DOPC Liposomal Formulation of Antimicrobial Peptide LL17-32 with Reduced Cytotoxicity: A Promising Carrier Against Porphyromonas gingivalis
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Preparation of LL17-32-Loaded/Unloaded Liposomes and Fluorescence Labelling
2.2.2. Particle Size and ζ-Potential Analysis
2.2.3. Transmission Electron Microscopy (TEM) Imaging
2.2.4. Determination of Association Efficacy (AE)
2.2.5. In Vitro RTF Release Study
2.2.6. Physical Stability in RTF and Basal Culture Medium
2.2.7. Liposome Membrane Fluidity
2.2.8. Bacteria-Killing Activity Against Porphyromonas gingivalis
2.2.9. FITC Labelling of Porphyromonas gingivalis
2.2.10. Treatment of LL17-32-Loaded Liposomes with P. gingivalis
2.2.11. Flow Cytometric Analysis of LL17-32-Loaded Liposomes to P. gingivalis
2.2.12. Mammalian Cell Culture
2.2.13. Cell Viability Assay
2.2.14. Cytotoxicity Assay
2.2.15. Statistical Analysis
3. Results and Discussion
3.1. Characterization of Particle Size, Polydispersity and ζ-Potential
3.2. Transmission Electron Microscopy (TEM) Imaging Studies
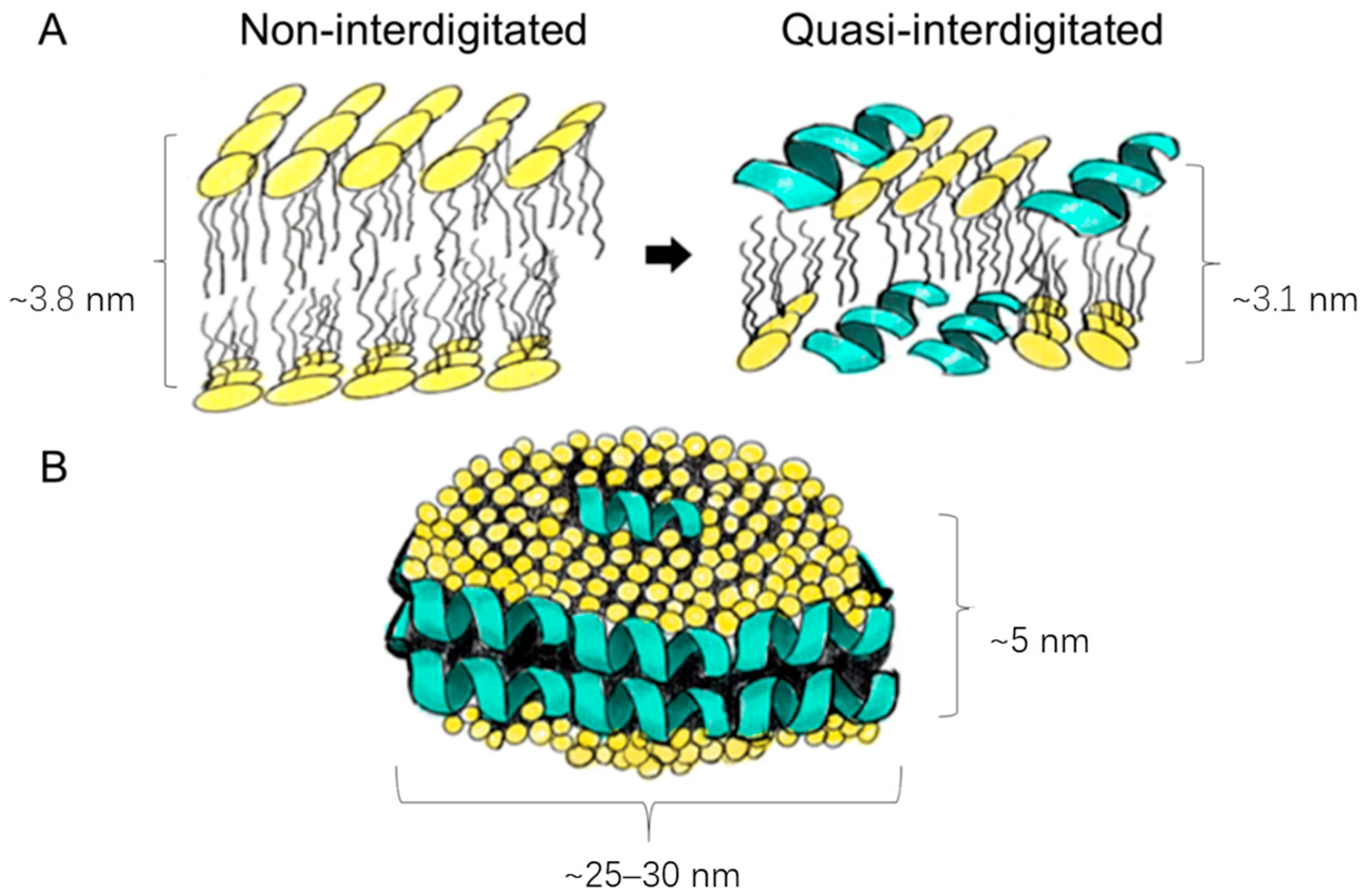
3.3. Liposome Membrane Fluidity and Structure
3.4. Determination of LL17-32 Association Efficiency (AE) and in Vitro Release in Reduced Transport Fluid (RTF)
3.5. Colloidal Stability in RTF and in Basal Culture Medium
3.6. Killing Activity on Porphyromonas gingivalis
3.7. Flow Cytometric Analysis of LL17-32-Loaded Liposomes Binding to P. gingivalis
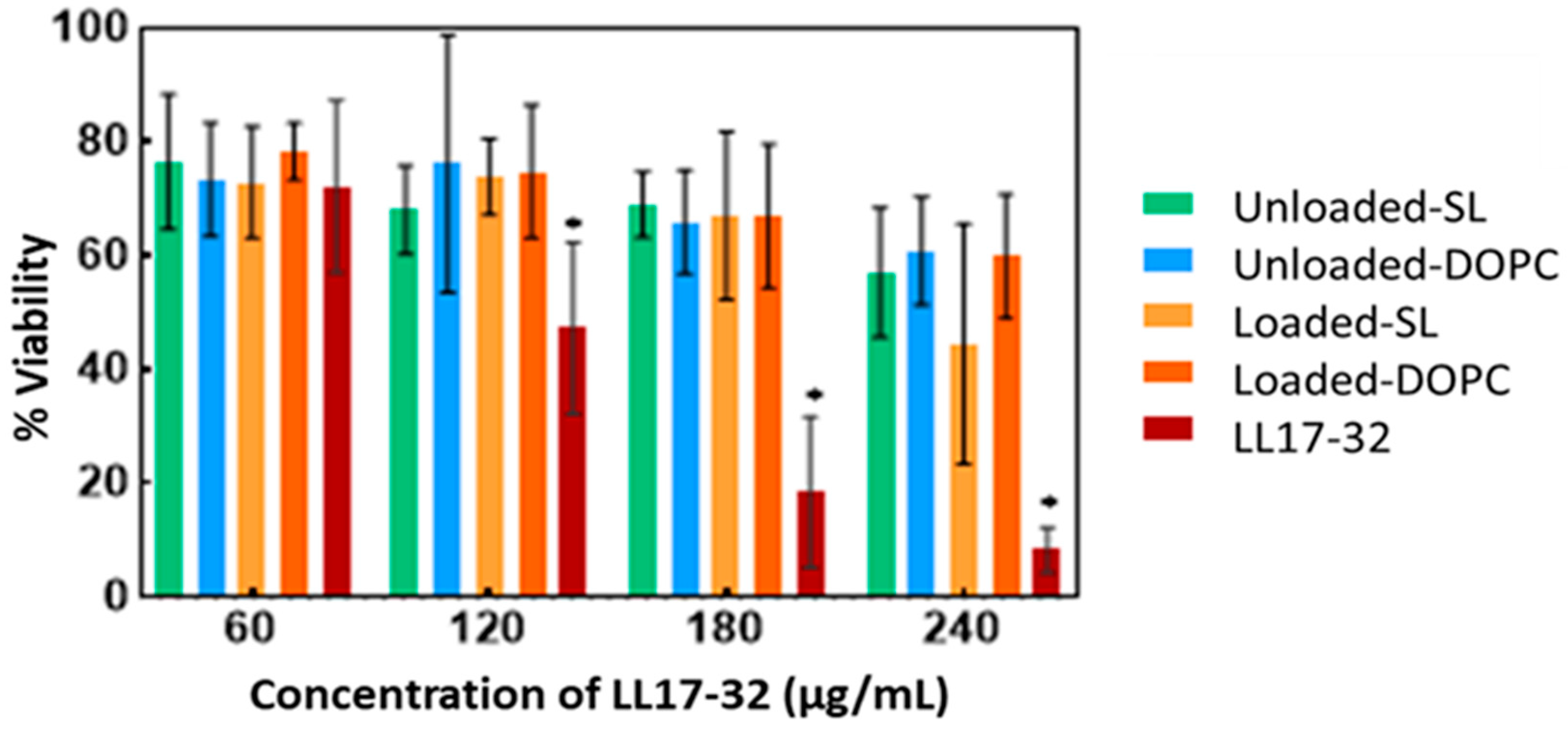
3.8. Cell Viability and Cytotoxicity
4. Conclusions and Outlooks
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Petersen, P.E.; Ogawa, H. The global burden of periodontal disease: Towards integration with chronic disease prevention and control. Periodontol. 2000 2012, 60, 15–39. [Google Scholar] [CrossRef]
- Kalhan, A.C.; Wong, M.L.; Allen, F.; Gao, X. Periodontal disease and systemic health: An update for medical practitioners. Ann. Acad. Med. Singap. 2022, 51, 567–574. [Google Scholar] [CrossRef]
- Dominy, S.S.; Lynch, C.; Ermini, F.; Benedyk, M.; Marczyk, A.; Konradi, A.; Nguyen, M.; Haditsch, U.; Raha, D.; Griffin, C. Porphyromonas gingivalis in Alzheimer’s disease brains: Evidence for disease causation and treatment with small-molecule inhibitors. Sci. Adv. 2019, 5, eaau3333. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.S. From focal sepsis to periodontal medicine: A century of exploring the role of the oral microbiome in systemic disease. J. Physiol. 2017, 595, 465–476. [Google Scholar] [CrossRef] [PubMed]
- Gnanasekaran, J.; Binder Gallimidi, A.; Saba, E.; Pandi, K.; Eli Berchoer, L.; Hermano, E.; Angabo, S.; Makkawi, H.a.; Khashan, A.; Daoud, A. Intracellular Porphyromonas gingivalis promotes the tumorigenic behavior of pancreatic carcinoma cells. Cancers 2020, 12, 2331. [Google Scholar] [CrossRef]
- Oliveira, F.A.F.; Forte, C.P.F.; de Barros Silva, P.G.n.; Lopes, C.B.; Montenegro, R.C.; Dos Santos, Â.K.C.R.; Sobrinho, C.R.M.R.; Mota, M.R.L.; Sousa, F.B.; Alves, A.P.N.N. Molecular analysis of oral bacteria in heart valve of patients with cardiovascular disease by real-time polymerase chain reaction. Medicine 2015, 94, e2067. [Google Scholar] [CrossRef]
- Maresz, K.J.; Hellvard, A.; Sroka, A.; Adamowicz, K.; Bielecka, E.; Koziel, J.; Gawron, K.; Mizgalska, D.; Marcinska, K.A.; Benedyk, M. Porphyromonas gingivalis facilitates the development and progression of destructive arthritis through its unique bacterial peptidylarginine deiminase (PAD). PLoS Pathog. 2013, 9, e1003627. [Google Scholar] [CrossRef]
- Krishnan, K.; Chen, T.; Paster, B.J. A practical guide to the oral microbiome and its relation to health and disease. Oral Dis. 2017, 23, 276–286. [Google Scholar] [CrossRef] [PubMed]
- Sudhakara, P.; Gupta, A.; Bhardwaj, A.; Wilson, A. Oral Dysbiotic Communities and Their Implications in Systemic Diseases. Dent. J. 2018, 6, 10. [Google Scholar] [CrossRef]
- How, K.Y.; Song, K.P.; Chan, K.G. Porphyromonas gingivalis: An Overview of Periodontopathic Pathogen below the Gum Line. Front. Microbiol. 2016, 7, 53. [Google Scholar] [CrossRef]
- Sakanaka, A.; Takeuchi, H.; Kuboniwa, M.; Amano, A. Dual lifestyle of Porphyromonas gingivalis in biofilm and gingival cells. Microb. Pathog. 2016, 94, 42–47. [Google Scholar] [CrossRef]
- Hajishengallis, G.; Liang, S.; Payne, M.A.; Hashim, A.; Jotwani, R.; Eskan, M.A.; McIntosh, M.L.; Alsam, A.; Kirkwood, K.L.; Lambris, J.D.; et al. Low-abundance biofilm species orchestrates inflammatory periodontal disease through the commensal microbiota and complement. Cell Host Microbe 2011, 10, 497–506. [Google Scholar] [CrossRef]
- Colombo, A.V.; da Silva, C.M.; Haffajee, A.; Colombo, A.P. Identification of intracellular oral species within human crevicular epithelial cells from subjects with chronic periodontitis by fluorescence in situ hybridization. J. Periodontal Res. 2007, 42, 236–243. [Google Scholar] [CrossRef] [PubMed]
- Murakami, J.; Kato, T.; Kawai, S.; Akiyama, S.; Amano, A.; Morisaki, I. Cellular motility of Down syndrome gingival fibroblasts is susceptible to impairment by Porphyromonas gingivalis invasion. J. Periodontol. 2008, 79, 721–727. [Google Scholar] [CrossRef]
- Zhang, W.; Ju, J.; Rigney, T.; Tribble, G. Integrin α5β1-fimbriae binding and actin rearrangement are essential for Porphyromonas gingivalis invasion of osteoblasts and subsequent activation of the JNK pathway. BMC Microbiol. 2013, 13, 5. [Google Scholar] [CrossRef]
- Kato, T.; Kawai, S.; Nakano, K.; Inaba, H.; Kuboniwa, M.; Nakagawa, I.; Tsuda, K.; Omori, H.; Ooshima, T.; Yoshimori, T.; et al. Virulence of Porphyromonas gingivalis is altered by substitution of fimbria gene with different genotype. Cell Microbiol. 2007, 9, 753–765. [Google Scholar] [CrossRef]
- Lamont, R.J.; Yilmaz, O. In or out: The invasiveness of oralbacteria. Periodontol. 2000 2002, 30, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Amano, A. Disruption of epithelial barrier and impairment of cellular function by Porphyromonas gingivalis. Front. Biosci. 2007, 12, 3965–3974. [Google Scholar] [CrossRef]
- Amano, A.; Furuta, N.; Tsuda, K. Host membrane trafficking for conveyance of intracellular oral pathogens. Periodontol. 2000 2010, 52, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Hajishengallis, G.; Lamont, R.J. Beyond the red complex and into more complexity: The polymicrobial synergy and dysbiosis (PSD) model of periodontal disease etiology. Mol. Oral. Microbiol. 2012, 27, 409–419. [Google Scholar] [CrossRef]
- Grassi, L.; Maisetta, G.; Esin, S.; Batoni, G. Combination Strategies to Enhance the Efficacy of Antimicrobial Peptides against Bacterial Biofilms. Front. Microbiol. 2017, 8, 2409. [Google Scholar] [CrossRef]
- Ouhara, K.; Komatsuzawa, H.; Yamada, S.; Shiba, H.; Fujiwara, T.; Ohara, M.; Sayama, K.; Hashimoto, K.; Kurihara, H.; Sugai, M. Susceptibilities of periodontopathogenic and cariogenic bacteria to antibacterial peptides, β-defensins and LL37, produced by human epithelial cells. J. Antimicrob. Chemother. 2005, 55, 888–896. [Google Scholar] [CrossRef]
- Ji, S.; Hyun, J.; Park, E.; Lee, B.L.; Kim, K.K.; Choi, Y. Susceptibility of various oral bacteria to antimicrobial peptides and to phagocytosis by neutrophils. J. Periodontal Res. 2007, 42, 410–419. [Google Scholar] [CrossRef]
- De Brucker, K.; Delattin, N.; Robijns, S.; Steenackers, H.; Verstraeten, N.; Landuyt, B.; Luyten, W.; Schoofs, L.; Dovgan, B.; Frohlich, M.; et al. Derivatives of the mouse cathelicidin-related antimicrobial peptide (CRAMP) inhibit fungal and bacterial biofilm formation. Antimicrob. Agents Chemother. 2014, 58, 5395–5404. [Google Scholar] [CrossRef]
- Ho, M.H.; Lamont, R.J.; Xie, H. Identification of Streptococcus cristatus peptides that repress expression of virulence genes in Porphyromonas gingivalis. Sci. Rep. 2017, 7, 1413. [Google Scholar] [CrossRef]
- Wakabayashi, H.; Yamauchi, K.; Kobayashi, T.; Yaeshima, T.; Iwatsuki, K.; Yoshie, H. Inhibitory effects of lactoferrin on growth and biofilm formation of Porphyromonas gingivalis and Prevotella intermedia. Antimicrob. Agents Chemother. 2009, 53, 3308–3316. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.Y.; Cheng, J.W.; Yu, H.Y.; Lin, L.; Chih, Y.H.; Pan, Y.P. Efficacy of a novel antimicrobial peptide against periodontal pathogens in both planktonic and polymicrobial biofilm states. Acta Biomater. 2015, 25, 150–161. [Google Scholar] [CrossRef] [PubMed]
- Li, J.Y.; Wang, X.J.; Wang, L.N.; Ying, X.X.; Ren, X.; Liu, H.Y.; Xu, L.; Ma, G.W. High in vitro antibacterial activity of Pac-525 against Porphyromonas gingivalis biofilms cultured on titanium. BioMed Res. Int. 2015, 2015, 909870. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Jia, L.; Zhang, Q.; Zhou, X.; Liu, Z.; Li, B.; Zhu, Z.; Wang, F.; Yu, C.; Zhang, Q.; et al. A novel antimicrobial peptide against dental-caries-associated bacteria. Anaerobe 2017, 47, 165–172. [Google Scholar] [CrossRef]
- Daep, C.A.; James, D.M.; Lamont, R.J.; Demuth, D.R. Structural characterization of peptide-mediated inhibition of Porphyromonas gingivalis biofilm formation. Infect. Immun. 2006, 74, 5756–5762. [Google Scholar] [CrossRef]
- Zhang, O.L.; Niu, J.Y.; Yin, I.X.; Yu, O.Y.; Mei, M.L.; Chu, C.H. Antibacterial properties of the antimicrobial peptide gallic acid-polyphemusin I (GAPI). Antibiotics 2023, 12, 1350. [Google Scholar] [CrossRef]
- Jiang, S.J.; Xiao, X.; Zheng, J.; Lai, S.; Yang, L.; Li, J.; Liu, C.; Yang, Y.; Mu, Y. Antibacterial and antibiofilm activities of novel antimicrobial peptide DP7 against the periodontal pathogen Porphyromonas gingivalis. J. Appl. Microbiol. 2022, 133, 1052–1062. [Google Scholar] [CrossRef] [PubMed]
- Gorr, S.-U.; Chen, R.; Abrahante, J.E.; Joyce, P.B. The oral pathogen Porphyromonas gingivalis gains tolerance to the antimicrobial peptide DGL13K by synonymous mutations in hagA. PLoS ONE 2024, 19, e0312200. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Chen, J.; Kong, F.; Zhang, Y.; Yi, J.; Li, Y.; Hu, M.; Wang, D. Polypeptide of Inonotus hispidus extracts alleviates periodontitis through suppressing inflammatory bone loss. Int. J. Biol. Macromol. 2025, 287, 138350. [Google Scholar] [CrossRef]
- Han, J.; Meade, J.; Devine, D.; Sadeghpour, A.; Rappolt, M.; Goycoolea, F.M. Chitosan-coated liposomal systems for delivery of antibacterial peptide LL17-32 to Porphyromonas gingivalis. Heliyon 2024, 10, e34554. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Li, Y.; Han, H.; Miller, D.W.; Wang, G. Solution structures of human LL-37 fragments and NMR-based identification of a minimal membrane-targeting antimicrobial and anticancer region. J. Am. Chem. Soc. 2006, 128, 5776–5785. [Google Scholar] [CrossRef]
- Weber, G.; Farris, F.J. Synthesis and spectral properties of a hydrophobic fluorescent probe: 6-propionyl-2-(dimethylamino) naphthalene. Biochemistry 1979, 18, 3075–3078. [Google Scholar] [CrossRef]
- Kaiser, H.J.; Lingwood, D.; Levental, I.; Sampaio, J.L.; Kalvodova, L.; Rajendran, L.; Simons, K. Order of lipid phases in model and plasma membranes. Proc. Natl. Acad. Sci. USA 2009, 106, 16645–16650. [Google Scholar] [CrossRef]
- Curtis, M.A.; Percival, R.S.; Devine, D.; Darveau, R.P.; Coats, S.R.; Rangarajan, M.; Tarelli, E.; Marsh, P.D.; McCormick, B.A. Temperature-Dependent Modulation ofPorphyromonas gingivalisLipid A Structure and Interaction with the Innate Host Defenses. Infect. Immun. 2011, 79, 1187–1193. [Google Scholar] [CrossRef]
- Pathirana, R.D.; O’Brien-Simpson, N.M.; Visvanathan, K.; Hamilton, J.A.; Reynolds, E.C. Flow cytometric analysis of adherence of Porphyromonas gingivalis to oral epithelial cells. Infect. Immun. 2007, 75, 2484–2492. [Google Scholar] [CrossRef]
- Nielsen, J.E.; Bjornestad, V.A.; Pipich, V.; Jenssen, H.; Lund, R. Beyond structural models for the mode of action: How natural antimicrobial peptides affect lipid transport. J. Colloid. Interface Sci. 2021, 582, 793–802. [Google Scholar] [CrossRef]
- Sevcsik, E.; Pabst, G.; Jilek, A.; Lohner, K. How lipids influence the mode of action of membrane-active peptides. Biochim. Biophys. Acta (BBA) 2007, 1768, 2586–2595. [Google Scholar] [CrossRef] [PubMed]
- de Miguel Catalina, A.; Forbrig, E.; Kozuch, J.; Nehls, C.; Paulowski, L.; Gutsmann, T.; Hildebrandt, P.; Mroginski, M.A. The C-Terminal VPRTES Tail of LL-37 Influences the Mode of Attachment to a Lipid Bilayer and Antimicrobial Activity. Biochemistry 2019, 58, 2447–2462. [Google Scholar] [CrossRef]
- Ron-Doitch, S.; Sawodny, B.; Kuhbacher, A.; David, M.M.N.; Samanta, A.; Phopase, J.; Burger-Kentischer, A.; Griffith, M.; Golomb, G.; Rupp, S. Reduced cytotoxicity and enhanced bioactivity of cationic antimicrobial peptides liposomes in cell cultures and 3D epidermis model against HSV. J. Control. Release 2016, 229, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Were, L.M.; Bruce, B.D.; Davidson, P.M.; Weiss, J. Size, stability, and entrapment efficiency of phospholipids nanocapsules containing polypeptide antimicrobials. J. Agric. Food Chem. 2003, 51, 8073–8079. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Seara, H.; Rog, T.; Karttunen, M.; Vattulainen, I. Interplay of Unsaturated Phospholipids and Cholesterol in Membrane Micro-domains. Biophys. J. 2008, 95, 1027–1039. [Google Scholar] [CrossRef]
- Kaddah, S.; Khreich, N.; Kaddah, F.; Charcosset, C.; Greige-Gerges, H. Cholesterol modulates the liposome membrane fluidity and permeability for a hydrophilic molecule. Food Chem. Toxicol. 2018, 113, 40–48. [Google Scholar] [CrossRef]
- Karal, M.A.S.; Mokta, N.A.; Levadny, V.; Belaya, M.; Ahmed, M.; Ahamed, M.K.; Ahammed, S. Effects of cholesterol on the size distribution and bending modulus of lipid vesicles. PLoS ONE 2022, 17, e0263119. [Google Scholar] [CrossRef]
- Henzler-Wildman, K.A.; Martinez, G.V.; Brown, M.F.; Ramamoorthy, A. Perturbation of the hydrophobic core of lipid bilayers by the human antimicrobial peptide LL-37. Biochemistry 2004, 43, 8459–8469. [Google Scholar] [CrossRef]
- Afonin, S.; Grage, S.L.; Ieronimo, M.; Wadhwani, P.; Ulrich, A.S. Temperature-dependent transmembrane insertion of the amphiphilic peptide PGLa in lipid bilayers observed by solid state 19F NMR spectroscopy. J. Am. Chem. Soc. 2008, 130, 16512–16514. [Google Scholar] [CrossRef]
- Burck, J.; Roth, S.; Wadhwani, P.; Afonin, S.; Kanithasen, N.; Strandberg, E.; Ulrich, A.S. Conformation and membrane orientation of amphiphilic helical peptides by oriented circular dichroism. Biophys. J. 2008, 95, 3872–3881. [Google Scholar] [CrossRef]
- Glaser, R.W.; Sachse, C.; Durr, U.H.; Wadhwani, P.; Afonin, S.; Strandberg, E.; Ulrich, A.S. Concentration-dependent realignment of the antimicrobial peptide PGLa in lipid membranes observed by solid-state 19F-NMR. Biophys. J. 2005, 88, 3392–3397. [Google Scholar] [CrossRef]
- Rapaport, D.; Hague, G.R.; Pouny, Y.; Shai, Y. pH-and ionic strength-dependent fusion of phospholipid vesicles induced by pardaxin analogs or by mixtures of charge-reversed peptides. Biochemistry 1993, 32, 3291–3297. [Google Scholar] [CrossRef]
- Tremouilhac, P.; Strandberg, E.; Wadhwani, P.; Ulrich, A.S. Conditions affecting the re-alignment of the antimicrobial peptide PGLa in membranes as monitored by solid state 2H-NMR. Biochim. Biophys. Acta (BBA) 2006, 1758, 1330–1342. [Google Scholar] [CrossRef]
- Mitchell, D.J.; Steinman, L.; Kim, D.T.; Fathman, C.G.; Rothbard, J.B. Polyarginine enters cells more efficiently than other polycationic homopolymers. J. Pept. Res. 2000, 56, 318–325. [Google Scholar] [CrossRef] [PubMed]
- Jobin, M.-L.; Alves, I.D. On the importance of electrostatic interactions between cell penetrating peptides and membranes: A pathway toward tumor cell selectivity? Biochimie 2014, 107, 154–159. [Google Scholar] [CrossRef] [PubMed]
- Shah, D.O. Interaction of uranyl ions with phospholipid and cholesterol monolayers. J. Colloid. Interface Sci. 1969, 29, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Los, D.A.; Murata, N. Membrane fluidity and its roles in the perception of environmental signals. Biochim. Biophys. Acta (BBA) 2004, 1666, 142–157. [Google Scholar] [CrossRef]
- Bompard, J.; Rosso, A.; Brizuela, L.; Mebarek, S.; Blum, L.J.; Trunfio-Sfarghiu, A.-M.; Lollo, G.; Granjon, T.; Girard-Egrot, A.; Maniti, O. Membrane fluidity as a new means to selectively target cancer cells with fusogenic lipid carriers. Langmuir 2020, 36, 5134–5144. [Google Scholar] [CrossRef]
- Hammond, J.; Richards, C.J.; Ko, Y.; Jonker, T.; Åberg, C.; Roos, W.H.; Lira, R.B. Membrane Fusion-Based Drug Delivery Liposomes Transiently Modify the Material Properties of Synthetic and Biological Membranes. Small 2025, 21, 2408039. [Google Scholar] [CrossRef]
- Paramshetti, S.; Angolkar, M.; Talath, S.; Osmani, R.A.M.; Spandana, A.; Al Fatease, A.; Hani, U.; Ramesh, K.; Singh, E. Unravelling the in vivo dynamics of liposomes: Insights into biodistribution and cellular membrane interactions. Life Sci. 2024, 346, 122616. [Google Scholar] [CrossRef] [PubMed]
- Ulrich, A.S.; Sami, M.; Watts, A. Hydration of DOPC bilayers by differential scanning calorimetry. Biochim. Biophys. Acta (BBA)-Biomembr. 1994, 1191, 225–230. [Google Scholar] [CrossRef]
- Arora, A.; Raghuraman, H.; Chattopadhyay, A. Influence of cholesterol and ergosterol on membrane dynamics: A fluorescence approach. Biochem. Biophys. Res. Commun. 2004, 318, 920–926. [Google Scholar] [CrossRef]
- De Vequi-Suplicy, C.C.; Benatti, C.R.; Lamy, M.T. Laurdan in fluid bilayers: Position and structural sensitivity. J. Fluoresc. 2006, 16, 431–439. [Google Scholar] [CrossRef]
- Henderson, J.M.; Iyengar, N.S.; Lam, K.L.H.; Maldonado, E.; Suwatthee, T.; Roy, I.; Waring, A.J.; Lee, K.Y.C. Beyond electrostatics: Antimicrobial peptide selectivity and the influence of cholesterol-mediated fluidity and lipid chain length on protegrin-1 activity. Biochim. Biophys. Acta (BBA)-Biomembr. 2019, 1861, 182977. [Google Scholar] [CrossRef]
- Chakraborty, S.; Doktorova, M.; Molugu, T.R.; Heberle, F.A.; Scott, H.L.; Dzikovski, B.; Nagao, M.; Stingaciu, L.R.; Standaert, R.F.; Barrera, F.N.; et al. How cholesterol stiffens unsaturated lipid membranes. Proc. Natl. Acad. Sci. USA 2020, 117, 21896–21905. [Google Scholar] [CrossRef]
- Veatch, S.L.; Soubias, O.; Keller, S.L.; Gawrisch, K. Critical fluctuations in domain-forming lipid mixtures. Proc. Natl. Acad. Sci. USA 2007, 104, 17650–17655. [Google Scholar] [CrossRef] [PubMed]
- Strömstedt, A.A.; Ringstad, L.; Schmidtchen, A.; Malmsten, M. Interaction between amphiphilic peptides and phospholipid membranes. Curr. Opin. Colloid. Interface Sci. 2010, 15, 467–478. [Google Scholar] [CrossRef]
- Ringstad, L.; Schmidtchen, A.; Malmsten, M. Effect of peptide length on the interaction between consensus peptides and DOPC/DOPA bilayers. Langmuir 2006, 22, 5042–5050. [Google Scholar] [CrossRef] [PubMed]
- Ringstad, L.; Kacprzyk, L.; Schmidtchen, A.; Malmsten, M. Effects of topology, length, and charge on the activity of a kininogen-derived peptide on lipid membranes and bacteria. Biochim. Biophys. Acta (BBA)-Gen. Subj. 2007, 1768, 715–727. [Google Scholar] [CrossRef]
- Ringstad, L.; Andersson Nordahl, E.; Schmidtchen, A.; Malmsten, M. Composition effect on peptide interaction with lipids and bacteria: Variants of C3a peptide CNY21. Biophys. J. 2007, 92, 87–98. [Google Scholar] [CrossRef]
- Lasic, D.D. Liposomes: From Physics to Applications; Elsevier Science Limited: Amsterdam, The Netherlands, 1993. [Google Scholar]
- Laridi, R.; Kheadr, E.E.; Benech, R.O.; Vuillemard, J.C.; Lacroix, C.; Fliss, I. Liposome encapsulated nisin Z: Optimization, stability and release during milk fermentation. Int. Dairy J. 2003, 13, 325–336. [Google Scholar] [CrossRef]
- El-Jastimi, R.; Lafleur, M. Structural characterization of free and membrane-bound nisin by infrared spectroscopy. Biochim. Biophys. Acta (BBA)-Biomembr. 1997, 1324, 151–158. [Google Scholar] [CrossRef]
- El Jastimi, R.; Lafleur, M. Nisin promotes the formation of non-lamellar inverted phases in unsaturated phosphatidylethanolamines. Biochim. Biophys. Acta (BBA)-Biomembr. 1999, 1418, 97–105. [Google Scholar] [CrossRef][Green Version]
- Sennato, S.; Truzzolillo, D.; Bordi, F. Aggregation and stability of polyelectrolyte-decorated liposome complexes in water–salt media. Soft Matter 2012, 8, 9384–9395. [Google Scholar] [CrossRef]
- Sabin, J.; Prieto, G.; Ruso, J.M.; Hidalgo-Alvarez, R.; Sarmiento, F. Size and stability of liposomes: A possible role of hydration and osmotic forces. Eur. Phys. J. Soft Matter Biol. Phys. 2006, 20, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Boge, L.; Umerska, A.; Matougui, N.; Bysell, H.; Ringstad, L.; Davoudi, M.; Eriksson, J.; Edwards, K.; Andersson, M. Cubosomes post-loaded with antimicrobial peptides: Characterization, bactericidal effect and proteolytic stability. Int. J. Pharm. 2017, 526, 400–412. [Google Scholar] [CrossRef]
- Johansson, J.; Gudmundsson, G.H.; Rottenberg, M.E.; Berndt, K.D.; Agerberth, B. Conformation-dependent antibacterial activity of the naturally occurring human peptide LL-37. J. Biol. Chem. 1998, 273, 3718–3724. [Google Scholar] [CrossRef]
- Wang, G.; Li, Y.; Li, X. Correlation of three-dimensional structures with the antibacterial activity of a group of peptides designed based on a nontoxic bacterial membrane anchor. J. Biol. Chem. 2005, 280, 5803–5811. [Google Scholar] [CrossRef]
- Hugo, A.A.; Tymczyszyn, E.E.; Gomez-Zavaglia, A.; Perez, P.F. Effect of human defensins on lactobacilli and liposomes. J. Appl. Microbiol. 2012, 113, 1491–1497. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Gitter, B.; Ruger, R.; Albrecht, V.; Wieland, G.D.; Fahr, A. Wheat germ agglutinin modified liposomes for the photodynamic inactivation of bacteria. Photochem. Photobiol. 2012, 88, 548–556. [Google Scholar] [CrossRef]
- Yang, K.; Gitter, B.; Ruger, R.; Wieland, G.D.; Chen, M.; Liu, X.; Albrecht, V.; Fahr, A. Antimicrobial peptide-modified liposomes for bacteria targeted delivery of temoporfin in photodynamic antimicrobial chemotherapy. Photochem. Photobiol. Sci. 2011, 10, 1593–1601. [Google Scholar] [CrossRef] [PubMed]
- Sugano, M.; Morisaki, H.; Negishi, Y.; Endo-Takahashi, Y.; Kuwata, H.; Miyazaki, T.; Yamamoto, M. Potential effect of cationic liposomes on interactions with oral bacterial cells and biofilms. J. Liposome Res. 2016, 26, 156–162. [Google Scholar] [CrossRef]
- Fotakis, G.; Timbrell, J.A. In vitro cytotoxicity assays: Comparison of LDH, neutral red, MTT and protein assay in hepatoma cell lines following exposure to cadmium chloride. Toxicol. Lett. 2006, 160, 171–177. [Google Scholar] [CrossRef]
- Zhao, J.; Mao, S. Tuning the membrane fluidity of liposomes for desirable in vivo fate with enhanced drug delivery. In Advances in Biomembranes and Lipid Self-Assembly; Elsevier: Amsterdam, The Netherlands, 2021; Volume 34, pp. 67–106. [Google Scholar]
- Rai, A.; Ferrao, R.; Palma, P.; Patricio, T.; Parreira, P.; Anes, E.; Tonda-Turo, C.; Martins, M.C.L.; Alves, N.; Ferreira, L. Antimicrobial peptide-based materials: Opportunities and challenges. J. Mater. Chem. B 2022, 10, 2384–2429. [Google Scholar] [CrossRef]
- Duarte-Mata, D.I.; Salinas-Carmona, M.C. Antimicrobial peptides immune modulation role in intracellular bacterial infection. Front. Immunol. 2023, 14, 1119574. [Google Scholar] [CrossRef] [PubMed]
- Campos, J.V.; Pontes, J.T.C.; Canales, C.S.C.; Roque-Borda, C.A.; Pavan, F.R. Advancing nanotechnology: Targeting biofilm-forming bacteria with antimicrobial peptides. BME Front. 2025, 6, 0104. [Google Scholar] [CrossRef] [PubMed]








| DOPC Liposomes + P. gingivalis | MFI GeoMean (DiD-A) | SL Liposomes + P. gingivalis | MFI GeoMean (DiD-A) |
|---|---|---|---|
| Unstained DOPC Liposome + FITC-P. gingivalis | 110 ± 82 | Unstained SL Liposome + FITC-P. gingivalis | 105 ± 72 |
| 1× DiD-DOPC Liposome + FITC-P. gingivalis | 2609 ± 1003 | 1× DiD-SL Liposome + FITC-P. gingivalis | 767 ± 559 |
| 2× DiD-DOPC Liposome + FITC-P. gingivalis | 7551 ± 3394 | 2× DiD-SL Liposome + FITC-P. gingivalis | 1094 ± 790 |
| 4× DiD-DOPC Liposome + FITC-P. gingivalis | 15,690 ± 9361 | 4× DiD-SL Liposome + FITC-P. gingivalis | 1855 ± 1347 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, J.; Meade, J.L.; Goycoolea, F.M. DOPC Liposomal Formulation of Antimicrobial Peptide LL17-32 with Reduced Cytotoxicity: A Promising Carrier Against Porphyromonas gingivalis. Pharmaceutics 2025, 17, 1424. https://doi.org/10.3390/pharmaceutics17111424
Han J, Meade JL, Goycoolea FM. DOPC Liposomal Formulation of Antimicrobial Peptide LL17-32 with Reduced Cytotoxicity: A Promising Carrier Against Porphyromonas gingivalis. Pharmaceutics. 2025; 17(11):1424. https://doi.org/10.3390/pharmaceutics17111424
Chicago/Turabian StyleHan, Jinyang, Josephine L. Meade, and Francisco M. Goycoolea. 2025. "DOPC Liposomal Formulation of Antimicrobial Peptide LL17-32 with Reduced Cytotoxicity: A Promising Carrier Against Porphyromonas gingivalis" Pharmaceutics 17, no. 11: 1424. https://doi.org/10.3390/pharmaceutics17111424
APA StyleHan, J., Meade, J. L., & Goycoolea, F. M. (2025). DOPC Liposomal Formulation of Antimicrobial Peptide LL17-32 with Reduced Cytotoxicity: A Promising Carrier Against Porphyromonas gingivalis. Pharmaceutics, 17(11), 1424. https://doi.org/10.3390/pharmaceutics17111424








