Drug-Loaded Lipid-Core Micelles in Mucoadhesive Films as a Novel Dosage Form for Buccal Administration of Poorly Water-Soluble and Biological Drugs
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of Rhodamine 123- and Insulin-Loaded Lipid-Core Micelles
2.3. Characterization of Rhodamine 123- and Insulin-Loaded Lipid-Core Micelles
2.4. In Vitro Release Study and Kinetic Modeling of Rhodamine 123- and Insulin-Loaded Lipid-Core Micelles
2.5. Elaboration of Rhodamine 123- and Insulin-Loaded Lipid-Core Micelles Mucoadhesive Films
2.6. Characterization of Hydroxypropyl Methylcellulose Mucoadhesive Films
2.6.1. Mechanical Properties
2.6.2. Mucoadhesive Properties
2.6.3. Scanning Electron Microscopy
2.7. Ex Vivo Permeation of Rhodamine 123- and Insulin-Loaded Lipid-Core Micelles Mucoadhesive Films
2.8. Statistical and Similarity Analysis
3. Results
3.1. Synthesis and Physical-Chemical Characterization of Rhodamine 123- and Insulin-Loaded Lipid-Core Micelles
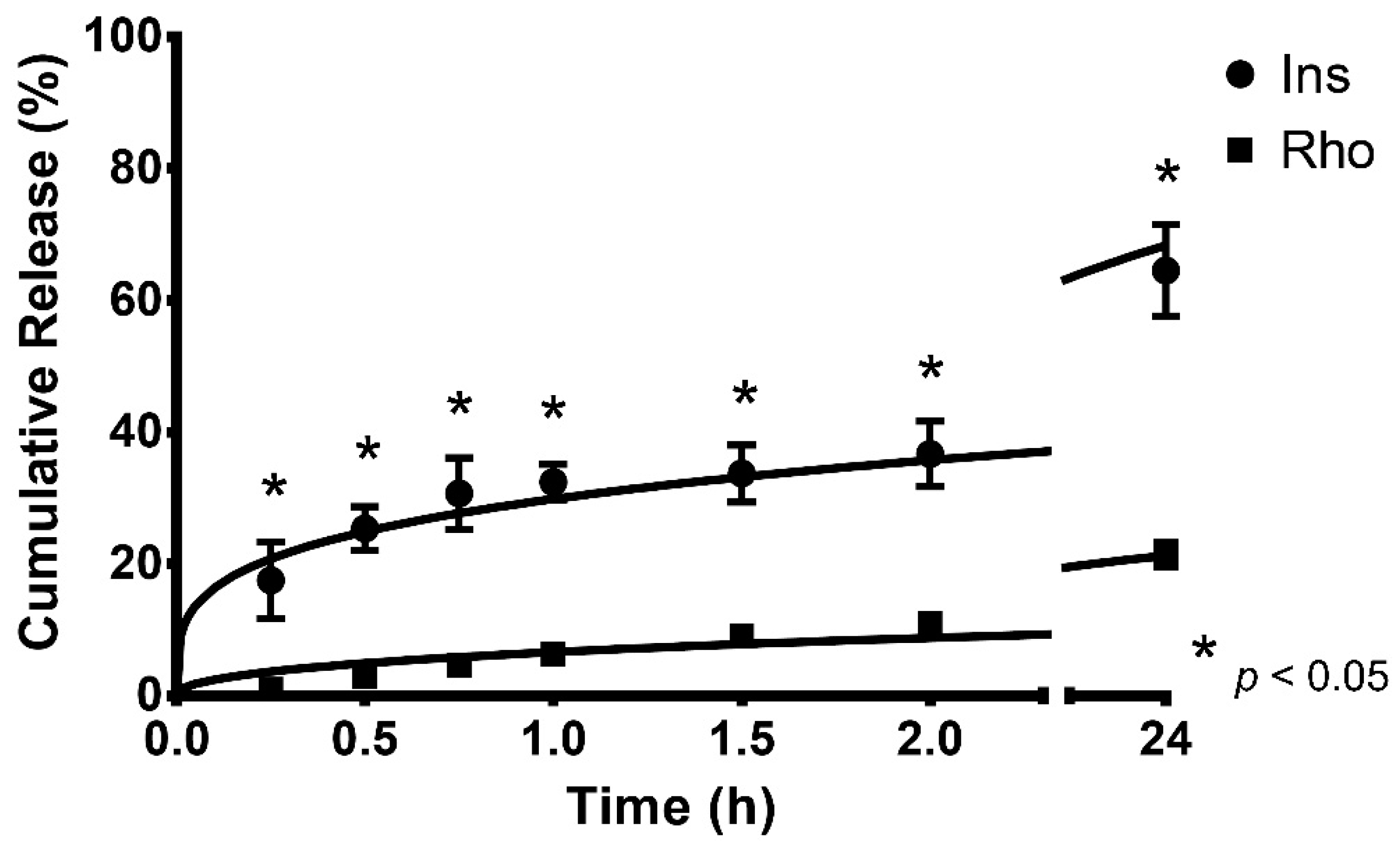
3.2. In Vitro Release Profiles and Kinetic Modeling for Rhodamine 123- and Insulin-Loaded Lipid-Core Micelles
3.3. Mechanical and Mucoadhesive Properties of Non-Loaded and Loaded Mucoadhesive Films
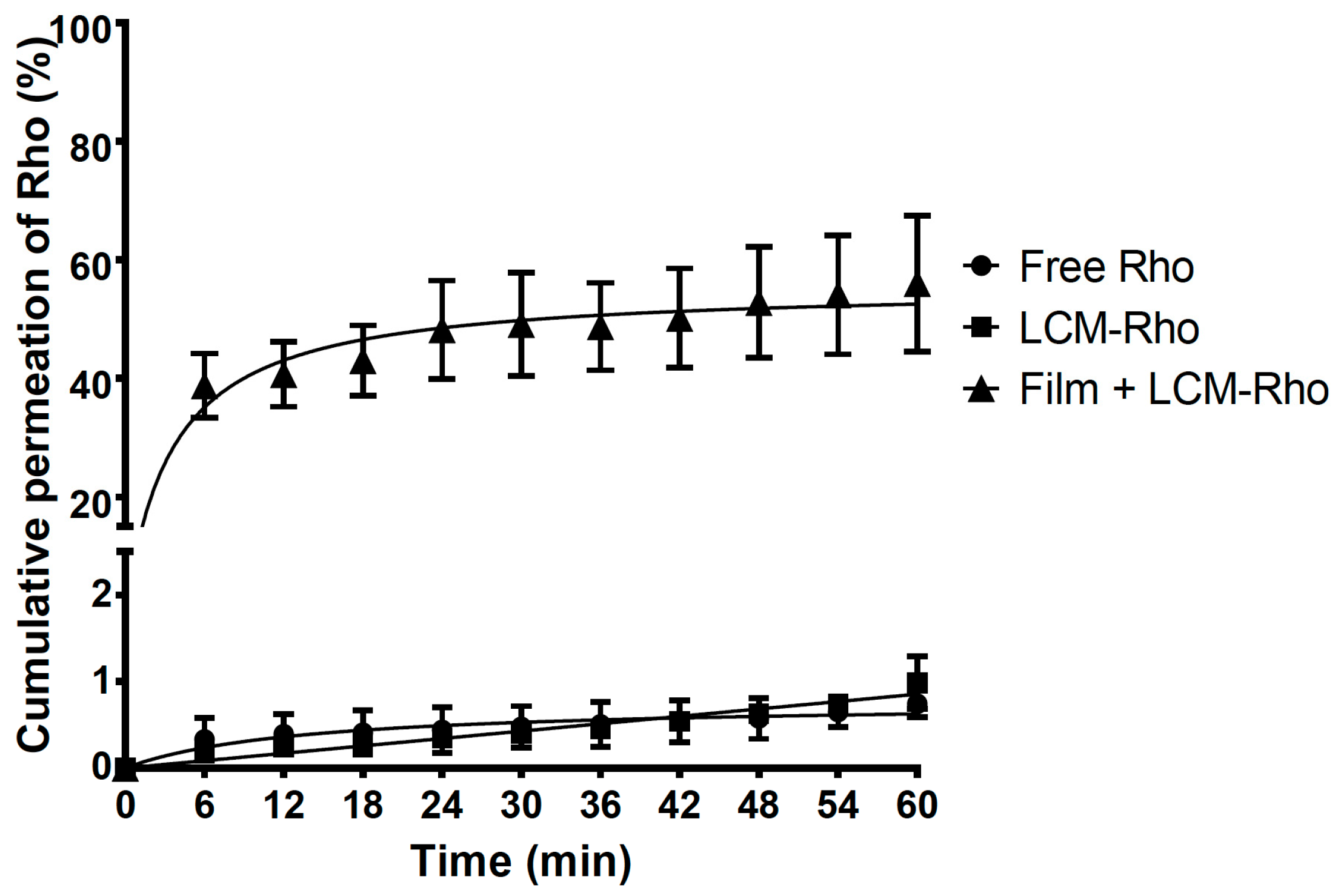
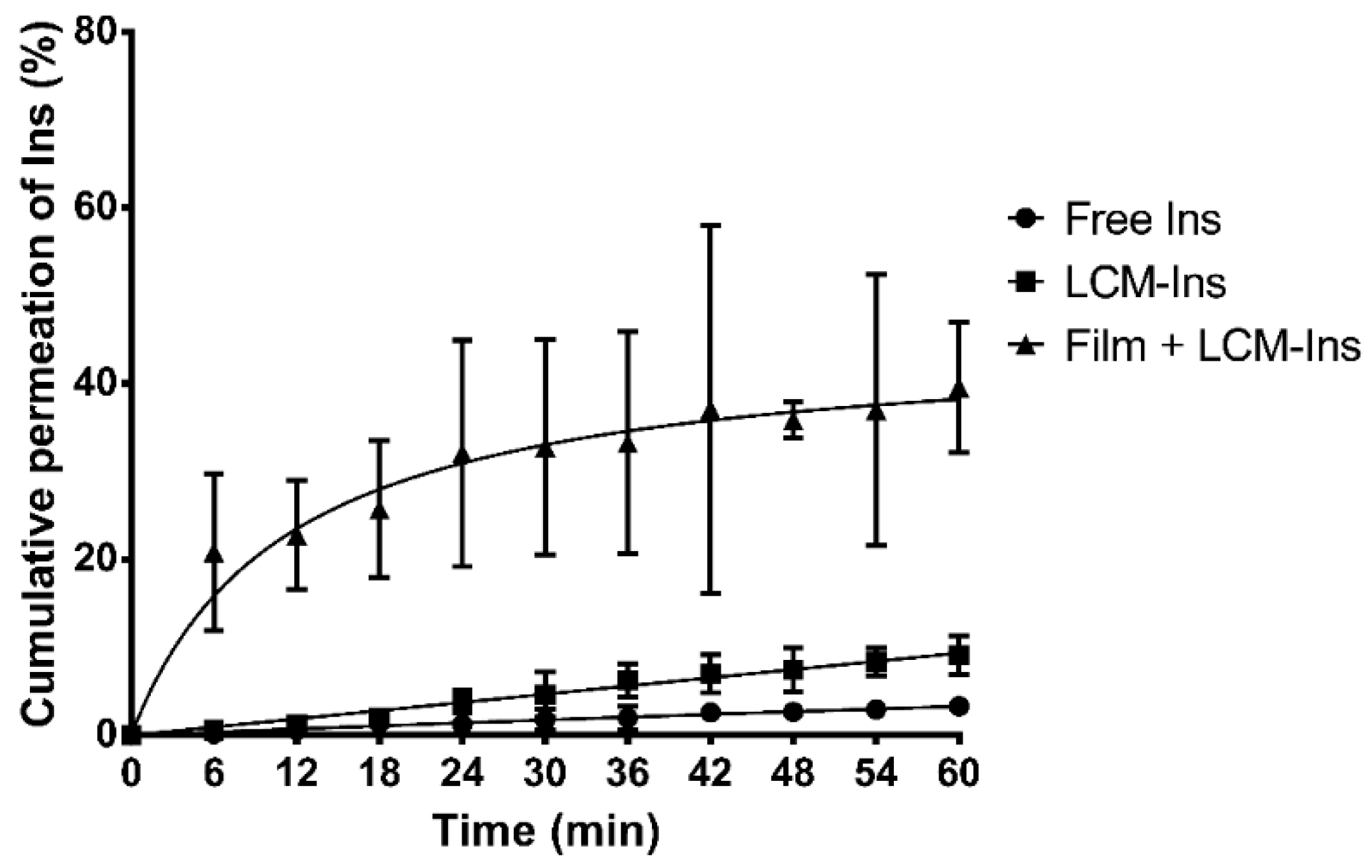
3.4. Ex Vivo Permeation for Rhodamine 123- and Insulin-Loaded Lipid-Core Micelles Using Mucoadhesive Films
4. Discussion
4.1. Physical-Chemical Characterization of Lipid-Core Micelles
4.2. In Vitro Release Profiles of Rhodamine 123 and Insulin
4.3. Mechanical and Mucoadhesive Properties of Films
4.4. Ex Vivo Permeation of Rhodamine 123 and Insulin
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Bardal, S.K.; Waechter, J.E.; Martin, D.S. Chapter 2—Pharmacokinetics. In Applied Pharmacology; Bardal, S.K., Waechter, J.E., Martin, D.S., Eds.; Content Repository Only: Philadelphia, PA, USA, 2011; pp. 17–34. ISBN 978-1-4377-0310-8. [Google Scholar]
- Kawabata, Y.; Wada, K.; Nakatani, M.; Yamada, S.; Onoue, S. Formulation design for poorly water-soluble drugs based on biopharmaceutics classification system: Basic approaches and practical applications. Int. J. Pharm. 2011, 420, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Kestenbaum, M.G.; Vilches, A.O.; Messersmith, S.; Connor, S.R.; Fine, P.G.; Murphy, B.; Davis, M.; Muir, J.C. Alternative Routes to Oral Opioid Administration in Palliative Care: A Review and Clinical Summary. Pain Med. 2014, 15, 1129–1153. [Google Scholar] [CrossRef] [PubMed]
- Caon, T.; Pan, Y.; Simões, C.M.O.; Nicolazzo, J.A. Exploiting the Buccal Mucosa as an Alternative Route for the Delivery of Donepezil Hydrochloride. J. Pharm. Sci. 2014, 103, 1643–1651. [Google Scholar] [CrossRef] [PubMed]
- Brown, M.B.; Patel, V.F. Buccal and sublingual drug delivery. In Drug Delivery: Fundamentals & Applications; Taylor & Francis Group: Oxfordshire, UK, 2017; pp. 201–214. [Google Scholar]
- Fonseca-Santos, B.; Chorilli, M. An overview of polymeric dosage forms in buccal drug delivery: State of art, design of formulations and their in vivo performance evaluation. Mater. Sci. Eng. C 2018, 86, 129–143. [Google Scholar] [CrossRef] [PubMed]
- Shojaei, A.H. Buccal mucosa as a route for systemic drug delivery: A review. J. Pharm. Pharm. Sci. 2000, 1, 15–30. [Google Scholar]
- Drugs@FDA: FDA-Approved Drugs. Available online: https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm (accessed on 20 February 2020).
- Costa, J.S.R.; Cruvinel, K.D.O.; Oliveira-Nascimento, L. A mini-review on drug delivery through wafer technology: Formulation and manufacturing of buccal and oral lyophilizates. J. Adv. Res. 2019, 20, 33–41. [Google Scholar] [CrossRef]
- Portero, A.; Teijeiro-Osorio, D.; Alonso, M.J.; Remuñán-López, C. Development of chitosan sponges for buccal administration of insulin. Carbohydr. Polym. 2007, 68, 617–625. [Google Scholar] [CrossRef]
- Baus, R.A.; Zahir-Jouzdani, F.; Dünnhaupt, S.; Atyabi, F.; Bernkop-Schnürch, A. Mucoadhesive hydrogels for buccal drug delivery: In vitro-in vivo correlation study. Eur. J. Pharm. Biopharm. 2019, 142, 498–505. [Google Scholar] [CrossRef]
- Alopaeus, J.F.; Hellfritzsch, M.; Gutowski, T.; Scherließ, R.; Almeida, A.; Sarmento, B.; Skalko-Basnet, N.; Tho, I. Mucoadhesive buccal films based on a graft co-polymer—A mucin-retentive hydrogel scaffold. Eur. J. Pharm. Sci. 2020, 142, 105142. [Google Scholar] [CrossRef]
- Pozzilli, P.; Manfrini, S.; Costanza, F.; Coppolino, G.; Cavallo, M.G.; Fioriti, E.; Modi, P. Biokinetics of buccal spray insulin in patients with type 1 diabetes. Metabolism 2005, 54, 930–934. [Google Scholar] [CrossRef]
- Rahbarian, M.; Mortazavian, E.; Dorkoosh, F.A.; Tehrani, M.R. Preparation, evaluation and optimization of nanoparticles composed of thiolated triethyl chitosan: A potential approach for buccal delivery of insulin. J. Drug Deliv. Sci. Technol. 2018, 44, 254–263. [Google Scholar] [CrossRef]
- Hua, S. Advances in Nanoparticulate Drug Delivery Approaches for Sublingual and Buccal Administration. Front. Pharmacol. 2019, 10, 1328. [Google Scholar] [CrossRef] [PubMed]
- Karki, S.; Kim, H.; Na, S.-J.; Shin, D.; Jo, K.; Lee, J. Thin films as an emerging platform for drug delivery. Asian J. Pharm. Sci. 2016, 11, 559–574. [Google Scholar] [CrossRef]
- Puri, A.; Loomis, K.; Smith, B.; Lee, J.-H.; Yavlovich, A.; Heldman, E.; Blumenthal, R. Lipid-Based Nanoparticles as Pharmaceutical Drug Carriers: From Concepts to Clinic. Crit. Rev. Ther. Drug Carr. Syst. 2009, 26, 523–580. [Google Scholar] [CrossRef]
- Torchilin, V. Lipid-core micelles for targeted drug delivery. Curr. Drug Deliv. 2005, 2, 319–327. [Google Scholar] [CrossRef]
- Hillery, A.M.; Park, K. Historical Introduction to the Field of Controlled Drug Delivery. In Drug Delivery: Fundamentals & Applications; Taylor & Francis Group: Oxfordshire, UK, 2017; pp. 1–21. [Google Scholar]
- Gao, Z.; Lukyanov, A.N.; Singhal, A.A.; Torchilin, V.P. Diacyllipid-Polymer Micelles as Nanocarriers for Poorly Soluble Anticancer Drugs. Nano Lett. 2002, 2, 979–982. [Google Scholar] [CrossRef]
- Wang, J.; Mongayt, D.A.; Lukyanov, A.N.; Levchenko, T.S.; Torchilin, V. Preparation and in vitro synergistic anticancer effect of Vitamin K3 and 1,8-diazabicyclo[5,4,0]undec-7-ene in poly(ethylene glycol)-diacyllipid micelles. Int. J. Pharm. 2004, 272, 129–135. [Google Scholar] [CrossRef]
- Mu, L.; Elbayoumi, T.; Torchilin, V. Mixed micelles made of poly (ethylene glycol)-phosphatidylethanolamine conjugate and d-alpha-tocopheryl polyethylene glycol 1000 succinate as pharmaceutical nanocarriers for camptothecin. Int. J. Pharm. 2005, 306, 142–149. [Google Scholar] [CrossRef]
- Dave, V.; Yadav, R.B.; Kushwaha, K.; Yadav, S.; Sharma, S.; Agrawal, U. Lipid-polymer hybrid nanoparticles: Development & statistical optimization of norfloxacin for topical drug delivery system. Bioact. Mater. 2017, 2, 269–280. [Google Scholar] [CrossRef]
- Fritz, H.F.; Ortiz, A.C.; Velaga, S.P.; Morales, J.O. Preparation of a novel lipid-core micelle using a low-energy emulsification method. Drug Deliv. Transl. Res. 2018, 8, 1807–1814. [Google Scholar] [CrossRef]
- Yu, F.; Li, Y.; Liu, C.S.; Chen, Q.; Wang, G.H.; Guo, W.; Wu, X.E.; Li, D.H.; Wu, W.D.; Chen, X.D. Enteric-coated capsules filled with mono-disperse micro-particles containing PLGA-lipid-PEG nanoparticles for oral delivery of insulin. Int. J. Pharm. 2015, 484, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Fuentes, M.; Torres, D.; Alonso, M.J. New surface-modified lipid nanoparticles as delivery vehicles for salmon calcitonin. Int. J. Pharm. 2005, 296, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Minato, S.; Iwanaga, K.; Kakemi, M.; Yamashita, S.; Oku, N. Application of polyethyleneglycol (PEG)-modified liposomes for oral vaccine: Effect of lipid dose on systemic and mucosal immunity. J. Control. Release 2003, 89, 189–197. [Google Scholar] [CrossRef]
- Heyes, J.; Palmer, L.; Chan, K.; Giesbrecht, C.; Jeffs, L.; MacLachlan, I. Lipid Encapsulation Enables the Effective Systemic Delivery of Polyplex Plasmid DNA. Mol. Ther. 2007, 15, 713–720. [Google Scholar] [CrossRef]
- Zhang, Y.; Huo, M.; Zhou, J.; Zou, A.; Li, W.; Yao, C.; Xie, S. DDSolver: An Add-In Program for Modeling and Comparison of Drug Dissolution Profiles. AAPS J. 2010, 12, 263–271. [Google Scholar] [CrossRef]
- Montenegro-Nicolini, M.; Reyes, P.E.; Jara, M.O.; Vuddanda, P.R.; Neira-Carrillo, A.; Butto, N.; Velaga, S.P.; Morales, J. The Effect of Inkjet Printing over Polymeric Films as Potential Buccal Biologics Delivery Systems. AAPS PharmSciTech 2018, 19, 3376–3387. [Google Scholar] [CrossRef]
- Montenegro-Nicolini, M.; Miranda, V.; Morales, J.O. Inkjet Printing of Proteins: An Experimental Approach. AAPS J. 2016, 19, 234–243. [Google Scholar] [CrossRef]
- Castro, P.; Madureira, R.; Sarmento, B.; Pintado, M.; Madureira, A.R. Tissue-based in vitro and ex vivo models for buccal permeability studies. In Concepts and Models for Drug Permeability Studies; Elsevier BV: Amsterdam, The Netherlands, 2016; pp. 189–202. [Google Scholar]
- Zhang, L.; Alfano, J.; Race, D.; Davé, R.N. Zero-order release of poorly water-soluble drug from polymeric films made via aqueous slurry casting. Eur. J. Pharm. Sci. 2018, 117, 245–254. [Google Scholar] [CrossRef]
- Mottram, L.F.; Forbes, S.; Ackley, B.D.; Peterson, B.R. Hydrophobic analogues of rhodamine B and rhodamine 101: Potent fluorescent probes of mitochondria in living C. elegans. Beilstein J. Org. Chem. 2012, 8, 2156–2165. [Google Scholar] [CrossRef]
- Sabnis, R.W. Handbook of Biological Dyes and Stains; Wiley: Hoboken, NJ, USA, 2010. [Google Scholar]
- Loftsson, T. Chapter 4–Drug Pharmacokinetics after Alternative Routes of Administration. In Essential Pharmacokinetics; Loftsson, T., Ed.; Academic Press: Boston, MA, USA, 2015; pp. 105–118. ISBN 978-0-12-801411-0. [Google Scholar]
- Blanco, E.; Shen, H.; Ferrari, M. Principles of nanoparticle design for overcoming biological barriers to drug delivery. Nat. Biotechnol. 2015, 33, 941–951. [Google Scholar] [CrossRef]
- Nicolazzo, J.A.; Mannila, J. Buccal Delivery Systems. In Biodrug Delivery Systems; Informa UK Limited: Colchester, UK, 2016; pp. 135–153. [Google Scholar]
- Ganesan, P.; Narayanasamy, D. Lipid nanoparticles: Different preparation techniques, characterization, hurdles, and strategies for the production of solid lipid nanoparticles and nanostructured lipid carriers for oral drug delivery. Sustain. Chem. Pharm. 2017, 6, 37–56. [Google Scholar] [CrossRef]
- Protein Calculator Protein Calculator v3.4. Available online: http://protcalc.sourceforge.net/ (accessed on 9 February 2019).
- Gumustas, M.; Sengel-Turk, C.T.; Gumustas, A.; Ozkan, S.A.; Uslu, B. Chapter 5—Effect of polymer-based nanoparticles on the assay of antimicrobial drug delivery systems. In Multifunctional Systems for Combined Delivery, Biosensing and Diagnostics; Grumezescu, A.M., Ed.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 67–108. ISBN 978-0-323-52725-5. [Google Scholar]
- Nutan, M.T.H.; Reddy, I.K. General principles of suspensions. In Pharmaceutical Suspensions: From Formulacion Development to Manufacturing; Springer: New York, NY, USA, 2010; pp. 39–65. ISBN 978-1-4419-1087-5. [Google Scholar]
- Shi, J. Steric Stabilization; Croup Inorganic Materials Science–Literature Review; The Ohio State University: Columbus, OH, USA, 2002. [Google Scholar]
- Honary, S.; Zahir, F. Effect of Zeta Potential on the Properties of Nano-Drug Delivery Systems—A Review (Part 2). Trop. J. Pharm. Res. 2013, 12, 265–273. [Google Scholar] [CrossRef]
- Arnida, M.M.; Janát-Amsbury, M.; Ray, A.; Peterson, C.M.; Ghandehari, H. Geometry and surface characteristics of gold nanoparticles influence their biodistribution and uptake by macrophages. Eur. J. Pharm. Biopharm. 2011, 77, 417–423. [Google Scholar] [CrossRef] [PubMed]
- Santos, J.; Sousa, F.; Queiroz, J.; Costa, D. Rhodamine based plasmid DNA nanoparticles for mitochondrial gene therapy. Colloids Surf. B Biointerfaces 2014, 121, 129–140. [Google Scholar] [CrossRef]
- He, H.; Lu, Y.; Qi, J.; Zhao, W.; Dong, X.; Wu, W. Biomimetic thiamine- and niacin-decorated liposomes for enhanced oral delivery of insulin. Acta Pharm. Sin. B 2018, 8, 97–105. [Google Scholar] [CrossRef]
- Boushra, M.; Tous, S.; Fetih, G.; Xue, H.-Y.; Wong, H.L. Development of bi-polymer lipid hybrid nanocarrier (BLN) to improve the entrapment and stability of insulin for efficient oral delivery. J. Drug Deliv. Sci. Technol. 2019, 49, 632–641. [Google Scholar] [CrossRef]
- Yang, C.; Lu, D.; Liu, Z. How PEGylation Enhances the Stability and Potency of Insulin: A Molecular Dynamics Simulation. Biochemistry 2011, 50, 2585–2593. [Google Scholar] [CrossRef]
- Svensson, A.; Neves, C.; Cabane, B. Hydration of an amphiphilic excipient, Gelucire 44/14. Int. J. Pharm. 2004, 281, 107–118. [Google Scholar] [CrossRef]
- Tosi, G.; Costantino, L.; Rivasi, F.; Ruozi, B.; Leo, E.; Vergoni, A.; Tacchi, R.; Bertolini, A.; Vandelli, M.A.; Forni, F. Targeting the central nervous system: In vivo experiments with peptide-derivatized nanoparticles loaded with Loperamide and Rhodamine-123. J. Control. Release 2007, 122, 1–9. [Google Scholar] [CrossRef]
- Park, S.-J.; Gil Choi, S.; Davaa, E.; Park, J.-S. Encapsulation enhancement and stabilization of insulin in cationic liposomes. Int. J. Pharm. 2011, 415, 267–272. [Google Scholar] [CrossRef]
- Bruschi, M.L. (Ed.) 5—Mathematical models of drug release. In Strategies to Modify the Drug Release from Pharmaceutical Systems; Woodhead Publishing: Cambridge, UK, 2015; pp. 63–86. ISBN 978-0-08-100092-2. [Google Scholar]
- Miao, J.; Tsige, M.; Taylor, P.L. Generalized model for the diffusion of solvents in glassy polymers: From Fickian to Super Case II. J. Chem. Phys. 2017, 147, 44904. [Google Scholar] [CrossRef] [PubMed]
- Ritger, P.L.; Peppas, N.A. A simple equation for description of solute release I. Fickian and non-fickian release from non-swellable devices in the form of slabs, spheres, cylinders or discs. J. Control. Release 1987, 5, 23–36. [Google Scholar] [CrossRef]
- Gao, Y.; Zuo, J.; Bou-Chacra, N.; Pinto, T.D.J.A.; Clas, S.-D.; Walker, R.B.; Löbenberg, R. In VitroRelease Kinetics of Antituberculosis Drugs from Nanoparticles Assessed Using a Modified Dissolution Apparatus. BioMed Res. Int. 2013, 2013, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Son, G.-H.; Lee, B.-J.; Cho, C.-W. Mechanisms of drug release from advanced drug formulations such as polymeric-based drug-delivery systems and lipid nanoparticles. J. Pharm. Investig. 2017, 47, 287–296. [Google Scholar] [CrossRef]
- El-Say, K.M.; Hosny, K.M. Optimization of carvedilol solid lipid nanoparticles: An approach to control the release and enhance the oral bioavailability on rabbits. PLoS ONE 2018, 13, e0203405. [Google Scholar] [CrossRef]
- Panigrahi, K.C.; Patra, C.N.; Jena, G.K.; Ghose, D.; Jena, J.; Panda, S.K.; Sahu, M. Gelucire: A versatile polymer for modified release drug delivery system. Futur. J. Pharm. Sci. 2018, 4, 102–108. [Google Scholar] [CrossRef]
- Dungan, S.R. Interfacial science and the creation of nanoparticles. In Nanotechnology and Functional Foods; Wiley: Hoboken, NJ, USA, 2015; pp. 52–68. [Google Scholar]
- Soleimani-Gorgani, A. 14. Inkjet printing. In Printing on Polymers; Izdebska, J., Thomas, S., Eds.; William Andrew Publishing: Norwich, NY, USA, 2016; pp. 231–246. ISBN 978-0-323-37468-2. [Google Scholar]
- Nayak, L.; Mohanty, S.; Nayak, S.K.; Ramadoss, A. A review on inkjet printing of nanoparticle inks for flexible electronics. J. Mater. Chem. C 2019, 7, 8771–8795. [Google Scholar] [CrossRef]
- HP 61XL High Yield Black Original Ink Cartridge, CH563WN#140. Available online: https://store.hp.com/us/en/pdp/hp-61xl-high-yield-black-original-ink-cartridge-p-ch563wn-140--1 (accessed on 5 April 2020).
- Pandey, S.P.; Shukla, T.; Dhote, V.K.; Mishra, D.K.; Maheshwari, R.; Tekade, R.K. Use of polymers in controlled release og active agents. In Basic Fundamentals of Drug Delivery; Academic Press: Cambridge, MA, USA, 2018; pp. 114–173. ISBN 978-0-12-817910-9. [Google Scholar]
- Phadtare, D.; Phadtare, G.; Asawat, M. Hypromellose: A choice of polymer in extended release tablet formulation. World J. Pharm. Pharm. Sci. 2014, 3, 551–566. [Google Scholar]
- Brindle, L.; Krochta, J. Physical Properties of Whey Protein-Hydroxypropylmethylcellulose Blend Edible Films. J. Food Sci. 2008, 73, E446–E454. [Google Scholar] [CrossRef]
- Bodini, R.B.; Lapa-Guimarães, J.; Monaco-Lourenço, C.A.; De Carvalho, R.A. Effect of starch and hydroxypropyl methylcellulose polymers on the properties of orally disintegrating films. J. Drug Deliv. Sci. Technol. 2019, 51, 403–410. [Google Scholar] [CrossRef]
- Ghadermazi, R.; Hamdipour, S.; Sadeghi, K.; Ghadermazi, R.; Asl, A.K. Effect of various additives on the properties of the films and coatings derived from hydroxypropyl methylcellulose—A review. Food Sci. Nutr. 2019, 7, 3363–3377. [Google Scholar] [CrossRef] [PubMed]
- De Moura, M.; Avena-Bustillos, R.; McHugh, T.; Krochta, J.; Mattoso, L.H.C. Properties of Novel Hydroxypropyl Methylcellulose Films Containing Chitosan Nanoparticles. J. Food Sci. 2008, 73, N31–N37. [Google Scholar] [CrossRef] [PubMed]
- Uddhav, B.; Kajal, G.; Shalaka, D.; Sanjeevani, A. Miken Bhavsar In vitro study of mucoadhesive strength of polymers for mucoadhesive drug delivery systems. Int. J. Curr. Pharm. Res. 2009, 1, 42–46. [Google Scholar]
- Yu, T.; Andrews, G.P.; Jones, D.S. Mucoadhesion and Characterization of Mucoadhesive Properties. In Mucosal Delivery of Biopharmaceuticals; Springer Science and Business Media LLC.: Berlin/Heidelberg, Germany, 2014; pp. 35–58. [Google Scholar]
- Mašková, E.; Kubová, K.; Raimi-Abraham, B.T.; Vllasaliu, D.; Vohlídalová, E.; Turanek, J.; Mašek, J. Hypromellose–A traditional pharmaceutical excipient with modern applications in oral and oromucosal drug delivery. J. Control. Release 2020, 324, 695–727. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, Y. Viscoelastic Properties for Sol-Gel Transition. Rheology 2012, 29–58. [Google Scholar] [CrossRef]
- Boddupalli, B.M.; Mohammed, Z.N.K.; Nath, R.A.; Banji, D. Mucoadhesive drug delivery system: An overview. J. Adv. Pharm. Technol. Res. 2010, 1, 381–387. [Google Scholar] [CrossRef]
- Akinosho, H.; Hawkins, S.; Wicker, L. Hydroxypropyl methylcellulose substituent analysis and rheological properties. Carbohydr. Polym. 2013, 98, 276–281. [Google Scholar] [CrossRef]
- Mortazavian, E.; Dorkoosh, F.A.; Rafiee-Tehrani, M. Design, characterization and ex vivo evaluation of chitosan film integrating of insulin nanoparticles composed of thiolated chitosan derivative for buccal delivery of insulin. Drug Dev. Ind. Pharm. 2014, 40, 691–698. [Google Scholar] [CrossRef]
- Joshi, S.C. Sol-Gel Behavior of Hydroxypropyl Methylcellulose (HPMC) in Ionic Media Including Drug Release. Materials 2011, 4, 1861–1905. [Google Scholar] [CrossRef]
- Dawson, D.V.; Drake, D.R.; Hill, J.R.; Brogden, K.A.; Fischer, C.L.; Wertz, P.W. Organization, barrier function and antimicrobial lipids of the oral mucosa. Int. J. Cosmet. Sci. 2013, 35, 220–223. [Google Scholar] [CrossRef]
- Santi, P.; Pescina, S.; Nicoli, S.; Santi, P. New Insights on the Mechanism of Fatty Acids as Buccal Permeation Enhancers. Pharm. 2018, 10, 201. [Google Scholar] [CrossRef]
- Nicolazzo, J.A.; Reed, B.L.; Finnin, B.C. Buccal penetration enhancers—How do they really work? J. Control. Release 2005, 105, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Shah, S.; Tahir, M.; Safdar, A.; Riaz, R.; Shahzad, Y.; Rabbani, M.; Karim, S.; Murtaza, G. Effect of Permeation Enhancers on the Release Behavior and Permeation Kinetics of Novel Tramadol Lotions. Trop. J. Pharm. Res. 2013, 12, 27–32. [Google Scholar] [CrossRef]
- Caon, T.; Simões, C.M.O. Effect of Freezing and Type of Mucosa on Ex Vivo Drug Permeability Parameters. AAPS PharmSciTech 2011, 12, 587–592. [Google Scholar] [CrossRef]
- Sonia, T.A.; Sharma, C.P. Routes of administration of insulin. In Oral Delivery of Insulin; Elsevier BV: Amsterdam, The Netherlands, 2014; pp. 59–112. [Google Scholar]
- Morales, J.O.; Huang, S.; Williams, R.O.; McConville, J.T.; Williams, I.R.O. Films loaded with insulin-coated nanoparticles (ICNP) as potential platforms for peptide buccal delivery. Colloids Surf. B Biointerfaces 2014, 122, 38–45. [Google Scholar] [CrossRef]
- Thakral, S.; Thakral, N.K.; Majumdar, D.K. Eudragit: A technology evaluation. Expert Opin. Drug Deliv. 2012, 10, 131–149. [Google Scholar] [CrossRef]
- Cai, Z.; Wang, Y.; Zhu, L.-J.; Liu, Z.-Q. Nanocarriers: A General Strategy for Enhancement of Oral Bioavailability of Poorly Absorbed or Pre-Systemically Metabolized Drugs. Curr. Drug Metab. 2010, 11, 197–207. [Google Scholar] [CrossRef]

| LCMs | HD (nm) | PdI | ZP (mV) | EE (%) | DL (%) |
|---|---|---|---|---|---|
| Rho | 26.2 ± 2.5 | 0.251 ± 0.114 | −12.4 ± 3.6 | 98.3 ± 0.3 | 0.29 ± 0.03 |
| Ins | 16.6 ± 1.0 | 0.202 ± 0.049 | −2.6 ± 1.1 | 94.3 ± 3.8 | 0.03 ± 0.00 |
| Kinetic Model | Correlation Coefficient | Rho | Ins |
|---|---|---|---|
| Zero Order | 0.896 ± 0.025 | 0.939 ± 0.013 | |
| 0.447 ± 0.065 | 1.495 ± 0.203 | ||
| First Order | 0.909 ± 0.026 | 0.971 ± 0.003 | |
| 0.005 ± 0.001 | 0.058 ± 0.031 | ||
| Higuchi | 0.957 ± 0.016 | 0.979 ± 0.003 | |
| 3.567 ± 0.495 | 12.246 ± 1.638 | ||
| Korsmeyer-Peppas | 0.969 ± 0.012 | 0.989 ± 0.007 | |
| 5.332 ± 1.417 | 29.855 ± 4.167 | ||
| 0.401 ± 0.150 | 0.261 ± 0.014 |
| Films | Thickness (μm) | TS (MPa) | EB (%) | EM (MPa) |
|---|---|---|---|---|
| NL-HPMC | 78.70 ± 10.30 | 8.23 ± 0.96 | 73.63 ± 8.28 | 4.40 ± 0.98 |
| L-HPMC | 79.45 ± 11.39 | 7.45 ± 0.42 | 77.23 ± 8.80 | 4.37 ± 0.68 |
| Films | DF (mN) | WoA (mN/mm) |
|---|---|---|
| NL-EC | 5.27 ± 1.10 A | 13.95 ± 5.42 A |
| NL-HPMC | 30.86 ± 2.72 B | 32.32 ± 9.53 A |
| L-HPMC | 19.41 ± 4.70 C | 21.92 ± 10.78 A |
| Parameters | Rhodamine 123 | Insulin | ||||
|---|---|---|---|---|---|---|
| Solution | LCMs | Films | Solution | LCMs | Films | |
| 0.00029 ± 0.00004 A | 0.00024 ± 0.00004 A | 0.01720 ± 0.00318 B | 0.05293 ± 0.01245 A | 0.14838 ± 0.02679 A | 0.39803 ± 0.28664 B | |
| 0.036 ± 0.006 A | 0.044 ± 0.006 A | 2.599 ± 0.481 B | 0.165 ± 0.039 A | 0.462 ± 0.083 A | 1.239 ± 0.892 B | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chou, W.-H.; Galaz, A.; Jara, M.O.; Gamboa, A.; Morales, J.O. Drug-Loaded Lipid-Core Micelles in Mucoadhesive Films as a Novel Dosage Form for Buccal Administration of Poorly Water-Soluble and Biological Drugs. Pharmaceutics 2020, 12, 1168. https://doi.org/10.3390/pharmaceutics12121168
Chou W-H, Galaz A, Jara MO, Gamboa A, Morales JO. Drug-Loaded Lipid-Core Micelles in Mucoadhesive Films as a Novel Dosage Form for Buccal Administration of Poorly Water-Soluble and Biological Drugs. Pharmaceutics. 2020; 12(12):1168. https://doi.org/10.3390/pharmaceutics12121168
Chicago/Turabian StyleChou, Wai-Houng, Ariel Galaz, Miguel O. Jara, Alexander Gamboa, and Javier O. Morales. 2020. "Drug-Loaded Lipid-Core Micelles in Mucoadhesive Films as a Novel Dosage Form for Buccal Administration of Poorly Water-Soluble and Biological Drugs" Pharmaceutics 12, no. 12: 1168. https://doi.org/10.3390/pharmaceutics12121168
APA StyleChou, W.-H., Galaz, A., Jara, M. O., Gamboa, A., & Morales, J. O. (2020). Drug-Loaded Lipid-Core Micelles in Mucoadhesive Films as a Novel Dosage Form for Buccal Administration of Poorly Water-Soluble and Biological Drugs. Pharmaceutics, 12(12), 1168. https://doi.org/10.3390/pharmaceutics12121168







