Gold—Polyoxoborates Nanocomposite Prohibits Adsorption of Bacteriophages on Inner Surfaces of Polypropylene Labware and Protects Samples from Bacterial and Yeast Infections
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. BOA Surface Modification
2.3. Bacteriophages
2.4. Bacteria
2.5. Yeasts
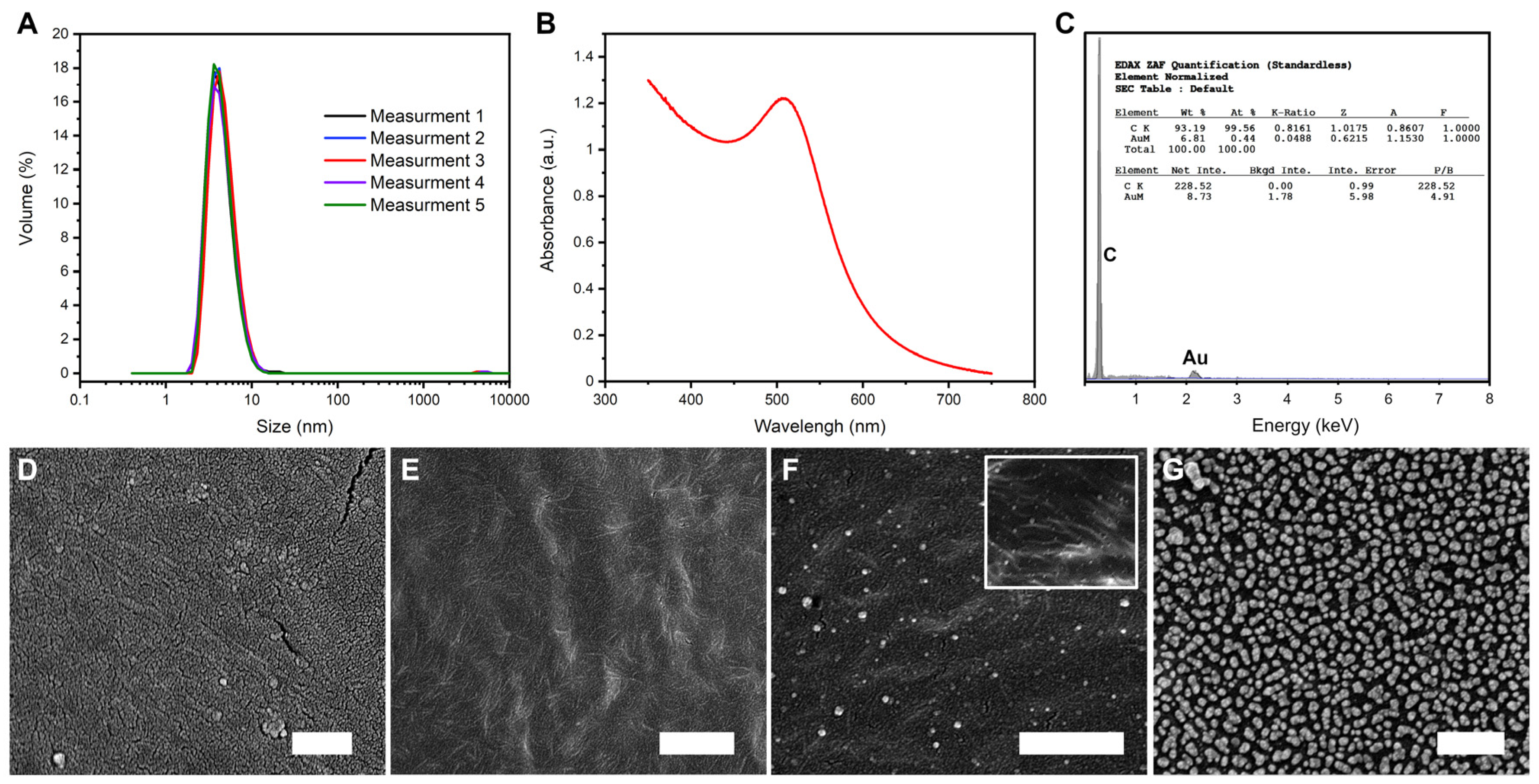
2.6. Instrumentation
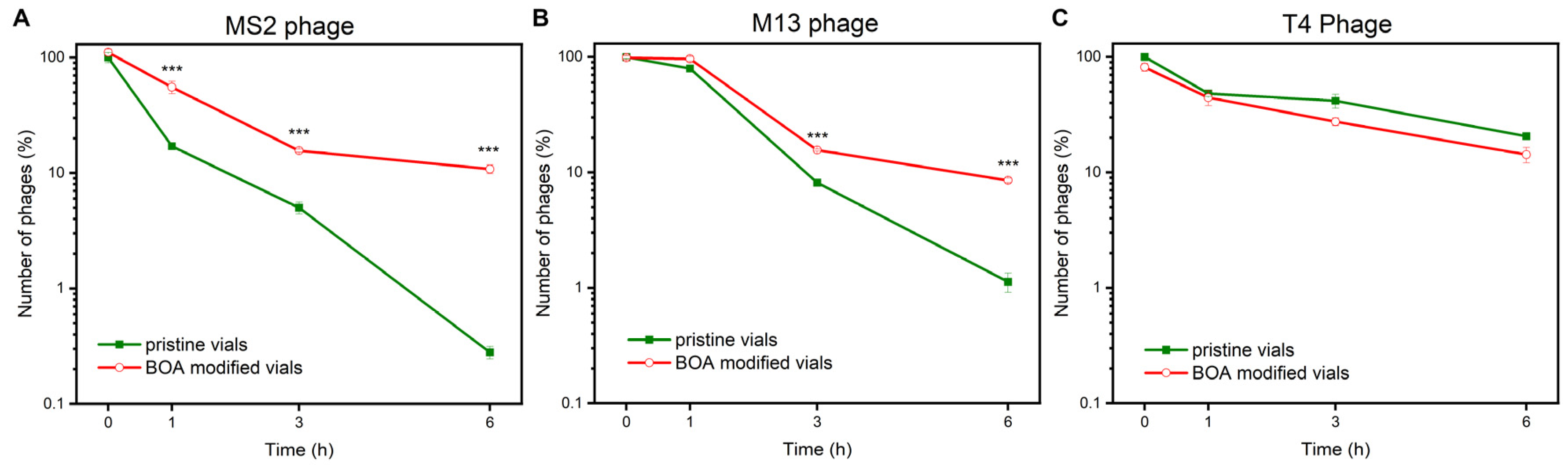
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sulakvelidze, A.; Alavidze, Z.J.; Morris, J.G., Jr. Bacteriophage Therapy. Antimicrob. Agents Chemother. 2001, 45, 649–659. [Google Scholar] [CrossRef] [Green Version]
- Parfitt, T. Georgia: An unlikely stronghold for bacteriophage therapy. Lancet 2005, 365, 2166–2167. [Google Scholar] [CrossRef]
- Wright, A.; Hawkins, C.H.; Änggård, E.E.; Harper, D.R. A controlled clinical trial of a therapeutic bacteriophage preparation in chronic otitis due to antibiotic-resistant Pseudomonas aeruginosa; A preliminary report of efficacy. Clin. Otolaryngol. 2009, 34, 349–357. [Google Scholar] [CrossRef] [PubMed]
- Leitner, L.; Ujmajuridze, A.; Chanishvili, N.; Goderdzishvili, M.; Chkonia, I.; Rigvava, S.; Chkhotua, A.; Changashvili, G.; McCallin, S.; Schneider, M.P.; et al. Intravesical bacteriophages for treating urinary tract infections in patients undergoing transurethral resection of the prostate: A randomised, placebo-controlled, double-blind clinical trial. Lancet Infect. Dis. 2020, 3099, 1–10. [Google Scholar] [CrossRef]
- Speck, P.; Smithyman, A. Safety and efficacy of phage therapy via the intravenous route. FEMS Microbiol. Lett. 2015, 363, 1–5. [Google Scholar] [CrossRef]
- Aslam, S.; Lampley, E.; Wooten, D.; Karris, M.; Benson, C.; Strathdee, S.; Schooley, R.T. Lessons learned from the first 10 consecutive cases of intravenous bacteriophage therapy to treat multidrug-resistant bacterial infections at a single center in the United States. Open Forum Infect. Dis. 2020, 7. [Google Scholar] [CrossRef]
- Jault, P.; Leclerc, T.; Jennes, S.; Pirnay, J.P.; Que, Y.-A.; Resch, G.; Rousseau, A.F.; Ravat, F.; Carsin, H.; Le Floch, R.; et al. Efficacy and tolerability of a cocktail of bacteriophages to treat burn wounds infected by Pseudomonas aeruginosa (PhagoBurn): A randomised, controlled, double-blind phase 1/2 trial. Lancet Infect. Dis. 2018, 19, 35–45. [Google Scholar] [CrossRef]
- Brigati, J.R.; Petrenko, V.A. Thermostability of landscape phage probes. Anal. Bioanal. Chem. 2005, 382, 1346–1350. [Google Scholar] [CrossRef] [PubMed]
- Nobrega, F.L.; Costa, A.R.; Santos, J.F.; Siliakus, M.F.; Van Lent, J.W.M.; Kengen, S.W.M.; Azeredo, J.; Kluskens, L.D. Genetically manipulated phages with improved pH resistance for oral administration in veterinary medicine. Sci. Rep. 2016, 6, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Olofsson, L.; Ankarloo, J.; Nicholls, I.A. Phage viability in organic media: Insights into phage stability. J. Mol. Recognit. 1998, 11, 91–93. [Google Scholar] [CrossRef]
- Olofsson, L.; Ankarloo, J.; Andersson, P.O.; Nicholls, I.A. Filamentous bacteriophage stability in non-aqueous media. Chem. Biol. 2001, 8, 661–671. [Google Scholar] [CrossRef] [Green Version]
- Jończyk-Matysiak, E.; Łodej, N.; Kula, D.; Owczarek, B.; Orwat, F.; Międzybrodzki, R.; Neuberg, J.; Bagińska, N.; Weber-Dąbrowska, B.; Górski, A. Factors determining phage stability/activity: Challenges in practical phage application. Expert Rev. Anti. Infect. Ther. 2019, 17, 583–606. [Google Scholar] [CrossRef] [PubMed]
- Richter, Ł.; Księżarczyk, K.; Paszkowska, K.; Janczuk-Richter, M.; Niedziółka-Jönsson, J.; Gapiński, J.; Łoś, M.; Hołyst, R.; Paczesny, J. Adsorption of bacteriophages on polypropylene labware affects the reproducibility of phage research. Sci. Rep. 2021, 11, 7387. [Google Scholar] [CrossRef] [PubMed]
- Rabe, M.; Verdes, D.; Seeger, S. Understanding protein adsorption phenomena at solid surfaces. Adv. Colloid Interface Sci. 2011, 162, 87–106. [Google Scholar] [CrossRef] [Green Version]
- Woodcock, S.E.; Johnson, W.C.; Chen, Z. Collagen adsorption and structure on polymer surfaces observed by atomic force microscopy. J. Colloid Interface Sci. 2005, 292, 99–107. [Google Scholar] [CrossRef]
- Krisdhasima, V.; McGuire, J.; Sproull, R. Surface hydrophobic influences on β-lactoglobulin adsorption kinetics. J. Colloid Interface Sci. 1992, 154, 337–350. [Google Scholar] [CrossRef]
- Malik, D.J.; Sokolov, I.J.; Vinner, G.K.; Mancuso, F.; Cinquerrui, S.; Vladisavljevic, G.T.; Clokie, M.R.J.; Garton, N.J.; Stapley, A.G.F.; Kirpichnikova, A. Formulation, stabilisation and encapsulation of bacteriophage for phage therapy. Adv. Colloid Interface Sci. 2017, 249, 100–133. [Google Scholar] [CrossRef] [Green Version]
- Rios, A.C.; Vila, M.M.D.C.; Lima, R.; Del Fiol, F.S.; Tubino, M.; Teixeira, J.A.; Balcão, V.M. Structural and functional stabilization of bacteriophage particles within the aqueous core of a W/O/W multiple emulsion: A potential biotherapeutic system for the inhalational treatment of bacterial pneumonia. Process Biochem. 2018, 64, 177–192. [Google Scholar] [CrossRef] [Green Version]
- Leung, V.; Szewczyk, A.; Chau, J.; Hosseinidoust, Z.; Groves, L.; Hawsawi, H.; Anany, H.; Griffiths, M.W.; Monsur Ali, M.; Filipe, C.D.M. Long-Term Preservation of Bacteriophage Antimicrobials Using Sugar Glasses. ACS Biomater. Sci. Eng. 2018, 4, 3802–3808. [Google Scholar] [CrossRef]
- Leung, V.; Groves, L.; Szewczyk, A.; Hosseinidoust, Z.; Filipe, C.D.M. Long-Term Antimicrobial Activity of Phage-Sugar Glasses is Closely Tied to the Processing Conditions. ACS Omega 2018, 3, 18295–18303. [Google Scholar] [CrossRef] [Green Version]
- Meyer, A.; Greene, M.; Kimmelshue, C.; Cademartiri, R. Stabilization of T4 bacteriophage at acidic and basic pH by adsorption on paper. Colloids Surfaces B Biointerfaces 2017, 160, 169–176. [Google Scholar] [CrossRef]
- Puapermpoonsiri, U.; Ford, S.J.; van der Walle, C.F. Stabilization of bacteriophage during freeze drying. Int. J. Pharm. 2010, 389, 168–175. [Google Scholar] [CrossRef]
- Wolfson, L.J.; Gasse, F.; Lee-Martin, S.P.; Lydon, P.; Magan, A.; Tibouti, A.; Johns, B.; Hutubessy, R.; Salama, P.; Okwo-Bele, J.M. Estimating the costs of achieving the WHO-UNICEF Global Immunization Vision and Strategy, 2006-2015. Bull. World Health Organ. 2008, 86, 27–39. [Google Scholar] [CrossRef]
- Braun, L.T.J.; Jezek, J.; Peterson, S.; Tyagi, A.; Perkins, S.; Sylvester, D.; Guy, M.; Lal, M.; Priddy, S.; Plzak, H.; et al. Characterization of a thermostable hepatitis B vaccine formulation. Vaccine 2009. [Google Scholar] [CrossRef] [PubMed]
- Walter, T.S.; Ren, J.; Tuthill, T.J.; Rowlands, D.J.; Stuart, D.I.; Fry, E.E. A plate-based high-throughput assay for virus stability and vaccine formulation. J. Virol. Methods 2012, 185, 166–170. [Google Scholar] [CrossRef] [Green Version]
- Croyle, M.A.; Cheng, X.; Wilson, J.M. Development of formulations that enhance physical stability of viral vectors for gene therapy. Gene Ther. 2001, 8, 1281–1290. [Google Scholar] [CrossRef] [Green Version]
- Evans, R.K.; Nawrocki, D.K.; Isopi, L.A.; Williams, D.M.; Casimiro, D.R.; Chin, S.; Chen, M.; Zhu, D.M.; Shiver, J.W.; Volkin, D.B. Development of stable liquid formulations for adenovirus-based vaccines. J. Pharm. Sci. 2004. [Google Scholar] [CrossRef]
- Rexroad, J.; Wiethoff, C.M.; Green, A.P.; Kierstead, T.D.; Scott, M.O.; Middaugh, C.R. Structural stability of adenovirus type 5. J. Pharm. Sci. 2003. [Google Scholar] [CrossRef] [PubMed]
- Stewart, M.; Ward, S.J.; Drew, J. Use of adenovirus as a model system to illustrate a simple method using standard equipment and inexpensive excipients to remove live virus dependence on the cold-chain. Vaccine 2014. [Google Scholar] [CrossRef] [PubMed]
- Pelliccia, M.; Andreozzi, P.; Paulose, J.; D’Alicarnasso, M.; Cagno, V.; Donalisio, M.; Civra, A.; Broeckel, R.M.; Haese, N.; Jacob Silva, P.; et al. Additives for vaccine storage to improve thermal stability of adenoviruses from hours to months. Nat. Commun. 2016, 7, 13520. [Google Scholar] [CrossRef]
- Wybrańska, K.; Paczesny, J.; Serejko, K.; Sura, K.; Włodyga, K.; Dzięcielewski, I.; Jones, S.T.; Śliwa, A.; Wybrańska, I.; Hołyst, R.; et al. Gold-oxoborate nanocomposites and their biomedical applications. ACS Appl. Mater. Interfaces 2015, 7, 3931–3939. [Google Scholar] [CrossRef]
- Gilcrease, E.; Williams, R.; Goel, R. Evaluating the effect of silver nanoparticles on bacteriophage lytic infection cycle-a mechanistic understanding. Water Res. 2020, 181, 115900. [Google Scholar] [CrossRef]
- Gokulan, K.; Bekele, A.; Drake, K.; Khare, S. Responses of intestinal virome to silver nanoparticles: Safety assessment by classical virology, whole-genome sequencing and bioinformatics approaches. Int. J. Nanomedicine 2018, Volume 13, 2857–2867. [Google Scholar] [CrossRef] [Green Version]
- Shimabuku, Q.L.; Ueda-Nakamura, T.; Bergamasco, R.; Fagundes-Klen, M.R. Chick-Watson kinetics of virus inactivation with granular activated carbon modified with silver nanoparticles and/or copper oxide. Process Saf. Environ. Prot. 2018, 117, 33–42. [Google Scholar] [CrossRef]
- Cho, M.; Cates, E.L.; Kim, J.-H. Inactivation and surface interactions of MS-2 bacteriophage in a TiO2 photoelectrocatalytic reactor. Water Res. 2011, 45, 2104–2110. [Google Scholar] [CrossRef]
- Syngouna, V.I.; Chrysikopoulos, C.V. Inactivation of MS2 bacteriophage by titanium dioxide nanoparticles in the presence of quartz sand with and without ambient light. J. Colloid Interface Sci. 2017, 497, 117–125. [Google Scholar] [CrossRef] [PubMed]
- De Gusseme, B.; Sintubin, L.; Baert, L.; Thibo, E.; Hennebel, T.; Vermeulen, G.; Uyttendaele, M.; Verstraete, W.; Boon, N. Biogenic Silver for Disinfection of Water Contaminated with Viruses. Appl. Environ. Microbiol. 2010, 76, 1082–1087. [Google Scholar] [CrossRef] [Green Version]
- Shimabuku, Q.L.; Arakawa, F.S.; Fernandes Silva, M.; Ferri Coldebella, P.; Ueda-Nakamura, T.; Fagundes-Klen, M.R.; Bergamasco, R. Water treatment with exceptional virus inactivation using activated carbon modified with silver (Ag) and copper oxide (CuO) nanoparticles. Environ. Technol. 2017, 38, 2058–2069. [Google Scholar] [CrossRef]
- Mazurkow, J.M.; Yüzbasi, N.S.; Domagala, K.W.; Pfeiffer, S.; Kata, D.; Graule, T. Nano-Sized Copper (Oxide) on Alumina Granules for Water Filtration: Effect of Copper Oxidation State on Virus Removal Performance. Environ. Sci. Technol. 2020, 54, 1214–1222. [Google Scholar] [CrossRef]
- Cheng, R.; Kang, M.; Zhuang, S.; Wang, S.; Zheng, X.; Pan, X.; Shi, L.; Wang, J. Removal of bacteriophage f2 in water by Fe/Ni nanoparticles: Optimization of Fe/Ni ratio and influencing factors. Sci. Total Environ. 2019, 649, 995–1003. [Google Scholar] [CrossRef] [PubMed]
- Martin, M.N.; Basham, J.I.; Chando, P.; Eah, S.K. Charged gold nanoparticles in non-polar solvents: 10-min synthesis and 2D self-assembly. Langmuir 2010, 26, 7410–7417. [Google Scholar] [CrossRef]
- Sambrook, J.; Russel, D.W. Molecular Cloning: A Laboratory Manual; Cold Spring Harbor Press: Cold Spring Harbor, NY, USA, 2001. [Google Scholar]
- Bishop, M.; Shahid, N.; Yang, J.; Barron, A.R. Determination of the mode and efficacy of the cross-linking of guar by borate using MAS 11 B NMR of borate cross-linked guar in combination with solution 11 B NMR of model systems. Dalt. Trans. 2004, 2621–2634. [Google Scholar] [CrossRef]
- Davidson, M.G.; Hughes, A.K.; Marder, T.B.; Wade, K. Contemporary Boron Chemistry; Special Publications; Davidson, M.G., Wade, K., Marder, T.B., Hughes, A.K., Eds.; Royal Society of Chemistry: Cambridge, UK, 2000; ISBN 978-0-85404-835-9. [Google Scholar]
- Farafonov, V.S.; Nerukh, D. MS2 bacteriophage capsid studied using all-atom molecular dynamics. Interface Focus 2019, 9. [Google Scholar] [CrossRef]
- Coulliette, A.D.; Perry, K.A.; Fisher, E.M.; Edwards, J.R.; Shaffer, R.E.; Noble-Wang, J. MS2 Coliphage as a Surrogate for 2009 Pandemic Influenza A (H1N1) Virus (pH1N1) in Surface Survival Studies on N95 Filtering Facepiece Respirators. J. Int. Soc. Respir. Prot. 2014, 21, 14–22. [Google Scholar]
- Turgeon, N.; Michel, K.; Ha, T.L.; Robine, E.; Moineau, S.; Duchaine, C. Resistance of aerosolized bacterial viruses to four germicidal products. PLoS ONE 2016, 11, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Turgeon, N.; Toulouse, M.J.; Martel, B.; Moineau, S.; Duchaine, C. Comparison of five bacteriophages as models for viral aerosol studies. Appl. Environ. Microbiol. 2014. [Google Scholar] [CrossRef] [Green Version]
- Moon, J.-S.; Kim, W.-G.; Kim, C.; Park, G.-T.; Heo, J.; Yoo, S.Y.; Oh, J.-W. M13 Bacteriophage-Based Self-Assembly Structures and Their Functional Capabilities. Mini. Rev. Org. Chem. 2015, 12, 271–281. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salivar, W.O.; Tzagoloff, H.; Pratt, D. Some physical-chemical and biological properties of the rod-shaped coliphage M13. Virology 1964, 24, 359–371. [Google Scholar] [CrossRef]
- Sidhu, S.S. Engineering M13 for phage display. Biomol. Eng. 2001, 18, 57–63. [Google Scholar] [CrossRef]
- Ackermann, H.W. 5500 Phages examined in the electron microscope. Arch. Virol. 2007, 152, 227–243. [Google Scholar] [CrossRef] [PubMed]
- Keen, E.C. A century of phage research: Bacteriophages and the shaping of modern biology. BioEssays 2015. [Google Scholar] [CrossRef]
- Armanious, A.; Aeppli, M.; Jacak, R.; Refardt, D.; Sigstam, T.; Kohn, T.; Sander, M. Viruses at Solid-Water Interfaces: A Systematic Assessment of Interactions Driving Adsorption. Environ. Sci. Technol. 2016, 50, 732–743. [Google Scholar] [CrossRef]
- Passaretti, P.; Sun, Y.; Dafforn, T.R.; Oppenheimer, P.G. Determination and characterisation of the surface charge properties of the bacteriophage M13 to assist bio-nanoengineering. RSC Adv. 2020, 10, 25385–25392. [Google Scholar] [CrossRef]
- Richter, Ł.; Matuła, K.; Leśniewski, A.; Kwaśnicka, K.; Łoś, J.; Łoś, M.; Paczesny, J.; Hołyst, R. Ordering of bacteriophages in the electric field: Application for bacteria detection. Sensors Actuators B Chem. 2016, 224, 233–240. [Google Scholar] [CrossRef]
- Richter, Ł.; Bielec, K.; Leśniewski, A.; Łoś, M.; Paczesny, J.; Hołyst, R. Dense Layer of Bacteriophages Ordered in Alternating Electric Field and Immobilized by Surface Chemical Modification as Sensing Element for Bacteria Detection. ACS Appl. Mater. Interfaces 2017, 9, 19622–19629. [Google Scholar] [CrossRef] [PubMed]
- Hahladakis, J.N.; Velis, C.A.; Weber, R.; Iacovidou, E.; Purnell, P. An overview of chemical additives present in plastics: Migration, release, fate and environmental impact during their use, disposal and recycling. J. Hazard. Mater. 2018, 344, 179–199. [Google Scholar] [CrossRef] [PubMed]
- Grzeskowiak, R.; Gerke, N.; Ag, E. Leachables: Minimizing the Influence of Plastic Consumables on the Laboratory Workflows; Eppendorf: Hamburg, Germany, 2015; Volume 26, pp. 1–6. [Google Scholar]
- Garvie, E.I. the Growth of Escherichia Coli in Buffer Substrate and Distilled Water. J. Bacteriol. 1955, 69, 393–398. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Greulich, C.; Braun, D.; Peetsch, A.; Siebers, B.; Ko, M. The toxic effect of silver ions and silver nanoparticles towards bacteria and human cells occurs in the same concentration range. RSC Adv. 2012, 2, 6981–6987. [Google Scholar] [CrossRef]
- Leifert, C.; Woodward, S. Laboratory contamination management: The requirement for microbiological quality assurance. Plant Cell. Tissue Organ Cult. 1998, 52, 83–88. [Google Scholar] [CrossRef]
- Hernández, A.; Pérez-Nevado, F.; Ruiz-Moyano, S.; Serradilla, M.J.; Villalobos, M.C.; Martín, A.; Córdoba, M.G. Spoilage yeasts: What are the sources of contamination of foods and beverages? Int. J. Food Microbiol. 2018, 286, 98–110. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wdowiak, M.; Ochirbat, E.; Paczesny, J. Gold—Polyoxoborates Nanocomposite Prohibits Adsorption of Bacteriophages on Inner Surfaces of Polypropylene Labware and Protects Samples from Bacterial and Yeast Infections. Viruses 2021, 13, 1206. https://doi.org/10.3390/v13071206
Wdowiak M, Ochirbat E, Paczesny J. Gold—Polyoxoborates Nanocomposite Prohibits Adsorption of Bacteriophages on Inner Surfaces of Polypropylene Labware and Protects Samples from Bacterial and Yeast Infections. Viruses. 2021; 13(7):1206. https://doi.org/10.3390/v13071206
Chicago/Turabian StyleWdowiak, Mateusz, Enkhlin Ochirbat, and Jan Paczesny. 2021. "Gold—Polyoxoborates Nanocomposite Prohibits Adsorption of Bacteriophages on Inner Surfaces of Polypropylene Labware and Protects Samples from Bacterial and Yeast Infections" Viruses 13, no. 7: 1206. https://doi.org/10.3390/v13071206
APA StyleWdowiak, M., Ochirbat, E., & Paczesny, J. (2021). Gold—Polyoxoborates Nanocomposite Prohibits Adsorption of Bacteriophages on Inner Surfaces of Polypropylene Labware and Protects Samples from Bacterial and Yeast Infections. Viruses, 13(7), 1206. https://doi.org/10.3390/v13071206





