aquifer recharge potentially increases the level of the aquifer, which could result in more artesian springs at higher elevations than previously existed. The appearance of these springs would further increase the potential for lands—at higher elevation—to be converted to flooded terraces.
- This conversion likely induced localized regime shifts in estuaries and nearshore reefs from predator-dominated to herbivore-dominated. In theory, this may have been achieved through the development of aquaculture technologies. The emergence of such technologies was likely enabled by the flooded-field system, which mobilized nutrients and then transported them to coastal areas. The water passing through this flooded-field system was presumably enriched due to both a direct and indirect increase in organic matter, and anaerobic soils that mobilized otherwise fixed phosphorous into water systems. This aquaculture system had several classes of fish ponds, including those that walled in large areas of near-shore reef. These walls trapped the enriched water, thus containing algal blooms which allowed for the farming of herbivorous fish within them, while maintaining the health of the reef outside of the walls. This effectively expanded and stabilized estuary habitat—another highly productive ecotone—from a relatively limited to a very broad area. The success of this technology hinged on the management strategies which included methodological removal of top predators.
7.6. Substitution of a Social-Ecological Keystone Alters the Structure of Social-Ecological Systems
8. Conclusions
8.1. On Keystone and Redundant Components within Social-Ecological Systems
8.2. On Biocultural Diversity and Resilience in Social-Ecological Systems
8.3. On the Model of the Hawaiian Social-Ecological System
8.4. Biocultural Restoration of the Hawaiian Social-Ecological System
8.5. Future Research
- Assessing the percentage of total land area associated with each biocultural functional group to classify between keystone, dominant, and redundant components within social-ecological systems.
- Exploring the functional groups relating to animal husbandry, and assessing dominance in the context of functional groups.
- Expanding these methods to the entire biocultural resource spectrum of a social-ecological unit, which in the Hawaiian archipelago extends from the mountains to sea.
- Assessing the viability of utilizing social-ecological keystones to induce a regime shift back towards the state of abundance known in the Hawaiian language as, “‘āina momona” or biocultural resource abundance.
Supplementary Materials
Supplementary File 1Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Balée, W. The research program of historical ecology. Annu. Rev. Anthropol. 2006, 35, 75–98. [Google Scholar] [CrossRef]
- Berkes, F.; Colding, J.; Folke, C. (Eds.) Navigating Social-Ecological Systems: Building Resilience for Complexity and Change; Cambridge University Press: Cambridge, UK, 2003. [Google Scholar]
- Berkes, F.; Folke, C. (Eds.) Linking Social and Ecological Systems: Management Practices and Social Mechanisms for Building Resilience; Cambridge University Press: Cambridge, UK, 1998. [Google Scholar]
- Ostrom, E. A General Framework for Analyzing Sustainability of Social-Ecological Systems. Science 2009, 325, 419–422. [Google Scholar] [CrossRef] [PubMed]
- McGinnis, M.D.; Ostrom, E. Social-ecological system framework: Initial changes and continuing challenges. Ecol. Soc. 2014, 19, 30. [Google Scholar] [CrossRef]
- Pieter, B.; Schlüter, M.; Sendzimir, J. A Framework for Analyzing, Comparing, and Diagnosing Social-Ecological Systems. Ecol. Soc. 2015, 20, 18. [Google Scholar]
- Sterling, E.J.; Filardi, C.; Newell, J.; Albert, S.; Alvira, D.; Bergamini, N.; Betley, E.; Blair, M.; Boseto, D.; Burrows, K.; et al. Biocultural approaches to sustainability indicators: Bridging local and global scales to foster human adaptive capacity and ecological resilience. Nat. Ecol. Evol. 2017. [Google Scholar] [CrossRef]
- Walker, B.; Holling, C.S.; Carpenter, S.R.; Kinzig, A. Resilience, adaptability and transformability in social-ecological systems. Ecol. Soc. 2004, 9, 5. [Google Scholar] [CrossRef]
- Berkes, F. Restoring Unity: The Concept of Marine Social-Ecological Systems. In World Fisheries: A Social-Ecological Analysis; Ommer, R.E., Perry, R.I., Cochrane, K., Cury, P., Eds.; Wiley-Blackwell: Oxford, UK, 2011. [Google Scholar]
- Cristancho, S.; Vining, J. Culturally defined keystone species. Hum. Ecol. Rev. 2004, 11, 153–162. [Google Scholar]
- Garibaldi, A.; Turner, N. Cultural keystone species: Implications for ecological conservation and restoration. Ecol. Soc. 2004, 9, 1. [Google Scholar] [CrossRef]
- Brosi, B.J.; Balick, M.; Wolkow, R.; Lee, R.; Kostka, M.; Raynor, W.; Gallen, R.; Raynor, A.; Lee, L.D. Quantifying cultural erosion and its relationship to biodiversity conservation: Canoe-making knowledge in Pohnpei, Micronesia. Conserv. Biol. 2007, 21, 875–879. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, M.L.T.; Wieting, J.; Doherty, K.T. Vegetation analysis of urban ethnic markets shows supermarket generalists and Chinatown ethnic-specialist vendors. Ethnobot. Res. Appl. 2008, 6, 63–85. [Google Scholar] [CrossRef]
- Anderies, J.M.; Janssen, M.A. Sustaining the Commons; Center for the Study of Institutional Diversity, Arizona State University: Tempe, AZ, USA, 2013. [Google Scholar]
- Norgaard, R.B. Sociosystem and ecosystem coevolution in the Amazon. J. Environ. Econ. Manag. 1981, 8, 238–254. [Google Scholar] [CrossRef]
- Winter, K.; McClatchey, W. Quantifying Evolution of Cultural Interactions with Plants: Implications for Managing Diversity for Resilience in Social-Ecological System. In Ethnobotany: A Focus on Brazil. Functional Ecosystems and Communities; Albuquerque, U.P., Ed.; Global Science Books Ltd.: Isleworth, UK; Ikenobe, Japan, 2008; Volume 2, pp. 1–10. [Google Scholar]
- Winter, K.B.; McClatchey, W. The Quantum Co-evolution Unit: An Example of Awa (Piper methysticum G. Foster) in Hawaiian Culture. Econ. Bot. 2009, 63, 353–362. [Google Scholar] [CrossRef]
- Winter, K.B. Kalo [Hawaiian Kalo: Colocasia esculenta (L.) Schott] Varieties: An Assessment of Ancient and Modern Synonymy and Diversity. Ethnobot. Res. Appl. 2012, 10, 423–447. [Google Scholar]
- Kirch, P.V. Hawaii as a model for human ecodynamics. Am. Anthropol. 2007, 109, 8–26. [Google Scholar] [CrossRef]
- Wilmshurst, J.M.; Hunt, T.L.; Lipo, C.P.; Anderson, A.J. High-precision radiocarbon dating shows recent and rapid initial human colonization of East Polynesia. Proc. Natl. Acad. Sci. USA 2011, 108, 1815–1820. [Google Scholar] [CrossRef] [PubMed]
- Kamakau, S.M. Ruling Chiefs of Hawaii; Kamehameha Schools Press: Honolulu, HI, USA, 1961. [Google Scholar]
- Kame’eleihiwa, L. Native Land and Foreign Desires; Bishop Museum Press: Honolulu, HI, USA, 1992. [Google Scholar]
- Cachola-Abad, C.K. Evaluating the orthodox dual settlement model for the Hawaiian Islands: An analysis of artefact distribution and Hawaiian oral traditions. In The Evolution and Organization of Prehistoric Society in Polynesia; Graves, M.W., Green, R.C., Eds.; New Zealand Archaeological Association Monograph: Auckland, New Zealand, 1993; Volume 19, pp. 13–32. [Google Scholar]
- Cachola-Abad, C.K. An Analysis of Hawaiian Oral Traditions: Descriptions and Explanations of the Evolution of Hawaiian Socio-Political Complexity. Doctoral Dissertation, University of Hawaii at Mānoa, Honolulu, HI, USA, 2000. [Google Scholar]
- Winter, K.B.; Beamer, K.; Vaughan, M.B.; Friedlander, A.M.; Kido, M.; Whitehead, A.N.; Akutagawa, M.K.H.; Kurashima, N.; Lucas, M.P.; Nyberg, B. The Moku System: Managing biocultural resources for abundance within social-ecological regions in Hawaiʻi. Sustainability 2018, in press. [Google Scholar]
- Power, M.E.; Tilman, D.; Estes, J.A.; Menge, B.A.; Bond, W.J.; Mills, L.S.; Daily, G.; Castilla, J.C.; Lubchenco, J.; Paine, R.T. Challenges in the quest for keystones. BioScience 1996, 46, 609–620. [Google Scholar] [CrossRef]
- Ladefoged, T.N.; Kirch, P.V.; Gon, S.M.; Chadwick, O.A., III; Hartshorn, A.S.; Vitousek, P.M. Opportunities and constraints for intensive agriculture in the Hawaiian archipelago prior to European contact. J. Archaeol. Sci. 2009, 36, 2374–2383. [Google Scholar] [CrossRef]
- Kirch, P.V.; Zimmerer, K.S. Roots of Conflict; School for Advanced Research Press: Santa Fe, NM, USA, 2011. [Google Scholar]
- Winter, K.B.; Lucas, M. Spatial modeling of social-ecological management zones of the alii era on the island of Kauai with implications for large-scale biocultural conservation and forest restoration efforts in Hawaii. Pac. Sci. 2017, 71, 457–477. [Google Scholar] [CrossRef]
- Paine, R.T. The Pisaster-Tegula Interaction: Prey, Patches, Predator Food Preference, and Intertidal Community Structure. Ecology 1969, 50, 950–961. [Google Scholar] [CrossRef]
- Davic, R.D. Linking Keystone Species and Functional Groups: A New Operational Definition of the Keystone Species Concept. Conserv. Ecol. 2003, 7, r11. [Google Scholar] [CrossRef]
- Von Bertalanffy, L. General System Theory; George Braziller, Inc.: New York, NY, USA, 1968; Volume 41973, p. 40. [Google Scholar]
- Pimm, S.L.; Gilpin, M.E. Theoretical issues in conservation biology. In Perspectives in Ecological Theory; Roughgarden, J., May, R., Levin, S., Eds.; Princeton University Press: Princeton, NJ, USA, 1989; pp. 287–305. [Google Scholar]
- Christianou, M.; Ebenman, B. Keystone Species and Vulnerable Species in Ecological Communities: Strong or Weak Interactors? J. Theoret. Biol. 2005, 235, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Folke, C.; Carpenter, S.; Walker, B.; Scheffer, M.; Elmqvist, T.; Gunderson, L.; Holling, C.S. Regime shifts, resilience, and biodiversity in ecosystem management. Annu. Rev. Ecol. Evol. Syst. 2004, 35, 557–581. [Google Scholar] [CrossRef]
- Rhindos, D. Darwinism and its role in the explanation of domestication. In Foraging and Farming: The Evolution of Plant Exploitation; Harris, D.R., Hillman, G.C., Eds.; Unwin Hyman: London, UK, 1989; pp. 27–41. [Google Scholar]
- Lincoln, N.K.; Vitousek, P.M. Indigenous Polynesian agriculture in Hawaii. Oxford Resear. Encycl. Environ. Sci. 2017. [Google Scholar] [CrossRef]
- Kirch, P.V. The Wet and the Dry: Irrigation and Agricultural Intensification in Polynesia; University of Chicago Press: Chicago, IL, USA, 1994. [Google Scholar]
- Lincoln, N.K.; Rossen, J.; Vitousek, P.; Kahoonei, J.; Shapiro, D.; Kalawe, K.; Pai, M.; Marshall, K.; Meheula, K. Restoration of ‘Āina Malo‘o on Hawai‘i Island: Expanding Biocultural Relationships. Sustainability 2018. under review. [Google Scholar]
- Holling, C.S. Resilience and stability of ecological systems. Annu. Rev. Ecol. Syst. 1973, 4, 1–23. [Google Scholar] [CrossRef]
- Resilience Alliance. 2002. Available online: https://www.resilience.org/about-resilience/ (accessed on 30 June 2018).
- Holling, C.S. Engineering resilience versus ecological resilience. In Engineering within Ecological Constraints; Schulze, P.C., Ed.; National Academy Press: Washington, DC, USA, 1996. [Google Scholar]
- Elmqvist, T.; Folke, C.; Nystrom, M.; Peterson, G.; Bengtsson, J.; Walker, B.; Norberg, J. Response Diversity and Ecosystem Resilience. Front. Ecol. Environ. 2003, 1, 488–494. [Google Scholar] [CrossRef]
- Naeem, S. Species Redundancy and Ecosystem Reliability. Conserv. Biol. 1998, 12, 39–45. [Google Scholar] [CrossRef]
- Scheffer, M.; Carpenter, S.; Foley, J.A.; Folke, C.; Walker, B. Catastrophic Shifts in Ecosystems. Nature 2001, 413, 591–596. [Google Scholar] [CrossRef] [PubMed]
- Fosberg, F.R. (Ed.) Man’s Place in Island Ecosystems: A Symposium; Bishop Museum Press: Honolulu, HI, USA, 1962; 264p. [Google Scholar]
- Vitousek, P. Nutrient Cycling and Limitation: Hawaii as a Model; Princeton University Press: Princeton, NJ, USA, 2004. [Google Scholar]
- Stannard, D.E. Before the Horror: The Population of Hawaii on the Eve of Western Contact; University of Hawaii Press: Honolulu, HI, USA, 1994. [Google Scholar]
- Swanson, D.A. The Number of Native Hawaiians and Part Hawaiians in Hawaii, 1778 to 1900: Demographic Estimates by Age, with Discussion; University of California: Riverside, CA, USA, 2015. [Google Scholar]
- Hommon, R.J. The Ancient Hawaiian State: Origins of a Political Society; Oxford University Press: Oxford, UK, 2013. [Google Scholar]
- Max-Neef, M.; Elizalde, A.; Hopenhayn, M.; Herrera, F.; Zemelman, H.; Jataba, J.; Weinstein, L. Human Scale Development: A Hope for the Future. Dev. Dial. 1989, 1, 5–80. [Google Scholar]
- Max-Neef, M.; Elizalde, A.; Hopenhayn, M. Human Scale Development: Conception, Application and Further Reflections; The Apex Press: New York, NY, USA, 1991; 114p. [Google Scholar]
- Ii, J.P. Fragments of Hawaiian History; Bishop Museum Press: Honolulu, HI, USA, 1959. [Google Scholar]
- Handy, E.S.C.; Handy, E.G.; Pukui, M.K. Native Planters in Old Hawaii: Their Life, Lore, and Environment; Bishop Museum Press: Honolulu, HI, USA, 1972. [Google Scholar]
- Kamakau, S.M. Ka Hana a Ka Poe Kahiko; Translated from the Newspaper Ke Au Okoa; Bishop Museum Press: Honolulu, HI, USA, 1976. [Google Scholar]
- Kamakau, S.M. Ka Poe Kahiko; Translated from the Newspaper Ke Au Okoa; Bishop Museum Press: Honolulu, HI, USA, 1991. [Google Scholar]
- Desha, S.L. Kamehameha and His Warrior Kekuhaupio; Frazier, Frances N., Translator; Kamehameha Schools Press: Honolulu, HI, USA, 2000. [Google Scholar]
- Malo, D. Ka Moolelo Hawaii: Hawaiian Traditions; Chun, Malcolm, Translator; First People’s Publications: Honolulu, HI, USA, 2006; 274p. [Google Scholar]
- Kepelino. Kepelino’s Traditions of Hawaii; Beckwith, M.W., Ed.; Bishop Museum Press: Honolulu, HI, USA, 2007; 206p. [Google Scholar]
- Berger, W.H.; Parker, F.L. Diversity of planktonic Foraminifera in deep sea sedimanets. Science 1970, 168, 1345–1347. [Google Scholar] [CrossRef] [PubMed]
- McNaughton, S.J. Structure and Function in California Grasslands. Ecology 1968, 49, 962–972. [Google Scholar] [CrossRef]
- Simpson, E.H. Measurement of diversity. Nature 1949, 163, 688. [Google Scholar] [CrossRef]
- Abbott, I.A. Lāau Hawaii Traditional Hawaiian Uses of Plants; Bishop Museum Press: Honolulu, HI, USA, 1992. [Google Scholar]
- Kelly, M. Ahupua‘a Fishponds and Loi: A Film for Our Time; Nā Maka o ka Āina: Honolulu, HI, USA, 1992. [Google Scholar]
- Handy, E.S.C. The Hawaiian Planter: His Plants, Methods and Areas of Cultivation; Bishop Museum Press: Honolulu, HI, USA, 1940; 227p. [Google Scholar]
- Molnar, Z.; Berkes, F. Role of traditional ecological knowledge in linking cultural and natural capital in cultural landscapes. In Reconnecting Natural and Cultural Capital: Contributions from Science and Policy; Paracchini, M.L., Zingari, P.C., Blasi, C., Eds.; European Union: Luxembourg, 2018; pp. 183–193. [Google Scholar]
- Burney, D.A.; Kikuchi, W. A millennium of human activity at Makauwahi Cave, Mahaulepu, Kauai. Hum. Ecol. 2006, 34, 219–247. [Google Scholar] [CrossRef]
- Burney, D.A.; James, H.F.; Burney, L.P.; Olson, S.L.; Kikuchi, W.; Wagner, W.L.; Burney, M.; McCloskey, D.; Kikuchi, D.; Grady, F.V.; et al. Fossil evidence from a diverse biota from Kauai and its transformation since human arrival. Ecol. Monogr. 2001, 71, 615–641. [Google Scholar]
- Kurashima, N.; Jeremiah, J.; Ticktin, T. I ka wa ma mua: The value of a historical ecology approach to ecological restoration in Hawaii. Pac. Sci. 2017, 71, 437–456. [Google Scholar] [CrossRef]
- Scheffer, M.; Carpenter, S.R.; Lenton, T.M.; Bascompte, J.; Brock, W.; Dakos, V.; Van de Koppel, J.; Van de Leemput, I.A.; Levin, S.A.; Van Nes, E.H.; et al. Anticipating critical transitions. Science 2012, 388, 344–348. [Google Scholar] [CrossRef] [PubMed]

| Cropping System | Dominance of Cropping Systems | Domination by Species Assemblage | Associated Crop Species |
|---|---|---|---|
| Rain-fed | 0.441 | 0.263 | 11 |
| Agroforestry | 0.288 | 0.110 | 17 |
| Flooded-field | 0.272 | 0.731 | 7 |
| Latin Name | Hawaiian Name | Dominance in Rain-Fed Systems | Dominance in Agroforestry | Dominance in Flooded Systems |
|---|---|---|---|---|
| Aleurites molaccanus | Kukui | - | 0.186 | - |
| Artocarpus altilis | ‘Ulu | - | 0.089 | - |
| Broussonetia papyrifera | Wauke | 0.013 | 0.066 | 0.016 |
| Cocos nucifera | Niu | - | 0.094 | - |
| Colocasia esculenta | Kalo | 0.352 | 0.178 | 0.852 |
| Cordia subcordata | Kou | - | 0.010 | - |
| Cordyline fruticosa | Kī | 0.005 | 0.028 | 0.014 |
| Curcuma domestica | ‘Ōlena | 0.005 | 0.015 | - |
| Dioscorea alata | ‘Uhi | 0.073 | 0.036 | - |
| Dioscorea bulbifera | Hoi | - | 0.028 | - |
| Dioscorea pentaphylla | Pi‘a | - | 0.008 | - |
| Ipomoea batatas | ‘Uala | 0.349 | - | - |
| Lageneria siceraria L. vulgaris | Ipu | 0.086 | - | - |
| Musa ssp. | Mai‘a | 0.023 | 0.099 | 0.059 |
| Pandanus tectorius | Hala | - | 0.084 | - |
| Piper methysticum | ‘Awa | 0.040 | 0.008 | 0.016 |
| Saccharum offinarum | Kō | 0.043 | 0.003 | 0.024 |
| Schizostachyum glaucifolium | ‘Ohe | - | 0.041 | - |
| Tacca leontopetaloides | Pia | 0.010 | 0.028 | 0.019 |
| Count | 11 | 17 | 7 |
| Biocultural Functional Group | Dominance of Hawaiian Agriculture (DIS) | Domination of Group by Crop (DIBP) | Number of Associated Crops | Dominant Crop |
|---|---|---|---|---|
| Complex carbohydrates for food | 0.211 | 0.336 | 9 | Kalo |
| Affiliated with deities | 0.129 | 0.132 | 12 | Kalo |
| Ceremonial plants for religious practice | 0.108 | 0.277 | 8 | ‘Awa |
| Wood (timber, fuel, vessel, music, misc.) | 0.098 | 0.184 | 11 | Kukui/Hau |
| Famine food for a resilient food system | 0.078 | 0.121 | 14 | Kalo |
| Medicinal applications | 0.069 | 0.105 | 17 | ‘Awa |
| Leaves for weaving or thatch material | 0.059 | 0.459 | 4 | Hala |
| Fibers for clothing | 0.037 | 1.000 | 4 | Wauke |
| Simple carbohydrate for food | 0.037 | 0.274 | 6 | Niu |
| Mulch for agriculture | 0.028 | 0.378 | 4 | Kukui |
| Relates to the family system | 0.029 | 0.746 | 2 | Kalo |
| Oil for culinary uses and healing | 0.026 | 0.501 | 2 | Kukui |
| Drink for refreshment and recreation | 0.023 | 0.453 | 3 | Niu |
| Genesis story with the culture | 0.020 | 1.000 | 1 | Kalo |
| Leafy greens for food | 0.019 | 0.600 | 3 | Kalo |
| Fibers for cordage | 0.018 | 0.257 | 5 | Kalo |
| Dye for visual attraction | 0.006 | 0.510 | 2 | ‘Ōlena |
| Glue/resin source | 0.002 | 1.000 | 2 | ‘Ulu |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Winter, K.B.; Lincoln, N.K.; Berkes, F. The Social-Ecological Keystone Concept: A Quantifiable Metaphor for Understanding the Structure, Function, and Resilience of a Biocultural System. Sustainability 2018, 10, 3294. https://doi.org/10.3390/su10093294
Winter KB, Lincoln NK, Berkes F. The Social-Ecological Keystone Concept: A Quantifiable Metaphor for Understanding the Structure, Function, and Resilience of a Biocultural System. Sustainability. 2018; 10(9):3294. https://doi.org/10.3390/su10093294
Chicago/Turabian StyleWinter, Kawika B., Noa Kekuewa Lincoln, and Fikret Berkes. 2018. "The Social-Ecological Keystone Concept: A Quantifiable Metaphor for Understanding the Structure, Function, and Resilience of a Biocultural System" Sustainability 10, no. 9: 3294. https://doi.org/10.3390/su10093294
APA StyleWinter, K. B., Lincoln, N. K., & Berkes, F. (2018). The Social-Ecological Keystone Concept: A Quantifiable Metaphor for Understanding the Structure, Function, and Resilience of a Biocultural System. Sustainability, 10(9), 3294. https://doi.org/10.3390/su10093294







