Importance of the Host Phenotype on the Preservation of the Genetic Diversity in Codling Moth Granulovirus
Abstract
1. Introduction
2. Materials and Methods
2.1. Insects
2.2. Viruses
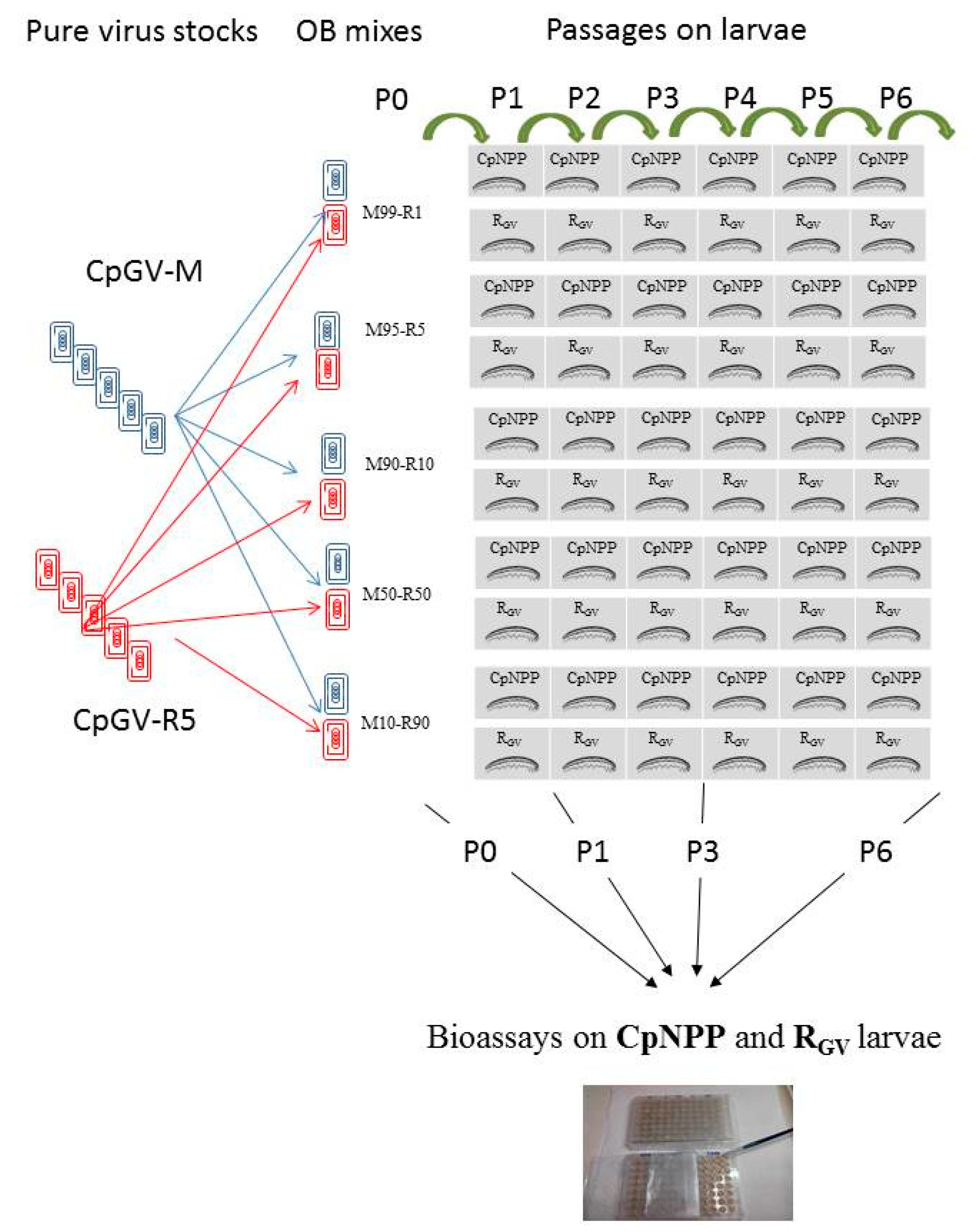
2.3. Viral Populations
2.4. Successive Passages of the Different Viral Lineages
2.5. Bioassays
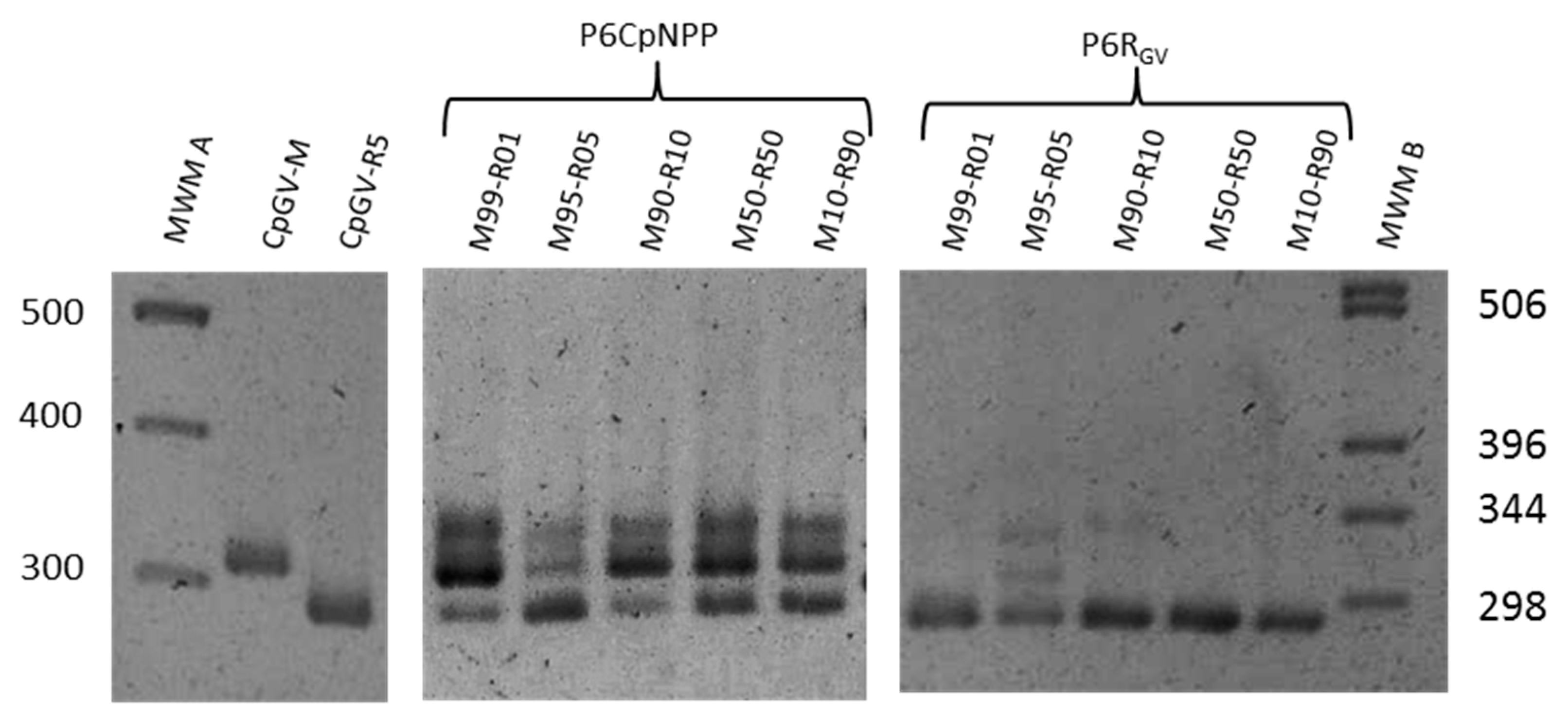
2.6. Estimating the Relative Proportions of Each Genotype by PCR
3. Results
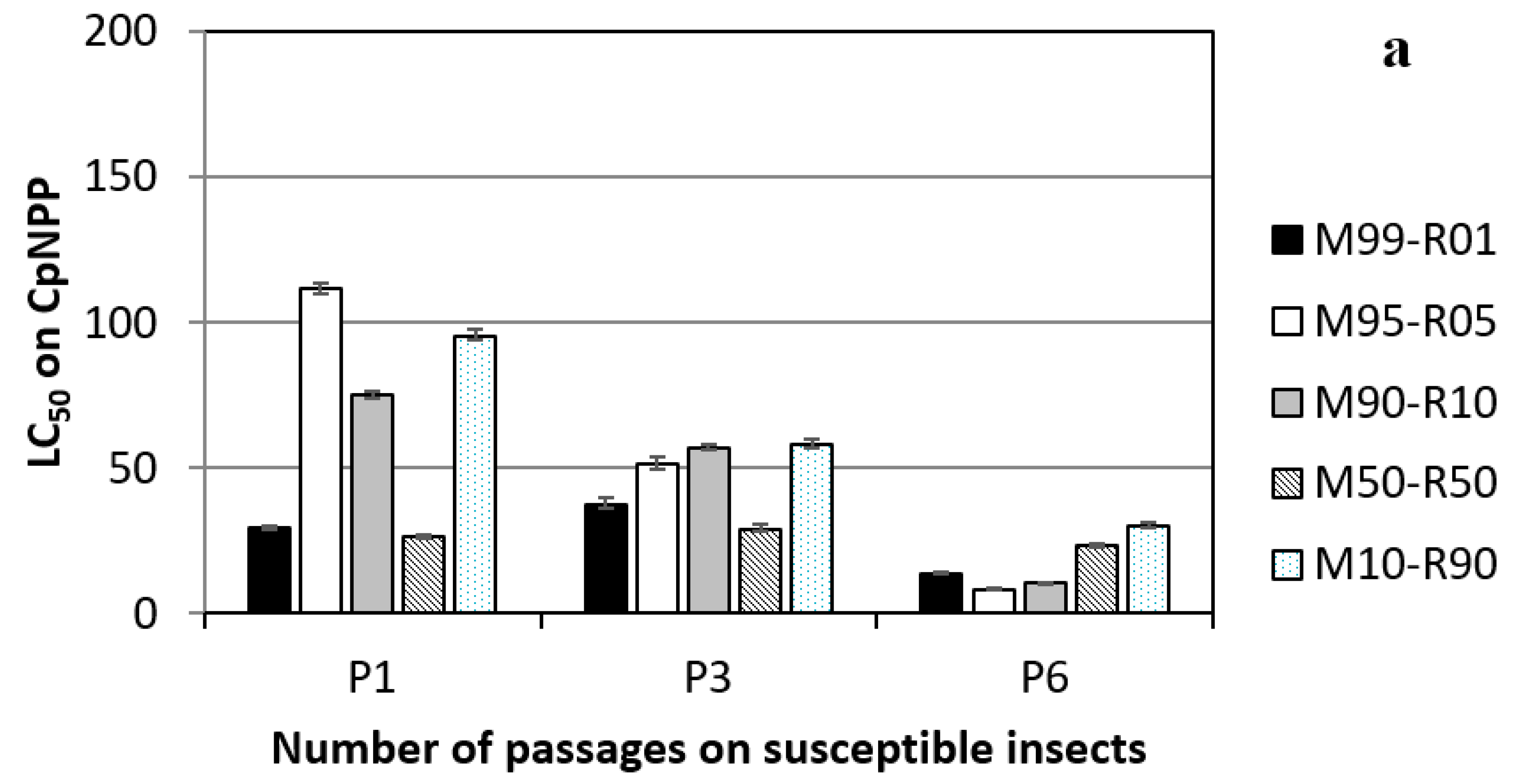
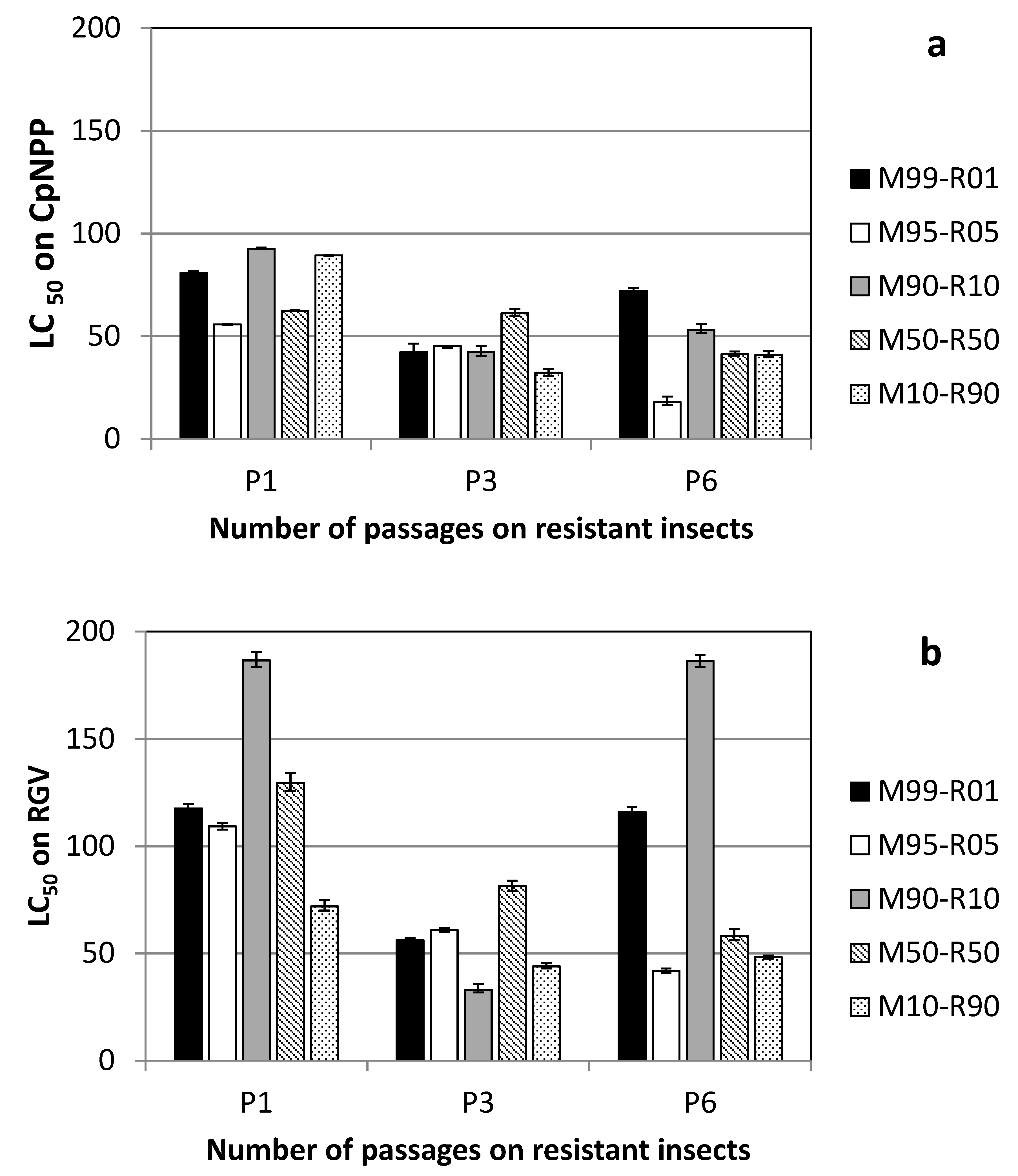
3.1. Lineages Obtained on Susceptible Insects
3.2. Lineages Obtained on Resistant Insects
3.3. Genomic Composition of the Virus Populations
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Cross, J.V.; Solomon, M.G.; Chandler, D.; Jarret, P.; Richardson, P.N.; Winstanley, D.; Bathon, H.; Huber, J.; Keller, B.; Langenbruch, G.A.; et al. Biocontrol of Pests of Apples and Pears in Northern and Central Europe: 1. Microbial Agents and Nematodes. Biocontrol Sci. Technol. 1999, 9, 125–149. [Google Scholar] [CrossRef]
- Willett, M.J.; Neven, L.; Miller, C.E. The occurrence of codling moth in low latitude countries: Validation of pest distribution reports. Hort. Technol. 2009, 19, 633–637. [Google Scholar] [CrossRef]
- Reyes, M.; Franck, P.; Charmillot, P.J.; Ioriatti, C.; Olivares, J.; Pasqualini, E.; Sauphanor, B. Diversity of insecticide resistance mechanisms and spectrum in European populations of the codling moth, Cydia pomonella. Pest Manag. Sci. 2007, 63, 890–902. [Google Scholar] [CrossRef] [PubMed]
- Gröner, A. Specificity and safety of baculoviruses. In The Biology of Baculoviruses; Granados, R.R., Federici, B.A., Eds.; Academic Press: San Diego, CA, USA, 1986; Volume 1, pp. 177–202. [Google Scholar]
- Tanada, Y. A granulosis virus of the codling moth, Carpocapsa pomonella (Linnaeus) (Olethreutidae, Lepidoptera). J. Insect Pathol. 1964, 6, 378–380. [Google Scholar]
- Luque, T.; Finch, R.; Crook, N.; O’Reilly, D.R.; Winstanley, D. The complete sequence of the Cydia pomonella granulovirus genome. J. Gen. Virol. 2001, 82, 2531–2547. [Google Scholar] [CrossRef]
- Lacey, L.A.; Thompson, D.; Vincent, C.; Arthurs, S.P. Codling moth granulovirus: A comprehensive review. Biocontrol Sci. Technol. 2008, 18, 639–663. [Google Scholar] [CrossRef]
- Fritsch, E.; Undorf-Span, K.; Kienle, J.; Zebitz, C.P.W.; Huber, J. Codling moth granulovirus: Variations in the susceptibility of local codling moth populations. Nachr. Dtsch. Pflanzenschutzd. 2005, 57, 29–34. [Google Scholar]
- Sauphanor, B.; Berling, M.; Toubon, J.F.; Reyes, M.; Delnatte, J.; Allemoz, P. Carpocapse des pommes: Cas de résistance au virus de la granulose en vergers biologiques. Phytoma Def. Veg. 2006, 590, 24–27. [Google Scholar]
- Schmitt, A.; Bisutti, I.L.; Ladurner, E.; Benuzzi, M.; Sauphanor, B.; Kienzle, J.; Zingg, D.; Undorf-Spahn, K.; Fritsch, E.; Huber, J.; et al. The occurrence and distribution of resistance of codling moth to Cydia pomonella granulovirus in Europe. J. Appl. Entomol. 2013, 137, 641–649. [Google Scholar] [CrossRef]
- Sauer, A.J.; Schulze-Bopp, S.; Fritsch, E.; Undorf-Spahn, K.; Jehle, J.A. A third type of resistance to Cydia pomonella granulovirus in codling moths shows a mixed Z-Linked and autosomal inheritance pattern. Appl. Environ. Microbiol. 2017, 83, e01036-17. [Google Scholar] [CrossRef]
- Clavijo, G.; Williams, T.; Simón, O.; Muñoz, D.; Cerutti, M.; López-Ferber, M.; Caballero, P. Mixtures of complete and pif1- and pif2-deficient genotypes are required for increased potency of an insect nucleopolyhedrovirus. J. Virol. 2009, 83, 5127–5136. [Google Scholar] [CrossRef] [PubMed]
- Bilimoria, S.L. Taxonomy and identification of baculoviruses. In The Biology of Baculoviruses; Granados, R.B., Federici, B.A., Eds.; CRC Press: Boca Raton, FL, USA, 1986; Volume 1, pp. 37–60. [Google Scholar]
- López-Ferber, M.; Simón, O.; Williams, T.; Caballero, P. Defective or effective? Mutualistic interactions between virus genotypes. Proc. R. Soc. Lond. B 2003, 270, 2249–2255. [Google Scholar] [CrossRef] [PubMed]
- Cory, J.S.; Green, B.M.; Paul, R.K.; Hunter-Fujita, F. Genotypic and phenotypic diversity of a baculovirus population within an individual insect host. J. Invertebr. Pathol. 2005, 89, 101–111. [Google Scholar] [CrossRef] [PubMed]
- Harvey, J.P.; Volkman, L.E. Biochemical and biological variation of Cydia pomonella (codling moth) granulosis virus. Virology 1988, 124, 21–34. [Google Scholar] [CrossRef]
- Crook, N.E.; Spencer, R.A.; Payne, C.C.; Leisy, D.J. Variation in Cydia pomonella Granulosis Virus Isolates and Physical Maps of the DNA from Three Variants. J. Gen. Virol. 1985, 66, 2423–2430. [Google Scholar] [CrossRef]
- Vickers, J.M.; Cory, J.S.; Entwistle, P.F. DNA characterization of eight geographic isolates of granulosis viruses from the potato tuber moth Phthorimaea operculella (Zeller) (Lepidoptera, Gelechiidae). J. Invertebr. Pathol. 1991, 57, 334–342. [Google Scholar] [CrossRef]
- Jehle, J.A.; Fritsch, E.; Huber, J.; Backhaus, H. Intra-specific and inter-specific recombination of tortricid-specific granuloviruses during co-infection in insect larvae. Arch. Virol. 2003, 148, 1317–1333. [Google Scholar]
- Eberle, K.E.; Asser-Kaiser, S.; Sayed, S.M.; Nguyen, H.T.; Jehle, J.A. Overcoming the resistance of codling moth against conventional Cydia pomonella granulovirus (CpGV-M) by a new isolate CpGV-I12. J. Invertebr. Pathol. 2008, 98, 293–298. [Google Scholar] [CrossRef]
- Berling, M.; Blachère-López, C.; Soubabère, O.; Léry, X.; Bonhomme, A.; Sauphanor, B.; López-Ferber, M. Cydia pomonella granulovirus genotypes overcome virus resistance in the codling moth and improve virus efficiency by selection against resistant hosts. Appl. Environ. Microbiol. 2009, 75, 925–930. [Google Scholar] [CrossRef]
- Gebhardt, M.M.; Eberle, K.E.; Radtke, P.; Jehle, J.A. Baculovirus resistance in codling moth is virus isolate-dependent and the consequence of a mutation in viral gene pe38. PNAS 2014, 111, 15711–15716. [Google Scholar] [CrossRef]
- Rezapanah, M.; Shojai-Estabragh, S.; Huber, J.; Jehle, J.A. Molecular characterization of new isolates of Cydia pomonella granulovirus from Iran. J. Pest. Sci. 2008, 81, 187–191. [Google Scholar] [CrossRef]
- Asser Kaiser, S.; Radtke, P.; El-Salamouny, S.; Winstanley, D.; Jehle, J.A. Baculovirus resistance in codling moth (Cydia pomonella L.) caused by early block of virus replication. Virology 2011, 410, 360–367. [Google Scholar] [CrossRef]
- Read, A.F.; Taylor, L.H. The ecology of genetically diverse infections. Science 2001, 292, 1099–1102. [Google Scholar] [CrossRef] [PubMed]
- Mideo, N. Parasite adaptations to within-host competition. Trends Parasitol. 2009, 25, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Bull, J.C.; Godfray, H.C.; O’Reilly, D.R. Persistence of an occlusion-negative recombinant nucleopolyhedrovirus in Trichoplusia ni indicates high multiplicity of cellular infection. Appl. Environ. Microbiol. 2001, 67, 5204–5209. [Google Scholar] [CrossRef]
- Clavijo, G.; Williams, T.; Muñoz, D.; Caballero, P.; López-Ferber, M. Mixed genotype transmission bodies and virions contribute to the maintenance of diversity in an insect virus. Proc. R. Soc. B 2010, 277, 943–951. [Google Scholar] [CrossRef] [PubMed]
- Franck, S.A. Deadly partnerships. Nature 2003, 425, 251–252. [Google Scholar] [CrossRef] [PubMed]
- Graillot, B.; Bayle, S.; Blachère-López, C.; Besse, S.; Siegwart, M.; López-Ferber, M. Biological Characteristics of Experimental Genotype Mixtures of Cydia Pomonella Granulovirus (CpGV): Ability to Control Susceptible and Resistant Pest Populations. Viruses 2016, 8, 147. [Google Scholar] [CrossRef] [PubMed]
- Graillot, B.; Berling, M.; Blachère-López, C.; Siegwart, M.; Besse, S.; López-Ferber, M. Progressive adaptation of a CpGV isolate to codling moth populations resistant to CpGV-M. Viruses 2014, 6, 5135–5144. [Google Scholar] [CrossRef]
- Graillot, B.; Berling, M.; Blachere-López, C.; Siegwart, M.; Besse, S.; López-Ferber, M. Correction: Graillot, B.; et al. Progressive Adaptation of a CpGV Isolate to Codling Moth Populations Resistant to CpGV-M. Viruses 2014, 6, 5135–5144. Viruses 2015, 7, 6313–6315. [Google Scholar] [CrossRef]
- Finney, J.R. Probit Analysis, 3rd ed.; Cambridge University Press: Cambridge, UK, 1971. [Google Scholar]
- POLO PLUS, version 2.0; LeOra Software: Berkeley, CA, USA, 1987.
- Konopka, J.K.; Scott, I.M.; NcNeil, J.N. Cost of insecticide resistance in Cydia pomonella (Lepidoptera: Tortricidae). J. Econ. Entomol. 2012, 105, 872–877. [Google Scholar] [CrossRef] [PubMed]
- Undorf-Spahn, K.; Fritsch, E.; Huber, J.; Kienzle, J.; Zebitz, C.P.; Jehle, J.A. High stability and no fitness costs of the resistance of codling moth to Cydia pomonella granulovirus (CpGV-M). J. Invertebr. Pathol. 2012, 111, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Beperet, I.; Irons, S.L.; Simón, O.; King, L.A.; Williams, T.; Possee, R.D.; López-Ferber, M.; Caballero, P. Superinfection exclusion in alphabaculovirus infections is concomitant with actin reorganization. J. Virol. 2014, 88, 3548–3556. [Google Scholar] [CrossRef] [PubMed]
- Croizier, G.; Ribeiro, H.C.T. Recombination as a possible major cause of genetic heterogeneity in Anticarsia gemmatalis nuclear polyhedrosis virus wild populations. Virus Res. 1992, 26, 183–196. [Google Scholar] [CrossRef]
- Andermatt France. Madex Pro Technical Notice. Available online: https://www.andermatt.fr/img/cms/Fiche%20produit/Madex-Pro-FP.pdf (accessed on 5 May 2019).
- Anonymous. Carpovirusine Evo 2 Technical Notice; Arysta Lifesciences: Pau, France, 2019. [Google Scholar]
- Glen, D.M.; Payne, C.C. Production and field evaluation of codling moth granulosis virus for control of Cydia pomonella in the United Kingdom. Ann. Appl. Biol. 1984, 104, 87–98. [Google Scholar] [CrossRef]
- Ballard, J.; Ellis, D.J.; Payne, C.C. Uptake of granulovirus from the surface of apples and leaves by first instar larvae of the codling moth, Cydia pomonella. Biocontrol Sci. Technol. 2000, 10, 617–625. [Google Scholar] [CrossRef]
- Jehle, J.A.; Sauer, A.J.; Gueli Aletti, G.; Fritsch, E.; undorf-Spahn, K. Granulovirusresistenz beim Apfelwickler und neue resistenzbrechende Granulovirus-Isolate. Öko-Obstbau 2016, 2, 16–18. [Google Scholar]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Graillot, B.; Blachere-López, C.; Besse, S.; Siegwart, M.; López-Ferber, M. Importance of the Host Phenotype on the Preservation of the Genetic Diversity in Codling Moth Granulovirus. Viruses 2019, 11, 621. https://doi.org/10.3390/v11070621
Graillot B, Blachere-López C, Besse S, Siegwart M, López-Ferber M. Importance of the Host Phenotype on the Preservation of the Genetic Diversity in Codling Moth Granulovirus. Viruses. 2019; 11(7):621. https://doi.org/10.3390/v11070621
Chicago/Turabian StyleGraillot, Benoit, Christine Blachere-López, Samantha Besse, Myriam Siegwart, and Miguel López-Ferber. 2019. "Importance of the Host Phenotype on the Preservation of the Genetic Diversity in Codling Moth Granulovirus" Viruses 11, no. 7: 621. https://doi.org/10.3390/v11070621
APA StyleGraillot, B., Blachere-López, C., Besse, S., Siegwart, M., & López-Ferber, M. (2019). Importance of the Host Phenotype on the Preservation of the Genetic Diversity in Codling Moth Granulovirus. Viruses, 11(7), 621. https://doi.org/10.3390/v11070621





