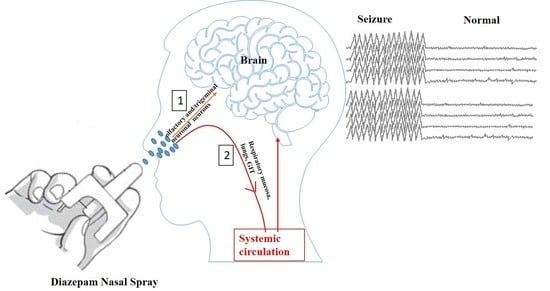
A Short Review on the Intranasal Delivery of Diazepam for Treating Acute Repetitive Seizures
Abstract
1. Introduction
2. Treatment Options for ARS
3. Diazepam for Intranasal Administration
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cereghino, J.J. Identification and treatment of acute repetitive seizures in children and adults. Curr. Treat. Options Neurol. 2007, 9, 249–255. [Google Scholar] [CrossRef]
- Cereghino, J.J.; Cloyd, J.C.; Kuzniecky, R.I.; The North American Diastat Study Group. Rectal diazepam gel for treatment of acute repetitive seizures in adults. Arch. Neurol. 2002, 59, 1915–1920. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Buelow, J.M.; Shafer, P.; Shinnar, R.; Austin, J.; Dewar, S.; Long, L.; O’Hara, K.; Santilli, N. Perspectives on seizure clusters: Gaps in lexicon, awareness, and treatment. Epilepsy Behav. 2016, 57, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Yu, W.; Lü, Y. The causes of new-onset epilepsy and seizures in the elderly. Neuropsychiatr. Dis. Treat. 2016, 12, 1425. [Google Scholar] [CrossRef] [PubMed]
- Jafarpour, S.; Hirsch, L.J.; Gaínza-Lein, M.; Kellinghaus, C.; Detyniecki, K. Seizure cluster: Definition, prevalence, consequences, and management. Seizure 2019, 68, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Zack, M.M.; Kobau, R. National and State Estimates of the Numbers of Adults and Children with Active Epilepsy—United States, 2015. Mmwr. Morb. Mortal. Wkly. Rep. 2017, 66, 821–825. [Google Scholar] [CrossRef]
- World Health Organization. Epilepsy. Available online: https://www.who.int/health-topics/epilepsy#tab=tab_1 (accessed on 27 November 2020).
- Haut, S.R. Seizure clustering. Epilepsy Behav. 2006, 8, 50–55. [Google Scholar] [CrossRef]
- Komaragiri, A.; Detyniecki, K.; Hirsch, L.J. Seizure clusters: A common, understudied and undertreated phenomenon in refractory epilepsy. Epilepsy Behav. 2016, 59, 83–86. [Google Scholar] [CrossRef]
- Chen, B.; Choi, H.; Hirsch, L.J.; Katz, A.; Legge, A.; Wong, R.A.; Jiang, A.; Kato, K.; Buchsbaum, R.; Detyniecki, K. Prevalence and risk factors of seizure clusters in adult patients with epilepsy. Epilepsy Res. 2017, 133, 98–102. [Google Scholar] [CrossRef]
- McKee, H.R.; Abou-Khalil, B. Outpatient pharmacotherapy and modes of administration for acute repetitive and prolonged seizures. CNS Drugs 2015, 29, 55–70. [Google Scholar] [CrossRef]
- Spencer, D. Hope for new treatments for acute repetitive seizures. Epilepsy Curr. 2014, 14, 147–149. [Google Scholar] [CrossRef] [PubMed]
- Ciccone, O.; Mathews, M.; Birbeck, G.L. Management of acute seizures in children: A review with special consideration of care in resource-limited settings. Afr. J. Emerg. Med. 2017, 7, S3–S9. [Google Scholar] [CrossRef] [PubMed]
- Greenfield, L.J., Jr. Molecular mechanisms of antiseizure drug activity at GABAA receptors. Seizure 2013, 22, 589–600. [Google Scholar] [CrossRef] [PubMed]
- Thompson, P.J.; Duncan, J.S. Cognitive decline in severe intractable epilepsy. Epilepsia 2005, 46, 1780–1787. [Google Scholar] [CrossRef] [PubMed]
- Kapur, J.; Macdonald, R.L. Rapid Seizure-Induced Reduction of Benzodiazepine and Zn2+ Sensitivity of Hippocampal Dentate Granule Cell GABAA Receptors. J. Neurosci. 1997, 17, 7532–7540. [Google Scholar] [CrossRef] [PubMed]
- Greenblatt, D.; Arendt, R.; Abernethy, D.R.; Giles, H.; Sellers, E.; Shader, R. In vitro quantitation of benzodiazepine lipophilicity: Relation to in vivo distribution. BJA Br. J. Anaesth. 1983, 55, 985–989. [Google Scholar] [CrossRef]
- Adeyemo, M.A.; Idowu, S.O. Correlation of lipophilicity descriptors with pharmacokinetic parameters of selected benzodiazepines. Afr. J. Biomed. Res. 2016, 19, 213–218. [Google Scholar]
- Agarwal, S.K.; Cloyd, J.C. Development of benzodiazepines for out-of-hospital management of seizure emergencies. Neurol. Clin. Pr. 2015, 5, 80–85. [Google Scholar] [CrossRef]
- Manno, E.M. Status epilepticus: Current treatment strategies. Neurohospitalist 2011, 1, 23–31. [Google Scholar] [CrossRef][Green Version]
- Rogawski, M.A.; Heller, A.H. Diazepam buccal film for the treatment of acute seizures. Epilepsy Behav. 2019, 101, 106537. [Google Scholar] [CrossRef]
- Ivaturi, V.D. Intranasal and Rectal Diazepam for Rescue Therapy: Assessment of Pharmacokinetics and Tolerability; Retrieved from the University of Minnesota Digital Conservancy: Twin Cities, MN, USA, 2020; Available online: http://hdl.handle.net/11299/100044 (accessed on 29 November 2020).
- Rey, E.; Tréluyer, J.-M.; Pons, G. Pharmacokinetic optimisation of benzodiazepine therapy for acute seizures. Clin. Pharmacokinet. 1999, 36, 409–424. [Google Scholar] [CrossRef] [PubMed]
- Maglalang, P.D.; Rautiola, D.; Siegel, R.A.; Fine, J.M.; Hanson, L.R.; Coles, L.D.; Cloyd, J.C. Rescue therapies for seizure emergencies: New modes of administration. Epilepsia 2018, 59, 207–215. [Google Scholar] [CrossRef]
- Detyniecki, K.; Van Ess, P.J.; Sequeira, D.J.; Wheless, J.W.; Meng, T.-C.; Pullman, W.E. Safety and efficacy of midazolam nasal spray in the outpatient treatment of patients with seizure clusters—A randomized, double-blind, placebo-controlled trial. Epilepsia 2019, 60, 1797–1808. [Google Scholar] [CrossRef]
- Anderson, M. Buccal midazolam for pediatric convulsive seizures: Efficacy, safety, and patient acceptability. Patient Prefer. Adherence 2013, 7, 27. [Google Scholar] [CrossRef]
- Sarma, A.K.; Khandker, N.; Kurczewski, L.; Brophy, G.M. Medical management of epileptic seizures: Challenges and solutions. Neuropsychiatr. Dis. Treat. 2016, 12, 467. [Google Scholar]
- Malu, C.K.K.; Kahamba, D.M.; Walker, T.D.; Mukampunga, C.; Musalu, E.M.; Kokolomani, J.; Mayamba, R.M.K.; Wilmshurst, J.M.; Dubru, J.-M.; Misson, J.-P. Efficacy of sublingual lorazepam versus intrarectal diazepam for prolonged convulsions in Sub-Saharan Africa. J. Child Neurol. 2014, 29, 895–902. [Google Scholar] [CrossRef] [PubMed]
- McIntyre, J.; Robertson, S.; Norris, E.; Appleton, R.; Whitehouse, W.P.; Phillips, B.; Martland, T.; Berry, K.; Collier, J.; Smith, S. Safety and efficacy of buccal midazolam versus rectal diazepam for emergency treatment of seizures in children: A randomised controlled trial. Lancet 2005, 366, 205–210. [Google Scholar] [CrossRef]
- Von Blomberg, A.; Kay, L.; Knake, S.; Fuest, S.; Zöllner, J.P.; Reif, P.S.; Herrmann, E.; Balaban, Ü.; Schubert-Bast, S.; Rosenow, F.; et al. Efficacy, Tolerability, and Safety of Concentrated Intranasal Midazolam Spray as Emergency Medication in Epilepsy Patients During Video-EEG Monitoring. CNS Drugs 2020, 34, 545–553. [Google Scholar] [CrossRef] [PubMed]
- Dhaliwal, J.S.; Saadabadi, A. Diazepam. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2019. [Google Scholar]
- Agarwal, S.K.; Kriel, R.L.; Brundage, R.C.; Ivaturi, V.D.; Cloyd, J.C. A pilot study assessing the bioavailability and pharmacokinetics of diazepam after intranasal and intravenous administration in healthy volunteers. Epilepsy Res. 2013, 105, 362–367. [Google Scholar] [CrossRef] [PubMed]
- Hogan, R.E.; Tarquinio, D.; Sperling, M.R.; Klein, P.; Miller, I.; Segal, E.B.; Rabinowicz, A.L.; Carrazana, E. Pharmacokinetics and safety of VALTOCO (NRL-1; diazepam nasal spray) in patients with epilepsy during seizure (ictal/peri-ictal) and nonseizure (interictal) conditions: A phase 1, open-label study. Epilepsia 2020, 61, 935–943. [Google Scholar] [CrossRef]
- Grower, M.F.; Russell, E.A., Jr.; Getter, L. Solubility of injectable valium in intravenous solutions. Anesth. Prog. 1978, 25, 158. [Google Scholar] [PubMed]
- Wermeling, D.P.H.; Miller, J.L.; Archer, S.M.; Manaligod, J.M.; Rudy, A.C. Bioavailability and pharmacokinetics of lorazepam after intranasal, intravenous, and intramuscular administration. J. Clin. Pharmacol. 2001, 41, 1225–1231. [Google Scholar] [CrossRef] [PubMed]
- Vyas, T.K.; Shahiwala, A.; Marathe, S.; Misra, A. Intranasal drug delivery for brain targeting. Curr. Drug Deliv. 2005, 2, 165–175. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, M.; Cloyd, J.C.; Siegel, R.A. A review of intranasal formulations for the treatment of seizure emergencies. J. Control. Release 2016, 237, 147–159. [Google Scholar] [CrossRef]
- Tanimoto, S.; Pesco Koplowitz, L.; Lowenthal, R.E.; Koplowitz, B.; Rabinowicz, A.L.; Carrazana, E. Evaluation of Pharmacokinetics and Dose Proportionality of Diazepam After Intranasal Administration of NRL-1 to Healthy Volunteers. Clin. Pharmacol. Drug Dev. 2020, 9, 719–727. [Google Scholar] [CrossRef]
- Maggio, E.T.; Pillion, D.J. High efficiency intranasal drug delivery using Intravail® alkylsaccharide absorption enhancers. Drug Deliv. Transl. Res. 2013, 3, 16–25. [Google Scholar] [CrossRef]
- Ali, J.; Ali, M.; Baboota, S.; Kaur Sahni, J.; Ramassamy, C.; Dao, L. Potential of nanoparticulate drug delivery systems by intranasal administration. Curr. Pharm. Des. 2010, 16, 1644–1653. [Google Scholar] [CrossRef]
- Arora, P.; Sharma, S.; Garg, S. Permeability issues in nasal drug delivery. Drug Discov. Today 2002, 7, 967–975. [Google Scholar] [CrossRef]
- Ugwoke, M.I.; Verbeke, N.; Kinget, R. The biopharmaceutical aspects of nasal mucoadhesive drug delivery. J. Pharm. Pharmacol. 2001, 53, 3–22. [Google Scholar] [CrossRef]
- Choi, H.-G.; Jung, J.-H.; Ryu, J.-M.; Yoon, S.-J.; Oh, Y.-K.; Kim, C.-K. Development of in situ-gelling and mucoadhesive acetaminophen liquid suppository. Int. J. Pharm. 1998, 165, 33–44. [Google Scholar] [CrossRef]
- Kaur, P.; Kim, K. Pharmacokinetics and brain uptake of diazepam after intravenous and intranasal administration in rats and rabbits. Int. J. Pharm. 2008, 364, 27–35. [Google Scholar] [CrossRef]
- Ivaturi, V.D.; Riss, J.R.; Kriel, R.L.; Cloyd, J.C. Pharmacokinetics and tolerability of intranasal diazepam and midazolam in healthy adult volunteers. Acta Neurol. Scand. 2009, 120, 353–357. [Google Scholar] [CrossRef] [PubMed]
- Schrier, L.; Zuiker, R.; Merkus, F.W.H.M.; Klaassen, E.S.; Guan, Z.; Tuk, B.; van Gerven, J.M.A.; van der Geest, R.; Groeneveld, G.J. Pharmacokinetics and pharmacodynamics of a new highly concentrated intranasal midazolam formulation for conscious sedation. Br. J. Clin. Pharmacol. 2017, 83, 721–731. [Google Scholar] [CrossRef] [PubMed]
- French, J.A. Benzo versus benzo: And the winner is…. Epilepsy Curr. 2011, 11, 143–144. [Google Scholar] [CrossRef] [PubMed]
- Lissauer, S.; Kenny, J.; Jefferis, O.; Wingfield, T.; Miller, A.; Chagaluka, G.; Kalilani-Phiri, L.; Molyneux, E. Buccal, intranasal or intravenous lorazepam for the treatment of acute convulsions in children in Malawi: An open randomized trial: Le lorazépam par voie orale, intranasale ou intraveineuse pour le traitement des convulsions aiguës chez l’enfant au Malawi: Étude ouverte randomisée. Afr. J. Emerg. Med. 2015, 5, 120–126. [Google Scholar]
- Gizurarson, S.; Gudbrandsson, F.K.; Jonsson, H.; Bechgaard, E. Intranasal administration of diazepam aiming at the treatment of acute seizures: Clinical trials in healthy volunteers. Biol. Pharm. Bull. 1999, 22, 425–427. [Google Scholar] [CrossRef][Green Version]
- Lindhardt, K.; Gizurarson, S.; Stefánsson, S.B.; Òlafsson, D.R.; Bechgaard, E. Electroencephalographic effects and serum concentrations after intranasal and intravenous administration of diazepam to healthy volunteers. Br. J. Clin. Pharm. 2001, 52, 521–527. [Google Scholar] [CrossRef]
- Ivaturi, V.D.; Riss, J.R.; Kriel, R.L.; Siegel, R.A.; Cloyd, J.C. Bioavailability and tolerability of intranasal diazepam in healthy adult volunteers. Epilepsy Res. 2009, 84, 120–126. [Google Scholar] [CrossRef]
- Lau, S.W.J.; Slattery, J.T. Absorption of diazepam and lorazepam following intranasal administration. Int. J. Pharm. 1989, 54, 171–174. [Google Scholar] [CrossRef]
- Bechgaard, E.; Gizurarson, S.; Hjortkjær, R.K. Pharmacokinetic and Pharmacodynamic Response after Intranasal Administration of Diazepam to Rabbits. J. Pharm. Pharmacol. 1997, 49, 747–750. [Google Scholar] [CrossRef]
- Platt, S.R.; Randell, S.C.; Scott, K.C.; Chrisman, C.L.; Hill, R.C.; Gronwall, R.R. Comparison of plasma benzodiazepine concentrations following intranasal and intravenous administration of diazepam to dogs. Am. J. Vet. Res. 2000, 61, 651–654. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Nandi, I.; Kim, K.H. Development of an ethyl laurate-based microemulsion for rapid-onset intranasal delivery of diazepam. Int. J. Pharm. 2002, 237, 77–85. [Google Scholar] [CrossRef]
- Lindhardt, K.; Ólafsson, D.R.; Gizurarson, S.; Bechgaard, E. Intranasal bioavailability of diazepam in sheep correlated to rabbit and man. Int. J. Pharm. 2002, 231, 67–72. [Google Scholar] [CrossRef]
- Hou, H. Development of Novel Intranasal Diazepam Formulations for the Treatment of Seizure Emergencies; University of Minnesota: Minneapolis, MN, USA, 2006. [Google Scholar]
- Jiao, C.-M.; Liu, H.-Z.; Jing, L.-Y.; Zhang, Y.; Li, S.-M. Preparation and evaluation of diazepam microemulsion for intranasal delivery. Chin. J. Pharm. 2008, 4, 1–284. [Google Scholar]
- Botner, S.; Sintov, A.C. Intranasal Delivery of Two Benzodiazepines, Midazolam and Diazepam, by a Microemulsion System. Pharmacol. Pharm. 2011, 9. [Google Scholar] [CrossRef]
- Musulin, S.E.; Mariani, C.L.; Papich, M.G. Diazepam pharmacokinetics after nasal drop and atomized nasal administration in dogs. J. Vet. Pharmacol. Ther. 2011, 34, 17–24. [Google Scholar] [CrossRef]
- Sheng, P.; Zhang, R.-T.; Wang, H.; Nie, H.; Fu, X.-H. The effect of l-menthol on the intranasal absorption to diazepam. Chin. J. Hosp. Pharm. 2012, 24, 172–182. [Google Scholar]
- Sheng, P.; Zhang, R.-T.; Wang, H.; Huang, Z.; Fu, X.-H. Intranasal Absorption Kinetics of Diazepam and Promoting Effect of Menthol on The Absorption of Diazepam. Eval. Anal. Drug Use Hosp. China 2013, 19, 34–40. [Google Scholar]
- Ivaturi, V.; Kriel, R.; Brundage, R.; Loewen, G.; Mansbach, H.; Cloyd, J. Bioavailability of Intranasal vs. Rectal Diazepam. Epilepsy Res. 2013, 103, 254–261. [Google Scholar] [CrossRef]
- Bream, G.; Leibowitz, M.; Wargin, B.; Abernathy, K.; Henney, H.; Ward, D. Assessment of Pharmacokinetic Linearity and Relative Bioavailability of an Intranasal Diazepam Formulation Compared with Diazepam Rectal Gel in Healthy Adult Subjects (P02. 214); AAN Enterprises: St. Paul, MN, USA, 2013. [Google Scholar]
- Sperling, M.R.; Haas, K.F.; Krauss, G.; Seif Eddeine, H.; Henney, H.R.; Rabinowicz, A.L.; Bream, G.; Squillacote, D.; Carrazana, E.J. Dosing feasibility and tolerability of intranasal diazepam in adults with epilepsy. Epilepsia 2014, 55, 1544–1550. [Google Scholar] [CrossRef]
- Henney, H.R.; Sperling, M.R.; Rabinowicz, A.L.; Bream, G.; Carrazana, E.J. Assessment of pharmacokinetics and tolerability of intranasal diazepam relative to rectal gel in healthy adults. Epilepsy Res. 2014, 108, 1204–1211. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, M.; Winter, T.; Lis, L.; Georg, G.I.; Siegel, R.A. Rapid Delivery of Diazepam from Supersaturated Solutions Prepared Using Prodrug/Enzyme Mixtures: Toward Intranasal Treatment of Seizure Emergencies. AAPS J. 2014, 16, 577–585. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Inokuchi, R.; Ohashi-Fukuda, N.; Nakamura, K.; Wada, T.; Gunshin, M.; Kitsuta, Y.; Nakajima, S.; Yahagi, N. Comparison of intranasal and intravenous diazepam on status epilepticus in stroke patients: A retrospective cohort study. Med. Baltim. 2015, 94, e555. [Google Scholar] [CrossRef] [PubMed]
- Sharma, D.; Sharma, R.K.; Sharma, N.; Gabrani, R.; Sharma, S.K.; Ali, J.; Dang, S. Nose-To-Brain Delivery of PLGA-Diazepam Nanoparticles. AAPS Pharmscitech 2015, 16, 1108–1121. [Google Scholar] [CrossRef] [PubMed]
- Ramreddy, S.; Janapareddi, K. Brain targeting of chitosan-based diazepam mucoadhesive microemulsions via nasal route: Formulation optimization, characterization, pharmacokinetic and pharmacodynamic evaluation. Drug Dev. Ind. Pharm. 2019, 45, 147–158. [Google Scholar] [CrossRef]
- Rautiola, D.; Maglalang, P.D.; Cheryala, N.; Nelson, K.M.; Georg, G.I.; Fine, J.M.; Svitak, A.L.; Faltesek, K.A.; Hanson, L.R.; Mishra, U.; et al. Intranasal Coadministration of a Diazepam Prodrug with a Converting Enzyme Results in Rapid Absorption of Diazepam in Rats. J. Pharmacol. Exp. Ther. 2019, 370, 796–805. [Google Scholar] [CrossRef]
- Hogan, R.; Sperling, M.; Klein, P.; Segal, E.; Carrazana, E. Pharmacokinetic Study of Valtoco™ (NRL-1; Diazepam Nasal Spray) in Patients with Epilepsy under Ictal and Inter-Ictal Conditions—Interim Report (P3.5-009); Neurology: Minneapolis, MN, USA, 2019; Volume 92, 46p. [Google Scholar]
- Hogan, R.E.; Gidal, B.E.; Koplowitz, B.; Koplowitz, L.P.; Lowenthal, R.E.; Carrazana, E. Bioavailability and safety of diazepam intranasal solution compared to oral and rectal diazepam in healthy volunteers. Epilepsia 2020, 61, 455–464. [Google Scholar] [CrossRef]
- Tarquinio, D.; Hogan, R.E.; Sperling, M.R.; Wheless, J.W.; Dlugos, D.; Miller, I.; Rabinowicz, A.L.; Carrazana, E. Safety and Tolerability of NRL-1, an Intranasal Formulation of Diazepam, in Subjects with Epilepsy in a Phase 1, Open-Label Study: Focus on Adverse Events Relevant to Clinicians and Patients (2044). Neurology 2020, 94, 2044. [Google Scholar]
- Miller, I.; Wheless, J.W.; Hogan, R.E.; Dlugos, D.; Biton, V.; Cascino, G.D.; Sperling, M.R.; Liow, K.; Vazquez, B.; Ayala, R.; et al. Safety and Tolerability of NRL-1, an Intranasal Formulation of Diazepam, in Relationship to Usage Frequency in Subjects with Epilepsy: Interim Results From a Phase 3, Open-label, Repeat Dose Study (1992). Neurology 2020, 94, 1992. [Google Scholar]
- Dean, P.; Santilli, N.; Wheless, J.W.; Vazquez, B.; Segal, E.B.; Miller, I.; Hogan, R.E.; Carrazana, E.; Rabinowicz, A.L. Low Rate of Medication Errors Supports the Ability of Patients and Caregivers to Administer NRL-1, an Intranasal Formulation of Diazepam: Interim Results from a Phase 3, Open-Label, Repeat Dose Study (1875). Neurology 2020, 94, 1875. [Google Scholar]
- Acorda. Development of a Nasal Form of Diazepam Stopped. Epilepsy Found. 2016, 214, 161–165. Available online: https://www.epilepsy.com/article/2016/5/development-nasal-form-diazepam-stopped (accessed on 29 November 2020).
- Neurelis, Inc. Neurelis Intranasal Diazepam Treatment for Epilepsy Granted Fast Track Designation by FDA; Neurelis, Inc.: San Diego, CA, USA, 2017. [Google Scholar]
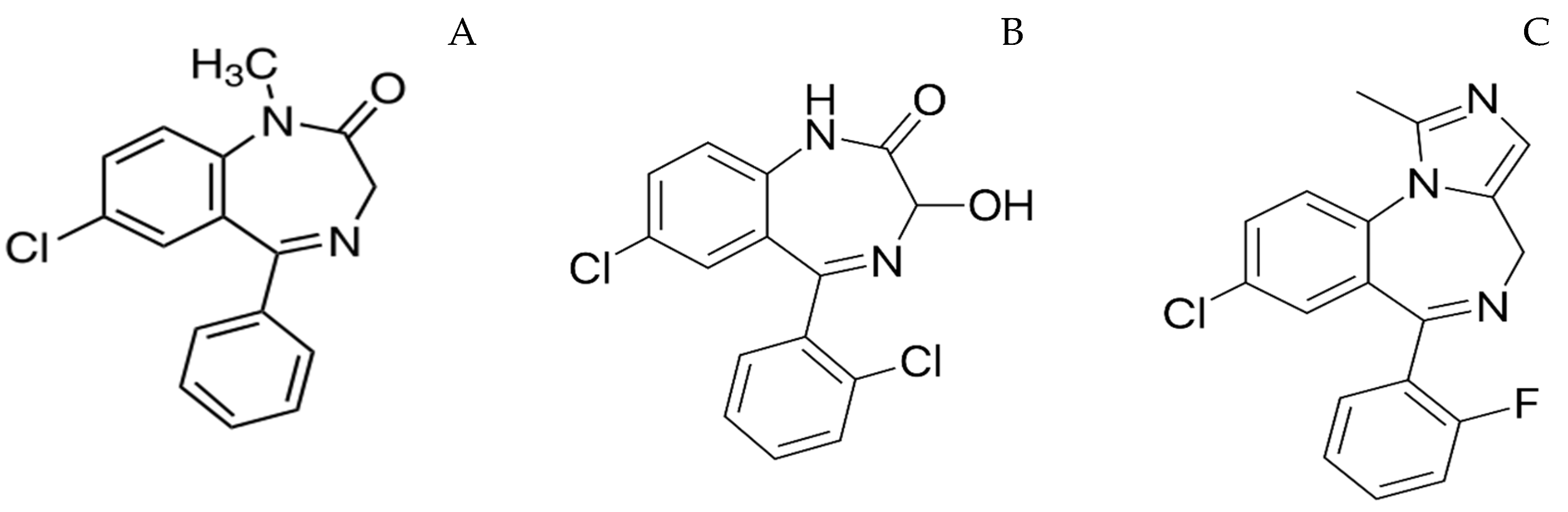
| Drug | Volume of Distribution (Vd), L/kg | Clearance (L/h/Kg) | Distribution Half-Life (t1/2α) (min) | Elimination Half-Life (t1/2β) (min) | Onset of Action (min) | Duration of Action |
|---|---|---|---|---|---|---|
| Diazepam | 0.89 ± 0.18 | 0.0388 ± 0.015 | 1.9–13.3 | 32.9 ± 8.8 | 1–3 | <2 h |
| Midazolam | 0.80 ± 0.19 | 0.42 ± 0.17 | 18.6 ± 14.4 | 2.4 ± 0.8 | ~2 | 3–4 h |
| Lorazepam | 1.14 ± 0.03 | 0.063 ± 0.009 | <11 | 14.3 ± 2.5 | 1–3 | <72 h |
| Route of Administration | Onset of Action (min) | Peak Plasma Levels (min) | Bioavailability |
|---|---|---|---|
| Oral | 15–60 | 30–90 | 100% |
| IM | 15–30 | 30–60 | 60% |
| Rectal | 5–10 | 10–45 | 80–90% |
| Intranasal | <5 | >60 | 97% |
| Drug | Brand Name | Route | Excipients |
|---|---|---|---|
| Diazepam | Valium® | Intravenous | 40% propylene glycol, 10% ethyl alcohol; 5% Na benzoate and, benzoic acid as buffers, and 1.5% benzyl alcohol as a preservative [34]. |
| Diazepam | Diastat® | Rectal | Propylene glycol, ethyl alcohol (10%), hydroxypropyl methylcellulose, sodium benzoate, benzyl alcohol (1.5%), benzoic acid and water. |
| Diazepam | Valtoco® (Available doses: 5 mg, 10 mg, 15 mg) | Intranasal | Benzyl alcohol (10.5 mg per 0.1 mL), dehydrated alcohol, n-dodecyl beta-d-maltoside, and vitamin E. |
| Lorazepam | Temesta®, solution for injection | Intravenous | Macrogol 400, benzyl alcohol 21 mg/ml, propylene glycol. |
| Lorazepam | Temesta Expidet® | Orodispersible | Gelatin, mannitol |
| Midazolam | Midazolam injection, USP | Intravenous | 0.8% sodium chloride and 0.01% edetate disodium, with 1% benzyl alcohol as preservative; the pH is adjusted to 3 to 3.6 with hydrochloric acid and, if necessary, sodium hydroxide |
| Midazolam | Seizalam™ | Intramuscular | 1% benzyl alcohol as preservative, 0.01% edetate disodium, and 0.8% sodium chloride, pH is adjusted to ~3. |
| Midazolam | Nayzilam® | Intranasal | Ethanol, PEG-6 methyl ether, polyethylene glycol 400, propylene glycol and purified water. |
| Midazolam hydrochloride | Buccolam® | Oromucosal solution | Sodium chloride, water for injections, hydrochloric acid (for pH adjustment and conversion of midazolam to the hydrochloride salt), sodium hydroxide (for pH adjustment |
| Midazolam maleate | Epistatus® | Oromucosal solution | Ethanol, saccharin sodium, glycerol purified water, sodium hydroxide (for pH adjustment), liquid maltitol |
| Formulations/Routes of Administration | Study Design | Subjects | Results | Conclusions | Ref. |
|---|---|---|---|---|---|
| Intranasal diazepam (10 mg) and lorazepam (4 mg) formulated using non-ionic surfactant (Cremophor EL) | Crossover trial | Healthy adults | Diazepam: Bioavailability = 84% and 72%, Tmax = 1 h, Cmax = 175 ng/mL. Peak concentration following intranasal administration was 27% to that of IV administration. Lorazepam: Bioavailability: 35–63%, Tmax = 0.5 to 4 h, Cmax = 18.7 ± 5.9 ng/mL, Cmax: 33–94% to that following IV administration. | Intranasal administration of diazepam and lorazepam would have limited potential in the acute treatment of seizures. | [52] |
| Intranasal diazepam in a mixture of 5% glycofurol and polyethylene glycol 200 versus commercial IV diazepam (Stesolid® Dumex-Alpharma), 2 mg dose | Open crossover trail | Healthy students | Intranasal diazepam: Cmax= 39 ± 17 ng/mL, tmax = 18 ± 11 min, t1/2 = 17.8 ± 15.5, AUC0–30min = 1095 ± 412 ng.min/mL. Intravenous diazepam: t1/2 = 14.4 ± 7.0, AUC0–30min = 2972 ± 980 ng.min/mL. | Intranasal diazepam could be an alternative to IV and rectal administration for treating acute seizures | [49] |
| Intranasal diazepam in polyethylene glycol 300 (4 mg and 7 mg dose) versus Stesolid Novum® intravenous diazepam (5 mg dose) | Double-blind, randomized, crossover design | Healthy volunteers | Mean differences between before and after drug administration values of P300-N100 amplitude differences were −0.9 (6.5, 4.7), −6.4 (−10.1, −2,7), −8.6 (−11.4, −5.8) and −9.6 (−12.1, −7.1) for placebo, 4 mg intranasal, 7 mg intranasal and 5 mg diazepam preparations, respectively. 4 and 7 mg intranasal diazepam formulations showed bioavailabilities of 45% and 42%, respectively. | Based on the electroencephalographic effects and blood concentration data, PEG300 may be used to deliver effective nasal dose of diazepam for the acute treatment of epilepsy | [50] |
| Intranasal diazepam in ten vehicles of different polarity to achieve tmax ≤ 5 min | - | Rabbits | Pure glycofurol 75, tetraethyleneglycol, poly(ethylene glycol) 200 and 30% glycofurol in tetraethyleneglycol showed very rapid pharmacodynamic response (1.5–3.5 min) compared to more polar liquids | Water-free low-molecular-weight glycols might be used as an alternative to IV injection for acute situations. | [53] |
| Intranasal diazepam versus IV diazepam (0.5 mg/kg) | Crossover design | Healthy adult greyhounds | IV: Cpeak: 1316 ± 216 µg/L, Tpeak was ≤ 3 min Intranasal: Cpeak: 448 ± 41 µg/L), Tpeak: 4.5 ± 1.5 min, Bioavailability: 80 ± 9% | Plasma concentrations exceed 300 µg/L (therapeutic concentration). Intranasal diazepam may be useful for treatment of seizures in dogs in place of IV administration | [54] |
| Intranasal ethyl laurate-based microemulsion systems of diazepam (1–2 mg/kg) and comparison with IV administration (1 mg/kg). | - | Rabbits | Tween 80–23.3%, propylene glycol–23.3% ethanol–15% H2O at 2 mg/kg dose resulted in rapid-onset of action (2–3 min) of diazepam with 50% bioavailability. | Ethyl laurate-based microemulsion of diazepam may be useful in the treatment of status epilepticus. | [55] |
| Diazepam intranasal (7 mg) versus diazepam intravenous (3 mg). Results were compared with rabbit and human data | Crossover design | Sheep | Mean nasal bioavailability, tmax and Cmax were 15 ± 8%, 5 ± 3 min and 934 ± 593 ng/mL, respectively. Bioavailability of diazepam in sheep was lower than rabbit (54%, p < 0.001) and man (34%, p < 0.05). | Correlation of bioavailability (rate and the extent of absorption) was not optimal between sheep, man and rabbit. | [56] |
| Supersaturated solution of diazepam in glycofurol/water for intranasal administration | - | MDCK epithelial cells as a nasal mucosa model | Steady-state flux of diazepam was obtained across MDCK epithelial cell monolayers from supersaturated solutions, which increased proportionally with increasing degree of saturation | Supersaturated diazepam solutions may be used for intranasal delivery | [57] |
| Diazepam was intravenously (1 mg/kg) or intranasally (2 mg/kg) administered to rats and rabbits | - | New Zealand white rabbits and Sprague–Dawley rats | Rats: Tmax = 5 min in rats, AUCbrain/AUCplasma ratios after IV (3.03 ± 0.07) and intranasal (3.00 ± 0.32) administration were nearly identical. Bioavailability in rat plasma (68.4%) and brain (67.7%) Rabbits: Tmax = 10 min, AUCbrain/AUCplasma ratios after intranasal administration (3.77 ± 0.17) were slightly lower than from IV administration (4.23 ± 0.08). Bioavailability in rabbit plasma (51.6%) and brain (45.9%) | No significant nose-to-brain transport (via olfactory epithelium) of diazepam was observed. Diazepam was mostly transported acorss the blood–brain barrier after intranasal administration. | [44] |
| Intranasal diazepam microemulsion | - | Bufo gargarizans | Miglyol 812 (8.0%), Tween 80 (21.3%), PEG400 (10.7%) and water (60.0%) containing microemulsion of diazepam showed only slight nasal ciliotoxicity | Microemulsions of Miglyol 812-Tween80-PEG400-water system with diazepam could be used for intranasal administration | [58] |
| 5 mg of diazepam and midazolam via both intranasal and IV routes | Four-way, randomized crossover trial. | Healthy adult volunteers | Diazepam: Cmax = 179.2° ng/mL, Tmax = 28.8 min Midazolam: Cmax = 62.8° ng/mL and Tmax 21.6 min. Intranasal administration resulted in rapid absorption with transient discomfort. Diazepam had a longer half-life, with an extended duration of action | Diazepam and midazolam were rapidly absorbed following intranasal administration with transient discomfort. | [45] |
| Intranasal formulation of diazepam (5 mg and 10 mg) in a glycofurol–water cosolvent system was investigated | Randomized, single-blind, three-way crossover | Healthy volunteers | The estimated bioavailability was 75% with pain and tolerability scores around 2–2.3 and 4.4–4.7 following administration of 5 and 10 mg doses, respectively | Intranasal diazepam provided a reasonable bioavailability, but was not well tolerated | [51] |
| Alcohol-free microemulsion system for intranasal delivery of diazepam or midazolam (2.5% by weight) | Randomized cross-over design | Rabbits | Diazepam: Cmax = 8.40 ± 3.00, Absolute bioavailability = 33.45 ± 12.36%, tmax = 18.33 ± 23.09 min Midazolam: Cmax = 46.62 ±17.38, tmax = 9.25 ± 6.75 min, Absolute bioavailability = 35.19 ±11.83% | Midazolam and diazepam microemulsion system could achieve rapid-onset of action following intranasal administration | [59] |
| Pharmacokinetics of diazepam following IV administration versus administration as intranasal drop versus atomized nasal administration | Randomized block design | Dogs | Mean diazepam concentrations following intranasal administration reached >300 ng/mL within 5 min in both groups. Diazepam bioavailability after intranasal drop and atomized nasal administration was 42% and 41%, respectively | Intranasal administration yielded rapid anticonvulsant concentrations of diazepam in dogs | [60] |
| Effect of l-menthol on absorption of intranasal diazepam | - | Mice | The effect of diazepam via intranasal administration was strengthened in the presence of l-menthol | Intranasal diazepam with l-menthol may result in sedative-hypnotic action and control epileptic seizures | [61] |
| Effect of menthol as a penetration enhancer on the absorption intranasal diazepam | Rabbit | At 0.2%, menthol increased the absorption of diazepam [k = (0.4424 ± 0.0023)/h] with quick absorption [t1/2 =(0.32 ± 0.07)h] | 0.2% menthol helped in the intranasal absorption of diazepam through passive diffusion | [62] | |
| Tolerability and pharmacokinetics of two intranasal diazepam formulations were compared with rectal gel (Diastat®) | Double blind, 4-period, 4-way crossover study | Healthy volunteers | Mean Cmax (± SD) was 181.8 ± 84.16, 151.3 ± 108.1 and 180.7 ± 82.1 ng/mL for Nas-A 10 mg, Nas-B 10 mg and Nas-B 13.4 mg respectively; while Cmax for the rectal gel was 160.9 ± 109.4 ng/mL. Median tmax was 0.75 h for all treatments. Intranasal formulations were well tolerated and exhibited relatively rapid but variable absorption with bioavailability of 70–90% compared to diazepam rectal gel | Intranasal diazepam could be an alternative to rectal diazepam | [63] |
| Dose proportionality of 5 mg and 20 mg of intranasal diazepam formulations. Relative bioavailability of 20 mg intranasal diazepam versus 20 mg rectal gel | Phase 1, single-center, randomized, open-label, three-period crossover study | Healthy subjects | Intranasal diazepam solutions (5 and 20 mg) showed dose proportionality with median time to Cmax of 1 h. Administration of a single dose of 20 mg intranasal diazepam resulted in similar plasma concentrations of diazepam and metabolite concentration, with less variability than with 20 mg rectal gel | Diazepam nasal solution (20 mg) showed comparable bioavailability as 20 mg rectal gel | [64] |
| Diazepam nasal spray (0.2 mg/kg) | Open-label study | Tmax of diazepam was 45 min with comparable dose-normalized mean Cmax and AUC0–12 values of diazepam among patients regardless of the timing of administration in relation to seizure. | Diazepam nasal spray could be used during the convulsive phase of tonic-clonic seizures or in the postictal periods following tonic-clonic or other seizure types. | [65] | |
| Intranasal diazepam formulation versus an equivalent dose of rectal diazepam (20 mg) | Phase 1, open-label, 3-period crossover study. | Healthy adults | Mean Cmax values of diazepam nasal spray and rectal gel were found to be 378 ± 106 and 328 ± 152 ng/mL, achieved at 1.0 and 1.5 h, respectively. Both intranasal and rectal diazepam were well tolerated with mild to moderate adverse events. | Single-dose of 20 mg diazepam nasal spray is tolerable and comparable in bioavailability to that of diazepam rectal gel. | [66] |
| Supersaturated diazepam solution using a prodrug/enzyme system (Avizafone, a peptide prodrug of diazepam, delivered with—Aspergillus oryzae protease) | - | Madin-Darby canine kidney II-wild type | Prodrug-protease mixtures upon apical exposure onto MDCKII-wt monolayers showed 2–17.6-fold higher diazepam flux (S = 1.3–15.3) compared to saturated diazepam (S = 0.7). | Intranasal avizafone-protease system with diazepam may provide rapid delivery. | [67] |
| Effectiveness of intranasal diazepam as an effective alternative to IV diazepam based on the medical records | Retrospective study | Stroke patients presenting with status epilepticus. | Intranasal diazepam was administered 9 times faster compared to IV diazepam resulting in about 3-fold reduction in the time to seizure activity termination following arrival at the hospital (3 min vs 9.5 min in the IV group, p = 0.030) | Intranasal diazepam could be a safe, quick and easier alternative to intravenous administration. | [68] |
| Diazepam-loaded poly(lactic-co-glycolic acid) nanoparticles | - | Rats | Gamma scintigraphy using technetium-99m-labeled (99mTc) showed a higher uptake of diazepam from nanoparticles compared to diazepam suspension in Sprague-Dawley rats. | PLGA nanoparticles of diazepam could be used in the treatment of status epilepticus | [69] |
| Mucoadhesive microemulsions of diazepam for intranasal administration versus Calmpose (i.v) and microemulsions | - | Rats | Diazepam microemulsion composed of oleic acid (5%), surfactant mixture (50%), water (45%), and chitosan (0.5%) showed significantly high flux of 846.96 ± 34 µg/cm2/h and AUCbrain = 1206.49 ± 145.8 compared to Calmpose (i.v) and microemulsion. | Mucoadhesive microemulsions showed higher absorption compared to IV administration | [70] |
| Coadministration of a hydrophilic diazepam prodrug (avizafone) and converting enzyme, human aminopeptidase B | - | Rats | Single doses of intranasal avizafone equivalent to 0.500, 1.00, and 1.50 mg/kg of diazepam resulted in 77.8% ± 6.0%, 112% ± 10%, and 114% ± 7% bioavailability with Cmax plasma concentrations 71.5 ± 9.3, 388 ± 31, and 355 ± 187 ng/mL; and tmax of 5, 8, and 5 min for each dose level, respectively. | Rapid and complete absorption by co-administering avizafone with aminopeptidase B | [71] |
| 1 dose period (5, 10, and 20 mg) followed by a 2-dose period (2 × 10 mg) with a minimum 28-day washout | Phase 1, open-label, randomized, crossover study | Healthy adult volunteers | Plasma concentration-time profiles showed similar patterns in a dose-dependent manner. The Cmax values of diazepam were 85.6, 133.6, and 235.3 ng/mL for 5-, 10-, and 20-mg doses, respectively. Dose-normalized AUC0–∞ values were comparable between the 2 × 10-mg and single 10-mg doses. | NRL-1 could be a potential therapy for managing seizure emergencies. | [38] |
| Valtoco™ (NRL-1; diazepam nasal spray) formulated with Intravail® A3 | Open-label study | Patients with epilepsy | Pharmacokinetic parameters in ictal/peri-ictal and inter-ictal conditions were similar (tmax: 3.31 ± 2.10 vs. 2.79 ± 1.89; Cmax: 156 ± 17 vs. 179 ± 18 ng/mL; AUC: 518 ± 30 vs. 566 ± 33 hr·ng/mL, respectively) | Valtoco™ showed a good safety profile in patients with epilepsy | [72] |
| Bioavailability and tolerability of intranasal diazepam containing Intravail® vs diazepam rectal gel | Phase 1, open-label, randomized, single-dose, crossover study | Healthy adult subjects | Tmax was similar for diazepam nasal spray and rectal gel, which were slower than oral diazepam in fasted individuals | Intravail® provided therapeutic levels of diazepam comparable to rectal diazepam with no damage to the nasal mucosa | [73] |
| Tolerability of NRL-1 (Valtoco®, diazepam nasal spray formulated with Intravail A3) and adverse events in patients | Open-label, Phase 3 study | Adults and children/adolescents with epilepsy | Of the 57 subjects, 17 subjects (29.8%) reported treatment emergent adverse events (TEAEs) with no treatment discontinuation. Treatment-related TEAEs were observed in 8 subjects (14%). Dysgeusia was reported in 3 subjects (5.3%) and nasal discomfort in 2 subjects) | NRL-1 demonstrated an acceptable safety/tolerability profile | [74] |
| Long-term safety and tolerability of NRL-1 (Valtoco®, diazepam nasal spray formulated with Intravail A3). A dose of 5, 10, 15, or 20 mg was administered based on patient weight | Phase 3, open-label, study | Patients (including adults and children/adolescents | Out of 132 enrolled subjects, NRL-1 was used moderately in 65 (49.2%) and frequently in 67 (50.8%) patients. Overall, 91 patients (68.9%) had TEAEs | Repeat dosing of NRL-1 showed an acceptable safety/tolerability profile similar to diazepam administered via other routes | [75] |
| Type of dosing errors and extent as a substitution for the ability of patients/caregivers to properly administer NRL-1 | Phase 3, open-label, study | Pediatric and adult patients with epilepsy | Patients/caregivers reported 31 dosing errors in 23 patients (1.2% of the administered 2486 doses). 80.6% of these errors were associated with doses requiring spray into both nostrils and 4 patients had multiple errors. Partial dosing errors were 48.4%, improper dosing errors were 12.9%, mechanical dosing time were 9.7% and 29.0% were other/unknown errors. | Most errors occurred when dose administration is required into both nostrils | [76] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boddu, S.H.S.; Kumari, S. A Short Review on the Intranasal Delivery of Diazepam for Treating Acute Repetitive Seizures. Pharmaceutics 2020, 12, 1167. https://doi.org/10.3390/pharmaceutics12121167
Boddu SHS, Kumari S. A Short Review on the Intranasal Delivery of Diazepam for Treating Acute Repetitive Seizures. Pharmaceutics. 2020; 12(12):1167. https://doi.org/10.3390/pharmaceutics12121167
Chicago/Turabian StyleBoddu, Sai H. S., and Sneha Kumari. 2020. "A Short Review on the Intranasal Delivery of Diazepam for Treating Acute Repetitive Seizures" Pharmaceutics 12, no. 12: 1167. https://doi.org/10.3390/pharmaceutics12121167
APA StyleBoddu, S. H. S., & Kumari, S. (2020). A Short Review on the Intranasal Delivery of Diazepam for Treating Acute Repetitive Seizures. Pharmaceutics, 12(12), 1167. https://doi.org/10.3390/pharmaceutics12121167