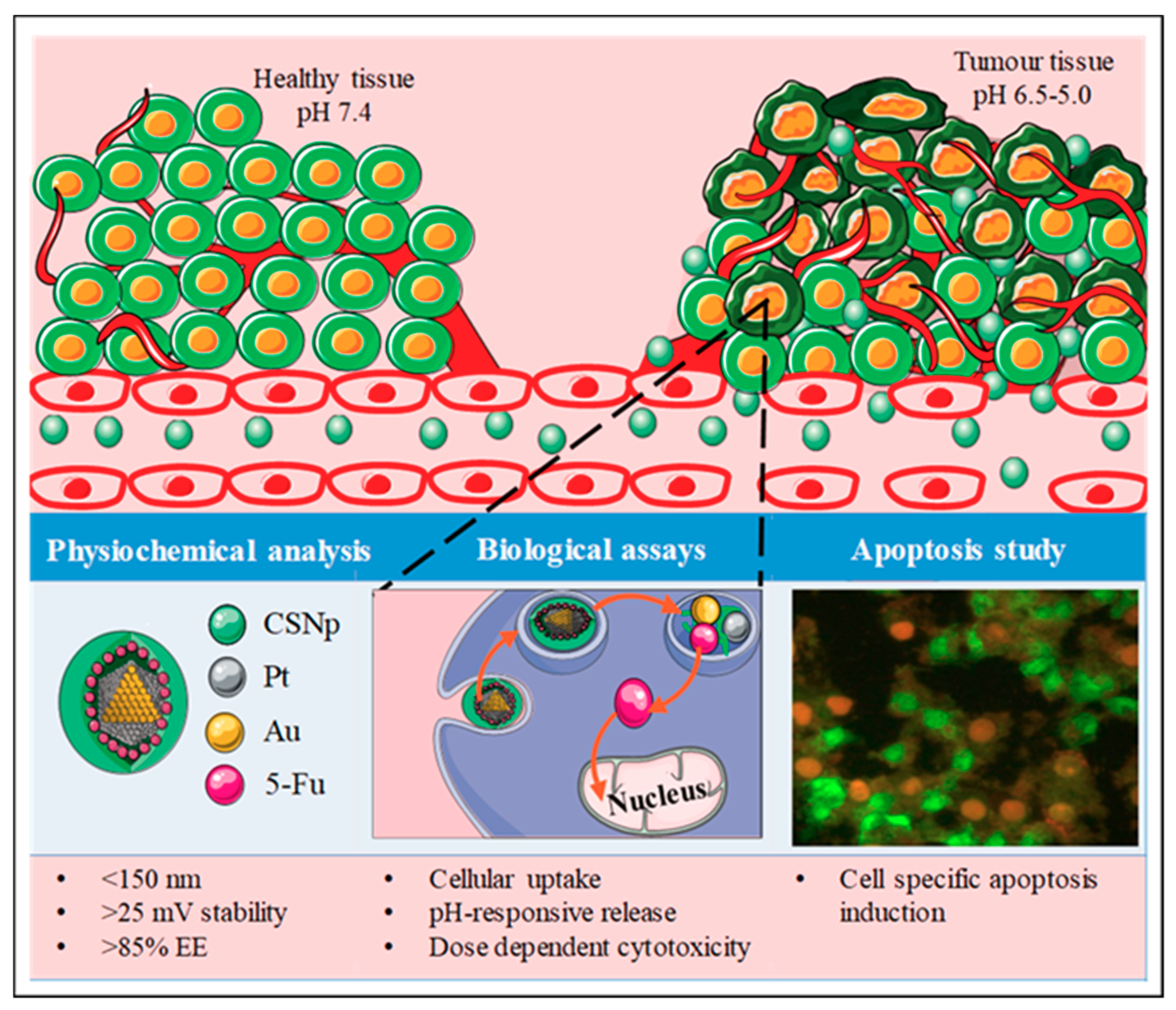
The Synergism of Platinum-Gold Bimetallic Nanoconjugates Enhances 5-Fluorouracil Delivery In Vitro
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Bimetallic PtAu Nanoparticles (PtAuBNps)
2.3. Preparation of Nanocomposites
2.4. Imaging, Nanoparticle Sizing and Zeta Potential Analysis
2.5. UV-Vis Spectrophotometry
2.6. Fourier Transform Infra-Red (FTIR) Spectroscopy
2.7. Drug Binding Studies
2.8. In Vitro Mucoadhesive Response
2.9. Pharmacokinetic Studies
2.10. In Vitro Cytotoxicity Assessment
2.11. Apoptosis Assay
2.12. Statistical Analysis
3. Results
3.1. Nanoparticle Morphology, Sizing and Zeta Potential
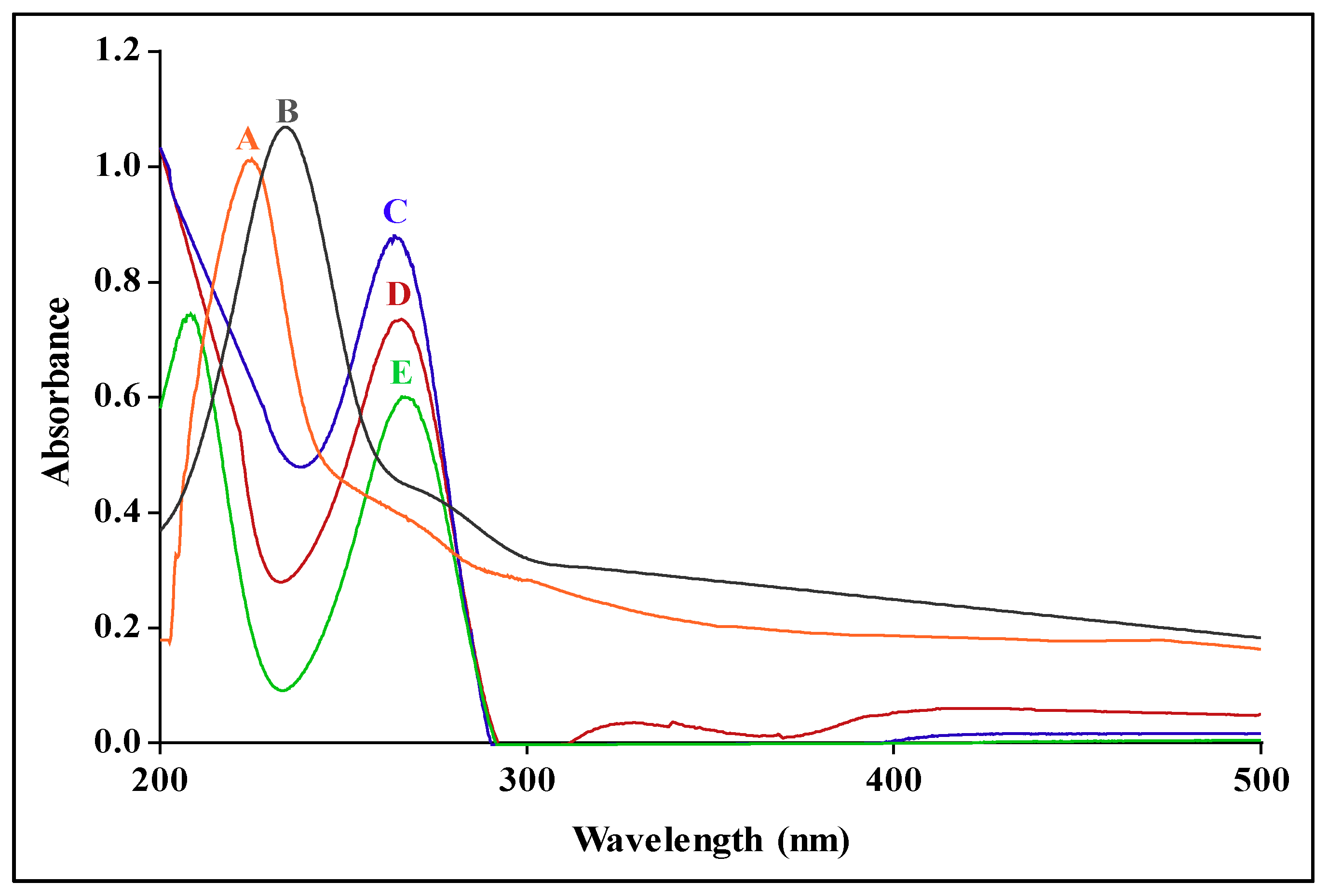
3.2. UV-Vis and FTIR Spectroscopy
3.3. Drug Binding Studies
3.4. In Vitro Mucoadhesive Response
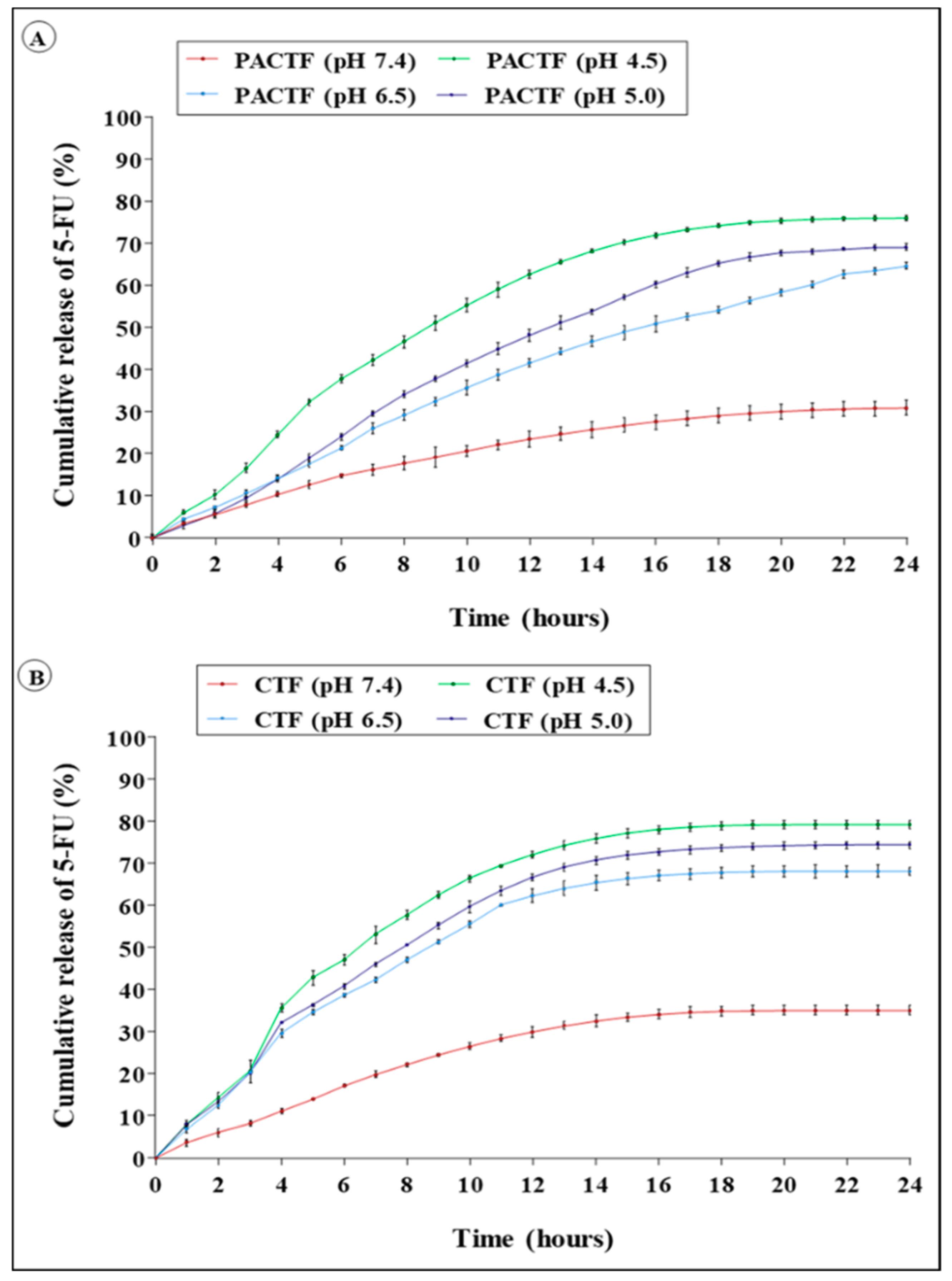
3.5. In Vitro Pharmacokinetics Studies
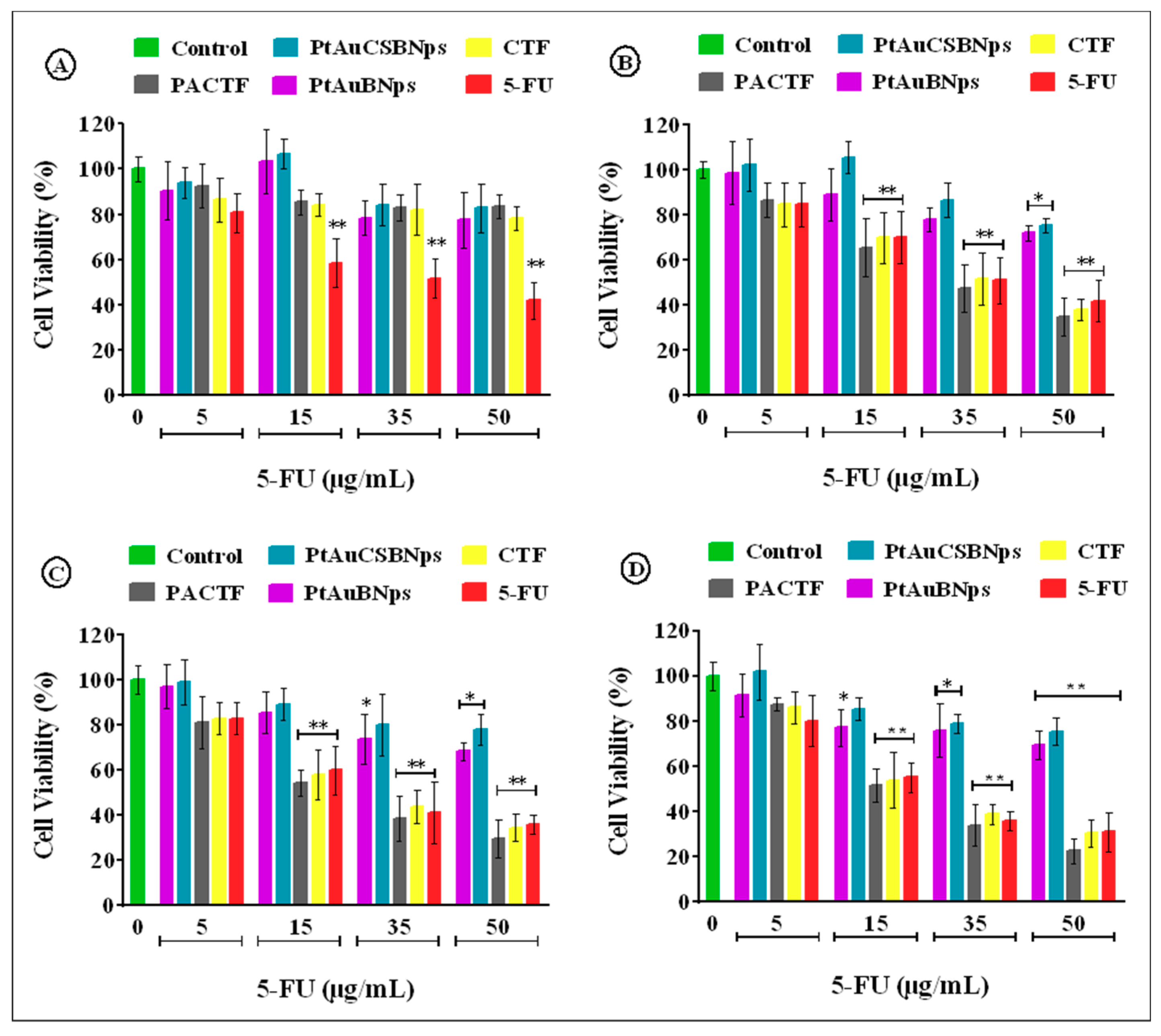
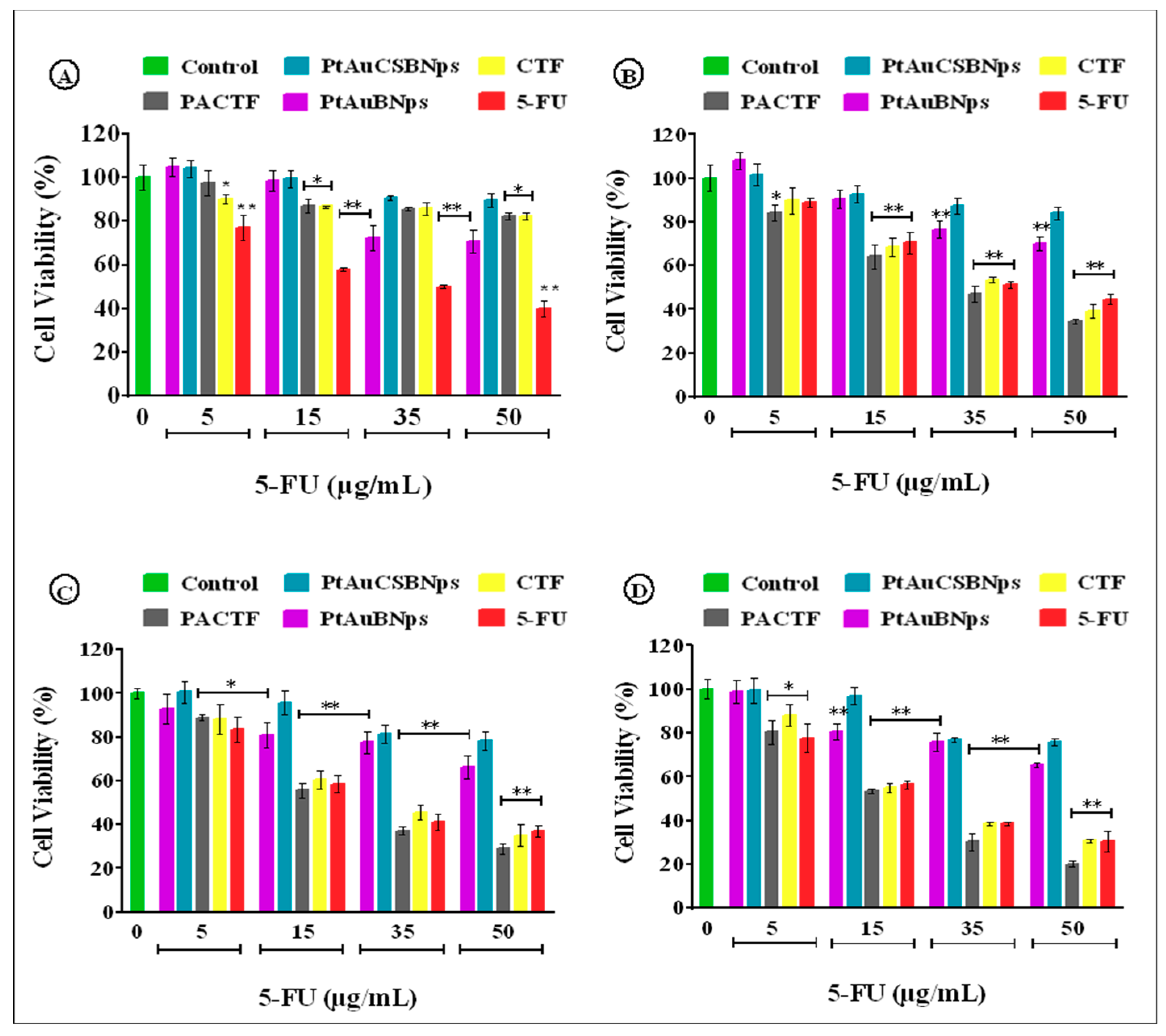
3.6. In Vitro Cytotoxicity
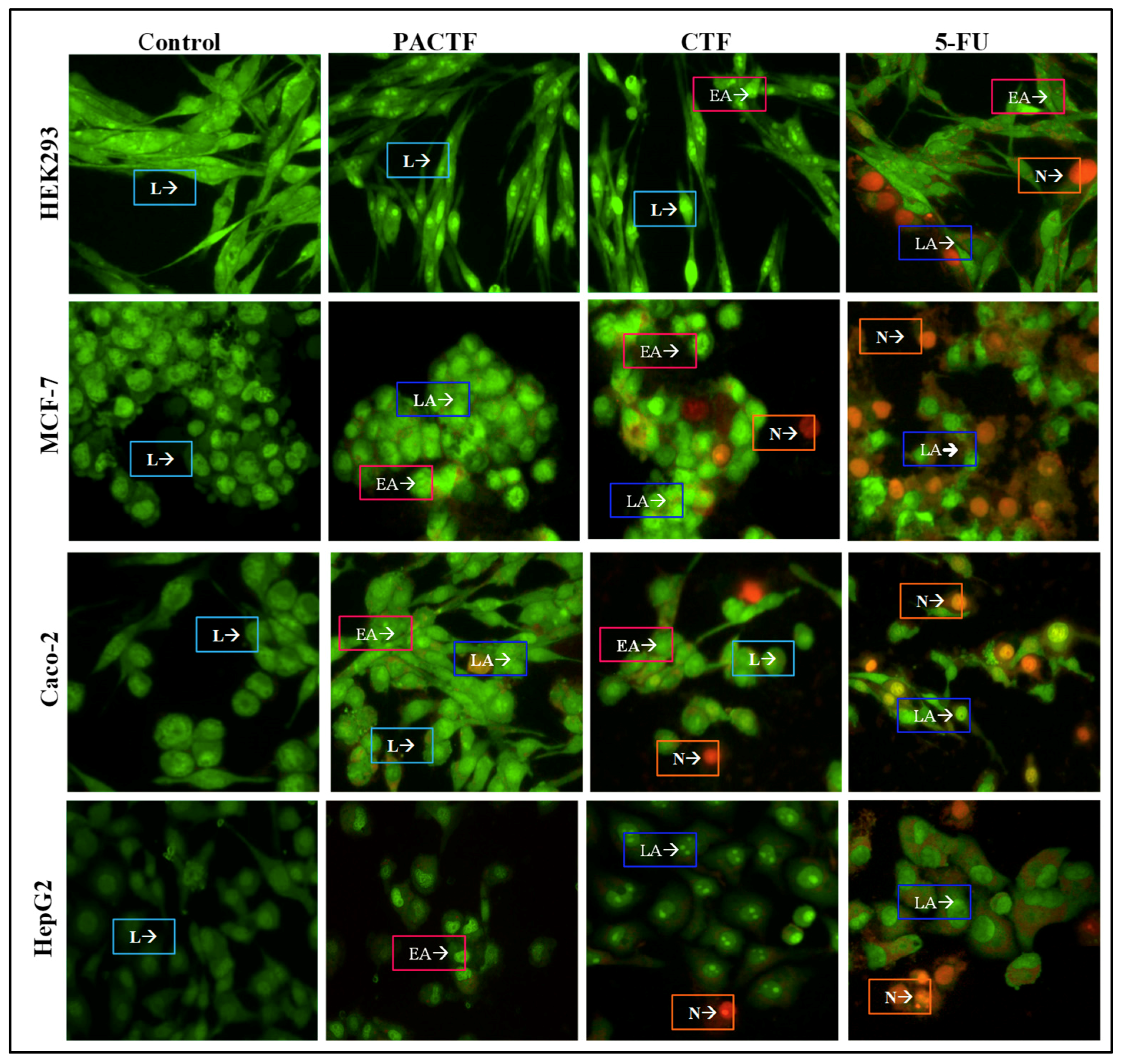
3.7. Apoptosis Induction Studies
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- El-Hammadi, M.M.; Delgado, Á.V.; Melguizo, C.; Prados, J.C.; Arias, J.L. Folic acid-decorated and PEGylated PLGA nanoparticles for improving the antitumour activity of 5-fluorouracil. Int. J. Pharm. 2017, 516, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Kevadiya, B.D.; Patel, T.A.; Jhala, D.D.; Thumbar, R.P.; Brahmbhatt, H.; Pandya, M.P.; Rajkumar, S.; Jena, P.K.; Joshi, G.V.; Gadhia, P.K.; et al. Layered inorganic nanocomposites: A promising carrier for 5-fluorouracil (5-FU). Eur. J. Pharm. Biopharm. 2012, 81, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Rejinold, N.S.; Thomas, R.G.; Muthiah, M.; Lee, H.J.; Jeong, Y.Y.; Park, I.-K.; Jayakumar, R. Breast Tumor Targetable Fe3O4 Embedded Thermo-Responsive Nanoparticles for Radiofrequency Assisted Drug Delivery. J. Biomed. Nanotechnol. 2016, 12, 43–55. [Google Scholar] [CrossRef] [PubMed]
- Reddy, A.B.; Manjula, B.; Jayaramudu, T.; Sadiku, E.; Babu, P.A.; Selvam, S.P. 5-Fluorouracil Loaded Chitosan-PVA/Na+. Nano-Micro Lett. 2016, 8, 260–269. [Google Scholar] [CrossRef] [PubMed]
- Couvreur, P.; Patrick, C. Nanoparticles in drug delivery: Past, present and future. Adv. Drug Deliv. Rev. 2013, 65, 21–23. [Google Scholar] [CrossRef] [PubMed]
- Lammers, T.; Kiessling, F.; Hennink, W.E.; Storm, G. Nanotheranostics and Image-Guided Drug Delivery: Current Concepts and Future Directions. Mol. Pharm. 2010, 7, 1899–1912. [Google Scholar] [CrossRef]
- Arvizo, R.R.; Bhattacharyya, S.; Kudgus, R.A.; Giri, K.; Bhattacharya, R.; Mukherjee, P. Intrinsic Therapeutic Applications of Noble Metal Nanoparticles: Past, Present and Future. Chem. Soc. Rev. 2012, 41, 2943–2970. [Google Scholar] [CrossRef] [PubMed]
- Misra, R.; Acharya, S.; Sahoo, S.K. Cancer nanotechnology: application of nanotechnology in cancer therapy. Drug Discov. Today 2010, 15, 842–850. [Google Scholar] [CrossRef]
- Buchtelova, H.; Dostalova, S.; Michalek, P.; Krizkova, S.; Strmiska, V.; Kopel, P.; Hynek, D.; Richtera, L.; Ridoskova, A.; Adam, P.; et al. Size-related cytotoxicological aspects of polyvinylpyrrolidone-capped platinum nanoparticles. Food Chem. Toxicol. 2017, 105, 337–346. [Google Scholar] [CrossRef]
- Yamada, M.; Foote, M.; Prow, T.W. Therapeutic gold, silver, and platinum nanoparticles. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2015, 7, 428–445. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, X.; Zhu, M.; Lin, G.; Liu, J.; Zhou, Z.; Tian, X.; Pan, Y. PEGylated Au@Pt Nanodendrites as Novel Theranostic Agents for Computed Tomography Imaging and Photothermal/Radiation Synergistic Therapy. ACS Appl. Mater. Interfaces 2016, 9, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.W.; Sie, E.J.; Liu, B.; Huan, C.H.A.; Sum, T.C.; Sun, H.D.; Fan, H.J. Surface plasmon enhanced band edge luminescence of ZnO nanorods by capping Au nanoparticles. Appl. Phys. Lett. 2010, 96, 071107. [Google Scholar] [CrossRef]
- Kumar, A.; Vimal, A.; Kumar, A. Why Chitosan? From properties to perspective of mucosal drug delivery. Int. J. Boil. Macromol. 2016, 91, 615–622. [Google Scholar] [CrossRef]
- Singla, A.; Chawla, M. Chitosan: some pharmaceutical and biological aspects—An update. J. Pharm. Pharmacol. 2001, 53, 1047–1067. [Google Scholar] [CrossRef]
- Elgadir, M.; Uddin, M.; Ferdosh, S.; Adam, A.; Chowdhury, A.J.K.; Sarker, M.I.; Ferdous, S. Impact of chitosan composites and chitosan nanoparticle composites on various drug delivery systems: A review. J. Food Drug Anal. 2015, 23, 619–629. [Google Scholar] [CrossRef] [PubMed]
- Chandran, P.R.; Sandhyarani, N. An electric field responsive drug delivery system based on chitosan–gold nanocomposites for site specific and controlled delivery of 5-fluorouracil. RSC Adv. 2014, 4, 44922–44929. [Google Scholar] [CrossRef]
- Maney, V.; Singh, M. An in vitro assessment of novel chitosan/bimetallic PtAu nanocomposites as delivery vehicles for doxorubicin. Nanomedicine 2017, 12, 2625–2640. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Rao, Y.; Chen, J.; Yang, V.C.; Liang, W. Polysorbate Cationic Synthetic Vesicle For Gene Delivery. J. Biomed. Mater. Res. Part A 2011, 96, 513–519. [Google Scholar] [CrossRef]
- Zhang, H.; Toshima, N. Synthesis of Au/Pt bimetallic nanoparticles with a Pt-rich shell and their high catalytic activities for aerobic glucose oxidation. J. Colloid Interface Sci. 2013, 394, 166–176. [Google Scholar] [CrossRef]
- Ekrami-Kakhki, M.S.; Khorasani-Motlagh, M.; Noroozifar, M. Platinum nanoparticles self-assembled onto chitosan membrane as anode for direct methanol fuel cell. J. Appl. Electrochem. 2001, 41, 527–534. [Google Scholar] [CrossRef]
- Khalil, M.M.; Mostafa, Y.M.; Torad, E. Biosynthesis and characterization of Pt and Au-Pt nanoparticles and their photo catalytic degradation of methylene blue. Int. J. Adv. Res. 2014, 2, 694–703. [Google Scholar]
- Singhvi, G.; Singh, M. Review: in-vitro drug release characterization models. Int. J. Pharm. Sci. Res. 2011, 2, 77–84. [Google Scholar]
- Patel, N.; Chotai, N.; Patel, J.; Soni, T.; Desai, J.; Patel, R. Comparison of In Vitro Dissolution Profiles of Oxcarbazepine-HP b-CD Tablet Formulations with Marketed Oxcarbazepine Tablets. Dissolution Technol. 2008, 15, 28–34. [Google Scholar] [CrossRef]
- Ramteke, K.; Dighe, P.; Kharat, A.; Patil, S. Mathematical models of drug dissolution: A review. Sch. Acad. J. Pharm. 2014, 3, 388–396. [Google Scholar]
- Testa, G.; Fontana, L.; Venditti, I.; Fratoddi, I. Functionalized platinum nanoparticles with surface charge trigged by pH: synthesis, characterization and stability studies. Beilstein J. Nanotechnol. 2016, 7, 1822–1828. [Google Scholar] [CrossRef] [PubMed]
- Nivethaa, E.A.K.; Dhanavel, S.; Vasu, C.A.; Narayanan, V.; Arumainathan, S. An in vitro cytotoxicity study of 5-fluorouracil encapsulated chitosan/gold nanocomposites towards MCF-7 cells. RSC Adv. 2015, 5, 1024–1032. [Google Scholar] [CrossRef]
- Sanyakamdhorn, S.; Agudelo, D.; Tajmir-Riahi, H.-A. Encapsulation of Antitumor Drug Doxorubicin and Its Analogue by Chitosan Nanoparticles. Biomacromolecules 2013, 14, 557–563. [Google Scholar] [CrossRef]
- Lawrie, G.; Keen, I.; Drew, B.; Chandler-Temple, A.; Rintoul, L.; Fredericks, P.; Grøndahl, L.; Lawrie, G. Interactions between Alginate and Chitosan Biopolymers Characterized Using FTIR and XPS. Biomacromolecules 2007, 8, 2533–2541. [Google Scholar] [CrossRef]
- Hou, J.; Yu, X.; Shen, Y.; Shi, Y.; Su, C.; Zhao, L. Triphenyl Phosphine-Functionalized Chitosan Nanoparticles Enhanced Antitumor Efficiency Through Targeted Delivery of Doxorubicin to Mitochondria. Nanoscale Res. Lett. 2017, 12, 415. [Google Scholar] [CrossRef]
- Mohammadpour Dounighi, N.; Eskandari, R.; Avadi, M.; Zolfagharian, H.; Mir Mohammad Sadeghi, A.; Rezayat, M. Preparation and in vitro characterization of chitosan nanoparticles containing Mesobuthus eupeus scorpion venom as an antigen delivery system. J. Venom. Anim. Toxins Incl. Trop. Dis. 2012, 18, 44–52. [Google Scholar]
- Akinyelu, J.; Singh, M. Chitosan Stabilized Gold-Folate-Poly(lactide-co-glycolide) Nanoplexes Facilitate Efficient Gene Delivery in Hepatic and Breast Cancer Cells. J. Nanosci. Nanotechnol. 2018, 18, 4478–4486. [Google Scholar] [CrossRef] [PubMed]
- Doostmohammadi, A.; Monshi, A.; Salehi, R.; Fathi, M.; Karbasi, S.; Pieles, U.; Daniels, A. Preparation, chemistry and physical properties of bone-derived hydroxyapatite particles having a negative zeta potential. Mater. Chem. Phys. 2012, 132, 446–452. [Google Scholar] [CrossRef]
- Chen, Z.; Zhang, C.; Tan, Y.; Zhou, T.; Ma, H.; Wan, C.; Lin, Y.; Li, K. Chitosan-functionalized gold nanoparti, cles for colorimetric detection of mercury ions based on chelation-induced aggregation. Microchim. Acta 2015, 182, 611–616. [Google Scholar] [CrossRef]
- Boyles, M.S.P.; Kristl, T.; Andosch, A.; Zimmermann, M.; Tran, N.; Casals, E.; Himly, M.; Puntes, V.; Huber, C.G.; Lütz-Meindl, U.; et al. Chitosan functionalisation of gold nanoparticles encourages particle uptake and induces cytotoxicity and pro-inflammatory conditions in phagocytic cells, as well as enhancing particle interactions with serum components. J. Nanobiotechnol. 2015, 13, 293. [Google Scholar] [CrossRef] [PubMed]
- Akinyelu, J.; Singh, M. Folate-tagged chitosan functionalised gold nanoparticles for enhanced delivery of 5-fluorouracil to cancer cells. Appl. Nanosci. 2019, 9, 7–17. [Google Scholar] [CrossRef]
- Hung, S.-F.; Yu, Y.-C.; Suen, N.-T.; Tzeng, G.-Q.; Tung, C.-W.; Hsu, Y.-Y.; Chang, C.-K.; Chan, T.-S.; Sheu, H.-S.; Lee, J.-F.; et al. The synergistic effect of a well-defined Au@Pt core–shell nanostructure toward photocatalytic hydrogen generation: interface engineering to improve the Schottky barrier and hydrogen-evolved kinetics. Chem. Commun. 2016, 52, 1567–1570. [Google Scholar] [CrossRef] [PubMed]
- Fan, F.-R.; Liu, D.-Y.; Wu, Y.-F.; Duan, S.; Xie, Z.-X.; Jiang, Z.-Y.; Tian, Z.-Q. Epitaxial Growth of Heterogeneous Metal Nanocrystals: From Gold Nano-octahedra to Palladium and Silver Nanocubes. J. Am. Chem. Soc. 2008, 130, 6949–6951. [Google Scholar] [CrossRef]
- Thanachayanont, C.; Hilborn, J.; Sugunan, A.; Dutta, J. Heavy-metal ion sensors using chitosan-capped gold nanoparticles. Sci. Technol. Adv. Mater. 2005, 6, 335–340. [Google Scholar]
- Guan, H.; Yu, J.; Chi, D. Label-free colorimetric sensing of melamine based on chitosan-stabilized gold nanoparticles probes. Food Control. 2013, 32, 35–41. [Google Scholar] [CrossRef]
- Wang, J.; Tauchi, Y.; Deguchi, Y.; Morimoto, K.; Tabata, Y.; Ikada, Y. Positively charged gelatin microspheres as gastric mucoadhesive drug delivery system for eradication of H. pylori. Drug Deliv. 2000, 7, 237–243. [Google Scholar]
- Nagarwal, R.C.; Kumar, R.; Pandit, J. Chitosan coated sodium alginate-chitosannanoparticles loaded with 5-FU for ocular delivery: In vitro characterization and in vivo study in rabbit eye. Eur. J. Pharm. Sci. 2012, 47, 678–685. [Google Scholar] [CrossRef] [PubMed]
- Pilicheva, B.; Zagorchev, P.; Uzunova, Y.; Kassarova, M. Development and in vitro Evaluation of mucoadhesive microsphere carriers for intranasal delivery of betahistine dihydrochloride. Int. J. Drug Deliv. 2013, 5, 389. [Google Scholar]
- Srivastava, G.; Walke, S.; Dhavale, D.; Gade, W.; Doshi, J.; Kumar, R.; Ravetkar, S.; Doshi, P. Tartrate/tripolyphosphate as co-crosslinker for water soluble chitosan used in protein antigens encapsulation. Int. J. Biol. Macromol. 2016, 91, 381–393. [Google Scholar] [CrossRef] [PubMed]
- Du, J.-Z.; Mao, C.-Q.; Yuan, Y.-Y.; Yang, X.-Z.; Wang, J. Tumor extracellular acidity-activated nanoparticles as drug delivery systems for enhanced cancer therapy. Biotechnol. Adv. 2014, 32, 789–803. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Yang, X.; Horte, S.; Kizhakkedathu, J.N.; Brooks, D.E. A pH and thermosensitive choline phosphate-based delivery platform targeted to the acidic tumor microenvironment. Biomaterials 2014, 35, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Kamaly, N.; Yameen, B.; Wu, J.; Farokhzad, O.C. Degradable Controlled-Release Polymers and Polymeric Nanoparticles: Mechanisms of Controlling Drug Release. Chem. Rev. 2016, 116, 2602–2663. [Google Scholar] [CrossRef] [PubMed]
- Kong, M.; Zuo, Y.; Wang, M.; Bai, X.; Feng, C.; Chen, X. Simply constructed chitosan nanocarriers with precise spatiotemporal control for efficient intracellular drug delivery. Carbohydr. Polym. 2017, 169, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Dubey, R.R.; Parikh, R.H. Two-stage optimization process for formulation of chitosan microspheres. AAPS PharmSciTech 2004, 5, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Tang, C.; Yin, C. Glycyrrhizin-modified O-carboxymethyl chitosan nanoparticles as drug vehicles targeting hepatocellular carcinoma. Biomaterials 2012, 33, 7594–7604. [Google Scholar] [CrossRef]
- Rafiei, P.; Haddadi, A. Pharmacokinetic Consequences of PLGA Nanoparticles in Docetaxel Drug Delivery. Pharm. Nanotechnol. 2017, 5, 3–23. [Google Scholar] [CrossRef]
- Islam, A.; Yasin, T. Controlled delivery of drug from pH sensitive chitosan/poly(vinyl alcohol)blend. Carbohydr. Polym. 2012, 88, 1055–1060. [Google Scholar] [CrossRef]
- Streubel, A.; Siepmann, J.; Peppas, N.; Bodmeier, R. Bimodal drug release achieved with multi-layer matrix tablets: transport mechanisms and device design. J. Control. Release 2000, 69, 455–468. [Google Scholar] [CrossRef]
- Keepers, Y.P.; Pizao, P.E.; Peters, G.J.; Van Ark-Otte, J.; Winograd, B.; Pinedo, H.M. Comparison of the sulforhodamine B protein and tetrazolium (MTT) assays for in vitro chemosensitivity testing. Eur. J. Cancer Clin. Oncol. 1991, 27, 897–900. [Google Scholar] [CrossRef]
- Moodley, T.; Singh, M. Polymeric Mesoporous Silica Nanoparticles for Enhanced Delivery of 5-Fluorouracil In Vitro. Pharmaceutics 2019, 11, 288. [Google Scholar] [CrossRef] [PubMed]
- Babaei, M.; Abnous, K.; Taghdisi, S.M.; Peivandi, M.T.; Alibolandi, M.; Farzad, S.A.; Ramezani, M. Synthesis of theranostic epithelial cell adhesion molecule targeted mesoporous silica nanoparticle with gold gatekeeper for hepatocellular carcinoma. Nanomedicine 2017, 12, 1261–1279. [Google Scholar] [CrossRef] [PubMed]
- Sahu, P.; Kashaw, S.K.; Jain, S.; Sau, S.; Iyer, A.K. Assessment of penetration potential of pH responsive double walled biodegradable nanogels coated with eucalyptus oil for the controlled delivery of 5-fluorouracil: In vitro and ex vivo studies. J. Control. Release 2017, 253, 122–136. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Liu, P.-C.; Liu, R.; Wu, X. Dual AO/EB Staining to Detect Apoptosis in Osteosarcoma Cells Compared with Flow Cytometry. Med Sci. Monit. Basic Res. 2015, 21, 15–20. [Google Scholar]
- Koff, J.L.; Ramachandiran, S.; Bernal-Mizrachi, L. A Time to Kill: Targeting Apoptosis in Cancer. Int. J. Mol. Sci. 2015, 16, 2942–2955. [Google Scholar] [CrossRef]
- Ćurčić, M.G.; Stanković, M.S.; Mrkalić, E.M.; Matović, Z.D.; Banković, D.D.; Cvetković, D.M.; Đačić, D.S.; Marković, S.D. Antiproliferative and proapoptotic activities of methanolic extracts from Ligustrum vulgare L. as an individual treatment and in combination with palladium complex. Int. J. Mol. Sci. 2012, 13, 2521–2534. [Google Scholar] [CrossRef]
- Fadok, V.A.; de Cathelineau, A.; Daleke, D.L.; Henson, P.M.; Bratton, D.L. Loss ofphospholipid asymmetry and surface exposure of phosphatidylserine is required for phagocytosis of apoptotic cells by macrophages and fibroblasts. J. Biol. Chem. 2001, 276, 1071–1077. [Google Scholar] [CrossRef]
- Bezabeh, T.; Mowat, M.R.A.; Jarolim, L.; Greenberg, A.H.; Smith, I.C.P. Detection of drug-induced apoptosis and necrosis in human cervical carcinoma cells using 1H NMR spectroscopy. Cell Death Differ. 2001, 8, 219–224. [Google Scholar] [CrossRef] [PubMed][Green Version]


| Kinetic Model | Equation |
|---|---|
| Zero-order | Rt = R0 + K0t |
| First-order | Ln Rt = lnR0+ K1t |
| Higuchi | Rt = KHt1/2 |
| Korsmeyer–Peppas | Rt/R∞ = Kktn |
| Sample | Particle Size (nm) | ζ Potential (mV) |
|---|---|---|
| PtAuBNps | 69.9 ± 3.2 | −21.5 ± 1.4 |
| PtAuCSBNps | 88.4 ± 10.8 | 58.2 ± 1.1 |
| PACTF | 108.6 ± 8.2 | 30.5 ± 0.6 |
| CTF | 118.8 ± 8.6 | 28.3 ± 2.6 |
| Sample | TDC (µg) | EE (%) | ADC (µg) | LC (%) |
|---|---|---|---|---|
| CTF | 139.21 | 87.24 | 121.45 | 23.24 |
| PACTF | 96.34 | 90.17 | 88.80 | 22.56 |
| Compound | Mucoadhesion (%) |
|---|---|
| PtAuBNps | 8.72 ± 1.67 |
| PtAuCSBNps | 86.24 ± 3.82 |
| PACTF | 68.74 ± 2.87 |
| CTF | 60.05 ± 4.21 |
| 5-FU | 21.51 ± 3.28 |
| Cell Lines | IC50 (µg/mL)—MTT Assay | IC50 (µg/mL)—SRB Assay | ||||
|---|---|---|---|---|---|---|
| PACTF | CTF | 5-FU | PACTF | CTF | 5-FU | |
| HEK293 | - | - | 31.79 | - | - | 30.84 |
| MCF-7 | 29.73 | 33.57 | 36.06 | 30.12 | 34.99 | 38.67 |
| HepG2 | 22.85 | 25.21 | 25.27 | 23.10 | 26.24 | 25.38 |
| Caco-2 | 18.98 | 21.99 | 20.41 | 19.25 | 22.58 | 21.19 |
| Cell Lines | Apoptosis Index | ||
|---|---|---|---|
| PACTF | CTF | 5-FU | |
| HEK293 | 0.034 | 0.052 | 0.389 |
| MCF-7 | 0.345 | 0.321 | 0.361 |
| HepG2 | 0.542 | 0.512 | 0.549 |
| Caco-2 | 0.621 | 0.549 | 0.624 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maney, V.; Singh, M. The Synergism of Platinum-Gold Bimetallic Nanoconjugates Enhances 5-Fluorouracil Delivery In Vitro. Pharmaceutics 2019, 11, 439. https://doi.org/10.3390/pharmaceutics11090439
Maney V, Singh M. The Synergism of Platinum-Gold Bimetallic Nanoconjugates Enhances 5-Fluorouracil Delivery In Vitro. Pharmaceutics. 2019; 11(9):439. https://doi.org/10.3390/pharmaceutics11090439
Chicago/Turabian StyleManey, Vareessh, and Moganavelli Singh. 2019. "The Synergism of Platinum-Gold Bimetallic Nanoconjugates Enhances 5-Fluorouracil Delivery In Vitro" Pharmaceutics 11, no. 9: 439. https://doi.org/10.3390/pharmaceutics11090439
APA StyleManey, V., & Singh, M. (2019). The Synergism of Platinum-Gold Bimetallic Nanoconjugates Enhances 5-Fluorouracil Delivery In Vitro. Pharmaceutics, 11(9), 439. https://doi.org/10.3390/pharmaceutics11090439






