Feasibility of Nonintubated Anesthesia for Lumboperitoneal Shunt Implantation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Anesthesia
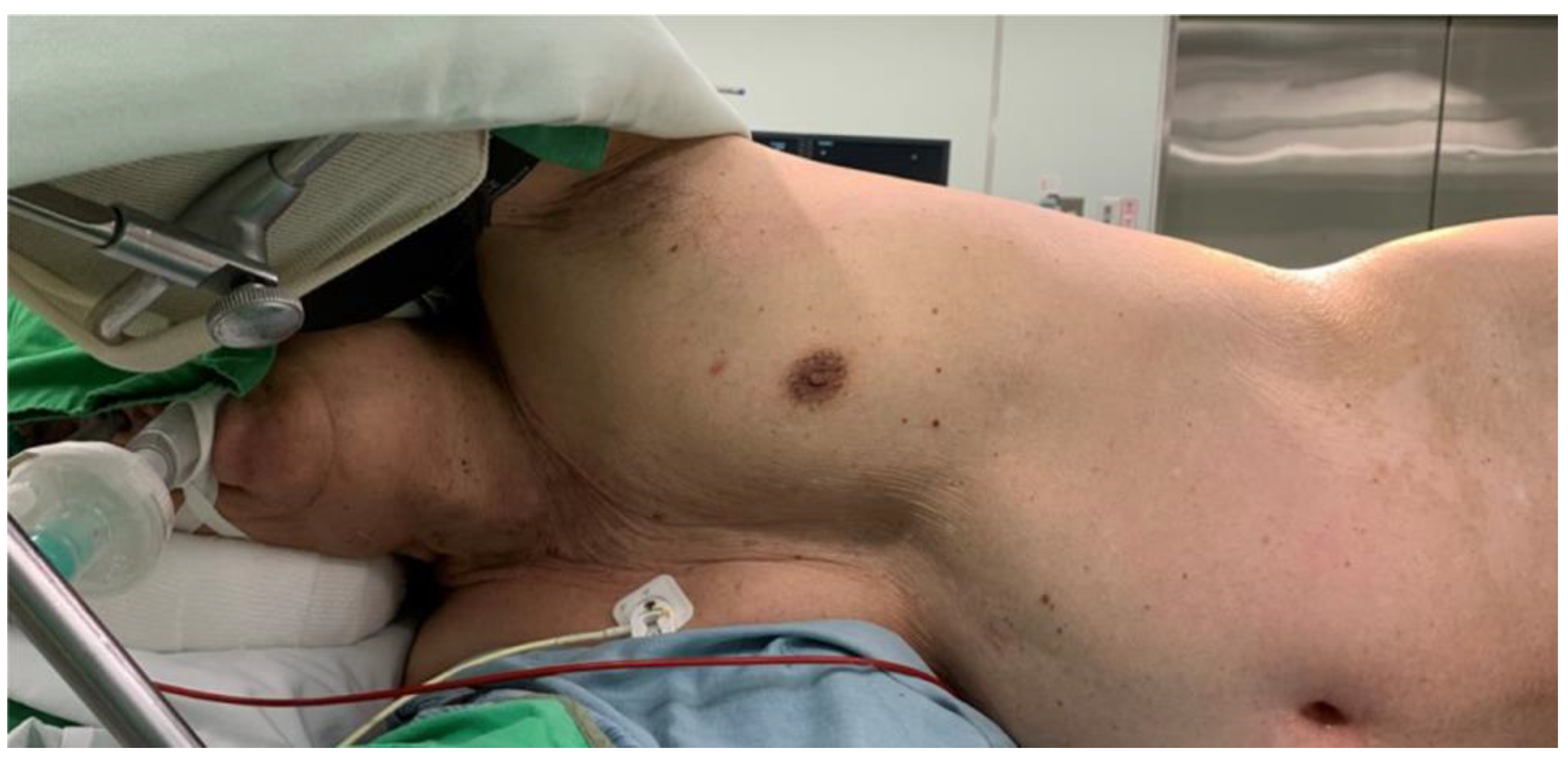
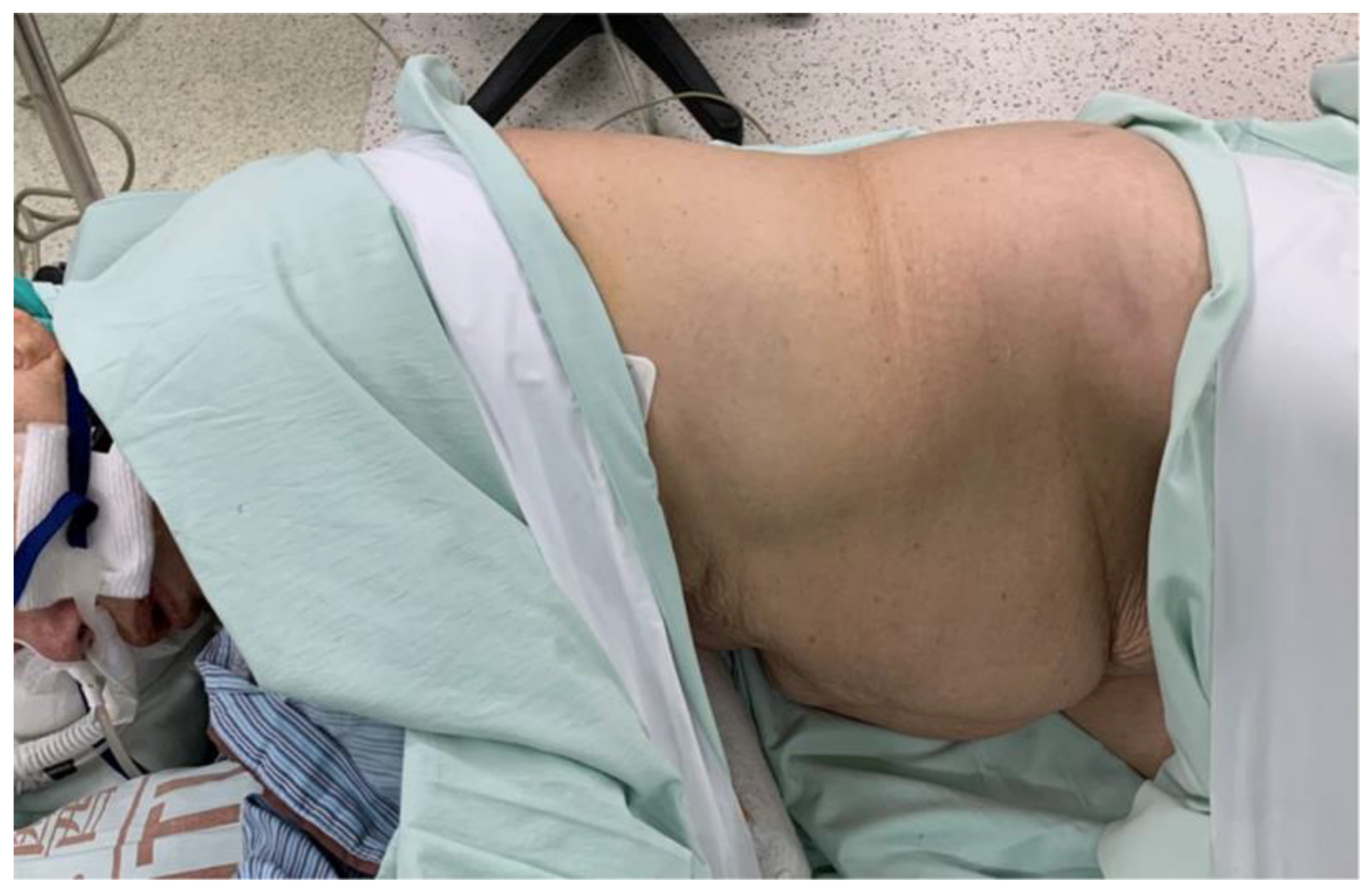
2.2. Operative Procedure
2.3. Data Collection and Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kiss, G.; Claret, A.; Desbordes, J.; Porte, H. Thoracic epidural anaesthesia for awake thoracic surgery in severely dyspnoeic patients excluded from general anaesthesia. Interact. Cardiovasc. Thorac. Surg. 2014, 19, 816–823. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de Albuquerque Medeiros, R.; Faresin, S.; Jardim, J. postoperative lung complications and mortality in patients with mild-to-moderate copd undergoing elective general surgery. Arch. Bronconeumol. 2001, 37, 227–234. [Google Scholar] [CrossRef]
- Whitehead, T.; Slutsky, A.S. The pulmonary physician in critical care * 7: Ventilator induced lung injury. Thorax 2002, 57, 635–642. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shprecher, D.; Schwalb, J.; Kurlan, R. Normal pressure hydrocephalus: Diagnosis and treatment. Curr. Neurol. Neurosci. Rep. 2008, 8, 371–376. [Google Scholar] [CrossRef]
- Gonzalez-Rivas, D.; Bonome, C.; Fieira, E.; Aymerich, H.; Fernandez, R.; Delgado, M.; Mendez, L.; de la Torre, M. Non-intubated video-assisted thoracoscopic lung resections: The future of thoracic surgery? Eur. J. Cardiothorac. Surg. 2016, 49, 721–731. [Google Scholar] [CrossRef] [PubMed]
- Pompeo, E. State of the art and perspectives in non-intubated thoracic surgery. Ann. Transl. Med. 2014, 2, 106. [Google Scholar]
- Peng, G.; Cui, F.; Ang, K.L.; Zhang, X.; Yin, W.; Shao, W.; Dong, Q.; Liang, L.; He, J. Non-intubated combined with video-assisted thoracoscopic in carinal reconstruction. J. Thorac. Dis. 2016, 8, 586–593. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.; Wang, K.; Qian, Z.; Zeng, L.; Gao, L. Lumboperitoneal and ventriculoperitoneal shunt surgery for posthemorrhagic communicating hydrocephalus: A comparison. World Neurosurg. 2019, 127, e638–e643. [Google Scholar] [CrossRef]
- Xie, D.; Chen, H.; Guo, X.; Liu, Y. Comparative study of lumboperitoneal shunt and ventriculoperitoneal shunt in the treatment of idiopathic normal pressure hydrocephalus. Am. J. Transl. Res. 2021, 13, 11917–11924. [Google Scholar]
- Goel, A.; Craven, C.; Matloob, S.; Thompson, S.; Watkins, L.; Toma, A. Csf-diverting shunts: Implications for abdominal and pelvic surgeons; a review and pragmatic overview. Ann. Med. Surg. (Lond.) 2019, 48, 100–104. [Google Scholar] [CrossRef]
- Sun, T.; Li, X.; Zhang, Q.; Zhou, Y.; Guan, J. Efficacy and safety of lumboperitoneal shunt in the treatment of all-cause communicating hydrocephalus: Analysis of risk factors of shunt failure. World Neurosurg. 2019, 132, e956–e962. [Google Scholar] [CrossRef] [PubMed]
- Duthel, R.; Nuti, C.; Motuo-Fotso, M.J.; Beauchesne, P.; Brunon, J. complications of lumboperitoneal shunts. A retrospective study of a series of 195 patients (214 procedures). Neurochirurgie 1996, 42, 83–89, discussion 89–90. [Google Scholar] [PubMed]
- Jeon, J.; Sung, S.; Moon, Y.; Koo, J.; Hyun, K.; Han, K.; Hwang, W. Comparison of early postoperative cytokine changes in patients undergoing intubated and non-intubated thoracic surgery: A randomized controlled trial. Interact. Cardiovasc. Thorac. Surg. 2021, 32, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.X.; Song, C.T.; Gao, Z.; Zhou, B.; Wang, H.B.; Gong, Q.; Li, B.; Guo, Q.; Li, H.F. A comparison of non-intubated video-assisted thoracic surgery with spontaneous ventilation and intubated video-assisted thoracic surgery: A meta-analysis based on 14 randomized controlled trials. J. Thorac. Dis. 2021, 13, 1624–1640. [Google Scholar] [CrossRef] [PubMed]
- Prisciandaro, E.; Bertolaccini, L.; Sedda, G.; Spaggiari, L. Non-intubated thoracoscopic lobectomies for lung cancer: An exploratory systematic review and meta-analysis. Interact. Cardiovasc. Thorac. Surg. 2020, 31, 499–506. [Google Scholar] [CrossRef]
- Adriani, J.; Naraghi, M.; Ward, M. Complications of endotracheal intubation. South Med. J. 1988, 81, 739–744. [Google Scholar] [CrossRef]
- Wang, M.L.; Galvez, C.; Chen, J.S.; Navarro-Martinez, J.; Bolufer, S.; Hung, M.H.; Hsu, H.H.; Cheng, Y.J. Non-intubated single-incision video-assisted thoracic surgery: A two-center cohort of 188 patients. J. Thorac. Dis. 2017, 9, 2587–2598. [Google Scholar] [CrossRef] [Green Version]
- Liu, J.; Cui, F.; Pompeo, E.; Gonzalez-Rivas, D.; Chen, H.; Yin, W.; Shao, W.; Li, S.; Pan, H.; Shen, J.; et al. The impact of non-intubated versus intubated anaesthesia on early outcomes of video-assisted thoracoscopic anatomical resection in non-small-cell lung cancer: A propensity score matching analysis. Eur. J. Cardiothorac. Surg. 2016, 50, 920–925. [Google Scholar] [CrossRef] [Green Version]
- Ambrogi, V.; Mineo, T.C. Vats biopsy for undetermined interstitial lung disease under non-general anesthesia: Comparison between uniportal approach under intercostal block vs. Three-ports in epidural anesthesia. J. Thorac. Dis. 2014, 6, 888–895. [Google Scholar]
- Hopkins, E.; Sharma, S. Physiology, Functional Residual Capacity; Statpearls: Treasure Island, FL, USA, 2021. [Google Scholar]
- Cammu, G.; De Witte, J.; De Veylder, J.; Byttebier, G.; Vandeput, D.; Foubert, L.; Vandenbroucke, G.; Deloof, T. Postoperative residual paralysis in outpatients versus inpatients. Anesth Analg. 2006, 102, 426–429. [Google Scholar] [CrossRef]
- Hayes, A.H.; Mirakhur, R.K.; Breslin, D.S.; Reid, J.E.; McCourt, K.C. Postoperative residual block after intermediate-acting neuromuscular blocking drugs. Anaesthesia 2001, 56, 312–318. [Google Scholar] [CrossRef] [PubMed]
- Farhan, H.; Moreno-Duarte, I.; McLean, D.; Eikermann, M. Residual paralysis: Does it influence outcome after ambulatory surgery? Curr. Anesthesiol. Rep. 2014, 4, 290–302. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sarton, E.; Teppema, L.; Nieuwenhuijs, D.; Matthes, H.W.; Kieffer, B.; Dahan, A. Opioid effect on breathing frequency and thermogenesis in mice lacking exon 2 of the mu-opioid receptor gene. Adv. Exp. Med. Biol. 2001, 499, 399–404. [Google Scholar] [PubMed]
- Henson, L.C.; Ward, D.S. Effects of anaesthetics and sedatives on the control of breathing. Ann. Acad. Med. Singap. 1994, 23, 125–129. [Google Scholar]
- Nakamoto, S.; Hirose, M. Prediction of early c-reactive protein levels after non-cardiac surgery under general anesthesia. PLoS ONE 2019, 14, e0226032. [Google Scholar] [CrossRef] [Green Version]
- Yu, M.G.; Jing, R.; Mo, Y.J.; Lin, F.; Du, X.K.; Ge, W.Y.; Dai, H.J.; Hu, Z.K.; Zhang, S.S.; Pan, L.H. Non-intubated anesthesia in patients undergoing video-assisted thoracoscopic surgery: A systematic review and meta-analysis. PLoS ONE 2019, 14, e0224737. [Google Scholar] [CrossRef] [Green Version]
- Saeg, A.A.A.; Alnori, H. Laryngeal injury and dysphonia after endotracheal intubation. J. Med. Life 2021, 14, 355–360. [Google Scholar] [CrossRef]
- Al-Rashed, S.; Kareem, H.; Kalra, N.; D’Antona, L.; Obeidat, M.; Patel, B.; Toma, A. Lumboperitoneal shunt insertion without fluoroscopy guidance: Accuracy of placement in a series of 107 procedures. F1000Research 2017, 6, 565. [Google Scholar] [CrossRef]
- Wahdan, A.S.; El-Refai, N.A.R.; Omar, S.H.; Moneem, S.A.A.; Mohamed, M.M.; Hussien, M.M. Endotracheal intubation in patients undergoing open abdominal surgery in the lateral position: A comparison between the intubating video stylet and fiberoptic intubating bronchoscopy. Korean J. Anesthesiol. 2021, 74, 234–241. [Google Scholar] [CrossRef]
- Cheng, K.I.; Chu, K.S.; Chau, S.W.; Ying, S.L.; Hsu, H.T.; Chang, Y.L.; Tang, C.S. Lightwand-assisted intubation of patients in the lateral decubitus position. Anesth Analg. 2004, 99, 279–283. [Google Scholar] [CrossRef] [Green Version]
- Adachi, Y.U.; Satomoto, M.; Higuchi, H. Tracheal intubation in the lateral position. Anesth Analg. 2004, 99, 952. [Google Scholar] [CrossRef] [PubMed]
| Case No. | Age/Sex | Diagnosis | Underlying Diseases | LVEF | Height, cm | Weight, kg | BMI, kg/m2 | Oxygen Delivery | Anesthesia Time (Min) | Operative Time (Min) | Complications |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 78/M | NPH | Laryngeal cancer | 62% | 157.8 | 52.9 | 21.2 | HFNC | 98 | 48 | None |
| 2 | 87/M | NPH | Stroke, DM, Parkinson | 51.8% | 150 | 49 | 21.8 | HFNC | 123 | 59 | None |
| 3 | 66/M | NPH | MI, HTN, CKD | 66.6% | 168 | 51.9 | 18.4 | HFNC | 109 | 64 | None |
| 4 | 75/F | NPH | C-HIVD | 71.2% | 154 | 43 | 18.1 | HFNC | 94 | 60 | None |
| 5 | 94/F | NPH | Heart failure | 61% | 168 | 51.9 | 18.4 | LMA | 95 | 48 | None |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, A.P.-H.; Tsai, F.-F.; Chen, C.-C.; Lee, T.-S.; Kuo, L.-T. Feasibility of Nonintubated Anesthesia for Lumboperitoneal Shunt Implantation. Clin. Pract. 2022, 12, 449-456. https://doi.org/10.3390/clinpract12030049
Huang AP-H, Tsai F-F, Chen C-C, Lee T-S, Kuo L-T. Feasibility of Nonintubated Anesthesia for Lumboperitoneal Shunt Implantation. Clinics and Practice. 2022; 12(3):449-456. https://doi.org/10.3390/clinpract12030049
Chicago/Turabian StyleHuang, Abel Po-Hao, Feng-Fang Tsai, Chien-Chia Chen, Tzong-Shiun Lee, and Lu-Ting Kuo. 2022. "Feasibility of Nonintubated Anesthesia for Lumboperitoneal Shunt Implantation" Clinics and Practice 12, no. 3: 449-456. https://doi.org/10.3390/clinpract12030049
APA StyleHuang, A. P.-H., Tsai, F.-F., Chen, C.-C., Lee, T.-S., & Kuo, L.-T. (2022). Feasibility of Nonintubated Anesthesia for Lumboperitoneal Shunt Implantation. Clinics and Practice, 12(3), 449-456. https://doi.org/10.3390/clinpract12030049








