Inhalable Protein Powder Prepared by Spray-Freeze-Drying Using Hydroxypropyl-β-Cyclodextrin as Excipient
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Design of Experiment by Factorial Design
2.3. Dry Powder Preparation by Spray-Freeze-Drying (SFD)
2.4. Quantification of HPβCD and BSA in Spray-Freeze-Dried Powder
2.5. High-Performance Liquid Chromatography (HPLC) and Size Exclusion Chromatography (SEC)
2.6. Particle Morphology by Scanning Electron Microscopy (SEM)
2.7. Particle Size Distribution Measurement by Laser Diffractometry
2.8. Aerosol Performance by Next-Generation Impactor (NGI)
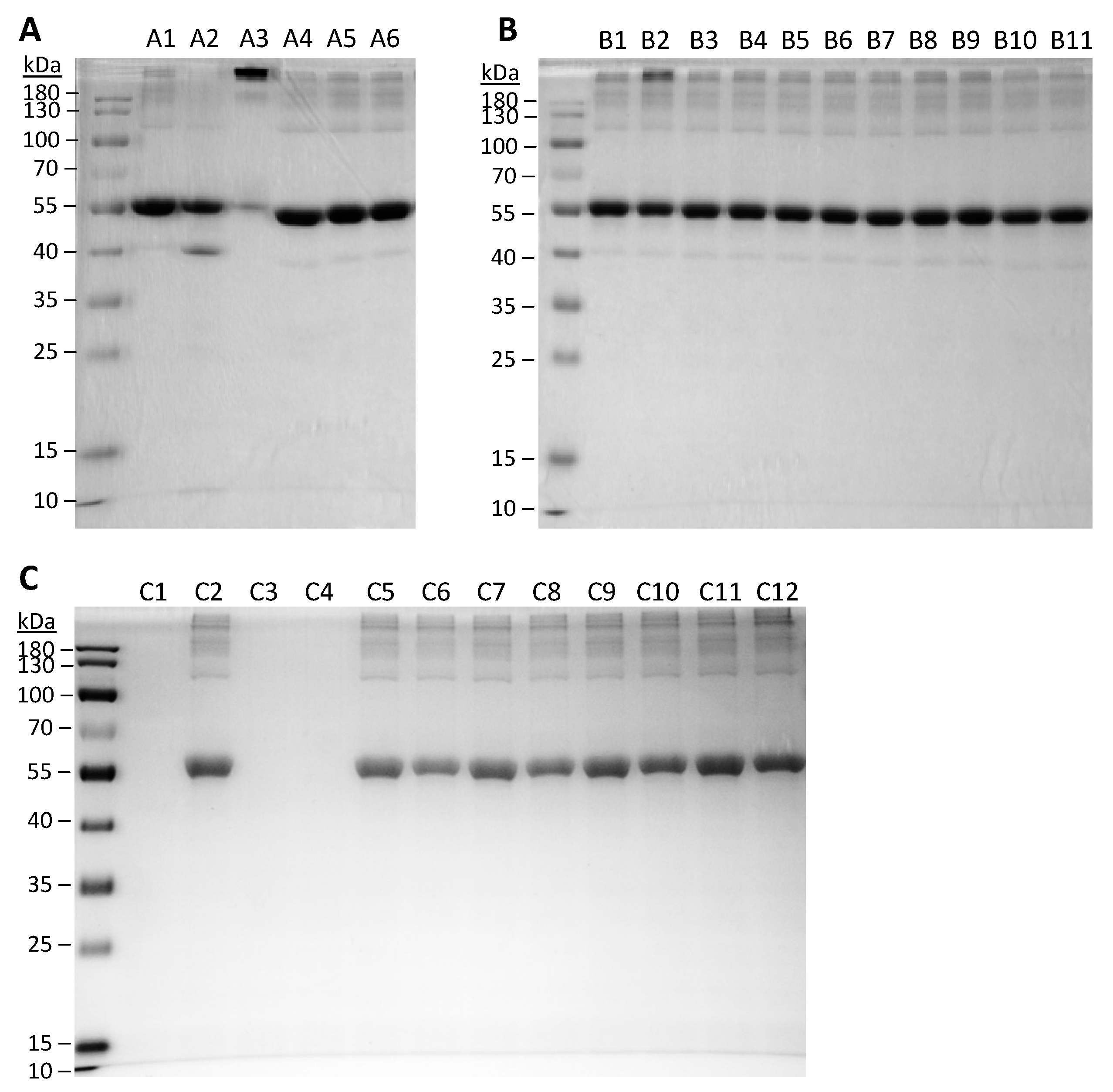
2.9. Protein Integrity by Sodium Dodecyl Sulfate-Polyacrylamide Gel Electrophoresis (SDS-PAGE)
2.10. Statistical Analysis
3. Results
3.1. Production Yield and Composition
3.2. Particle Morphology and Size Distribution
3.3. Aerosol Performance
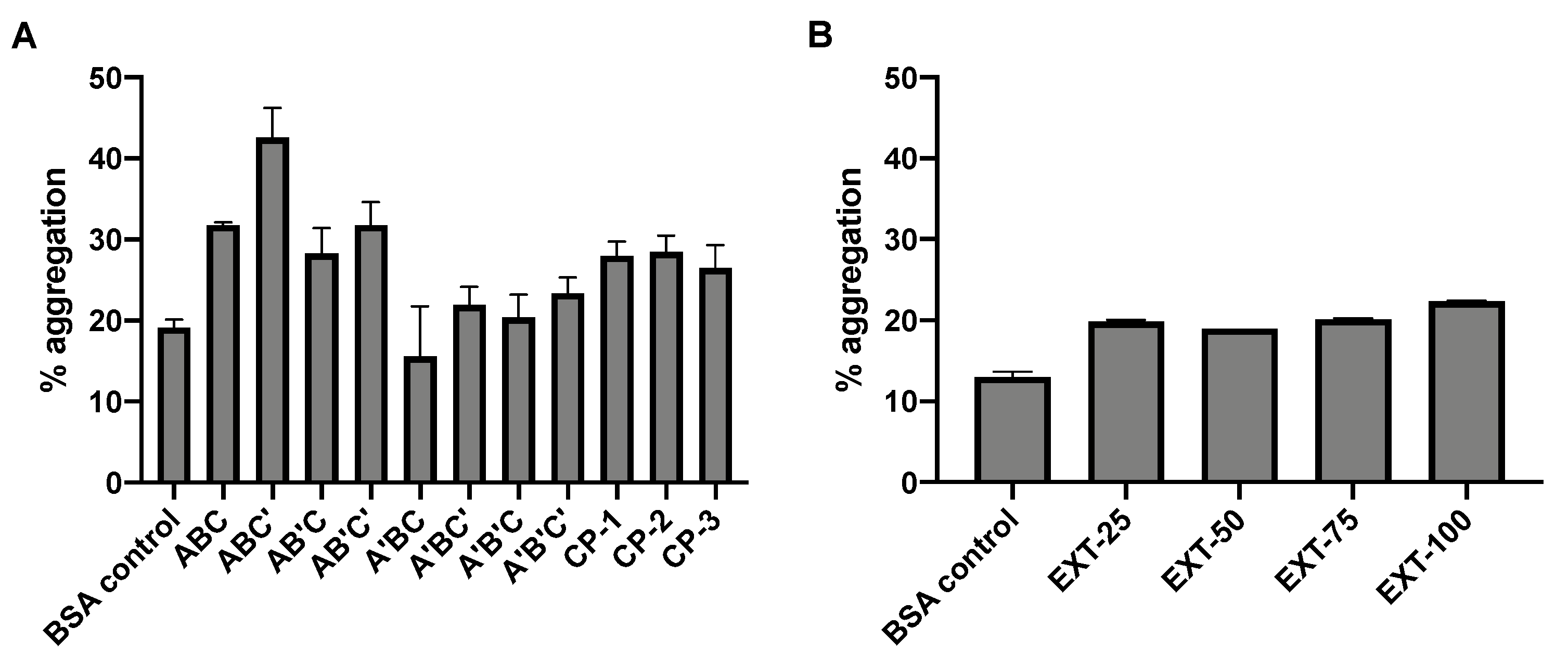
3.4. Protein Integrity and Aggregation
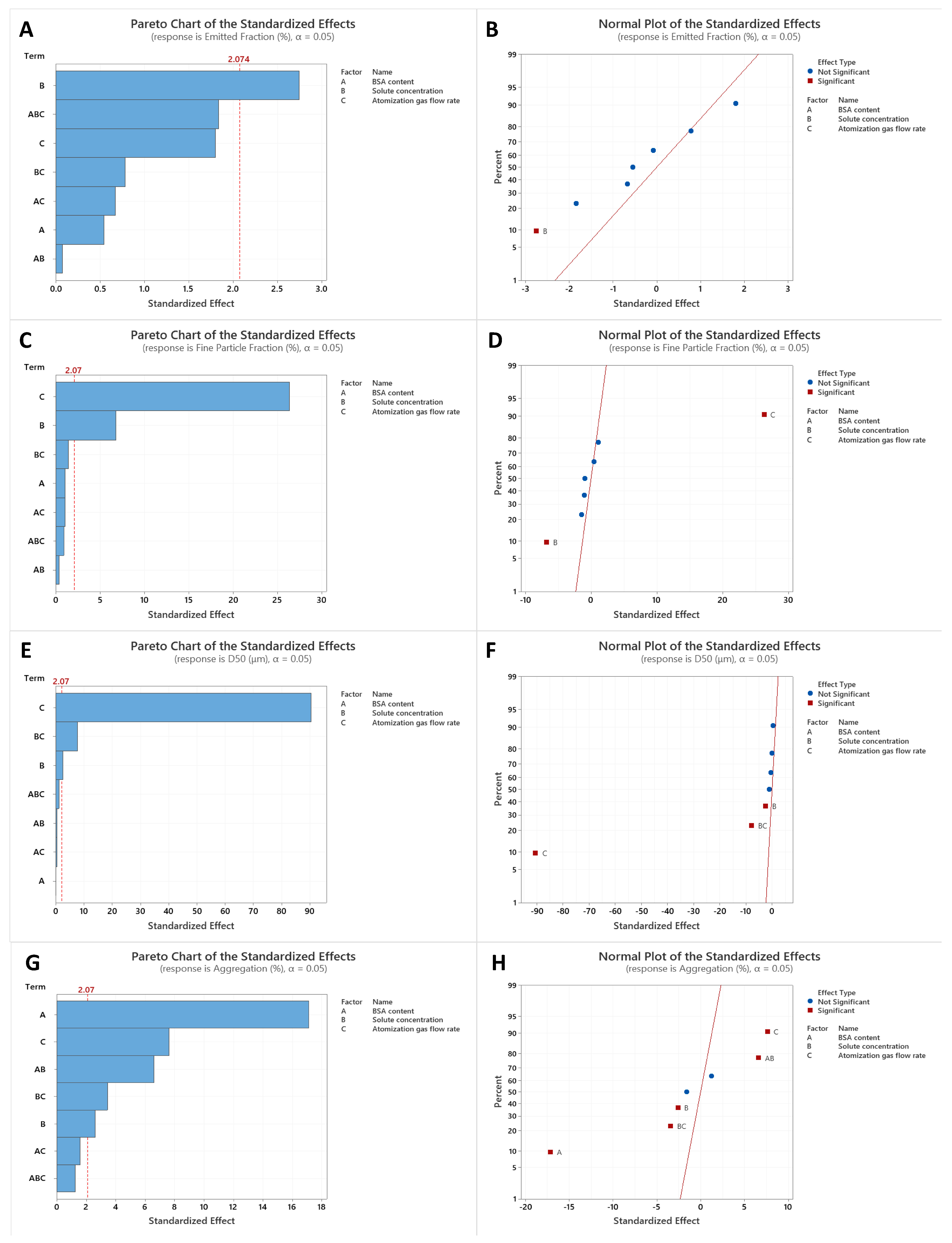
3.5. Factorial Design Analysis
3.6. Extended Formulations—The Effect of BSA Content
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Liang, W.; Pan, H.W.; Vllasaliu, D.; Lam, J.K.W. Pulmonary Delivery of Biological Drugs. Pharmaceutics 2020, 12, 1025. [Google Scholar] [CrossRef]
- Pelaia, C.; Crimi, C.; Vatrella, A.; Tinello, C.; Terracciano, R.; Pelaia, G. Molecular Targets for Biological Therapies of Severe Asthma. Front. Immunol. 2020, 11, 603312. [Google Scholar] [CrossRef] [PubMed]
- Secher, T.; Guilleminault, L.; Reckamp, K.; Amanam, I.; Plantier, L.; Heuze-Vourc’h, N. Therapeutic antibodies: A new era in the treatment of respiratory diseases? Pharmacol. Ther. 2018, 189, 149–172. [Google Scholar] [CrossRef] [PubMed]
- Ziaee, A.; Albadarin, A.B.; Padrela, L.; Femmer, T.; O’Reilly, E.; Walker, G. Spray drying of pharmaceuticals and biopharmaceuticals: Critical parameters and experimental process optimization approaches. Eur. J. Pharm. Sci. 2019, 127, 300–318. [Google Scholar] [CrossRef]
- Adali, M.B.; Barresi, A.A.; Boccardo, G.; Pisano, R. Spray Freeze-Drying as a Solution to Continuous Manufacturing of Pharmaceutical Products in Bulk. Processes 2020, 8, 709. [Google Scholar] [CrossRef]
- Borges Sebastiao, I.; Robinson, T.D.; Alexeenko, A. Atmospheric Spray Freeze-Drying: Numerical Modeling and Comparison With Experimental Measurements. J. Pharm. Sci. 2017, 106, 183–192. [Google Scholar] [CrossRef] [PubMed]
- Liang, W.; Chow, M.Y.T.; Chow, S.F.; Chan, H.-K.; Kwok, P.C.; Lam, J.K. Using two-fluid nozzle for spray freeze drying to produce porous powder formulation of naked siRNA for inhalation. Int. J. Pharm. 2018, 552, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Ly, A.; Carrigy, N.B.; Wang, H.; Harrison, M.; Sauvageau, D.; Martin, A.R.; Vehring, R.; Finlay, W.H. Atmospheric Spray Freeze Drying of Sugar Solution With Phage D29. Front. Microbiol. 2019, 10, 488. [Google Scholar] [CrossRef]
- Emami, F.; Vatanara, A.; Park, E.J.; Na, D.H. Drying Technologies for the Stability and Bioavailability of Biopharmaceuticals. Pharmaceutics 2018, 10, 131. [Google Scholar] [CrossRef]
- Wanning, S.; Suverkrup, R.; Lamprecht, A. Pharmaceutical spray freeze drying. Int. J. Pharm. 2015, 488, 136–153. [Google Scholar] [CrossRef]
- Grasmeijer, N.; Stankovic, M.; de Waard, H.; Frijlink, H.; Hinrichs, W. Unraveling protein stabilization mechanisms: Vitrification and water replacement in a glass transition temperature controlled system. Biochim. Biophys. Acta 2013, 1834, 763–769. [Google Scholar] [CrossRef]
- Mensink, M.A.; Frijlink, H.W.; van der Voort Maarschalk, K.; Hinrichs, W.L. How sugars protect proteins in the solid state and during drying (review): Mechanisms of stabilization in relation to stress conditions. Eur. J. Pharm. Biopharm. 2017, 114, 288–295. [Google Scholar] [CrossRef]
- Depreter, F.; Pilcer, G.; Amighi, K. Inhaled proteins: Challenges and perspectives. Int. J. Pharm. 2013, 447, 251–280. [Google Scholar] [CrossRef]
- Liu, J.; Ding, X.; Fu, Y.; Xiang, C.; Yuan, Y.; Zhang, Y.; Yu, P. Cyclodextrins based delivery systems for macro biomolecules. Eur. J. Med. Chem. 2021, 212, 113105. [Google Scholar] [CrossRef]
- Milani, S.; Faghihi, H.; Roulholamini Najafabadi, A.; Amini, M.; Montazeri, H.; Vatanara, A. Hydroxypropyl beta cyclodextrin: A water-replacement agent or a surfactant upon spray freeze-drying of IgG with enhanced stability and aerosolization. Drug Dev. Ind. Pharm. 2020, 46, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Poursina, N.; Vatanara, A.; Rouini, M.R.; Gilani, K.; Najafabadi, R. The effect of excipients on the stability and aerosol performance of salmon calcitonin dry powder inhalers prepared via the spray freeze drying process. Acta Pharm. 2016, 66, 207–218. [Google Scholar] [CrossRef] [PubMed]
- Pouya, M.A.; Daneshmand, B.; Aghababaie, S.; Faghihi, H.; Vatanara, A. Spray-Freeze Drying: A Suitable Method for Aerosol Delivery of Antibodies in the Presence of Trehalose and Cyclodextrins. AAPS PharmSciTech 2018, 19, 2247–2254. [Google Scholar] [CrossRef]
- Serno, T.; Geidobler, R.; Winter, G. Protein stabilization by cyclodextrins in the liquid and dried state. Adv. Drug Deliv. Rev. 2011, 63, 1086–1106. [Google Scholar] [CrossRef]
- Zhao, Z.; Zhang, X.; Cui, Y.; Huang, Y.; Huang, Z.; Wang, G.; Liang, R.; Pan, X.; Tao, L.; Wu, C. Hydroxypropyl-β-cyclodextrin as anti-hygroscopicity agent in amorphous lactose carriers for dry powder inhalers. Powder Technol. 2019, 358, 29–38. [Google Scholar] [CrossRef]
- Liao, Q.; Lam, I.C.H.; Lin, H.H.S.; Wan, L.T.; Lo, J.C.; Tai, W.; Kwok, P.C.; Lam, J.K. Effect of formulation and inhaler parameters on the dispersion of spray freeze dried voriconazole particles. Int. J. Pharm. 2020, 584, 119444. [Google Scholar] [CrossRef] [PubMed]
- Chow, A.H.; Tong, H.H.; Chattopadhyay, P.; Shekunov, B.Y. Particle engineering for pulmonary drug delivery. Pharm. Res. 2007, 24, 411–437. [Google Scholar] [CrossRef]
- Demoly, P.; Hagedoorn, P.; de Boer, A.H.; Frijlink, H.W. The clinical relevance of dry powder inhaler performance for drug delivery. Respir. Med. 2014, 108, 1195–1203. [Google Scholar] [CrossRef]
- Mahler, H.-C.; Friess, W.; Grauschopf, U.; Kiese, S. Protein aggregation: Pathways, induction factors and analysis. J. Pharm. Sci. 2009, 98, 2909–2934. [Google Scholar] [CrossRef]
- Sebastiao, I.B.; Bhatnagar, B.; Tchessalov, S.; Ohtake, S.; Plitzko, M.; Luy, B.; Alexeenko, A. Bulk Dynamic Spray Freeze-Drying Part 1: Modeling of Droplet Cooling and Phase Change. J. Pharm. Sci. 2019, 108, 2063–2074. [Google Scholar] [CrossRef]
- Liang, W.; Chan, A.Y.L.; Chow, M.Y.T.; Lo, F.F.; Qiu, Y.; Kwok, P.C.; Lam, J.K. Spray freeze drying of small nucleic acids as inhaled powder for pulmonary delivery. Asian J. Pharm. Sci. 2018, 13, 163–172. [Google Scholar] [CrossRef]
- Zhu, C.; Chen, J.; Yu, S.; Que, C.; Taylor, L.S.; Tan, W.; Wu, C.; Zhou, Q.T. Inhalable Nanocomposite Microparticles with Enhanced Dissolution and Superior Aerosol Performance. Mol. Pharm. 2020, 17, 3270–3280. [Google Scholar] [CrossRef] [PubMed]
- Ogienko, A.G.; Bogdanova, E.G.; Trofimov, N.A.; Myz, S.; Kolesov, B.; Yunoshev, A.; Zubikov, N.; Manakov, A.; Boldyrev, V.; Boldyreva, E. Large porous particles for respiratory drug delivery. Glycine-based formulations. Eur. J. Pharm. Sci. 2017, 110, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Bee, J.S.; Stevenson, J.L.; Mehta, B.; Svitel, J.; Pollastrini, J.; Platz, R.; Freund, E.; Carpenter, J.F.; Randolph, T.W. Response of a concentrated monoclonal antibody formulation to high shear. Biotechnol. Bioeng. 2009, 103, 936–943. [Google Scholar] [CrossRef]
- Duerkop, M.; Berger, E.; Dürauer, A.; Jungbauer, A. Impact of Cavitation, High Shear Stress and Air/Liquid Interfaces on Protein Aggregation. Biotechnol. J. 2018, 13, 1800062. [Google Scholar] [CrossRef] [PubMed]
- Ratanji, K.D.; Derrick, J.P.; Dearman, R.J.; Kimber, I. Immunogenicity of therapeutic proteins: Influence of aggregation. J. Immunotoxicol. 2014, 11, 99–109. [Google Scholar] [CrossRef]
- Minton, A.P. Influence of macromolecular crowding upon the stability and state of association of proteins: Predictions and observations. J. Pharm. Sci. 2005, 94, 1668–1675. [Google Scholar] [CrossRef] [PubMed]
- Ohtake, S.; Kita, Y.; Arakawa, T. Interactions of formulation excipients with proteins in solution and in the dried state. Adv. Drug Deliv. Rev. 2011, 63, 1053–1073. [Google Scholar] [CrossRef] [PubMed]
- Sarciaux, J.-M.; Mansour, S.; Hageman, M.J.; Nail, S.L. Effects of buffer composition and processing conditions on aggregation of bovine IgG during freeze-drying. J. Pharm. Sci. 1999, 88, 1354–1361. [Google Scholar] [CrossRef]
- Arsiccio, A.; Pisano, R. The Ice-Water Interface and Protein Stability: A Review. J. Pharm. Sci. 2020, 109, 2116–2130. [Google Scholar] [CrossRef] [PubMed]
- Mutukuri, T.T.; Wilson, N.E.; Taylor, L.S.; Topp, E.M.; Zhou, Q.T. Effects of drying method and excipient on the structure and physical stability of protein solids: Freeze drying vs. spray freeze drying. Int. J. Pharm. 2021, 594, 120169. [Google Scholar] [CrossRef] [PubMed]
- Serno, T.; Carpenter, J.F.; Randolph, T.W.; Winter, G. Inhibition of Agitation-Induced Aggregation of an IgG-Antibody by Hydroxypropyl-β-Cyclodextrin. J. Pharm. Sci. 2010, 99, 1193–1206. [Google Scholar] [CrossRef]

| Factor | Level | ||
|---|---|---|---|
| −1 | 0 | +1 | |
| A—BSA content (% w/w) | 2 | 6 | 10 |
| B—Solute concentration (% w/v) | 2.5 | 5 | 7.5 |
| C—Atomization gas flow rate (L/h) | 301 | 473 | 670 |
| Sample | A—BSA Content (% w/w) | B—Solute Concentration (% w/v) | C—Atomization Gas Flow Rate (L/h) |
|---|---|---|---|
| 23 full factorial design formulations | |||
| ABC | 2 | 2.5 | 301 |
| ABC’ | 2 | 2.5 | 670 |
| AB’C | 2 | 7.5 | 301 |
| AB’C’ | 2 | 7.5 | 670 |
| A’BC | 10 | 2.5 | 301 |
| A’BC’ | 10 | 2.5 | 670 |
| A’B’C | 10 | 7.5 | 301 |
| A’B’C’ | 10 | 7.5 | 670 |
| CP-1 | 6 | 5 | 473 |
| CP-2 | 6 | 5 | 473 |
| CP-3 | 6 | 5 | 473 |
| Extended formulations | |||
| EXT-0 | 0 | 5 | 473 |
| EXT-25 | 25 | 5 | 473 |
| EXT-50 | 50 | 5 | 473 |
| EXT-75 | 75 | 5 | 473 |
| EXT-100 | 100 | 5 | 473 |
| Sample | Production Yield (%) | BSA Content (%) | HPβCD Content (%) |
|---|---|---|---|
| ABC | 84.4 | N.A. | 102.7 ± 5.3 |
| ABC’ | 77.3 | N.A. | 96.9 ± 2.0 |
| AB’C | 75.3 | N.A. | 100.0 ± 1.3 |
| AB’C’ | 72.8 | N.A. | 95.7 ± 4.4 |
| A’BC | 78.5 | N.A. | 88.6 ± 4.5 |
| A’BC’ | 72.6 | N.A. | 86.4 ± 5.2 |
| A’B’C | 76.0 | N.A. | 93.4 ± 1.8 |
| A’B’C’ | 71.5 | N.A. | 91.6 ± 3.6 |
| CP-1 | 71.0 | N.A. | 97.4 ± 2.3 |
| CP-2 | 71.8 | N.A. | 93.5 ± 7.4 |
| CP-3 | 73.0 | N.A. | 96.3 ± 5.7 |
| EXT-0 | 91.1 | 0 | 94.8 ± 2.4 |
| EXT-25 | 93.8 | 27.4 ± 0.3 | 71.3 ± 0.2 |
| EXT-50 | 97.4 | 52.8 ± 0.2 | 46.8 ± 2.0 |
| EXT-75 | 97.6 | 76.0 ± 0.7 | 23.0 ± 0.1 |
| EXT-100 | 88.6 | 96.0 ± 0.5 | 0 |
| Formulation | Volumetric Diameter | EF (%) | FPF (%) | MMAD (µm) | |||
|---|---|---|---|---|---|---|---|
| D10 (µm) | D50 (µm) | D90 (µm) | Span | ||||
| ABC | 11.2 ± 0.2 | 30.2 ± 0.9 | 58.6 ± 1.6 | 1.6 ± 0.0 | 96.0 ± 0.78 | 23.0 ± 2.6 | 9.0 ± 1.8 |
| ABC’ | 4.1 ± 0.0 | 9.3 ± 0.2 | 20.2 ± 1.3 | 1.7 ± 0.1 | 96.0 ± 1.2 | 78.4 ± 1.1 | 1.0 ± 0.2 |
| AB’C | 6.7 ± 0.3 | 31.4 ± 1.1 | 60.0 ± 2.1 | 1.70 ± 0.0 | 93.4 ± 1.1 | 9.8 ± 2.5 | 13.6 ± 1.9 |
| AB’C’ | 2.8 ± 0.1 | 7.1 ± 0.2 | 17.4 ± 0.7 | 2.1 ± 0.0 | 95.9 ± 1.4 | 63.1 ± 3.6 | 1.9 ± 0.3 |
| A’BC | 10.8 ± 0.2 | 29.9 ± 1.0 | 57.9 ± 2.0 | 1.6 ± 0.0 | 95.2 ± 1.1 | 24.6 ± 4.7 | 8.2 ± 4.2 |
| A’BC’ | 4.3 ± 0.1 | 9.8 ± 0.1 | 21.3 ± 0.4 | 1.8 ± 0.0 | 96.3 ± 0.2 | 79.4 ± 1.0 | 1.0 ± 0.1 |
| A’B’C | 6.5 ± 0.2 | 31.4 ± 1.1 | 59.1 ± 1.4 | 1.7 ± 0.0 | 94.3 ± 2.5 | 16.7 ± 4.1 | 6.8 ± 1.2 |
| A’B’C’ | 2.8 ± 0.1 | 6.8 ± 0.1 | 15.9 ± 0.9 | 1.9 ± 0.1 | 94.4 ± 1.5 | 62.1 ± 3.6 | 1.9 ± 0.3 |
| CP-1 | 4.3 ± 0.1 | 10.8 ± 0.3 | 24.7 ± 1.0 | 1.9 ± 0.0 | 96.5 ± 0.5 | 60.4 ± 6.4 | 2.1 ± 0.5 |
| CP-2 | 4.3 ± 0.1 | 10.8 ± 0.2 | 24.8 ± 0.3 | 1.9 ± 0.0 | 95.4 ± 0.9 | 59.8 ± 8.1 | 2.0 ± 0.8 |
| CP-3 | 4.3 ± 0.0 | 10.6 ± 0.1 | 24.4 ± 0.3 | 1.9 ± 0.0 | 95.7 ± 1.1 | 61.1 ± 8.7 | 1.5 ± 0.2 |
| EXT-0 | 3.9 ± 0.1 | 9.9 ± 0.3 | 24.4 ± 0.9 | 2.1 ± 0.0 | 98.5 ± 0.6 | 60.5 ± 2.7 | 1.8 ± 0.2 |
| EXT-25 | 4.0 ± 0.0 | 10.4 ± 0.2 | 25.5 ± 0.5 | 2.1 ± 0.0 | 100 * | 65.5 ± 1.7 | 1.4 ± 0.0 |
| EXT-50 | 4.1 ± 0.0 | 11.1 ± 0.3 | 27.2 ± 1.2 | 2.1 ± 0.1 | 100 * | 63.6 ± 1.8 | 1.4 ± 0.1 |
| EXT-75 | 3.9 ± 0.1 | 10.8 ± 0.7 | 27.2 ± 1.9 | 2.2 ± 0.0 | 98.3 ± 0.7 | 54.7 ± 3.3 | 2.1 ± 0.3 |
| EXT-100 | 3.8 ± 0.0 | 11.7 ± 0.5 | 31.1 ± 2.0 | 2.3 ± 0.1 | 98.4 ± 0.4 | 52.3 ± 4.7 | 2.5 ± 0.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lo, J.C.K.; Pan, H.W.; Lam, J.K.W. Inhalable Protein Powder Prepared by Spray-Freeze-Drying Using Hydroxypropyl-β-Cyclodextrin as Excipient. Pharmaceutics 2021, 13, 615. https://doi.org/10.3390/pharmaceutics13050615
Lo JCK, Pan HW, Lam JKW. Inhalable Protein Powder Prepared by Spray-Freeze-Drying Using Hydroxypropyl-β-Cyclodextrin as Excipient. Pharmaceutics. 2021; 13(5):615. https://doi.org/10.3390/pharmaceutics13050615
Chicago/Turabian StyleLo, Jason C. K., Harry W. Pan, and Jenny K. W. Lam. 2021. "Inhalable Protein Powder Prepared by Spray-Freeze-Drying Using Hydroxypropyl-β-Cyclodextrin as Excipient" Pharmaceutics 13, no. 5: 615. https://doi.org/10.3390/pharmaceutics13050615
APA StyleLo, J. C. K., Pan, H. W., & Lam, J. K. W. (2021). Inhalable Protein Powder Prepared by Spray-Freeze-Drying Using Hydroxypropyl-β-Cyclodextrin as Excipient. Pharmaceutics, 13(5), 615. https://doi.org/10.3390/pharmaceutics13050615







