Potential Role of Global Longitudinal Strain in Cardiac and Oncological Patients Undergoing Cardio-Oncology Rehabilitation (CORE)
Abstract
1. Introduction
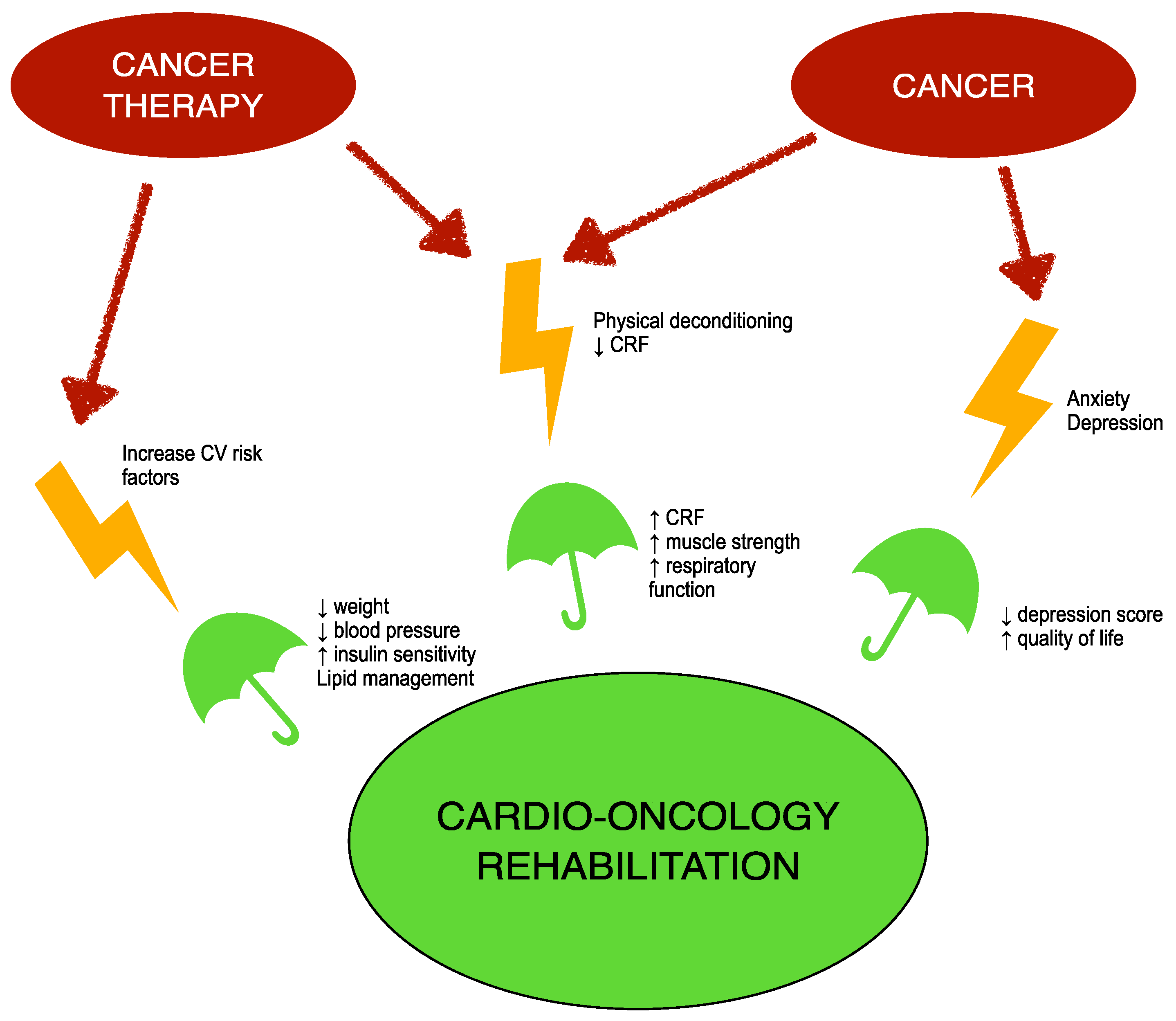
2. Cardio-Oncology Rehabilitation (CORE)
3. Global Longitudinal Strain (GLS), a New Technology for Detection of Systolic Disfunction
4. Effects of Exercise Training on Global Longitudinal Strain (GLS) in Patients with Cancer and Cardiac Disease
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Shapiro, C.L. Cancer Survivorship. N. Engl. J. Med. 2018, 379, 2438–2450. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer Statistics, 2021. CA Cancer J. Clin. 2021, 71, 7–33. [Google Scholar] [CrossRef]
- Patnaik, J.L.; Byers, T.; DiGuiseppi, C.; Dabelea, D.; Denberg, T.D. Cardiovascular disease competes with breast cancer as the leading cause of death for older females diagnosed with breast cancer: A retrospective cohort study. Breast Cancer Res. 2011, 13, R64. [Google Scholar] [CrossRef]
- Armenian, S.H.; Xu, L.; Ky, B.; Sun, C.; Farol, L.T.; Pal, S.K.; Douglas, P.S.; Bhatia, S.; Chao, C. Cardiovascular Disease Among Survivors of Adult-Onset Cancer: A Community-Based Retrospective Cohort Study. J. Clin. Oncol. 2016, 34, 1122–1130. [Google Scholar] [CrossRef]
- Lipshultz, S.E.; Adams, M.J.; Colan, S.D.; Constine, L.S.; Herman, E.H.; Hsu, D.T.; Hudson, M.M.; Kremer, L.C.; Landy, D.C.; Miller, T.L.; et al. Long-term cardiovascular toxicity in children, adolescents, and young adults who receive cancer therapy: Pathophysiology, course, monitoring, management, prevention, and research directions: A scientific statement from the American Heart Association. Circulation 2013, 128, 1927–1995. [Google Scholar] [CrossRef]
- Ezaz, G.; Long, J.B.; Gross, C.P.; Chen, J. Risk prediction model for heart failure and cardiomyopathy after adjuvant trastuzumab therapy for breast cancer. J. Am. Heart Assoc. 2014, 3, e000472. [Google Scholar] [CrossRef]
- Bhagat, A.; Kleinerman, E.S. Anthracycline-Induced Cardiotoxicity: Causes, Mechanisms, and Prevention. Adv. Exp. Med. Biol. 2020, 1257, 181–192. [Google Scholar] [CrossRef]
- Gernaat, S.A.; Ho, P.J.; Rijnberg, N.; Emaus, M.J.; Baak, L.M.; Hartman, M.; Grobbee, D.E.; Verkooijen, H.M. Risk of death from cardiovascular disease following breast cancer: A systematic review. Breast Cancer Res. Treat. 2017, 164, 537–555. [Google Scholar] [CrossRef]
- Guha, A.; Dey, A.K.; Al-Kindi, S.; Miller, P.E.; Ghosh, A.K.; Banerjee, A.; Lopez-Mattei, J.; Desai, N.R.; Patel, B.; Oliveira, G.H.; et al. Socio-Economic Burden of Myocardial Infarction Among Cancer Patients. Am. J. Cardiol. 2021, 141, 16–22. [Google Scholar] [CrossRef]
- Lancellotti, P.; Suter, T.M.; Lopez-Fernandez, T.; Galderisi, M.; Lyon, A.R.; Van der Meer, P.; Cohen Solal, A.; Zamorano, J.L.; Jerusalem, G.; Moonen, M.; et al. Cardio-Oncology Services: Rationale, organization, and implementation. Eur. Heart J. 2019, 40, 1756–1763. [Google Scholar] [CrossRef]
- Lyon, A.R.; López-Fernández, T.; Couch, L.S.; Asteggiano, R.; Aznar, M.C.; Bergler-Klein, J.; Boriani, G.; Cardinale, D.; Cordoba, R.; Cosyns, B.; et al. 2022 ESC Guidelines on cardio-oncology developed in collaboration with the European Hematology Association (EHA), the European Society for Therapeutic Radiology and Oncology (ESTRO) and the International Cardio-Oncology Society (IC-OS). Eur Heart J. 2022, 43, 4229–4361. [Google Scholar] [CrossRef]
- Herrmann, J.; Lenihan, D.; Armenian, S.; Barac, A.; Blaes, A.; Cardinale, D.; Carver, J.; Dent, S.; Ky, B.; Lyon, A.R.; et al. Defining cardiovascular toxicities of cancer therapies: An International Cardio-Oncology Society (IC-OS) consensus statement. Eur. Heart J. 2022, 43, 280–299. [Google Scholar] [CrossRef]
- Prescott, E.; Hippe, M.; Schnohr, P.; Hein, H.O.; Vestbo, J. Smoking and risk of myocardial infarction in women and men: Longitudinal population study. BMJ 1998, 316, 1043–1047. [Google Scholar] [CrossRef]
- Vineis, P.; Alavanja, M.; Buffler, P.; Fontham, E.; Franceschi, S.; Gao, Y.T.; Gupta, P.C.; Hackshaw, A.; Matos, E.; Samet, J.; et al. Tobacco and cancer: Recent epidemiological evidence. J. Natl. Cancer Inst. 2004, 96, 99–106. [Google Scholar] [CrossRef]
- Tabarés Seisdedos, R. Health Effects of Overweight and Obesity in 195 Countries over 25 Years. N. Engl. J. Med. 2017, 377, 13–27. [Google Scholar] [CrossRef]
- Chao, C.; Xu, L.; Bhatia, S.; Cooper, R.; Brar, S.; Wong, F.L.; Armenian, S.H. Cardiovascular Disease Risk Profiles in Survivors of Adolescent and Young Adult (AYA) Cancer: The Kaiser Permanente AYA Cancer Survivors Study. J. Clin. Oncol. 2016, 34, 1626–1633. [Google Scholar] [CrossRef]
- Gibson, T.M.; Li, Z.; Green, D.M.; Armstrong, G.T.; Mulrooney, D.A.; Srivastava, D.; Bhakta, N.; Ness, K.K.; Hudson, M.M.; Robison, L.L. Blood Pressure Status in Adult Survivors of Childhood Cancer: A Report from the St. Jude Lifetime Cohort Study. Cancer Epidemiol. Biomark. Prev. 2017, 26, 1705–1713. [Google Scholar] [CrossRef]
- Inoue, M.; Iwasaki, M.; Otani, T.; Sasazuki, S.; Noda, M.; Tsugane, S. Diabetes mellitus and the risk of cancer: Results from a large-scale population-based cohort study in Japan. Arch. Intern. Med. 2006, 166, 1871–1877. [Google Scholar] [CrossRef]
- Dehal, A.N.; Newton, C.C.; Jacobs, E.J.; Patel, A.V.; Gapstur, S.M.; Campbell, P.T. Impact of diabetes mellitus and insulin use on survival after colorectal cancer diagnosis: The Cancer Prevention Study-II Nutrition Cohort. J. Clin. Oncol. 2012, 30, 53–59. [Google Scholar] [CrossRef]
- Alexopoulos, C.G.; Blatsios, B.; Avgerinos, A. Serum lipids and lipoprotein disorders in cancer patients. Cancer 1987, 60, 3065–3070. [Google Scholar] [CrossRef]
- Armenian, S.H.; Sun, C.L.; Vase, T.; Ness, K.K.; Blum, E.; Francisco, L.; Venkataraman, K.; Samoa, R.; Wong, F.L.; Forman, S.J.; et al. Cardiovascular risk factors in hematopoietic cell transplantation survivors: Role in development of subsequent cardiovascular disease. Blood 2012, 120, 4505–4512. [Google Scholar] [CrossRef] [PubMed]
- Plana, J.C.; Galderisi, M.; Barac, A.; Ewer, M.S.; Ky, B.; Scherrer-Crosbie, M.; Ganame, J.; Sebag, I.A.; Agler, D.A.; Badano, L.P.; et al. Expert consensus for multimodality imaging evaluation of adult patients during and after cancer therapy: A report from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J. Am. Soc. Echocardiogr. 2014, 27, 911–939. [Google Scholar] [CrossRef] [PubMed]
- Awadalla, M.; Mahmood, S.S.; Groarke, J.D.; Hassan, M.Z.; Nohria, A.; Rokicki, A.; Murphy, S.P.; Mercaldo, N.D.; Zhang, L.; Zlotoff, D.A.; et al. Global Longitudinal Strain and Cardiac Events in Patients With Immune Checkpoint Inhibitor-Related Myocarditis. J. Am. Coll. Cardiol. 2020, 75, 467–478. [Google Scholar] [CrossRef]
- Venturini, E.; Gilchrist, S.; Corsi, E.; Cuomo, G.; D’Ambrosio, G.; Pacileo, M.; D’Andrea, A.; Canale, M.L.; Iannuzzo, G.; Sarullo, F.M.; et al. The core components of cardio-oncology rehabilitation. Panminerva Med. 2021, 63, 170–183. [Google Scholar] [CrossRef]
- Clarke, P.; Radford, K.; Coffey, M.; Stewart, M. Speech and swallow rehabilitation in head and neck cancer: United Kingdom National Multidisciplinary Guidelines. J. Laryngol. Otol. 2016, 130, S176–S180. [Google Scholar] [CrossRef]
- Reese, C.; Weis, J.; Schmucker, D.; Mittag, O. Development of practice guidelines for psychological interventions in the rehabilitation of patients with oncological disease (breast, prostate, or colorectal cancer): Methods and results. Psychooncology 2017, 26, 1513–1518. [Google Scholar] [CrossRef] [PubMed]
- Rock, C.L.; Doyle, C.; Demark-Wahnefried, W.; Meyerhardt, J.; Courneya, K.S.; Schwartz, A.L.; Bandera, E.V.; Hamilton, K.K.; Grant, B.; McCullough, M.; et al. Nutrition and physical activity guidelines for cancer survivors. CA Cancer J. Clin. 2012, 62, 243–274. [Google Scholar] [CrossRef]
- Anderson, L.; Thompson, D.R.; Oldridge, N.; Zwisler, A.D.; Rees, K.; Martin, N.; Taylor, R.S. Exercise-based cardiac rehabilitation for coronary heart disease. Cochrane Database Syst. Rev. 2016, 2016, CD001800. [Google Scholar] [CrossRef] [PubMed]
- Cuomo, G.; Di Lorenzo, A.; Tramontano, A.; Iannone, F.P.; D’Angelo, A.; Pezzella, R.; Testa, C.; Parlato, A.; Merone, P.; Pacileo, M.; et al. Exercise Training in Patients with Heart Failure: From Pathophysiology to Exercise Prescription. Rev. Cardiovasc. Med. 2022, 23, 144. [Google Scholar] [CrossRef]
- Braith, R.W.; Edwards, D.G. Neurohormonal abnormalities in heart failure: Impact of exercise training. Congest. Heart Fail. 2003, 9, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Giallauria, F.; Del Forno, D.; Pilerci, F.; De Lorenzo, A.; Manakos, A.; Lucci, R.; Vigorito, C. Improvement of heart rate recovery after exercise training in older people. J. Am. Geriatr. Soc. 2005, 53, 2037–2038. [Google Scholar] [CrossRef] [PubMed]
- Giallauria, F.; Lorenzo, A.D.; Pilerci, F.; Manakos, A.; Lucci, R.; Psaroudaki, M.; D’Agostino, M.; Forno, D.D.; Vigorito, C. Long-term effects of cardiac rehabilitation on end-exercise heart rate recovery after myocardial infarction. Eur. J. Cardiovasc. Prev. Rehabil. 2006, 13, 544–550. [Google Scholar] [CrossRef] [PubMed]
- Giallauria, F.; Lucci, R.; Pietrosante, M.; Gargiulo, G.; De Lorenzo, A.; D’Agostino, M.; Gerundo, G.; Abete, P.; Rengo, F.; Vigorito, C. Exercise-based cardiac rehabilitation improves heart rate recovery in elderly patients after acute myocardial infarction. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2006, 61, 713–717. [Google Scholar] [CrossRef] [PubMed]
- Giallauria, F.; Cirillo, P.; Lucci, R.; Pacileo, M.; De Lorenzo, A.; D’Agostino, M.; Moschella, S.; Psaroudaki, M.; Del Forno, D.; Orio, F.; et al. Left ventricular remodelling in patients with moderate systolic dysfunction after myocardial infarction: Favourable effects of exercise training and predictive role of N-terminal pro-brain natriuretic peptide. Eur. J. Cardiovasc. Prev. Rehabil. 2008, 15, 113–118. [Google Scholar] [CrossRef]
- Giallauria, F.; Galizia, G.; Lucci, R.; D’Agostino, M.; Vitelli, A.; Maresca, L.; Orio, F.; Vigorito, C. Favourable effects of exercise-based Cardiac Rehabilitation after acute myocardial infarction on left atrial remodeling. Int. J. Cardiol. 2009, 136, 300–306. [Google Scholar] [CrossRef]
- Cirillo, P.; Giallauria, F.; Pacileo, M.; Petrillo, G.; D’agostino, M.; Vigorito, C.; Chiariello, M. Increased high mobility group box-1 protein levels are associated with impaired cardiopulmonary and echocardiographic findings after acute myocardial infarction. J. Card. Fail. 2009, 15, 362–367. [Google Scholar] [CrossRef]
- Arbab-Zadeh, A.; Perhonen, M.; Howden, E.; Peshock, R.M.; Zhang, R.; Adams-Huet, B.; Haykowsky, M.J.; Levine, B.D. Cardiac remodeling in response to 1 year of intensive endurance training. Circulation 2014, 130, 2152–2161. [Google Scholar] [CrossRef]
- Giallauria, F.; Acampa, W.; Ricci, F.; Vitelli, A.; Maresca, L.; Mancini, M.; Grieco, A.; Gallicchio, R.; Xhoxhi, E.; Spinelli, L.; et al. Effects of exercise training started within 2 weeks after acute myocardial infarction on myocardial perfusion and left ventricular function: A gated SPECT imaging study. Eur. J. Prev. Cardiol. 2012, 19, 1410–1419. [Google Scholar] [CrossRef]
- Giallauria, F.; Acampa, W.; Ricci, F.; Vitelli, A.; Torella, G.; Lucci, R.; Del Prete, G.; Zampella, E.; Assante, R.; Rengo, G.; et al. Exercise training early after acute myocardial infarction reduces stress-induced hypoperfusion and improves left ventricular function. Eur. J. Nucl. Med. Mol. Imaging 2013, 40, 315–324. [Google Scholar] [CrossRef]
- Zampella, E.; Mannarino, T.; Gaudieri, V.; D’Antonio, A.; Giallauria, F.; Assante, R.; Cantoni, V.; Green, R.; Mainolfi, C.G.; Nappi, C.; et al. Effect of changes in perfusion defect size during serial stress myocardial perfusion imaging on cardiovascular outcomes in patients treated with primary percutaneous coronary intervention after myocardial infarction. J. Nucl. Cardiol. 2022, 29, 2624–2632. [Google Scholar] [CrossRef] [PubMed]
- Takura, T.; Ebata-Kogure, N.; Goto, Y.; Kohzuki, M.; Nagayama, M.; Oikawa, K.; Koyama, T.; Itoh, H. Cost-Effectiveness of Cardiac Rehabilitation in Patients with Coronary Artery Disease: A Meta-Analysis. Cardiol. Res. Pract. 2019, 2019, 1840894. [Google Scholar] [CrossRef] [PubMed]
- Bozkurt, B.; Fonarow, G.C.; Goldberg, L.R.; Guglin, M.; Josephson, R.A.; Forman, D.E.; Lin, G.; Lindenfeld, J.; O’Connor, C.; Panjrath, G.; et al. Cardiac Rehabilitation for Patients With Heart Failure: JACC Expert Panel. J. Am. Coll. Cardiol. 2021, 77, 1454–1469. [Google Scholar] [CrossRef] [PubMed]
- Gilchrist, S.C.; Barac, A.; Ades, P.A.; Alfano, C.M.; Franklin, B.A.; Jones, L.W.; La Gerche, A.; Ligibel, J.A.; Lopez, G.; Madan, K.; et al. Cardio-Oncology Rehabilitation to Manage Cardiovascular Outcomes in Cancer Patients and Survivors: A Scientific Statement From the American Heart Association. Circulation 2019, 139, e997–e1012. [Google Scholar] [CrossRef] [PubMed]
- Tonorezos, E.S.; Snell, P.G.; Moskowitz, C.S.; Eshelman-Kent, D.A.; Liu, J.E.; Chou, J.F.; Smith, S.M.; Dunn, A.L.; Church, T.S.; Oeffinger, K.C. Reduced cardiorespiratory fitness in adult survivors of childhood acute lymphoblastic leukemia. Pediatr. Blood Cancer 2013, 60, 1358–1364. [Google Scholar] [CrossRef]
- Peel, A.B.; Barlow, C.E.; Leonard, D.; DeFina, L.F.; Jones, L.W.; Lakoski, S.G. Cardiorespiratory fitness in survivors of cervical, endometrial, and ovarian cancers: The Cooper Center Longitudinal Study. Gynecol. Oncol. 2015, 138, 394–397. [Google Scholar] [CrossRef]
- Gong, J.; Payne, D.; Caron, J.; Bay, C.P.; McGregor, B.A.; Hainer, J.; Partridge, A.H.; Neilan, T.G.; Di Carli, M.; Nohria, A.; et al. Reduced Cardiorespiratory Fitness and Increased Cardiovascular Mortality After Prolonged Androgen Deprivation Therapy for Prostate Cancer. JACC CardioOncol. 2020, 2, 553–563. [Google Scholar] [CrossRef]
- Thomas, G.K.; Trankle, C.R.; Carbone, S.; Billingsley, H.; Van Tassell, B.W.; Evans, R.K.; Garten, R.; Weiss, E.; Abbate, A.; Canada, J.M. Diastolic Dysfunction Contributes to Impaired Cardiorespiratory Fitness in Patients with Lung Cancer and Reduced Lung Function Following Chest Radiation. Lung 2021, 199, 403–407. [Google Scholar] [CrossRef]
- Jones, L.W.; Watson, D.; Herndon, J.E.; Eves, N.D.; Haithcock, B.E.; Loewen, G.; Kohman, L. Peak oxygen consumption and long-term all-cause mortality in nonsmall cell lung cancer. Cancer 2010, 116, 4825–4832. [Google Scholar] [CrossRef]
- Scott, J.M.; Nilsen, T.S.; Gupta, D.; Jones, L.W. Exercise Therapy and Cardiovascular Toxicity in Cancer. Circulation 2018, 137, 1176–1191. [Google Scholar] [CrossRef]
- Lee, K.; Tripathy, D.; Demark-Wahnefried, W.; Courneya, K.S.; Sami, N.; Bernstein, L.; Spicer, D.; Buchanan, T.A.; Mortimer, J.E.; Dieli-Conwright, C.M. Effect of Aerobic and Resistance Exercise Intervention on Cardiovascular Disease Risk in Women With Early-Stage Breast Cancer: A Randomized Clinical Trial. JAMA Oncol. 2019, 5, 710–714. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.M.; Zabor, E.C.; Schwitzer, E.; Koelwyn, G.J.; Adams, S.C.; Nilsen, T.S.; Moskowitz, C.S.; Matsoukas, K.; Iyengar, N.M.; Dang, C.T.; et al. Efficacy of Exercise Therapy on Cardiorespiratory Fitness in Patients With Cancer: A Systematic Review and Meta-Analysis. J. Clin. Oncol. 2018, 36, 2297–2305. [Google Scholar] [CrossRef] [PubMed]
- Wallen, M.P.; Hennessy, D.; Brown, S.; Evans, L.; Rawstorn, J.C.; Wong Shee, A.; Hall, A. High-intensity interval training improves cardiorespiratory fitness in cancer patients and survivors: A meta-analysis. Eur. J. Cancer Care 2020, 29, e13267. [Google Scholar] [CrossRef] [PubMed]
- Courneya, K.S.; Sellar, C.M.; Stevinson, C.; McNeely, M.L.; Peddle, C.J.; Friedenreich, C.M.; Tankel, K.; Basi, S.; Chua, N.; Mazurek, A.; et al. Randomized controlled trial of the effects of aerobic exercise on physical functioning and quality of life in lymphoma patients. J. Clin. Oncol. 2009, 27, 4605–4612. [Google Scholar] [CrossRef] [PubMed]
- Jones, L.W.; Courneya, K.S.; Mackey, J.R.; Muss, H.B.; Pituskin, E.N.; Scott, J.M.; Hornsby, W.E.; Coan, A.D.; Herndon, J.E.; Douglas, P.S.; et al. Cardiopulmonary function and age-related decline across the breast cancer survivorship continuum. J. Clin. Oncol. 2012, 30, 2530–2537. [Google Scholar] [CrossRef]
- Lakoski, S.G.; Willis, B.L.; Barlow, C.E.; Leonard, D.; Gao, A.; Radford, N.B.; Farrell, S.W.; Douglas, P.S.; Berry, J.D.; DeFina, L.F.; et al. Midlife Cardiorespiratory Fitness, Incident Cancer, and Survival After Cancer in Men: The Cooper Center Longitudinal Study. JAMA Oncol. 2015, 1, 231–237. [Google Scholar] [CrossRef]
- Giallauria, F.; Gentile, M.; Chiodini, P.; Berrino, F.; Mattiello, A.; Maresca, L.; Vitelli, A.; Mancini, M.; Grieco, A.; Russo, A.; et al. Exercise training reduces high mobility group box-1 protein levels in women with breast cancer: Findings from the DIANA-5 study. Monaldi Arch. Chest Dis. 2014, 82, 61–67. [Google Scholar] [CrossRef]
- Giallauria, F.; Vitelli, A.; Maresca, L.; Santucci De Magistris, M.; Chiodini, P.; Mattiello, A.; Gentile, M.; Mancini, M.; Grieco, A.; Russo, A.; et al. Exercise training improves cardiopulmonary and endothelial function in women with breast cancer: Findings from the Diana-5 dietary intervention study. Intern. Emerg. Med. 2016, 11, 183–189. [Google Scholar] [CrossRef]
- Giallauria, F.; Maresca, L.; Vitelli, A.; Santucci de Magistris, M.; Chiodini, P.; Mattiello, A.; Gentile, M.; Mancini, M.; Grieco, A.; Russo, A.; et al. Exercise training improves heart rate recovery in women with breast cancer. Springerplus 2015, 4, 388. [Google Scholar] [CrossRef]
- Gordon, L.G.; Scuffham, P.; Battistutta, D.; Graves, N.; Tweeddale, M.; Newman, B. A cost-effectiveness analysis of two rehabilitation support services for women with breast cancer. Breast Cancer Res. Treat. 2005, 94, 123–133. [Google Scholar] [CrossRef]
- Mitchell, A.J.; Chan, M.; Bhatti, H.; Halton, M.; Grassi, L.; Johansen, C.; Meader, N. Prevalence of depression, anxiety, and adjustment disorder in oncological, haematological, and palliative-care settings: A meta-analysis of 94 interview-based studies. Lancet Oncol. 2011, 12, 160–174. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, A.J.; Ferguson, D.W.; Gill, J.; Paul, J.; Symonds, P. Depression and anxiety in long-term cancer survivors compared with spouses and healthy controls: A systematic review and meta-analysis. Lancet Oncol. 2013, 14, 721–732. [Google Scholar] [CrossRef] [PubMed]
- Bodurka-Bevers, D.; Basen-Engquist, K.; Carmack, C.L.; Fitzgerald, M.A.; Wolf, J.K.; De Moor, C.; Gershenson, D.M. Depression, anxiety, and quality of life in patients with epithelial ovarian cancer. Gynecol. Oncol. 2000, 78, 302–308. [Google Scholar] [CrossRef]
- Frick, E.; Tyroller, M.; Panzer, M. Anxiety, depression and quality of life of cancer patients undergoing radiation therapy: A cross-sectional study in a community hospital outpatient centre. Eur. J. Cancer Care 2007, 16, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Dolan, L.B.; Barry, D.; Petrella, T.; Davey, L.; Minnes, A.; Yantzi, A.; Marzolini, S.; Oh, P. The Cardiac Rehabilitation Model Improves Fitness, Quality of Life, and Depression in Breast Cancer Survivors. J. Cardiopulm. Rehabil. Prev. 2018, 38, 246–252. [Google Scholar] [CrossRef]
- Oberoi, S.; Yang, J.; Woodgate, R.L.; Niraula, S.; Banerji, S.; Israels, S.J.; Altman, G.; Beattie, S.; Rabbani, R.; Askin, N.; et al. Association of Mindfulness-Based Interventions With Anxiety Severity in Adults With Cancer: A Systematic Review and Meta-analysis. JAMA Netw. Open 2020, 3, e2012598. [Google Scholar] [CrossRef]
- Després, J.P. Body fat distribution and risk of cardiovascular disease: An update. Circulation 2012, 126, 1301–1313. [Google Scholar] [CrossRef]
- Chan, D.S.; Vieira, A.R.; Aune, D.; Bandera, E.V.; Greenwood, D.C.; McTiernan, A.; Rosenblatt, D.N.; Thune, I.; Vieira, R.; Norat, T. Body mass index and survival in women with breast cancer-systematic literature review and meta-analysis of 82 follow-up studies. Ann. Oncol. 2014, 25, 1901–1914. [Google Scholar] [CrossRef]
- Barbaresko, J.; Koch, M.; Schulze, M.B.; Nöthlings, U. Dietary pattern analysis and biomarkers of low-grade inflammation: A systematic literature review. Nutr. Rev. 2013, 71, 511–527. [Google Scholar] [CrossRef]
- Sofi, F.; Dinu, M.; Pagliai, G.; Cesari, F.; Gori, A.M.; Sereni, A.; Becatti, M.; Fiorillo, C.; Marcucci, R.; Casini, A. Low-Calorie Vegetarian Versus Mediterranean Diets for Reducing Body Weight and Improving Cardiovascular Risk Profile: CARDIVEG Study (Cardiovascular Prevention With Vegetarian Diet). Circulation 2018, 137, 1103–1113. [Google Scholar] [CrossRef]
- Naghshi, S.; Sadeghi, O.; Willett, W.C.; Esmaillzadeh, A. Dietary intake of total, animal, and plant proteins and risk of all cause, cardiovascular, and cancer mortality: Systematic review and dose-response meta-analysis of prospective cohort studies. BMJ 2020, 370, m2412. [Google Scholar] [CrossRef] [PubMed]
- Sase, K.; Kida, K.; Furukawa, Y. Cardio-Oncology rehabilitation-challenges and opportunities to improve cardiovascular outcomes in cancer patients and survivors. J. Cardiol. 2020, 76, 559–567. [Google Scholar] [CrossRef] [PubMed]
- Geyer, H.; Caracciolo, G.; Abe, H.; Wilansky, S.; Carerj, S.; Gentile, F.; Nesser, H.-J.; Khandheria, B.K.; Narula, J.; Sengupta, P.P. Assessment of myocardial mechanics using speckle tracking echocardiography: Fundamentals and clinical applications. J. Am. Soc. Echocardiogr. 2010, 23, 351–369, quiz 453–455. [Google Scholar] [CrossRef]
- Reisner, S.A.; Lysyansky, P.; Agmon, Y.; Mutlak, D.; Lessick, J.; Friedman, Z. Global longitudinal strain: A novel index of left ventricular systolic function. J. Am. Soc. Echocardiogr. 2004, 17, 630–633. [Google Scholar] [CrossRef] [PubMed]
- Yingchoncharoen, T.; Agarwal, S.; Popović, Z.B.; Marwick, T.H. Normal ranges of left ventricular strain: A meta-analysis. J. Am. Soc. Echocardiogr. 2013, 26, 185–191. [Google Scholar] [CrossRef]
- Joseph, G.; Zaremba, T.; Johansen, M.B.; Ekeloef, S.; Heiberg, E.; Engblom, H.; Jensen, S.E.; Sogaard, P. Echocardiographic global longitudinal strain is associated with infarct size assessed by cardiac magnetic resonance in acute myocardial infarction. Echo Res. Pract. 2019, 6, 81–89. [Google Scholar] [CrossRef]
- Yingchoncharoen, T.; Gibby, C.; Rodriguez, L.L.; Grimm, R.A.; Marwick, T.H. Association of myocardial deformation with outcome in asymptomatic aortic stenosis with normal ejection fraction. Circ. Cardiovasc. Imaging 2012, 5, 719–725. [Google Scholar] [CrossRef]
- Zhang, K.W.; French, B.; May Khan, A.; Plappert, T.; Fang, J.C.; Sweitzer, N.K.; Borlaug, B.A.; Chirinos, J.A.; St John Sutton, M.; Cappola, T.P.; et al. Strain improves risk prediction beyond ejection fraction in chronic systolic heart failure. J. Am. Heart Assoc. 2014, 3, e000550. [Google Scholar] [CrossRef]
- Sengeløv, M.; Jørgensen, P.G.; Jensen, J.S.; Bruun, N.E.; Olsen, F.J.; Fritz-Hansen, T.; Nochioka, K.; Biering-Sørensen, T. Global Longitudinal Strain Is a Superior Predictor of All-Cause Mortality in Heart Failure With Reduced Ejection Fraction. JACC Cardiovasc. Imaging 2015, 8, 1351–1359. [Google Scholar] [CrossRef]
- Stanton, T.; Leano, R.; Marwick, T.H. Prediction of all-cause mortality from global longitudinal speckle strain: Comparison with ejection fraction and wall motion scoring. Circ. Cardiovasc. Imaging 2009, 2, 356–364. [Google Scholar] [CrossRef]
- Russo, C.; Jin, Z.; Elkind, M.S.; Rundek, T.; Homma, S.; Sacco, R.L.; Di Tullio, M.R. Prevalence and prognostic value of subclinical left ventricular systolic dysfunction by global longitudinal strain in a community-based cohort. Eur. J. Heart Fail. 2014, 16, 1301–1309. [Google Scholar] [CrossRef] [PubMed]
- Biering-Sørensen, T.; Biering-Sørensen, S.R.; Olsen, F.J.; Sengeløv, M.; Jørgensen, P.G.; Mogelvang, R.; Shah, A.M.; Jensen, J.S. Global Longitudinal Strain by Echocardiography Predicts Long-Term Risk of Cardiovascular Morbidity and Mortality in a Low-Risk General Population: The Copenhagen City Heart Study. Circ. Cardiovasc. Imaging 2017, 10, e005521. [Google Scholar] [CrossRef] [PubMed]
- Cuomo, A.; Mercurio, V.; Varricchi, G.; Galdiero, M.R.; Rossi, F.W.; Carannante, A.; Arpino, G.; Formisano, L.; Bianco, R.; Carlomagno, C.; et al. Impact of a cardio-oncology unit on prevention of cardiovascular events in cancer patients. ESC Heart Fail. 2022, 9, 1666–1676. [Google Scholar] [CrossRef] [PubMed]
- Laufer-Perl, M.; Derakhshesh, M.; Milwidsky, A.; Mor, L.; Ravid, D.; Amrami, N.; Sherez, J.; Keren, G.; Topilsky, Y.; Arbel, Y. Usefulness of Global Longitudinal Strain for Early Identification of Subclinical Left Ventricular Dysfunction in Patients With Active Cancer. Am. J. Cardiol. 2018, 122, 1784–1789. [Google Scholar] [CrossRef] [PubMed]
- Negishi, K.; Negishi, T.; Hare, J.L.; Haluska, B.A.; Plana, J.C.; Marwick, T.H. Independent and incremental value of deformation indices for prediction of trastuzumab-induced cardiotoxicity. J. Am. Soc. Echocardiogr. 2013, 26, 493–498. [Google Scholar] [CrossRef] [PubMed]
- Santoro, C.; Arpino, G.; Esposito, R.; Lembo, M.; Paciolla, I.; Cardalesi, C.; de Simone, G.; Trimarco, B.; De Placido, S.; Galderisi, M. 2D and 3D strain for detection of subclinical anthracycline cardiotoxicity in breast cancer patients: A balance with feasibility. Eur. Heart J. Cardiovasc. Imaging 2017, 18, 930–936. [Google Scholar] [CrossRef]
- Fourati, N.; Charfeddine, S.; Chaffai, I.; Dhouib, F.; Farhat, L.; Boukhris, M.; Abid, L.; Kammoun, S.; Mnejja, W.; Daoud, J. Subclinical left ventricle impairment following breast cancer radiotherapy: Is there an association between segmental doses and segmental strain dysfunction? Int. J. Cardiol. 2021, 345, 130–136. [Google Scholar] [CrossRef]
- Chen, L.; Ta, S.; Wu, W.; Wang, C.; Zhang, Q. Prognostic and Added Value of Echocardiographic Strain for Prediction of Adverse Outcomes in Patients with Locally Advanced Non-Small Cell Lung Cancer after Radiotherapy. Ultrasound Med. Biol. 2019, 45, 98–107. [Google Scholar] [CrossRef]
- Oikonomou, E.K.; Kokkinidis, D.G.; Kampaktsis, P.N.; Amir, E.A.; Marwick, T.H.; Gupta, D.; Thavendiranathan, P. Assessment of Prognostic Value of Left Ventricular Global Longitudinal Strain for Early Prediction of Chemotherapy-Induced Cardiotoxicity: A Systematic Review and Meta-analysis. JAMA Cardiol. 2019, 4, 1007. [Google Scholar] [CrossRef]
- Negishi, T.; Thavendiranathan, P.; Negishi, K.; Marwick, T.H. Rationale and Design of the Strain Surveillance of Chemotherapy for Improving Cardiovascular Outcomes: The SUCCOUR Trial. JACC Cardiovasc. Imaging 2018, 11, 1098–1105. [Google Scholar] [CrossRef]
- Thavendiranathan, P.; Negishi, T.; Somerset, E.; Negishi, K.; Penicka, M.; Lemieux, J.; Aakhus, S.; Miyazaki, S.; Shirazi, M.; Galderisi, M.; et al. Strain-Guided Management of Potentially Cardiotoxic Cancer Therapy. J. Am. Coll. Cardiol. 2021, 77, 392–401. [Google Scholar] [CrossRef] [PubMed]
- Nolan, M.T.; Plana, J.C.; Thavendiranathan, P.; Shaw, L.; Si, L.; Marwick, T.H. Cost-effectiveness of strain-targeted cardioprotection for prevention of chemotherapy-induced cardiotoxicity. Int. J. Cardiol. 2016, 212, 336–345. [Google Scholar] [CrossRef] [PubMed]
- D’andrea, A.; Sperlongano, S.; Pacileo, M.; Venturini, E.; Iannuzzo, G.; Gentile, M.; Sperlongano, R.; Vitale, G.; Maglione, M.; Cice, G.; et al. New Ultrasound Technologies for Ischemic Heart Disease Assessment and Monitoring in Cardiac Rehabilitation. J. Clin. Med. 2020, 9, 3131. [Google Scholar] [CrossRef]
- Sahin, A.A.; Ozben, B.; Sunbul, M.; Yagci, I.; Sayar, N.; Cincin, A.; Gurel, E.; Tigen, K.; Basaran, Y. The effect of cardiac rehabilitation on blood pressure, and on left atrial and ventricular functions in hypertensive patients. J. Clin. Ultrasound 2020, 49, 456–465. [Google Scholar] [CrossRef] [PubMed]
- O’Driscoll, J.M.; Edwards, J.J.; Wiles, J.D.; Taylor, K.A.; Leeson, P.; Sharma, R. Myocardial work and left ventricular mechanical adaptations following isometric exercise training in hypertensive patients. Eur. J. Appl. Physiol. 2022, 122, 727–734. [Google Scholar] [CrossRef] [PubMed]
- Malfatto, G.; Revera, M.; Branzi, G.; Ciambellotti, F.; Giglio, A.; Blengino, S.; Oldani, M.; Facchini, C.; Parati, G.; Facchini, M. A brief period of intensive cardiac rehabilitation improves global longitudinal strain and diastolic function after a first uncomplicated myocardial infarction. Acta Cardiol. 2017, 72, 284–291. [Google Scholar] [CrossRef] [PubMed]
- D’Andrea, A.; Carbone, A.; Ilardi, F.; Pacileo, M.; Savarese, C.; Sperlongano, S.; Di Maio, M.; Giallauria, F.; Russo, V.; Bossone, E.; et al. Effects of High Intensity Interval Training Rehabilitation Protocol after an Acute Coronary Syndrome on Myocardial Work and Atrial Strain. Medicina 2022, 58, 453. [Google Scholar] [CrossRef]
- Trachsel, L.D.; David, L.P.; Gayda, M.; Henri, C.; Hayami, D.; Thorin-Trescases, N.; Thorin, E.; Blain, M.A.; Cossette, M.; Lalonge, J.; et al. The impact of high-intensity interval training on ventricular remodeling in patients with a recent acute myocardial infarction-A randomized training intervention pilot study. Clin. Cardiol. 2019, 42, 1222–1231. [Google Scholar] [CrossRef]
- Van De Heyning, C.M.; De Maeyer, C.; Pattyn, N.; Beckers, P.J.; Cornelissen, V.A.; Goetschalckx, K.; Possemiers, N.; Van Craenenbroeck, E.M.; Voigt, J.U.; Vanhees, L.; et al. Impact of aerobic interval training and continuous training on left ventricular geometry and function: A SAINTEX-CAD substudy. Int. J. Cardiol. 2018, 257, 193–198. [Google Scholar] [CrossRef]
- Eser, P.; Trachsel, L.D.; Marcin, T.; Herzig, D.; Freiburghaus, I.; De Marchi, S.; Zimmermann, A.; Schmid, J.P.; Wilhelm, M. Short- and Long-Term Effects of High-Intensity Interval Training vs. Moderate-Intensity Continuous Training on Left Ventricular Remodeling in Patients Early After ST-Segment Elevation Myocardial Infarction-The HIIT-EARLY Randomized Controlled Trial. Front. Cardiovasc. Med. 2022, 9, 869501. [Google Scholar] [CrossRef]
- Hojan, K.; Procyk, D.; Horyńska-Kęstowicz, D.; Leporowska, E.; Litwiniuk, M. The Preventive role of Regular Physical Training in Ventricular Remodeling, Serum Cardiac Markers, and Exercise Performance Changes in Breast Cancer in Women Undergoing Trastuzumab Therapy-An REH-HER Study. J. Clin. Med. 2020, 9, 1379. [Google Scholar] [CrossRef] [PubMed]
- Howden, E.J.; Bigaran, A.; Beaudry, R.; Fraser, S.; Selig, S.; Foulkes, S.; Antill, Y.; Nightingale, S.; Loi, S.; Haykowsky, M.J.; et al. Exercise as a diagnostic and therapeutic tool for the prevention of cardiovascular dysfunction in breast cancer patients. Eur. J. Prev. Cardiol. 2019, 26, 305–315. [Google Scholar] [CrossRef] [PubMed]
- Kirkham, A.A.; Eves, N.D.; Shave, R.E.; Bland, K.A.; Bovard, J.; Gelmon, K.A.; Virani, S.A.; McKenzie, D.C.; Stöhr, E.J.; Waburton, D.E.; et al. The effect of an aerobic exercise bout 24 h prior to each doxorubicin treatment for breast cancer on markers of cardiotoxicity and treatment symptoms: A RCT. Breast Cancer Res. Treat. 2018, 167, 719–729. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Balboa, E.; González-Salvado, V.; Rodríguez-Romero, B.; Martínez-Monzonís, A.; Pedreira-Pérez, M.; Palacios-Ozores, P.; López-López, R.; Peña-Gil, C.; González-Juanatey, J.R. A randomized trial to evaluate the impact of exercise-based cardiac rehabilitation for the prevention of chemotherapy-induced cardiotoxicity in patients with breast cancer: ONCORE study protocol. BMC Cardiovasc. Disord. 2021, 21, 165. [Google Scholar] [CrossRef]
| Advantages | Limitations |
|---|---|
| Cost-effective | Not available for all ultrasound devices |
| High reproducibility | Low image quality reduces measurements accuracy |
| Minimal angle dependence | Experienced operator required |
| Ability to measure strain in multiple LV segments from a single acquisition | Inter-vendor variability in strain measurements |
| High predictive power for mortality | Error in ROI estimation leads to GLS underestimation |
| Earlier detection of subclinical LV dysfunction | Increased value in response to preload increase |
| It is an independent stronger predictor of CTRCD |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cuomo, G.; Iannone, F.P.; Di Lorenzo, A.; Testa, C.; Ciccarelli, M.; Venturini, E.; Cesaro, A.; Pacileo, M.; Tagliamonte, E.; D’Andrea, A.; et al. Potential Role of Global Longitudinal Strain in Cardiac and Oncological Patients Undergoing Cardio-Oncology Rehabilitation (CORE). Clin. Pract. 2023, 13, 384-397. https://doi.org/10.3390/clinpract13020035
Cuomo G, Iannone FP, Di Lorenzo A, Testa C, Ciccarelli M, Venturini E, Cesaro A, Pacileo M, Tagliamonte E, D’Andrea A, et al. Potential Role of Global Longitudinal Strain in Cardiac and Oncological Patients Undergoing Cardio-Oncology Rehabilitation (CORE). Clinics and Practice. 2023; 13(2):384-397. https://doi.org/10.3390/clinpract13020035
Chicago/Turabian StyleCuomo, Gianluigi, Francesca Paola Iannone, Anna Di Lorenzo, Crescenzo Testa, Michele Ciccarelli, Elio Venturini, Arturo Cesaro, Mario Pacileo, Ercole Tagliamonte, Antonello D’Andrea, and et al. 2023. "Potential Role of Global Longitudinal Strain in Cardiac and Oncological Patients Undergoing Cardio-Oncology Rehabilitation (CORE)" Clinics and Practice 13, no. 2: 384-397. https://doi.org/10.3390/clinpract13020035
APA StyleCuomo, G., Iannone, F. P., Di Lorenzo, A., Testa, C., Ciccarelli, M., Venturini, E., Cesaro, A., Pacileo, M., Tagliamonte, E., D’Andrea, A., Vecchione, C., Vigorito, C., & Giallauria, F. (2023). Potential Role of Global Longitudinal Strain in Cardiac and Oncological Patients Undergoing Cardio-Oncology Rehabilitation (CORE). Clinics and Practice, 13(2), 384-397. https://doi.org/10.3390/clinpract13020035









