Exposure of Wild Ungulates to the Usutu and Tick-Borne Encephalitis Viruses in France in 2009–2014: Evidence of Undetected Flavivirus Circulation a Decade Ago
Abstract
1. Introduction
2. Materials and Methods
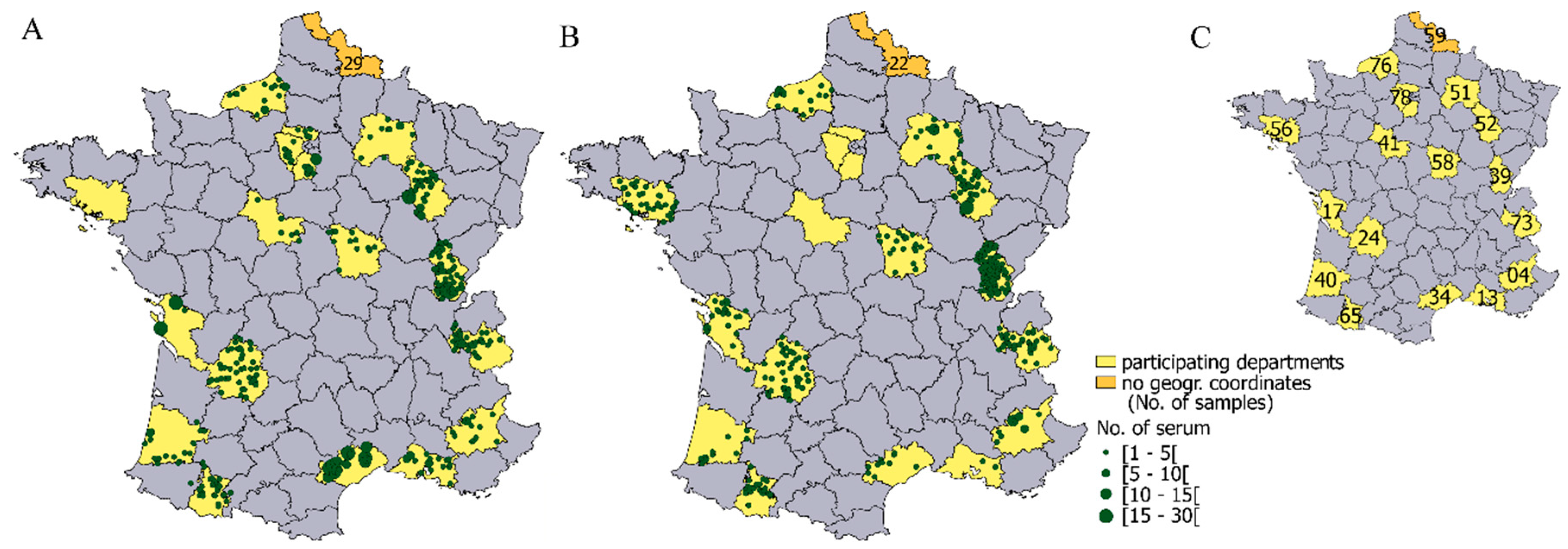
2.1. Data and Study Area
2.2. Laboratory Analysis
2.3. Laboratory Result Interpretation
2.4. Statistical Analyses
3. Results
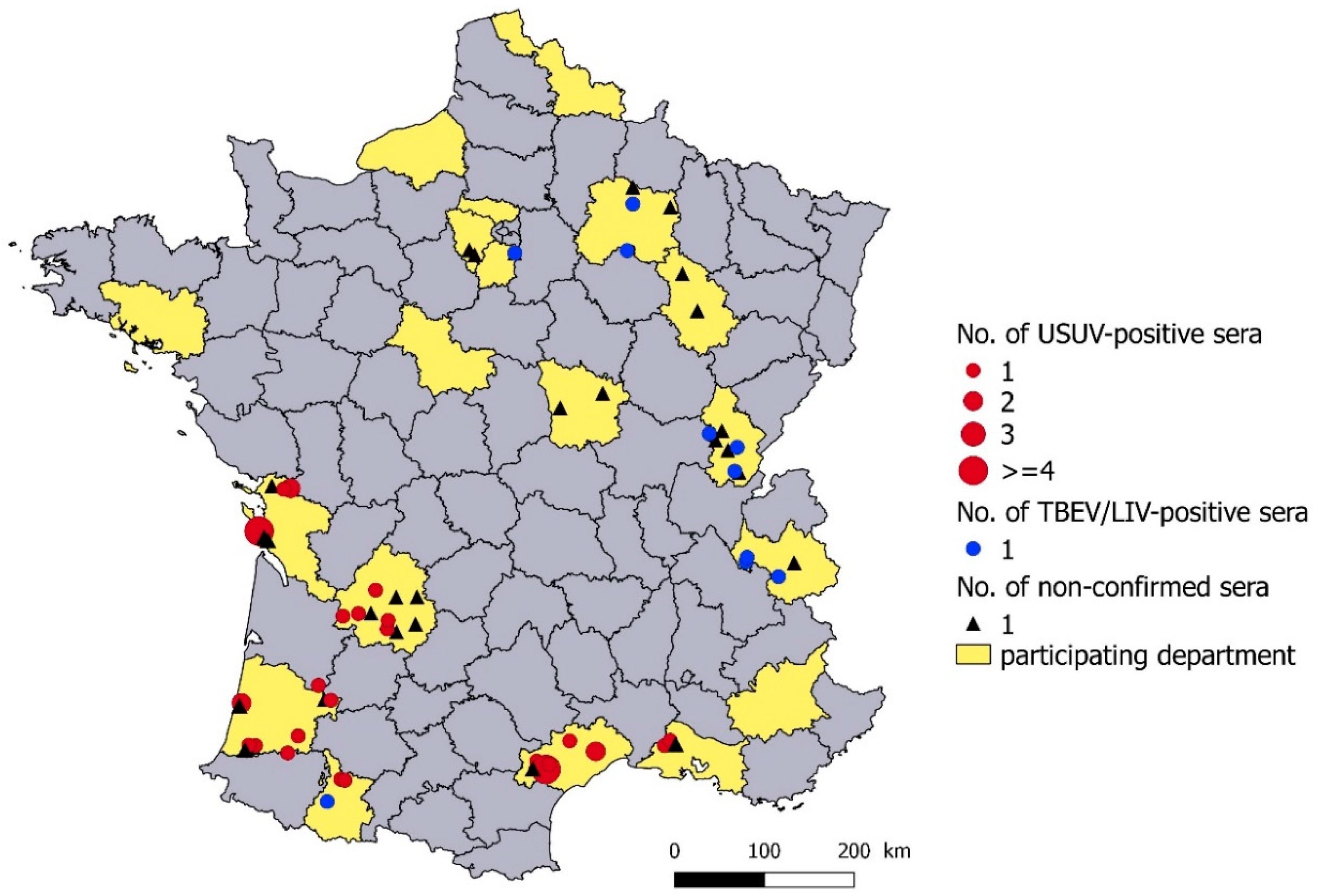
3.1. Serological Results
3.2. Geographical Distribution
3.3. Seroprevalence in USUV-Infected Departments
3.4. Seroprevalence in TBEV/LIV-Infected Departments in Eastern France
4. Discussion
4.1. Spatiotemporal Circulation of USUV in France
4.2. No Detection of WNV and BAGV Antibodies
4.3. TBEV Serocomplex Distribution
4.4. Unidentified Flaviviruses
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Weissenböck, H.; Hubálek, Z.; Bakonyi, T.; Nowotny, N. Zoonotic mosquito-borne flaviviruses: Worldwide presence of agents with proven pathogenicity and potential candidates of future emerging diseases. Vet. Microbiol. 2010, 140, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Clé, M.; Beck, C.; Salinas, S.; Lecollinet, S.; Gutierrez, S.; Van de Perre, P.; Baldet, T.; Foulongne, V.; Simonin, Y. Usutu virus: A new threat? Epidemiol. Infect. 2019, 147, e232. [Google Scholar] [CrossRef] [PubMed]
- Erber, W.; Schmitt, H.-J.; Janković, T.V. Chapter 12a: Epidemiology by country–an overview. Tick-Borne Enceph.-Book 2019. [Google Scholar] [CrossRef]
- Gilbert, L. Louping ill virus in the UK: A review of the hosts, transmission and ecological consequences of control. Exp. Appl. Acarol. 2016, 68, 363–374. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control Surveillance and Disease Data for West Nile Fever. Available online: https://ecdc.europa.eu/en/west-nile-fever/surveillance-and-disease-data (accessed on 16 July 2019).
- Beck, C.; Jimenez-Clavero, M.; Leblond, A.; Durand, B.; Nowotny, N.; Leparc-Goffart, I.; Zientara, S.; Jourdain, E.; Lecollinet, S. Flaviviruses in Europe: Complex circulation patterns and their consequences for the diagnosis and control of West Nile disease. Int. J. Environ. Res. Public. Health 2013, 10, 6049–6083. [Google Scholar] [CrossRef]
- Weissenböck, H.; Bakonyi, T.; Rossi, G.; Mani, P.; Nowotny, N. Usutu Virus, Italy, 1996. Emerg. Infect. Dis. 2013, 19, 274–277. [Google Scholar] [CrossRef]
- Nikolay, B. A review of West Nile and Usutu virus co-circulation in Europe: How much do transmission cycles overlap? Trans. R. Soc. Trop. Med. Hyg. 2015, 109, 609–618. [Google Scholar] [CrossRef]
- Cadar, D.; Lühken, R.; van der Jeugd, H.; Garigliany, M.; Ziegler, U.; Keller, M.; Lahoreau, J.; Lachmann, L.; Becker, N.; Kik, M.; et al. Widespread activity of multiple lineages of Usutu virus, western Europe, 2016. Euro Surveill 2017, 22, 30452. [Google Scholar] [CrossRef]
- Weidinger, P.; Kolodziejek, J.; Bakonyi, T.; Brunthaler, R.; Erdélyi, K.; Weissenböck, H.; Nowotny, N. Different dynamics of Usutu virus infections in Austria and Hungary, 2017–2018. Transbound. Emerg. Dis. 2019. [Google Scholar] [CrossRef]
- Bahuon, C.; Marcillaud-Pitel, C.; Bournez, L.; Leblond, A.; Beck, C.; Hars, J.; Leparc-Goffart, I.; L’Ambert, G.; Paty, M.-C.; Cavalerie, L.; et al. West Nile virus epizootics in the Camargue (France) in 2015 and reinforcement of surveillance and control networks: -EN- -FR- Épizootie due au virus de West Nile survenue en Camargue (France) en 2015 et renforcement des réseaux de surveillance et de contrôle -ES- Epizootia causada por el virus West Nile en la Camarga (Francia) en 2015 y refuerzo de las redes de vigilancia y control. Rev. Sci. Tech. OIE 2016, 35, 811–824. [Google Scholar]
- Maquart, M.; Dahmani, M.; Marié, J.-L.; Gravier, P.; Leparc-Goffart, I.; Davoust, B. First serological evidence of West Nile Virus in horses and dogs from Corsica island, France. Vector-Borne Zoonotic Dis. 2017, 17, 275–277. [Google Scholar] [CrossRef]
- Johnson, N.; Fernández de Marco, M.; Giovannini, A.; Ippoliti, C.; Danzetta, M.; Svartz, G.; Erster, O.; Groschup, M.; Ziegler, U.; Mirazimi, A.; et al. Emerging Mosquito-Borne Threats and the Response from European and Eastern Mediterranean Countries. Int. J. Environ. Res. Public. Health 2018, 15, 2775. [Google Scholar] [CrossRef]
- Jourdain, E.; Toussaint, Y.; Leblond, A.; Bicout, D.J.; Sabatier, P.; Gauthier-Clerc, M. Bird species potentially involved in introduction, amplification, and spread of West Nile Virus in a mediterranean wetland, the Camargue (Southern France). Vector-Borne Zoonotic Dis. 2007, 7, 15–33. [Google Scholar] [CrossRef]
- Vittecoq, M.; Lecollinet, S.; Jourdain, E.; Thomas, F.; Blanchon, T.; Arnal, A.; Lowenski, S.; Gauthier-Clerc, M. Recent circulation of West Nile Virus and potentially other closely related flaviviruses in Southern France. Vector-Borne Zoonotic Dis. 2013, 13, 610–613. [Google Scholar] [CrossRef]
- Lecollinet, S.; Blanchard, Y.; Manson, C.; Lowenski, S.; Laloy, E.; Quenault, H.; Touzain, F.; Lucas, P.; Eraud, C.; Bahuon, C.; et al. Dual emergence of Usutu virus in common blackbirds, Eastern France, 2015. Emerg. Infect. Dis. 2016, 22, 2225. [Google Scholar] [CrossRef]
- Eiden, M.; Gil, P.; Ziegler, U.; Rakotoarivony, I.; Marie, A.; Frances, B.; L’Ambert, G.; Simonin, Y.; Foulongne, V.; Groschup, M.H.; et al. Emergence of two Usutu virus lineages in Culex pipiens mosquitoes in the Camargue, France, 2015. Infect. Genet. Evol. 2018, 61, 151–154. [Google Scholar] [CrossRef]
- Beck, C.; Gonzales, G.; Decors, A.; Lemberger, K.; Lowenski, S.; Dumarest, D.; Lecollinet, S. Surveillance épidémiologique du virus Usutu dans l’avifaune. Virologie 2018, 22, 261–263. [Google Scholar]
- Simonin, Y.; Sillam, O.; Carles, M.J.; Gutierrez, S.; Gil, P.; Constant, O.; Martin, M.F.; Grard, G.; Van de Perre, P.; Salinas, S.; et al. Human Usutu Virus Infection with Atypical Neurologic Presentation, Montpellier, France, 2016. Emerg. Infect. Dis. 2018, 24, 875–878. [Google Scholar] [CrossRef]
- Velay, A.; Argemi, X.; Wendling, M.-J.; Martinot, M.; Hansmann, Y.; Fafi-Kremer, S. L’encéphalite à tique en France: Qu’en savons-nous aujourd’hui? Rev. Francoph. Lab. 2019, 2019, 34–43. [Google Scholar] [CrossRef]
- Botelho-Nevers, E.; Gagneux-Brunon, A.; Velay, A.; Guerbois-Galla, M.; Grard, G.; Bretagne, C.; Mailles, A.; Verhoeven, P.O.; Pozzetto, B.; Gonzalo, S.; et al. Tick-Borne Encephalitis in Auvergne-Rhône-Alpes Region, France, 2017–2018. Emerg. Infect. Dis. 2019, 25, 1944–1948. [Google Scholar] [CrossRef]
- Thorin, C.; Rigaud, E.; Capek, I.; André-Fontaine, G.; Oster, B.; Gastinger, G.; Abadia, G. Séroprévalence de la borréliose de Lyme et de l’encéphalite à tiques chez des professionnels exposés dans le Grand Est de la France. Médecine Mal. Infect. 2008, 38, 533–542. [Google Scholar] [CrossRef]
- Balseiro, A.; Royo, L.J.; Martínez, C.P.; Fernández de Mera, I.G.; Höfle, Ú.; Polledo, L.; Marreros, N.; Casais, R.; Marín, J.F.G. Louping ill in goats, Spain, 2011. Emerg. Infect. Dis. 2012, 18, 976–978. [Google Scholar] [CrossRef]
- Dobler, G. Zoonotic tick-borne flaviviruses. Vet. Microbiol. 2010, 140, 221–228. [Google Scholar] [CrossRef]
- Marin, M.S.; McKenzie, J.; Gao, G.F.; Reid, H.W.; Antoniadis, A.; Gould, E.A. The virus causing encephalomyelitis in sheep in Spain: A new member of the tick-borne encephalitis group. Res. Vet. Sci. 1995, 58, 11–13. [Google Scholar] [CrossRef]
- Gao, G.F.; Zanotto, P.M.; Holmes, E.C.; Reid, H.W.; Gould, E.A. Molecular variation, evolution and geographical distribution of louping ill virus. Acta Virol. 1997, 41, 259–268. [Google Scholar]
- Jensen, P.M.; Skarphedinsson, S.; Semenov, A. Densities of the tick (Ixodes ricinus) and coexistence of the Louping ill virus and tick borne encephalitis on the island of Bornholm. Ugeskr. Laeger 2004, 166, 2563–2565. [Google Scholar]
- García-Bocanegra, I.; Jurado-Tarifa, E.; Cano-Terriza, D.; Martínez, R.; Pérez-Marín, J.E.; Lecollinet, S. Exposure to West Nile virus and tick-borne encephalitis virus in dogs in Spain. Transbound. Emerg. Dis. 2018, 65, 765–772. [Google Scholar] [CrossRef]
- Vanhomwegen, J.; Beck, C.; Desprès, P.; Figuerola, A.; García, R.; Lecollinet, S.; López-Roig, M.; Manuguerra, J.-C.; Serra-Cobo, J. Circulation of zoonotic arboviruses in equine populations of Mallorca island (Spain). Vector-Borne Zoonotic Dis. 2017, 17, 340–346. [Google Scholar] [CrossRef]
- Randolph, S.E. The shifting landscape of tick-borne zoonoses: Tick-borne encephalitis and Lyme borreliosis in Europe. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2001, 356, 1045–1056. [Google Scholar] [CrossRef]
- Boadella, M.; Díez-Delgado, I.; Gutiérrez-Guzmán, A.V.; Höfle, U.; Gortázar, C. Do wild ungulates allow improved monitoring of flavivirus circulation in Spain? Vector-Borne Zoonotic Dis. 2012, 12, 490–495. [Google Scholar] [CrossRef]
- Gerth, H.-J.; Grimshandl, D.; Stage, B.; Döller, G.; Kunz, C. Roe deer as sentinels for endemicity of tick-borne encephalitis virus. Epidemiol. Infect. 1995, 115, 355–365. [Google Scholar] [CrossRef]
- Roelandt, S.; Suin, V.; Van Gucht, S.; Van der Stede, Y.; Roels, S. Comparative tick-borne encephalitis (virus) surveillance in Belgium 2009–2015: Experiences with diagnostic tests, sentinel species and surveillance designs. J Zoonotic Dis Public Health 2017, 1, 1–4. [Google Scholar]
- García-Bocanegra, I.; Paniagua, J.; Gutiérrez-Guzmán, A.V.; Lecollinet, S.; Boadella, M.; Arenas-Montes, A.; Cano-Terriza, D.; Lowenski, S.; Gortázar, C.; Höfle, U. Spatio-temporal trends and risk factors affecting West Nile virus and related flavivirus exposure in Spanish wild ruminants. BMC Vet. Res. 2016, 12, 249. [Google Scholar] [CrossRef]
- Gutiérrez-Guzmán, A.-V.; Vicente, J.; Sobrino, R.; Perez-Ramírez, E.; Llorente, F.; Höfle, U. Antibodies to West Nile virus and related flaviviruses in wild boar, red foxes and other mesomammals from Spain. Vet. Microbiol. 2012, 159, 291–297. [Google Scholar] [CrossRef]
- Labuda, M.; Elečková, E.; Ličková, M.; Sabó, A. Tick-borne encephalitis virus foci in Slovakia. Int. J. Med. Microbiol. 2002, 291, 43–47. [Google Scholar] [CrossRef]
- Geevarghese, G.; Shaikh, B.H.; Jacob, P.G.; Bhat, H.R. Persistence of haemagglutination-inhibition antibodies to JE and WN viruses in naturally infected domestic pigs in Karnataka State, India. Acta Virol. 1994, 38, 235–237. [Google Scholar]
- Llorente, F.; García-Irazábal, A.; Pérez-Ramírez, E.; Cano-Gómez, C.; Sarasa, M.; Vázquez, A.; Jiménez-Clavero, M.Á. Influence of flavivirus co-circulation in serological diagnostics and surveillance: A model of study using West Nile, Usutu and Bagaza viruses. Transbound. Emerg. Dis. 2019, 66, 2100–2106. [Google Scholar] [CrossRef]
- Beck, C.; Desprès, P.; Paulous, S.; Vanhomwegen, J.; Lowenski, S.; Nowotny, N.; Durand, B.; Garnier, A.; Blaise-Boisseau, S.; Guitton, E.; et al. A high-performance multiplex immunoassay for serodiagnosis of flavivirus-associated neurological diseases in horses. BioMed Res. Int. 2015, 2015, 1–13. [Google Scholar] [CrossRef]
- R Development Core team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- Niewiesk, S. Maternal Antibodies: Clinical Significance, Mechanism of Interference with Immune Responses, and Possible Vaccination Strategies. Front. Immunol. 2014, 5, 446. [Google Scholar] [CrossRef]
- Escribano-Romero, E.; Lupulović, D.; Merino-Ramos, T.; Blázquez, A.-B.; Lazić, G.; Lazić, S.; Saiz, J.-C.; Petrović, T. West Nile virus serosurveillance in pigs, wild boars, and roe deer in Serbia. Vet. Microbiol. 2015, 176, 365–369. [Google Scholar] [CrossRef]
- Hubálek, Z.; Juricová, Z.; Straková, P.; Blazejová, H.; Betásová, L.; Rudolf, I. Serological survey for West Nile virus in wild artiodactyls, Southern Moravia (Czech Republic). Vector-Borne Zoonotic Dis. 2017, 17, 654–657. [Google Scholar] [CrossRef]
- Juricová, Z.; Hubálek, Z. Serological surveys for arboviruses in the game animals of southern Moravia (Czech Republic). Folia Zool. 1999, 48, 185–189. [Google Scholar]
- Ziegler, U.; Fast, C.; Eiden, M.; Bock, S.; Schulze, C.; Hoeper, D.; Ochs, A.; Schlieben, P.; Keller, M.; Zielke, D.E.; et al. Evidence for an independent third Usutu virus introduction into Germany. Vet. Microbiol. 2016, 192, 60–66. [Google Scholar] [CrossRef]
- Sieg, M.; Schmidt, V.; Ziegler, U.; Keller, M.; Höper, D.; Heenemann, K.; Rückner, A.; Nieper, H.; Muluneh, A.; Groschup, M.H.; et al. Outbreak and Cocirculation of Three Different Usutu Virus Strains in Eastern Germany. Vector-Borne Zoonotic Dis. 2017, 17, 662–664. [Google Scholar] [CrossRef]
- Moreno, J.; Tenorio, A.; Figuerola, J.; Sánchez-Seco, M.P.; Ruiz, S.; Vázquez, A.; Magallanes, A.; Molero, F.; Herrero, L. West Nile and Usutu Viruses in Mosquitoes in Spain, 2008–2009. Am. J. Trop. Med. Hyg. 2011, 85, 178–181. [Google Scholar]
- Busquets, N.; Alba, A.; Allepuz, A.; Aranda, C.; Nuñez, J.I. Usutu Virus Sequences in Culex pipiens (Diptera: Culicidae), Spain. Emerg. Infect. Dis. 2008, 14, 861–863. [Google Scholar] [CrossRef]
- Decors, A.; Hars, J.; Faure, E.; Quintaine, T.; Chollet, J.-Y.; Rossi, S. Le réseau SAGIR: Un outil de vigilance vis-à-vis des agents pathogènes exotiques. Bull. Épidémiologique Santé Anim.-Aliment. 2014, 66, 35–39. [Google Scholar]
- Chvala, S.; Bakonyi, T.; Bukovsky, C.; Meister, T.; Brugger, K.; Rubel, F.; Nowotny, N.; Weissenböck, H. Monitoring of Usutu virus activity and spread by using dead bird surveillance in Austria, 2003–2005. Vet. Microbiol. 2007, 122, 237–245. [Google Scholar] [CrossRef]
- Steinmetz, H.W.; Bakonyi, T.; Weissenböck, H.; Hatt, J.-M.; Eulenberger, U.; Robert, N.; Hoop, R.; Nowotny, N. Emergence and establishment of Usutu virus infection in wild and captive avian species in and around Zurich, Switzerland—Genomic and pathologic comparison to other central European outbreaks. Vet. Microbiol. 2011, 148, 207–212. [Google Scholar] [CrossRef]
- Bakonyi, T.; Busquets, N.; Nowotny, N. Comparison of complete genome sequences of Usutu Virus strains detected in Spain, Central Europe, and Africa. Vector-Borne Zoonotic Dis. 2014, 14, 324–329. [Google Scholar] [CrossRef]
- Ziegler, U.; Jöst, H.; Müller, K.; Fischer, D.; Rinder, M.; Tietze, D.T.; Danner, K.-J.; Becker, N.; Skuballa, J.; Hamann, H.-P.; et al. Epidemic spread of Usutu virus in Southwest Germany in 2011 to 2013 and monitoring of wild birds for Usutu and West Nile viruses. Vector-Borne Zoonotic Dis. 2015, 15, 481–488. [Google Scholar] [CrossRef]
- Rijks, J.; Kik, M.; Slaterus, R.; Foppen, R.; Stroo, A.; IJzer, J.; Stahl, J.; Gröne, A.; Koopmans, M.; van der Jeugd, H.; et al. Widespread Usutu virus outbreak in birds in the Netherlands, 2016. Euro Surveill 2016, 21, 30391. [Google Scholar] [CrossRef]
- Murgue, B.; Murri, S.; Zientara, S.; Durand, B.; Durand, J.P.; Zeller, H. West Nile outbreak in horses in southern France, 2000: The return after 35 years. Emerg. Infect. Dis. 2001, 7, 692–696. [Google Scholar] [CrossRef]
- Del Giudice, P.; Schuffenecker, I.; Vandenbos, F.; Counillon, E.; Zellet, H. Human West Nile virus, France. Emerg. Infect. Dis. 2004, 10, 1885–1886. [Google Scholar] [CrossRef]
- Jourdain, E.; Gauthier-Clerc, M.; Sabatier, P.; Grège, O.; Greenland, T.; Leblond, A.; Lafaye, M.; Zeller, H.G. Magpies as hosts for West Nile Virus, Southern France. Emerg. Infect. Dis. 2008, 14, 158–160. [Google Scholar] [CrossRef]
- Sánchez-Gómez, A.; Amela, C.; Fernández-Carrión, E.; Martínez-Avilés, M.; Sánchez-Vizcaíno, J.M.; Sierra-Moros, M.J. Risk mapping of West Nile virus circulation in Spain, 2015. Acta Trop. 2017, 169, 163–169. [Google Scholar] [CrossRef]
- Aguero, M. Bagaza Virus in Partridges and Pheasants, Spain, 2010. Emerg. Infect. Dis. 2011, 17, 1498–1501. [Google Scholar] [CrossRef]
- Llorente, F.; Pérez-Ramírez, E.; Fernández-Pinero, J.; Soriguer, R.; Figuerola, J.; Jiménez-Clavero, M.Á. Flaviviruses in game birds, Southern Spain, 2011–2012. Emerg. Infect. Dis. 2013, 19, 1023–1025. [Google Scholar] [CrossRef]
- Llorente, F.; Pérez-Ramírez, E.; Fernández-Pinero, J.; Elizalde, M.; Figuerola, J.; Soriguer, R.C.; Jiménez-Clavero, M.Á. Bagaza virus is pathogenic and transmitted by direct contact in experimentally infected partridges, but is not infectious in house sparrows and adult mice. Vet. Res. 2015, 46, 93. [Google Scholar] [CrossRef]
- Bondre, V.P.; Sapkal, G.N.; Yergolkar, P.N.; Fulmali, P.V.; Sankararaman, V.; Ayachit, V.M.; Mishra, A.C.; Gore, M.M. Genetic characterization of Bagaza virus (BAGV) isolated in India and evidence of anti-BAGV antibodies in sera collected from encephalitis patients. J. Gen. Virol. 2009, 90, 2644–2649. [Google Scholar] [CrossRef]
- Ruiz-Fons, F.; Balseiro, A.; Willoughby, K.; Oleaga, Á.; Dagleish, M.P.; Pérez-Ramírez, E.; Havlíková, S.; Klempa, B.; Llorente, F.; Martín-Hernando, M.P. Clinical infection of Cantabrian chamois (Rupicapra pyrenaica parva) by louping ill virus: New concern for mountain ungulate conservation? Eur. J. Wildl. Res. 2014, 60, 691–694. [Google Scholar] [CrossRef]
- Holding, M.; Dowall, S.D.; Medlock, J.M.; Carter, D.P.; Pullan, S.T.; Lewis, J.; Vipond, R.; Rocchi, M.S.; Baylis, M.; Hewson, R. Tick-Borne Encephalitis Virus, United Kingdom. Emerg. Infect. Dis. 2020, 26. [Google Scholar] [CrossRef]
- Kuivanen, S.; Levanov, L.; Kareinen, L.; Sironen, T.; Jääskeläinen, A.J.; Plyusnin, I.; Zakham, F.; Emmerich, P.; Schmidt-Chanasit, J.; Hepojoki, J.; et al. Detection of novel tick-borne pathogen, Alongshan virus, in Ixodes ricinus ticks, south-eastern Finland, 2019. Euro Surveill 2019, 24. [Google Scholar] [CrossRef]
- Balling, A.; Plessow, U.; Beer, M.; Pfeffer, M. Prevalence of antibodies against tick-borne encephalitis virus in wild game from Saxony, Germany. Ticks Tick-Borne Dis. 2014, 5, 805–809. [Google Scholar] [CrossRef]
- Kiffner, C.; Vor, T.; Hagedorn, P.; Niedrig, M.; RüHe, F. Determinants of tick-borne encephalitis virus antibody presence in roe deer (Capreolus capreolus) sera. Med. Vet. Entomol. 2012, 26, 18–25. [Google Scholar] [CrossRef]
- Cisak, E.; Wójcik-Fatla, A.; Sroka, J.; Zając, V.; Bilska-Zając, E.; Chmurzyńska, E.; Dutkiewicz, J. Prevalence of tick-borne encephalitis virus antibodies in domestic and game animals from Eastern Poland. Bull. Vet. Inst. Pulawy 2012, 56, 275–278. [Google Scholar] [CrossRef]
- Gomez Martinez, C. Role of Cervids and Wild Boar on the Presence of Tick-Borne Encephalitis Virus in Sweden. Master’s Thesis, Department of Wildlife, Fish and Environmental Studies, Swedish University of Agricultural Sciences, Umea, Sweden, 2014; p. 17. [Google Scholar]
- Linden, A.; Wirtgen, M.; Nahayo, A.; Heyman, P.; Niedrig, M.; Schulze, Y. Tickborne encephalitis virus antibodies in wild cervids in Belgium. Vet. Rec. 2012, 170, 108. [Google Scholar] [CrossRef]
- Roelandt, S.; Suin, V.; der Stede, Y.V.; Lamoral, S.; Marche, S.; Tignon, M.; Saiz, J.C.; Escribano-Romero, E.; Casaer, J.; Brochier, B.; et al. First TBEV serological screening in Flemish wild boar. Infect. Ecol. Epidemiol. 2016, 6, 31099. [Google Scholar] [CrossRef]
- Tavernier, P.; Sys, S.U.; De Clercq, K.; De Leeuw, I.; Caij, A.B.; De Baere, M.; De Regge, N.; Fretin, D.; Roupie, V.; Govaerts, M.; et al. Serologic screening for 13 infectious agents in roe deer (Capreolus capreolus) in Flanders. Infect. Ecol. Epidemiol. 2015, 5, 29862. [Google Scholar] [CrossRef]
- Skarphédinsson, S.; Jensen, P.M.; Kristiansen, K. Survey of tickborne infections in Denmark. Emerg. Infect. Dis. 2005, 11, 1055–1061. [Google Scholar] [CrossRef]
- Van Wieren, S.E.; Hofmeester, T.R. The role of large herbivores in Ixodes ricinus and Borrelia burgdorferi s.l. dynamics. In Ecology and Control of Vector-Borne Diseases; Braks, M.A.H., van Wieren, S.E., Takken, W., Sprong, H., Eds.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2016; Volume 4, pp. 75–89. ISBN 978-90-8686-293-1. [Google Scholar]
- Földvári, G.; Široký, P.; Szekeres, S.; Majoros, G.; Sprong, H. Dermacentor reticulatus: A vector on the rise. Parasit. Vectors 2016, 9, 314. [Google Scholar] [CrossRef]
| Dpt | Wild Boar | Roe Deer | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No. of Samples | cELISA | USUV-Positive | TBEV/LIV-Positive | No. of Samples | cELISA | USUV-Positive | TBEV/LIV-Positive | |||||||||||
| Pos. or Dbt | % | MNT | MNT or MIA | % § | MNT | MNT or MIA | % § | Pos. or Dbt | % | MNT | MNT or MIA | % § | MNT | MNT or MIA | % § | |||
| 4 | 38 | 0 | 0% | - | - | 0 | - | - | 0% | 40 | 0 | 0% | - | - | 0% | - | - | 0% |
| 13 | 34 | 3 | 8.8% | 2 | 2 | 5.9% | 0 | 0 | 0% | 2 | 0 | 0% | - | - | 0% | - | - | 0% |
| 17 | 50 | 10 | 20.0% | 8 | 9 | 18.0% | 0 | 0 | 0% | 44 | 2 | 4.5% | 0 | 0 | 0% | 0 | 0 | 0% |
| 24 | 61 | 7 a | 11.5% | 2 | 3 | 4.9% | 0 | 0 | 0% | 60 | 3 | 5% | 2 | 2 | 3.3% | 0 | 0 | 0% |
| 34 | 212 | 11 | 5.2% | 9 | 10 | 4.7% | 0 | 0 | 0% | 19 | 0 | 0% | - | - | 0% | - | - | 0% |
| 39 | 128 | 3 | 2.3% | 0 | 0 | 0% | 1 | 2 | 1.6% | 176 | 4 | 2.3% | 0 | 0 | 0% | 0 | 1 | 0.6% |
| 40 | 30 | 10 | 33.3% | 7 | 8 | 26.7% | 0 | 0 | 0% | 31 | 1 | 3.2% | 0 | 0 | 0% | 0 | 0 | 0% |
| 41 | 8 | 0 | 0% | - | - | 0% | - | - | 0% | - | - | - | - | - | - | - | - | - |
| 51 | 26 | 2 | 7.7% | 0 | 0 | 0% | 0 | 2 | 7.7% | 54 | 2 | 3.7% | 0 | 0 | 0% | 0 | 0 | 0% |
| 52 | 127 | 1 | 0.8% | 0 | 0 | 0% | 0 | 0 | 0% | 85 | 1 | 1.2% | 0 | 0 | 0% | 0 | 0 | 0% |
| 56 | - | - | - | - | - | - | - | - | - | 33 | 0 | 0% | - | - | 0% | - | - | 0% |
| 58 | 26 | 0 | 0% | - | - | 0% | - | - | 0% | 25 | 2 | 8.0% | 0 | 0 | 0% | 0 | 0 | 0% |
| 59 | 29 | 0 | 0% | - | - | 0% | - | - | 0% | 22 | 0 | 0% | - | - | 0% | - | - | 0% |
| 65 | 37 | 3 | 8.1% | 2 | 2 | 5.4% | 0 | 1 | 2.7% | 40 | 0 | 0% | - | - | 0% | - | - | 0% |
| 73 | 85 | 3 | 3.5% | 0 | 0 | 0% | 3 | 3 | 3.5% | 90 | 1 | 1.1% | 0 | 0 | 0% | 0 | 0 | 0% |
| 76 | 29 | 0 | 0% | - | - | 0% | - | - | 0% | 37 | 0 | 0% | - | - | 0% | - | - | 0% |
| 78–91–95 | 94 | 4 b | 4.2% | 0 | 0 | 0% | 0 | 1 | 1.1% | - | - | - | - | - | - | - | - | - |
| TOTAL | 1014 | 57 | 5.6% | 30 | 34 | 3.4% | 4 | 9 | 0.9% | 758 | 16 | 2.1% | 2 | 2 | 0.3% | 0 | 1 | 0.1% |
| Confirmation Method | MIA | Total | |||
|---|---|---|---|---|---|
| Positive for USUV | Positive for TBEV/LIV | Negative | |||
| MNT | Positive for USUV | 26 | 0 | 6 | 32 |
| Positive for TBEV/LIV | 0 | 1 | 3 | 4 | |
| Negative | 3 | 5 | 22 | 30 | |
| Total | 29 | 6 | 31 | 66 | |
| Variables | No. of Individuals | No. of Positives | β | Error | OR | 95% CI Bootstrap | p |
|---|---|---|---|---|---|---|---|
| Species | |||||||
| roe deer | 196 | 2 | a | a | a | a | a |
| wild boar | 419 | 34 | 2.66 | 0.66 | 17.3 | 4.8–9.6.108 | <0.001 |
| Age Category | |||||||
| adult | 458 | 31 | a | a | a | a | a |
| juvenile | 157 | 5 | −1.28 | 0.52 | 0.3 | 0.1–0.6 | 0.01 |
| Sampling Period | |||||||
| 2009–2010 | 62 | 2 | a | a | a | a | a |
| 2010–2011 | 81 | 1 | −1.01 | 1.23 | 0.4 | 0–75.2 | 0.41 |
| 2011–2012 | 88 | 4 | 0.19 | 0.9 | 1.2 | 0.1–1.5.108 | 0.83 |
| 2012–2013 | 198 | 12 | 1.08 | 0.83 | 2.9 | 0.9–7.3.108 | 0.19 |
| 2013–2014 | 156 | 13 | 1.84 | 0.82 | 6.3 | 1.5–1.3.108 | 0.02 |
| 2014–2015 | 22 | 4 | 3.17 | 1.04 | 23.9 | 3.9–5.3.108 | 0.002 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bournez, L.; Umhang, G.; Faure, E.; Boucher, J.-M.; Boué, F.; Jourdain, E.; Sarasa, M.; Llorente, F.; Jiménez-Clavero, M.A.; Moutailler, S.; et al. Exposure of Wild Ungulates to the Usutu and Tick-Borne Encephalitis Viruses in France in 2009–2014: Evidence of Undetected Flavivirus Circulation a Decade Ago. Viruses 2020, 12, 10. https://doi.org/10.3390/v12010010
Bournez L, Umhang G, Faure E, Boucher J-M, Boué F, Jourdain E, Sarasa M, Llorente F, Jiménez-Clavero MA, Moutailler S, et al. Exposure of Wild Ungulates to the Usutu and Tick-Borne Encephalitis Viruses in France in 2009–2014: Evidence of Undetected Flavivirus Circulation a Decade Ago. Viruses. 2020; 12(1):10. https://doi.org/10.3390/v12010010
Chicago/Turabian StyleBournez, Laure, Gérald Umhang, Eva Faure, Jean-Marc Boucher, Franck Boué, Elsa Jourdain, Mathieu Sarasa, Francisco Llorente, Miguel A. Jiménez-Clavero, Sara Moutailler, and et al. 2020. "Exposure of Wild Ungulates to the Usutu and Tick-Borne Encephalitis Viruses in France in 2009–2014: Evidence of Undetected Flavivirus Circulation a Decade Ago" Viruses 12, no. 1: 10. https://doi.org/10.3390/v12010010
APA StyleBournez, L., Umhang, G., Faure, E., Boucher, J.-M., Boué, F., Jourdain, E., Sarasa, M., Llorente, F., Jiménez-Clavero, M. A., Moutailler, S., Lacour, S. A., Lecollinet, S., & Beck, C. (2020). Exposure of Wild Ungulates to the Usutu and Tick-Borne Encephalitis Viruses in France in 2009–2014: Evidence of Undetected Flavivirus Circulation a Decade Ago. Viruses, 12(1), 10. https://doi.org/10.3390/v12010010









