Mechanisms of Rhinovirus Neutralisation by Antibodies
Abstract
1. Introduction
2. Rhinoviruses
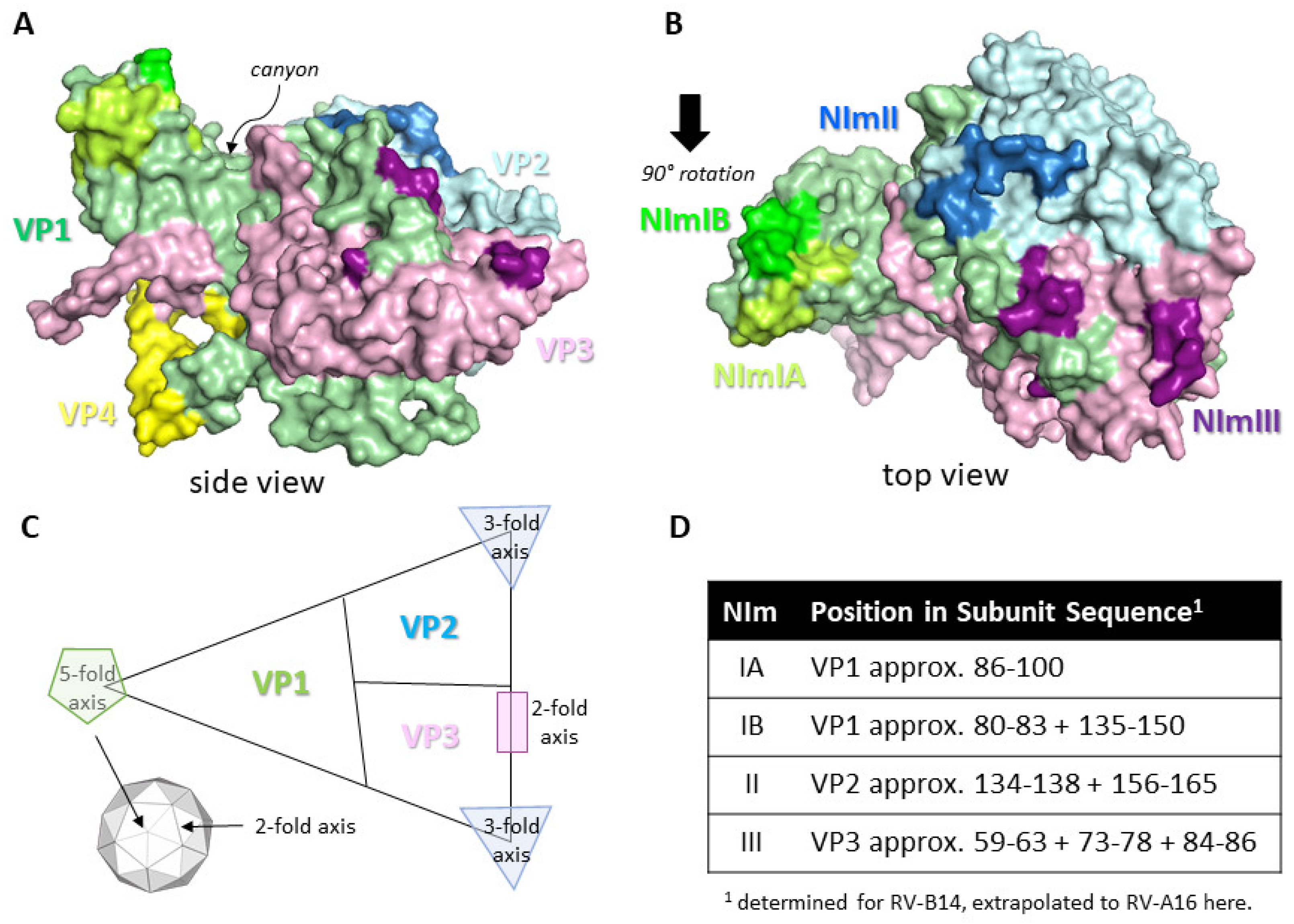
3. Capsid Protomer Structure, Receptor Binding and Neutralising Ab Sites
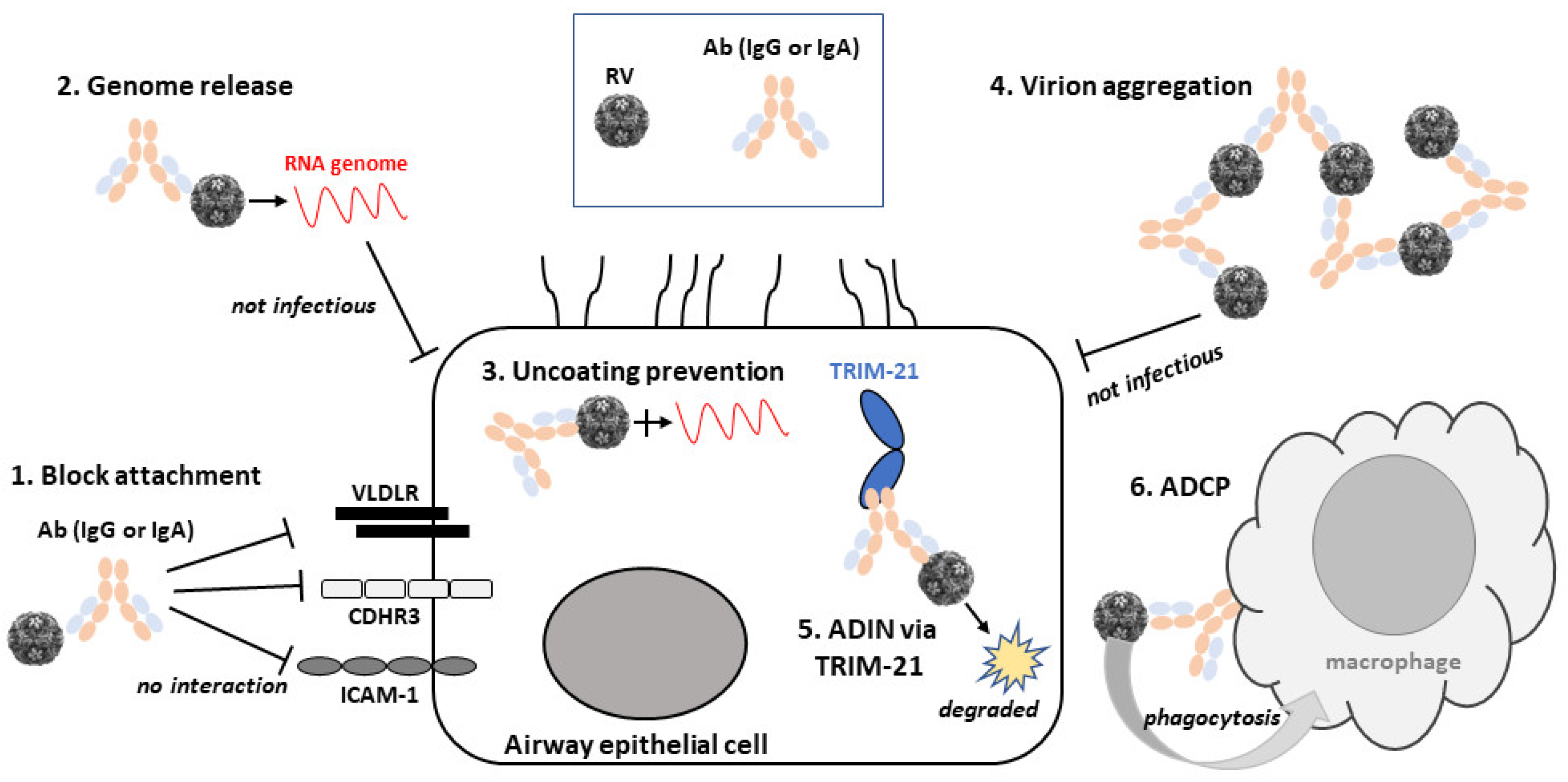
4. Mechanisms of RV Neutralisation by Abs
4.1. Abs That Block Attachment of RV to Cells
4.2. Ab Induction of RV Genome Release before Attachment to Cells
4.3. Prevention of Uncoating by Abs
4.4. Aggregation of RV by Ab Binding
4.5. Neutralisation Inside the Cell through TRIM21
4.6. Ab-Dependent Cellular Phagocytosis (ADCP)
5. Summary and Implications for RV Vaccine Development
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jacobs, S.E.; Lamson, D.M.; St George, K.; Walsh, T.J. Human rhinoviruses. Clin. Microbiol. Rev. 2013, 26, 135–162. [Google Scholar] [CrossRef]
- Greenberg, S.B. Respiratory consequences of rhinovirus infection. Arch. Intern. Med. 2003, 163, 278–284. [Google Scholar] [CrossRef]
- Johnston, S.L.; Pattemore, P.K.; Sanderson, G.; Smith, S.; Lampe, F.; Josephs, L.; Symington, P.; O’Toole, S.; Myint, S.H.; Tyrrell, D.A.; et al. Community study of role of viral infections in exacerbations of asthma in 9–11 year old children. BMJ (Clin. Res. Ed.) 1995, 310, 1225–1229. [Google Scholar] [CrossRef]
- Ritchie, A.I.; Farne, H.A.; Singanayagam, A.; Jackson, D.J.; Mallia, P.; Johnston, S.L. Pathogenesis of Viral Infection in Exacerbations of Airway Disease. Ann. Am. Thorac. Soc. 2015, 12 (Suppl. 2), S115–S132. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.J.; Arnold, J.C.; Fairchok, M.P.; Danaher, P.J.; McDonough, E.A.; Blair, P.J.; Garcia, J.; Halsey, E.S.; Schofield, C.; Ottolini, M.; et al. Epidemiologic, clinical, and virologic characteristics of human rhinovirus infection among otherwise healthy children and adults: Rhinovirus among adults and children. J. Clin. Virol. Off. Publ. Pan Am. Soc. Clin. Virol. 2015, 64, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Annamalay, A.A.; Khoo, S.K.; Jacoby, P.; Bizzintino, J.; Zhang, G.; Chidlow, G.; Lee, W.M.; Moore, H.C.; Harnett, G.B.; Smith, D.W.; et al. Prevalence of and risk factors for human rhinovirus infection in healthy aboriginal and non-aboriginal Western Australian children. Pediatr. Infect. Dis. J. 2012, 31, 673–679. [Google Scholar] [CrossRef]
- Price, W.H. The isolation of a new virus associated with respiratory clinical disease in humans. Proc. Natl. Acad. Sci. USA 1956, 42, 892–896. [Google Scholar] [CrossRef] [PubMed]
- Ortega, H.; Nickle, D.; Carter, L. Rhinovirus and asthma: Challenges and opportunities. Rev. Med. Virol. 2020, e2193. [Google Scholar] [CrossRef]
- McLean, G.R. Vaccine strategies to induce broadly protective immunity to rhinoviruses. Hum. Vaccines Immunother. 2020, 16, 684–686. [Google Scholar] [CrossRef] [PubMed]
- Williams, G.R.; Kubajewska, I.; Glanville, N.; Johnston, S.L.; McLean, G.R. The potential for a protective vaccine for rhinovirus infections. Expert Rev. Vaccines 2016, 15, 569–571. [Google Scholar] [CrossRef]
- Rossmann, M.G.; Arnold, E.; Erickson, J.W.; Frankenberger, E.A.; Griffith, J.P.; Hecht, H.J.; Johnson, J.E.; Kamer, G.; Luo, M.; Mosser, A.G.; et al. Structure of a human common cold virus and functional relationship to other picornaviruses. Nature 1985, 317, 145–153. [Google Scholar] [CrossRef]
- Greve, J.M.; Davis, G.; Meyer, A.M.; Forte, C.P.; Yost, S.C.; Marlor, C.W.; Kamarck, M.E.; McClelland, A. The major human rhinovirus receptor is ICAM-1. Cell 1989, 56, 839–847. [Google Scholar] [CrossRef]
- Hofer, F.; Gruenberger, M.; Kowalski, H.; Machat, H.; Huettinger, M.; Kuechler, E.; Blaas, D. Members of the low density lipoprotein receptor family mediate cell entry of a minor-group common cold virus. Proc. Natl. Acad. Sci. USA 1994, 91, 1839–1842. [Google Scholar] [CrossRef]
- Liu, Y.; Hill, M.G.; Klose, T.; Chen, Z.; Watters, K.; Bochkov, Y.A.; Jiang, W.; Palmenberg, A.C.; Rossmann, M.G. Atomic structure of a rhinovirus C, a virus species linked to severe childhood asthma. Proc. Natl. Acad. Sci. USA 2016, 113, 8997–9002. [Google Scholar] [CrossRef] [PubMed]
- Bochkov, Y.A.; Watters, K.; Ashraf, S.; Griggs, T.F.; Devries, M.K.; Jackson, D.J.; Palmenberg, A.C.; Gern, J.E. Cadherin-related family member 3, a childhood asthma susceptibility gene product, mediates rhinovirus C binding and replication. Proc. Natl. Acad. Sci. USA 2015, 112, 5485–5490. [Google Scholar] [CrossRef]
- Sherry, B.; Rueckert, R. Evidence for at least two dominant neutralization antigens on human rhinovirus 14. J. Virol. 1985, 53, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Sherry, B.; Mosser, A.G.; Colonno, R.J.; Rueckert, R.R. Use of monoclonal antibodies to identify four neutralization immunogens on a common cold picornavirus, human rhinovirus 14. J. Virol. 1986, 57, 246–257. [Google Scholar] [CrossRef] [PubMed]
- Appleyard, G.; Russell, S.M.; Clarke, B.E.; Speller, S.A.; Trowbridge, M.; Vadolas, J. Neutralization epitopes of human rhinovirus type 2. J. Gen. Virol. 1990, 71 Pt 6, 1275–1282. [Google Scholar] [CrossRef]
- Hastings, G.Z.; Speller, S.A.; Francis, M.J. Neutralizing antibodies to human rhinovirus produced in laboratory animals and humans that recognize a linear sequence from VP2. J. Gen. Virol. 1990, 71 Pt 12, 3055–3059. [Google Scholar] [CrossRef]
- Ping, L.H.; Jansen, R.W.; Stapleton, J.T.; Cohen, J.I.; Lemon, S.M. Identification of an immunodominant antigenic site involving the capsid protein VP3 of hepatitis A virus. Proc. Natl. Acad. Sci. USA 1988, 85, 8281–8285. [Google Scholar] [CrossRef] [PubMed]
- Pfaff, E.; Thiel, H.J.; Beck, E.; Strohmaier, K.; Schaller, H. Analysis of neutralizing epitopes on foot-and-mouth disease virus. J. Virol. 1988, 62, 2033–2040. [Google Scholar] [CrossRef] [PubMed]
- Dimmock, N.J. Mechanisms of neutralization of animal viruses. J. Gen. Virol. 1984, 65 Pt 6, 1015–1022. [Google Scholar] [CrossRef]
- VanBlargan, L.A.; Goo, L.; Pierson, T.C. Deconstructing the Antiviral Neutralizing-Antibody Response: Implications for Vaccine Development and Immunity. Microbiol. Mol. Biol. Rev. 2016, 80, 989–1010. [Google Scholar] [CrossRef] [PubMed]
- Dörner, T.; Radbruch, A. Antibodies and B cell memory in viral immunity. Immunity 2007, 27, 384–392. [Google Scholar] [CrossRef] [PubMed]
- Che, Z.; Olson, N.H.; Leippe, D.; Lee, W.M.; Mosser, A.G.; Rueckert, R.R.; Baker, T.S.; Smith, T.J. Antibody-mediated neutralization of human rhinovirus 14 explored by means of cryoelectron microscopy and X-ray crystallography of virus-Fab complexes. J. Virol. 1998, 72, 4610–4622. [Google Scholar] [CrossRef] [PubMed]
- Colonno, R.J.; Callahan, P.L.; Leippe, D.M.; Rueckert, R.R.; Tomassini, J.E. Inhibition of rhinovirus attachment by neutralizing monoclonal antibodies and their Fab fragments. J. Virol. 1989, 63, 36–42. [Google Scholar] [CrossRef]
- Thomas, A.A.; Brioen, P.; Boeyé, A. A monoclonal antibody that neutralizes poliovirus by cross-linking virions. J. Virol. 1985, 54, 7–13. [Google Scholar] [CrossRef]
- Smith, T.J.; Chase, E.S.; Schmidt, T.J.; Olson, N.H.; Baker, T.S. Neutralizing antibody to human rhinovirus 14 penetrates the receptor-binding canyon. Nature 1996, 383, 350–354. [Google Scholar] [CrossRef]
- Dong, Y.; Liu, Y.; Jiang, W.; Smith, T.J.; Xu, Z.; Rossmann, M.G. Antibody-induced uncoating of human rhinovirus B14. Proc. Natl. Acad. Sci. USA 2017, 114, 8017–8022. [Google Scholar] [CrossRef]
- Vrijsen, R.; Mosser, A.; Boeyé, A. Postabsorption neutralization of poliovirus. J. Virol. 1993, 67, 3126–3133. [Google Scholar] [CrossRef]
- Watkinson, R.E.; McEwan, W.A.; Tam, J.C.; Vaysburd, M.; James, L.C. TRIM21 Promotes cGAS and RIG-I Sensing of Viral Genomes during Infection by Antibody-Opsonized Virus. PLoS Pathog. 2015, 11, e1005253. [Google Scholar] [CrossRef]
- Fletcher, A.J.; James, L.C. Coordinated Neutralization and Immune Activation by the Cytosolic Antibody Receptor TRIM21. J. Virol. 2016, 90, 4856–4859. [Google Scholar] [CrossRef] [PubMed]
- Behzadi, M.A.; Choi, A.; Duehr, J.; Feyznezhad, R.; Upadhyay, C.; Schotsaert, M.; Palese, P.; Nachbagauer, R. A cross-reactive mouse monoclonal antibody against rhinovirus mediates phagocytosis in vitro. Sci. Rep. 2020, 10, 9750. [Google Scholar] [CrossRef] [PubMed]
- Tay, M.Z.; Wiehe, K.; Pollara, J. Antibody-Dependent Cellular Phagocytosis in Antiviral Immune Responses. Front. Immunol. 2019, 10, 332. [Google Scholar] [CrossRef] [PubMed]
- Edlmayr, J.; Niespodziana, K.; Popow-Kraupp, T.; Krzyzanek, V.; Focke-Tejkl, M.; Blaas, D.; Grote, M.; Valenta, R. Antibodies induced with recombinant VP1 from human rhinovirus exhibit cross-neutralisation. Eur. Respir. J. 2011, 37, 44–52. [Google Scholar] [CrossRef]
- McLean, G.R.; Walton, R.P.; Shetty, S.; Peel, T.J.; Paktiawal, N.; Kebadze, T.; Gogsadze, L.; Niespodziana, K.; Valenta, R.; Bartlett, N.W.; et al. Rhinovirus infections and immunisation induce cross-serotype reactive antibodies to VP1. Antivir. Res. 2012, 95, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.J.; Olson, N.H.; Cheng, R.H.; Chase, E.S.; Baker, T.S. Structure of a human rhinovirus-bivalently bound antibody complex: Implications for viral neutralization and antibody flexibility. Proc. Natl. Acad. Sci. USA 1993, 90, 7015–7018. [Google Scholar] [CrossRef]
- Smith, T.J.; Olson, N.H.; Cheng, R.H.; Liu, H.; Chase, E.S.; Lee, W.M.; Leippe, D.M.; Mosser, A.G.; Rueckert, R.R.; Baker, T.S. Structure of human rhinovirus complexed with Fab fragments from a neutralizing antibody. J. Virol. 1993, 67, 1148–1158. [Google Scholar] [CrossRef]
- Barnett, P.V.; Rowlands, D.J.; Parry, N.R. Characterization of monoclonal antibodies raised against a synthetic peptide capable of inducing a neutralizing response to human rhinovirus type 2. J. Gen. Virol. 1993, 74 Pt 7, 1295–1302. [Google Scholar] [CrossRef]
- Rossmann, M.G. The canyon hypothesis. Hiding the host cell receptor attachment site on a viral surface from immune surveillance. J. Biol. Chem. 1989, 264, 14587–14590. [Google Scholar] [CrossRef]
- Hewat, E.A.; Blaas, D. Structure of a neutralizing antibody bound bivalently to human rhinovirus 2. EMBO J. 1996, 15, 1515–1523. [Google Scholar] [CrossRef] [PubMed]
- Tormo, J.; Centeno, N.B.; Fontana, E.; Bubendorfer, T.; Fita, I.; Blaas, D. Docking of a human rhinovirus neutralizing antibody onto the viral capsid. Proteins 1995, 23, 491–501. [Google Scholar] [CrossRef]
- Hewat, E.A.; Marlovits, T.C.; Blaas, D. Structure of a neutralizing antibody bound monovalently to human rhinovirus 2. J. Virol. 1998, 72, 4396–4402. [Google Scholar] [CrossRef]
- Ye, X.; Fan, C.; Ku, Z.; Zuo, T.; Kong, L.; Zhang, C.; Shi, J.; Liu, Q.; Chen, T.; Zhang, Y.; et al. Structural Basis for Recognition of Human Enterovirus 71 by a Bivalent Broadly Neutralizing Monoclonal Antibody. PLoS Pathog. 2016, 12, e1005454. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhu, L.; Dang, M.; Hu, Z.; Gao, Q.; Yuan, S.; Sun, Y.; Zhang, B.; Ren, J.; Kotecha, A.; et al. Potent neutralization of hepatitis A virus reveals a receptor mimic mechanism and the receptor recognition site. Proc. Natl. Acad. Sci. USA 2017, 114, 770–775. [Google Scholar] [CrossRef]
- Lee, H.; Cifuente, J.O.; Ashley, R.E.; Conway, J.F.; Makhov, A.M.; Tano, Y.; Shimizu, H.; Nishimura, Y.; Hafenstein, S. A strain-specific epitope of enterovirus 71 identified by cryo-electron microscopy of the complex with fab from neutralizing antibody. J. Virol. 2013, 87, 11363–11370. [Google Scholar] [CrossRef]
- Moser, R.; Snyers, L.; Wruss, J.; Angulo, J.; Peters, H.; Peters, T.; Blaas, D. Neutralization of a common cold virus by concatemers of the third ligand binding module of the VLDL-receptor strongly depends on the number of modules. Virology 2005, 338, 259–269. [Google Scholar] [CrossRef] [PubMed]
- Blaas, D.; Fuchs, R. Mechanism of human rhinovirus infections. Mol. Cell. Pediatr. 2016, 3, 21. [Google Scholar] [CrossRef]
- Plevka, P.; Lim, P.Y.; Perera, R.; Cardosa, J.; Suksatu, A.; Kuhn, R.J.; Rossmann, M.G. Neutralizing antibodies can initiate genome release from human enterovirus 71. Proc. Natl. Acad. Sci. USA 2014, 111, 2134–2139. [Google Scholar] [CrossRef] [PubMed]
- Hewat, E.A.; Blaas, D. Nonneutralizing human rhinovirus serotype 2-specific monoclonal antibody 2G2 attaches to the region that undergoes the most dramatic changes upon release of the viral RNA. J. Virol. 2006, 80, 12398–12401. [Google Scholar] [CrossRef][Green Version]
- Zhu, L.; Xu, K.; Wang, N.; Cao, L.; Wu, J.; Gao, Q.; Fry, E.E.; Stuart, D.I.; Rao, Z.; Wang, J.; et al. Neutralization Mechanisms of Two Highly Potent Antibodies against Human Enterovirus 71. mBio 2018, 9, e01013-18. [Google Scholar] [CrossRef] [PubMed]
- Bottermann, M.; James, L.C. Intracellular Antiviral Immunity. Adv. Virus Res. 2018, 100, 309–354. [Google Scholar] [CrossRef]
- Foss, S.; Bottermann, M.; Jonsson, A.; Sandlie, I.; James, L.C.; Andersen, J.T. TRIM21-From Intracellular Immunity to Therapy. Front. Immunol. 2019, 10, 2049. [Google Scholar] [CrossRef] [PubMed]
- James, L.C.; Keeble, A.H.; Khan, Z.; Rhodes, D.A.; Trowsdale, J. Structural basis for PRYSPRY-mediated tripartite motif (TRIM) protein function. Proc. Natl. Acad. Sci. USA 2007, 104, 6200–6205. [Google Scholar] [CrossRef]
- Mallery, D.L.; McEwan, W.A.; Bidgood, S.R.; Towers, G.J.; Johnson, C.M.; James, L.C. Antibodies mediate intracellular immunity through tripartite motif-containing 21 (TRIM21). Proc. Natl. Acad. Sci. USA 2010, 107, 19985–19990. [Google Scholar] [CrossRef]
- Veerati, P.C.; Troy, N.M.; Reid, A.T.; Li, N.F.; Nichol, K.S.; Kaur, P.; Maltby, S.; Wark, P.A.B.; Knight, D.A.; Bosco, A.; et al. Airway Epithelial Cell Immunity Is Delayed During Rhinovirus Infection in Asthma and COPD. Front. Immunol. 2020, 11, 974. [Google Scholar] [CrossRef]
- Neidich, S.D.; Fong, Y.; Li, S.S.; Geraghty, D.E.; Williamson, B.D.; Young, W.C.; Goodman, D.; Seaton, K.E.; Shen, X.; Sawant, S.; et al. Antibody Fc effector functions and IgG3 associate with decreased HIV-1 risk. J. Clin. Investig. 2019, 129, 4838–4849. [Google Scholar] [CrossRef]
- Chromikova, V.; Tan, J.; Aslam, S.; Rajabhathor, A.; Bermudez-Gonzalez, M.; Ayllon, J.; Simon, V.; García-Sastre, A.; Salaun, B.; Nachbagauer, R.; et al. Activity of human serum antibodies in an influenza virus hemagglutinin stalk-based ADCC reporter assay correlates with activity in a CD107a degranulation assay. Vaccine 2020, 38, 1953–1961. [Google Scholar] [CrossRef]
- Jenks, J.A.; Goodwin, M.L.; Permar, S.R. The Roles of Host and Viral Antibody Fc Receptors in Herpes Simplex Virus (HSV) and Human Cytomegalovirus (HCMV) Infections and Immunity. Front. Immunol. 2019, 10, 2110. [Google Scholar] [CrossRef] [PubMed]
- Barclay, W.S.; al-Nakib, W.; Higgins, P.G.; Tyrrell, D.A. The time course of the humoral immune response to rhinovirus infection. Epidemiol. Infect. 1989, 103, 659–669. [Google Scholar] [CrossRef] [PubMed]
- Potaczek, D.P.; Unger, S.D.; Zhang, N.; Taka, S.; Michel, S.; Akdağ, N.; Lan, F.; Helfer, M.; Hudemann, C.; Eickmann, M.; et al. Development and characterization of DNAzyme candidates demonstrating significant efficiency against human rhinoviruses. J. Allergy Clin. Immunol. 2019, 143, 1403–1415. [Google Scholar] [CrossRef] [PubMed]
- McLean, G.R. Developing a vaccine for human rhinoviruses. J. Vaccines Immun. 2014, 2, 16–20. [Google Scholar] [CrossRef] [PubMed][Green Version]
| RV-A | RV-B | RV-C | |
|---|---|---|---|
| Members | 80 serotypes | 32 serotypes | 57 types 3 |
| Features | Includes all minor 1 group RVs (10) and most of the major 2 group RVs (70) | All 32 are major group | 1st discovery in 2007 and numbers continue to expand |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Touabi, L.; Aflatouni, F.; McLean, G.R. Mechanisms of Rhinovirus Neutralisation by Antibodies. Viruses 2021, 13, 360. https://doi.org/10.3390/v13030360
Touabi L, Aflatouni F, McLean GR. Mechanisms of Rhinovirus Neutralisation by Antibodies. Viruses. 2021; 13(3):360. https://doi.org/10.3390/v13030360
Chicago/Turabian StyleTouabi, Lila, Faryal Aflatouni, and Gary R. McLean. 2021. "Mechanisms of Rhinovirus Neutralisation by Antibodies" Viruses 13, no. 3: 360. https://doi.org/10.3390/v13030360
APA StyleTouabi, L., Aflatouni, F., & McLean, G. R. (2021). Mechanisms of Rhinovirus Neutralisation by Antibodies. Viruses, 13(3), 360. https://doi.org/10.3390/v13030360







