Development of an E2 ELISA Methodology to Assess Chikungunya Seroprevalence in Patients from an Endemic Region of Mexico
Abstract
:1. Introduction
2. Materials and Methods
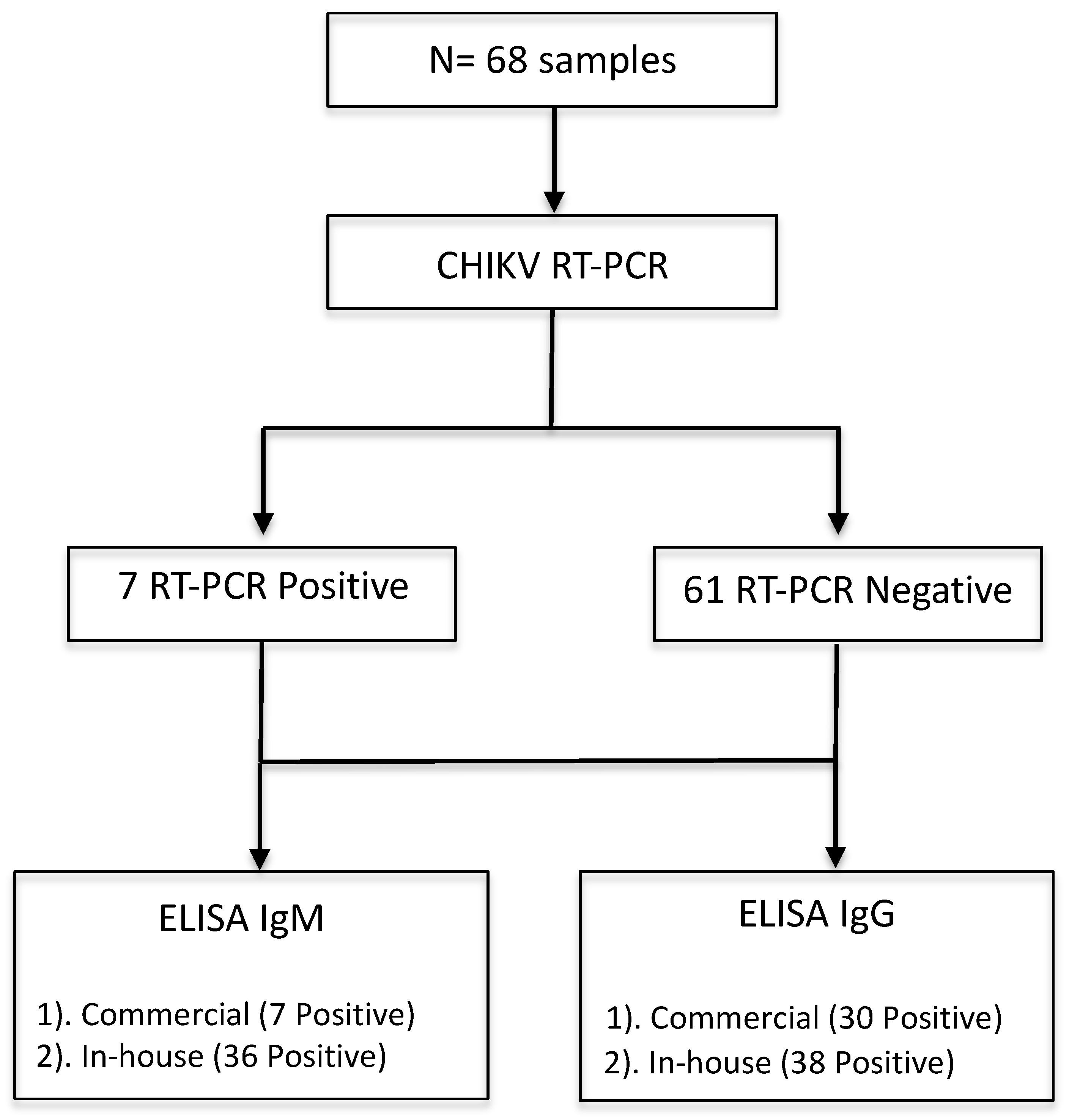
2.1. Study Design
2.2. Sample Collection
2.3. Real-Time Quantitative Reverse Transcription PCR (qRT-PCR)
2.4. Production and Purification of Recombinant CHIKV E2 Protein
2.5. Detection of Anti-CHIKV IgM and IgG by in-House ELISA
2.6. Detection of Anti-CHIKV IgM and IgG by Commercial ELISA Kit
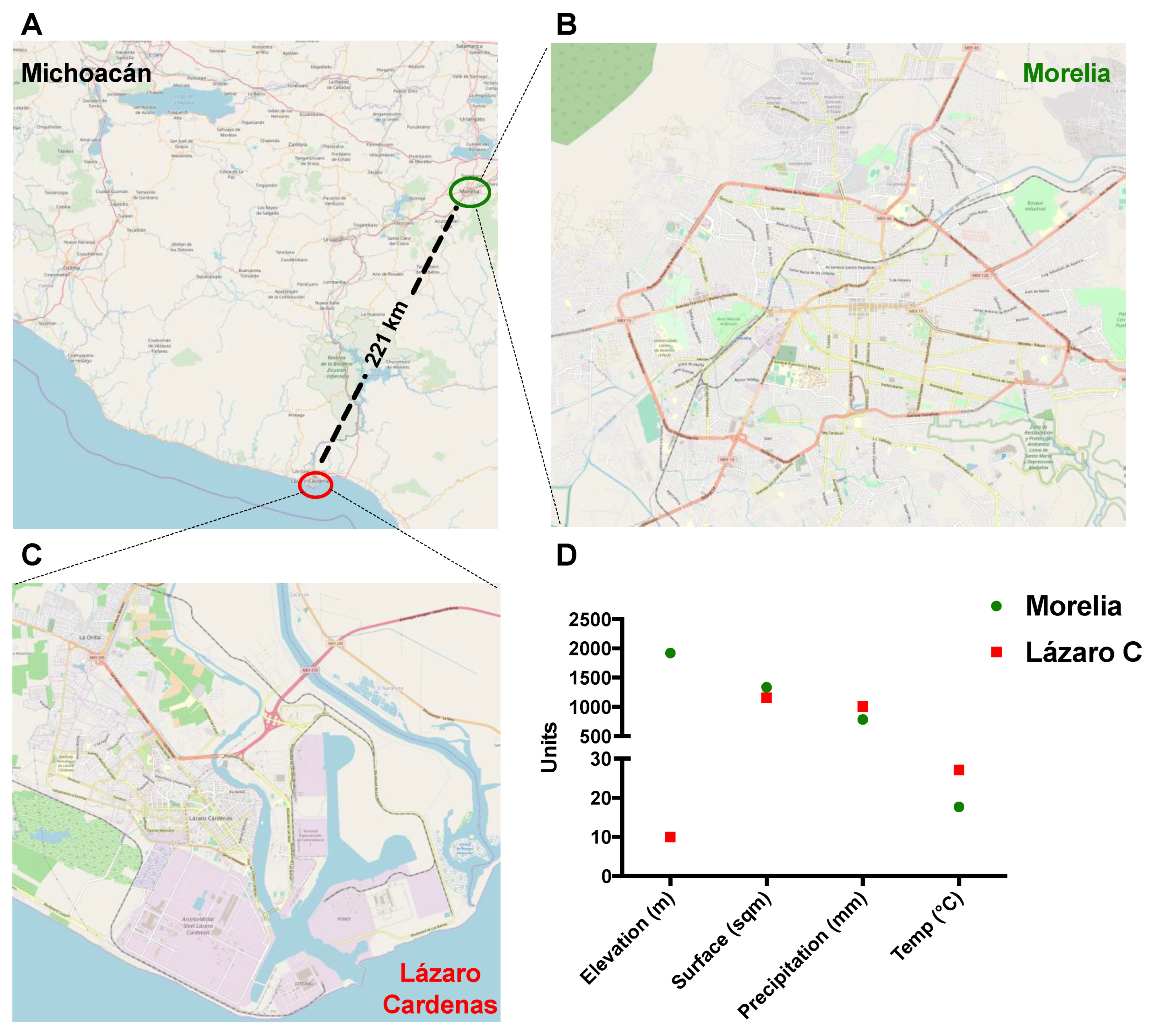
2.7. Maps and Geographical Data
2.8. Ethics Approval and Consent to Participate
2.9. Data Analysis and Statistics
3. Results
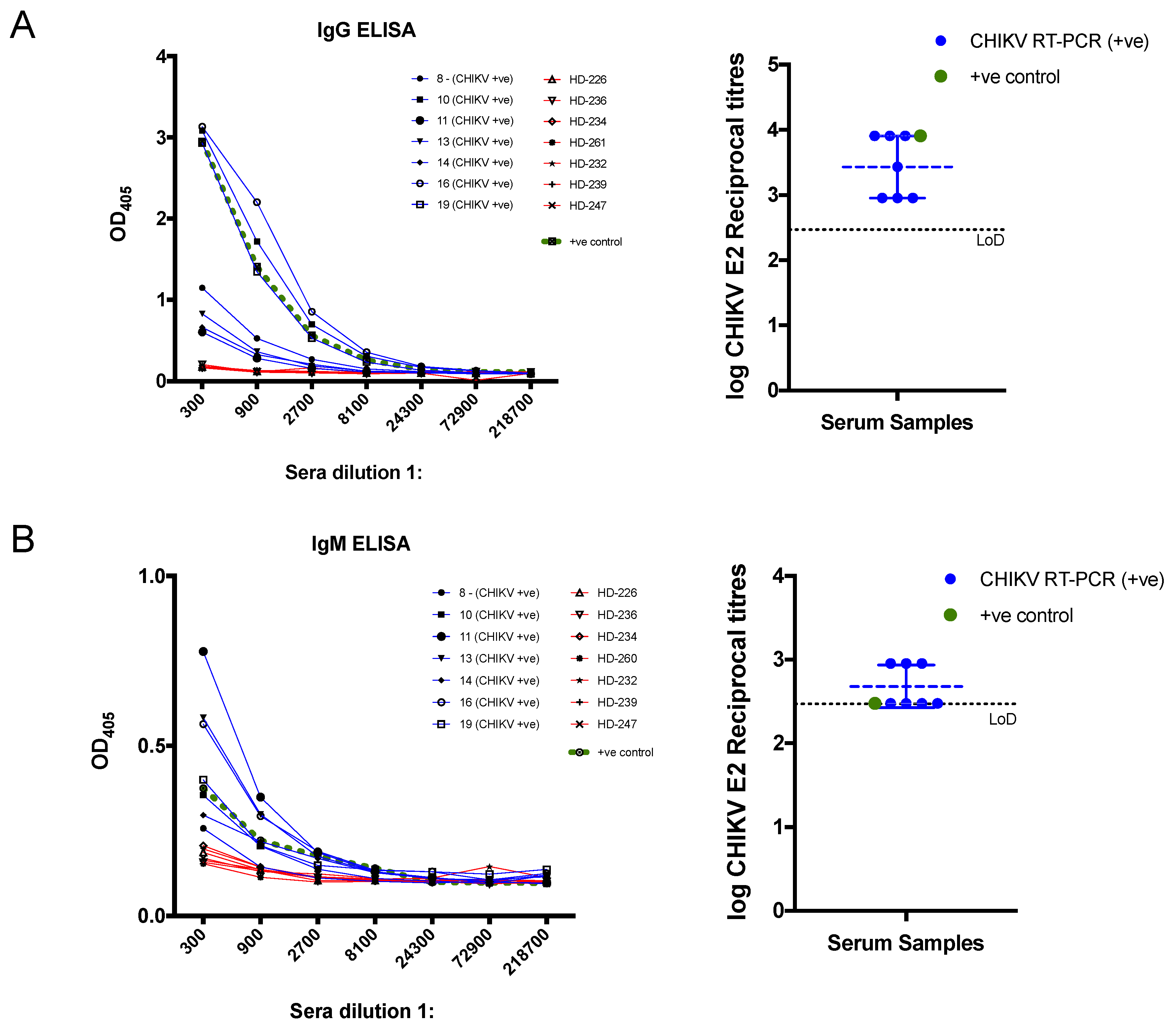
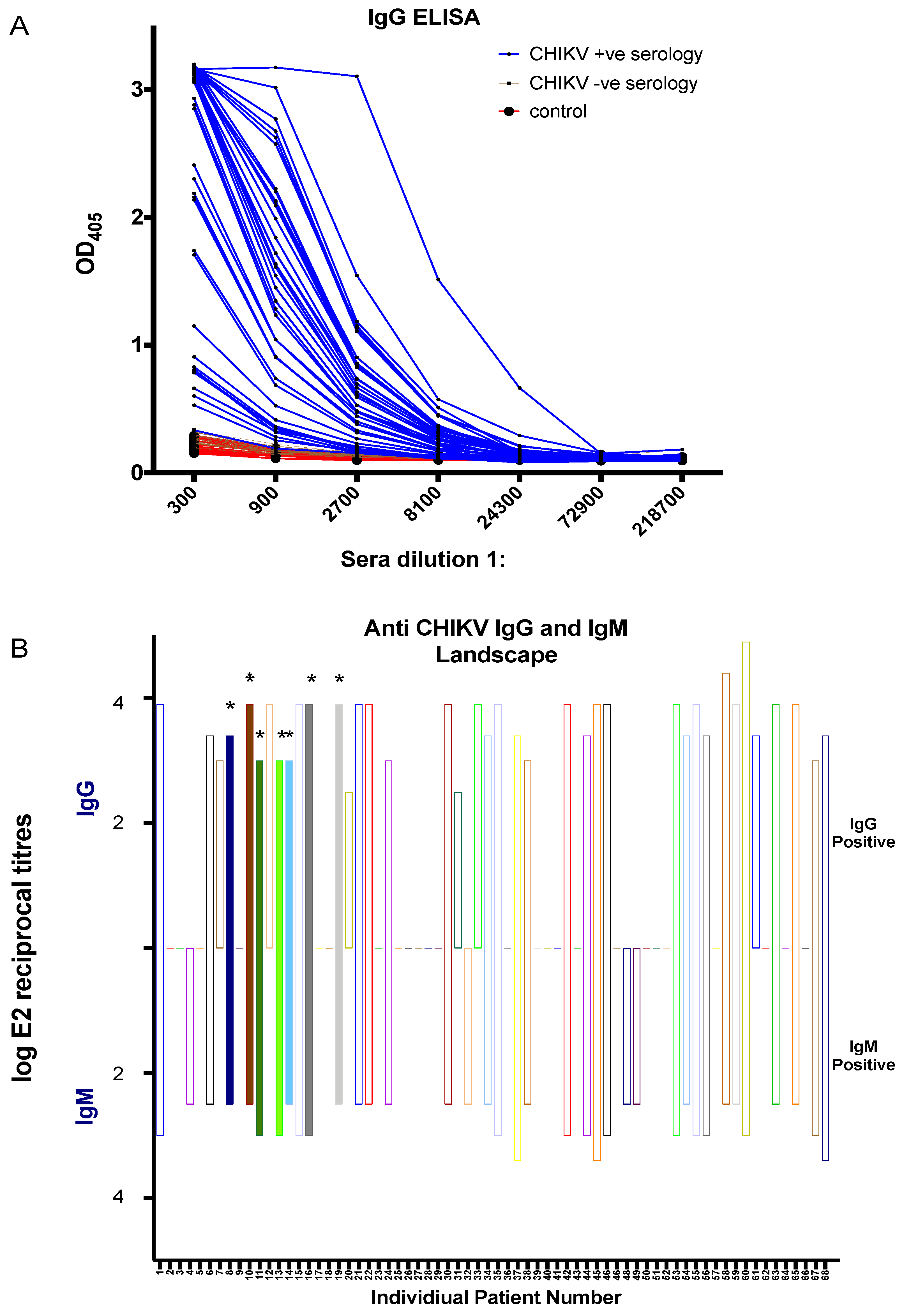
3.1. Development and Evaluation of CHIKV Recombinant E2 Based in-House ELISA
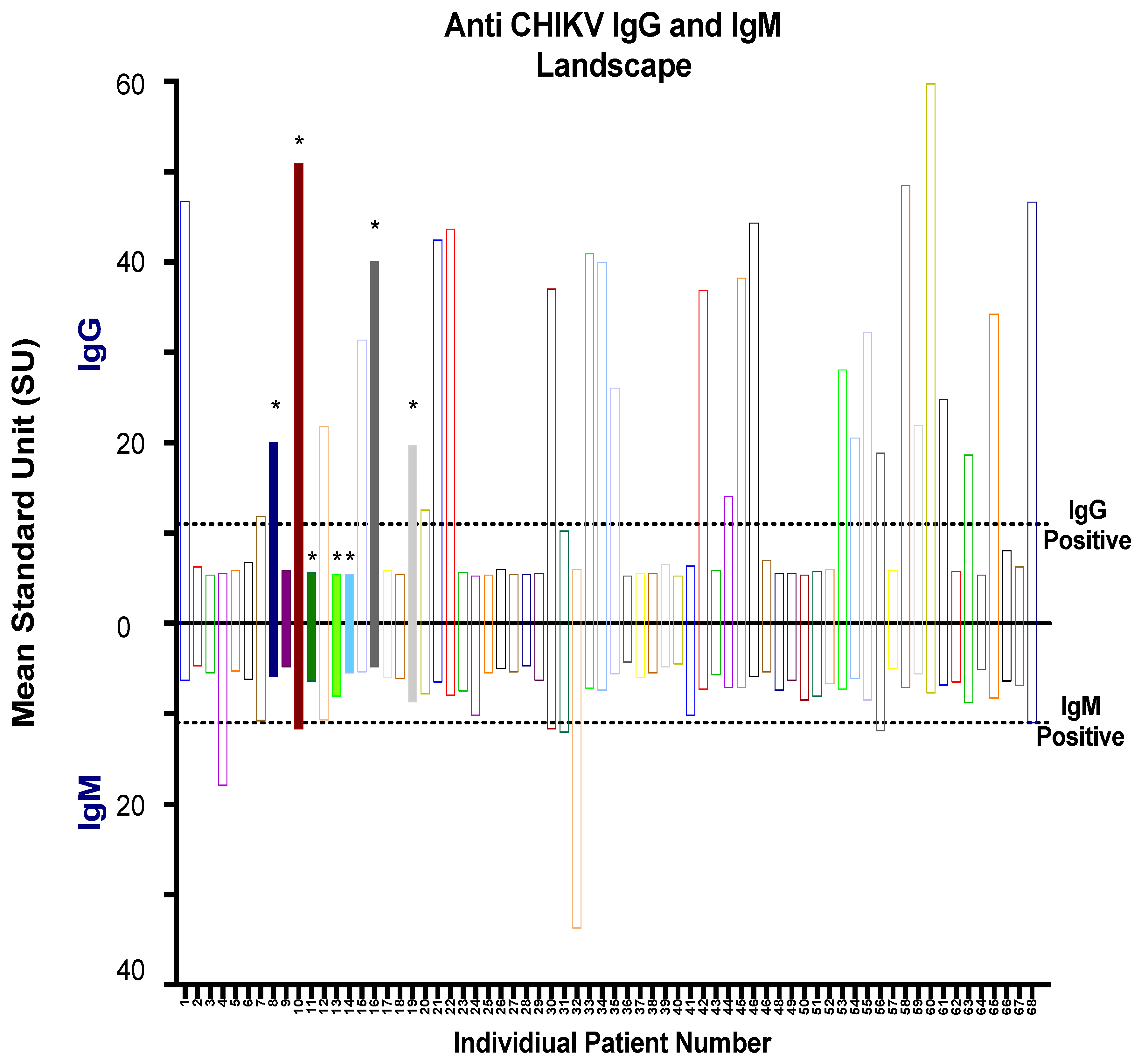
3.2. CHIKV Serological Diagnosis Using the Commercial ELISA Kits
3.3. CHIKV Serological Diagnosis Using the in-House ELISA
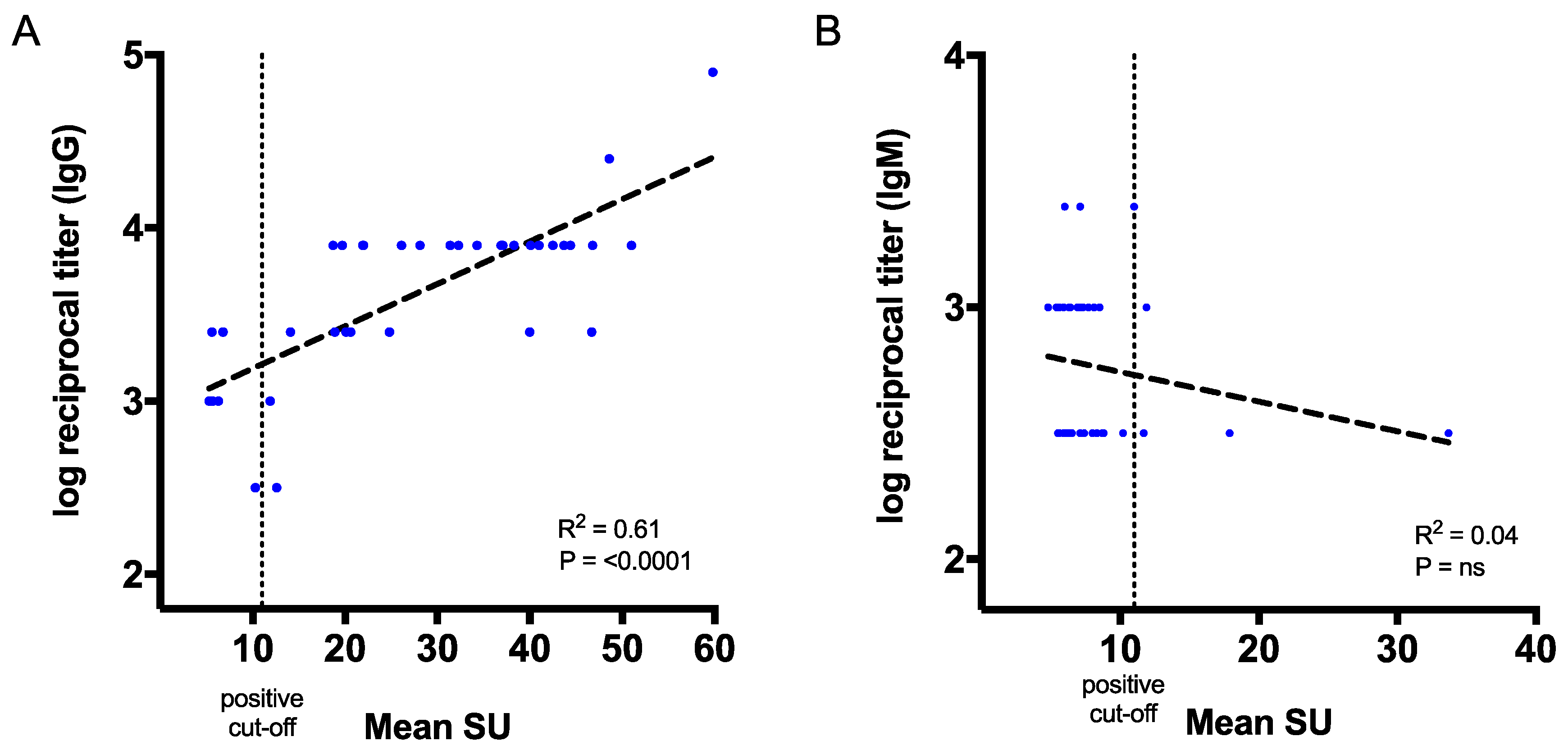
3.4. Correlation of CHIKV Antibody Titres with the Commercial Kits
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Strauss, J.H.; Strauss, E.G. The alphaviruses: Gene expression, replication, and evolution. Microbiol. Mol. Biol. Rev. 1994, 58, 491–562. [Google Scholar]
- Khan, A.H.; Morita, K.; del Carmen Parquet, M.; Hasebe, F.; Mathenge, E.G.M.; Igarashi, A. Complete nucleotide sequence of chikungunya virus and evidence for an internal polyadenylation site. J. Gen. Virol. 2002, 83, 3075–3084. [Google Scholar] [CrossRef] [PubMed]
- Pialoux, G.; Gaüzère, B.-A.; Jauréguiberry, S.; Strobel, M. Chikungunya, an epidemic arbovirosis. Lancet Infect. Dis. 2007, 7, 319–327. [Google Scholar] [CrossRef]
- Mason, P.J.; Haddow, A.J. An epidemic of virus disease in Southern Province, Tanganyika Territory, in 1952-53; an additional note on Chikungunya virus isolations and serum antibodies. Trans. R. Soc. Trop. Med. Hyg. 1957, 51, 238–240. [Google Scholar] [CrossRef]
- Bodenmann, P.; Genton, B. Chikungunya: An epidemic in real time. Lancet 2006, 368, 258. [Google Scholar] [CrossRef]
- Simizu, B.; Yamamoto, K.; Hashimoto, K.; Ogata, T. Structural proteins of Chikungunya virus. J. Virol. 1984, 51, 254–258. [Google Scholar]
- Powers, A.M.; Brault, A.C.; Shirako, Y.; Strauss, E.G.; Kang, W.; Strauss, J.H.; Weaver, S.C. Evolutionary relationships and systematics of the alphaviruses. J. Virol. 2001, 75, 10118–10131. [Google Scholar] [CrossRef] [PubMed]
- Higashi, N.; Matsumoto, A.; Tabata, K.; Nagatomo, Y. Electron microscope study of development of Chikungunya virus in green monkey kidney stable (VERO) cells. Virology 1967, 33, 55–69. [Google Scholar] [CrossRef]
- Cheng, R.H.; Kuhn, R.J.; Olson, N.H.; Rossmann, M.G.; Choi, H.K.; Smith, T.J.; Baker, T.S. Nucleocapsid and glycoprotein organization in an enveloped virus. Cell 1995, 80, 621–630. [Google Scholar] [CrossRef]
- Voss, J.E.; Vaney, M.-C.; Duquerroy, S.; Vonrhein, C.; Girard-Blanc, C.; Crublet, E.; Thompson, A.; Bricogne, G.; Rey, F.A. Glycoprotein organization of Chikungunya virus particles revealed by X-ray crystallography. Nature 2010, 468, 709–712. [Google Scholar] [CrossRef]
- Leparc-Goffart, I.; Nougairede, A.; Cassadou, S.; Prat, C.; Lamballerie, X. de Chikungunya in the Americas. Lancet 2014, 383, 514. [Google Scholar] [CrossRef]
- Weaver, S.C. Arrival of Chikungunya Virus in the New World: Prospects for Spread and Impact on Public Health. PLoS Negl. Trop. Dis. 2014, 8, e2921. [Google Scholar] [CrossRef]
- Fischer, M.; Staples, J.E. Notes from the field: Chikungunya virus spreads in the Americas-Caribbean and South America 2013–2014. MMWR 2014, 63, 500–501. [Google Scholar]
- Weaver, S.C.; Osorio, J.E.; Livengood, J.A.; Chen, R.; Stinchcomb, D.T. Chikungunya virus and prospects for a vaccine. Expert Rev. Vaccines 2012, 11, 1087–1101. [Google Scholar] [CrossRef] [PubMed]
- Reddy, V.; Ravi, V.; Desai, A.; Parida, M.; Powers, A.M.; Johnson, B.W. Utility of IgM ELISA, TaqMan real-time PCR, reverse transcription PCR, and RT-LAMP assay for the diagnosis of Chikungunya fever. J. Med. Virol. 2012, 84, 1771–1778. [Google Scholar] [CrossRef]
- Hasebe, F.; Parquet, M.C.; Pandey, B.D.; Mathenge, E.G.M.; Morita, K.; Balasubramaniam, V.; Saat, Z.; Yusop, A.; Sinniah, M.; Natkunam, S.; et al. Combined detection and genotyping of Chikungunya virus by a specific reverse transcription-polymerase chain reaction. J. Med. Virol. 2002, 67, 370–374. [Google Scholar] [CrossRef] [PubMed]
- Telles, J.-N.; Le Roux, K.; Grivard, P.; Vernet, G.; Michault, A. Evaluation of real-time nucleic acid sequence-based amplification for detection of Chikungunya virus in clinical samples. J. Med. Microbiol. 2009, 58, 1168–1172. [Google Scholar] [CrossRef] [PubMed]
- Johnson, B.W.; Russell, B.J.; Goodman, C.H. Laboratory Diagnosis of Chikungunya Virus Infections and Commercial Sources for Diagnostic Assays. J. Infect. Dis. 2016, 214, S471–S474. [Google Scholar] [CrossRef]
- Kosasih, H.; Widjaja, S.; Surya, E.; Hadiwijaya, S.H.; Butarbutar, D.P.; Jaya, U.A.; Alisjahbana, B.; Williams, M. Evaluation of two IgM rapid immunochromatographic tests during circulation of Asian lineage Chikungunya virus. Southeast Asian J. Trop. Med. Public Health 2012, 43, 55–61. [Google Scholar] [PubMed]
- Azami, N.A.M.; Salleh, S.A.; Shah, S.A.; Neoh, H.; Othman, Z.; Zakaria, S.Z.S.; Jamal, R. Emergence of chikungunya seropositivity in healthy Malaysian adults residing in outbreak-free locations: Chikungunya seroprevalence results from the Malaysian Cohort. BMC Infect. Dis. 2013, 13, 67. [Google Scholar] [CrossRef]
- Yap, G.; Pok, K.-Y.; Lai, Y.-L.; Hapuarachchi, H.-C.; Chow, A.; Leo, Y.-S.; Tan, L.-K.; Ng, L.-C. Evaluation of Chikungunya Diagnostic Assays: Differences in Sensitivity of Serology Assays in Two Independent Outbreaks. PLoS Negl. Trop. Dis. 2010, 4, e753. [Google Scholar] [CrossRef] [PubMed]
- Rianthavorn, P.; Wuttirattanakowit, N.; Prianantathavorn, K.; Limpaphayom, N.; Theamboonlers, A.; Poovorawan, Y. Evaluation of a rapid assay for detection of IgM antibodies to chikungunya. Southeast Asian J. Trop. Med. Public Health 2010, 41, 92–96. [Google Scholar] [PubMed]
- Pierro, A.; Rossini, G.; Gaibani, P.; Finarelli, A.C.; Moro, M.L.; Landini, M.P.; Sambri, V. Persistence of anti-chikungunya virus-specific antibodies in a cohort of patients followed from the acute phase of infection after the 2007 outbreak in Italy. New Microbes New Infect. 2015, 7, 23–25. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.P.; Suresh, A.; Vanamail, P.; Sabesan, S.; Krishnamoorthy, K.G.; Mathew, J.; Jose, V.T.; Jambulingam, P. Chikungunya virus outbreak in Kerala, India, 2007: A seroprevalence study. Mem. Inst. Oswaldo Cruz 2011, 106, 912–916. [Google Scholar] [CrossRef]
- Niedrig, M.; Zeller, H.; Schuffenecker, I.; Drosten, C.; Emmerich, P.; Rumer, L.; Donoso-Mantke, O. International diagnostic accuracy study for the serological detection of chikungunya virus infection. Clin. Microbiol. Infect. 2009, 15, 880–884. [Google Scholar] [CrossRef] [PubMed]
- López-Camacho, C.; Chan Kim, Y.; Blight, J.; Lazaro Moreli, M.; Montoya-Diaz, E.; Huiskonen, J.T.; Mareike Kümmerer, B.; Reyes-Sandoval, A. Assessment of Immunogenicity and Neutralisation Efficacy of Viral-Vectored Vaccines Against Chikungunya Virus. Viruses 2019, 11, 322. [Google Scholar] [CrossRef]
- Prat, C.M.; Flusin, O.; Panella, A.; Tenebray, B.; Lanciotti, R.; Leparc-Goffart, I. Evaluation of Commercially Available Serologic Diagnostic Tests for Chikungunya Virus. Emerg. Infect. Dis. 2014, 20, 2129–2132. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.; Dhanwani, R.; Kumar, J.S.; Rao, P.V.L.; Parida, M. Comparative evaluation of the diagnostic potential of recombinant envelope proteins and native cell culture purified viral antigens of Chikungunya virus. J. Med. Virol. 2014, 86, 1169–1175. [Google Scholar] [CrossRef]
- Hierholzer, J.C.; Killington, R.A.; Stokes, A. Preparation of antigens. In Virology Methods Manual; Elsevier: Amsterdam, The Netherlands, 1996. [Google Scholar]
- Erasmus, J.H.; Needham, J.; Raychaudhuri, S.; Diamond, M.S.; Beasley, D.W.C.; Morkowski, S.; Salje, H.; Salas, I.F.; Kim, D.Y.; Frolov, I.; et al. Utilization of an Eilat Virus-Based Chimera for Serological Detection of Chikungunya Infection. PLoS Negl. Trop. Dis. 2015, 9, e0004119. [Google Scholar] [CrossRef]
- Fumagalli, M.J.; de Souza, W.M.; Espósito, D.L.A.; Silva, A.; Romeiro, M.F.; Martinez, E.Z.; da Fonseca, B.A.L.; Figueiredo, L.T.M. Enzyme-linked immunosorbent assay using recombinant envelope protein 2 antigen for diagnosis of Chikungunya virus. Virol. J. 2018, 15, 112. [Google Scholar] [CrossRef] [PubMed]
- Cho, B.; Jeon, B.-Y.; Kim, J.; Noh, J.; Kim, J.; Park, M.; Park, S. Expression and Evaluation of Chikungunya Virus E1 and E2 Envelope Proteins for Serodiagnosis of Chikungunya Virus Infection. Yonsei Med. J. 2008, 49, 828–835. [Google Scholar] [CrossRef]
- Kumar, P.; Pok, K.-Y.; Tan, L.-K.; Angela, C.; Leo, Y.-S.; Ng, L.-C. Development and evaluation of baculovirus-expressed Chikungunya virus E1 envelope proteins for serodiagnosis of Chikungunya infection. J. Virol. Methods 2014, 206, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.C.; Lopez-Camacho, C.; Nettleship, J.E.; Rahman, N.; Hill, M.L.; Silva-Reyes, L.; Ortiz-Martinez, G.; Figueroa-Aguilar, G.; Mar, M.A.; Vivanco-Cid, H.; et al. Optimization of Zika virus envelope protein production for ELISA and correlation of antibody titers with virus neutralization in Mexican patients from an arbovirus endemic region. Virol. J. 2018, 15, 193. [Google Scholar] [CrossRef]
- Nettleship, J.E.; Rahman-Huq, N.; Owens, R.J. The production of glycoproteins by transient expression in Mammalian cells. In High Throughput Protein Expression and Purification; Methods in Molecular Biology; Springer: Berlin, Germany, 2009; pp. 245–263. [Google Scholar]
- Lancaster, C.; Pristatsky, P.; Hoang, V.M.; Casimiro, D.R.; Schwartz, R.M.; Rustandi, R.; Ha, S. Characterization of N-glycosylation profiles from mammalian and insect cell derived chikungunya VLP. J. Chromatogr. B 2016, 1032, 218–223. [Google Scholar] [CrossRef] [PubMed]
- Azami, N.A.M.; Moi, M.L.; Takasaki, T. Neutralization Assay for Chikungunya Virus Infection: Plaque Reduction Neutralization Test. In Chikungunya Virus; Methods in Molecular Biology; Springer: Berlin, Germany, 2016; pp. 273–282. [Google Scholar]
- Weaver, S.C.; Winegar, R.; Manger, I.D.; Forrester, N.L. Alphaviruses: Population genetics and determinants of emergence. Antivir. Res. 2012, 94, 242–257. [Google Scholar] [CrossRef] [PubMed]
- Navarro, J.-C.; Carrera, J.-P.; Liria, J.; Auguste, A.J.; Weaver, S.C. Alphaviruses in Latin America and the introduction of chikungunya virus. In Human Virology in Latin America; Springer: Berlin, Germany, 2017; pp. 169–192. [Google Scholar]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, Y.C.; López-Camacho, C.; Garcia-Larragoiti, N.; Cano-Mendez, A.; Hernandez-Flores, K.G.; Domínguez-Alemán, C.A.; Antonieta Mar, M.; Vivanco-Cid, H.; Viveros-Sandoval, M.E.; Reyes-Sandoval, A. Development of an E2 ELISA Methodology to Assess Chikungunya Seroprevalence in Patients from an Endemic Region of Mexico. Viruses 2019, 11, 407. https://doi.org/10.3390/v11050407
Kim YC, López-Camacho C, Garcia-Larragoiti N, Cano-Mendez A, Hernandez-Flores KG, Domínguez-Alemán CA, Antonieta Mar M, Vivanco-Cid H, Viveros-Sandoval ME, Reyes-Sandoval A. Development of an E2 ELISA Methodology to Assess Chikungunya Seroprevalence in Patients from an Endemic Region of Mexico. Viruses. 2019; 11(5):407. https://doi.org/10.3390/v11050407
Chicago/Turabian StyleKim, Young Chan, César López-Camacho, Nallely Garcia-Larragoiti, Alan Cano-Mendez, Karina Guadalupe Hernandez-Flores, Carlos Alonso Domínguez-Alemán, Maria Antonieta Mar, Héctor Vivanco-Cid, Martha Eva Viveros-Sandoval, and Arturo Reyes-Sandoval. 2019. "Development of an E2 ELISA Methodology to Assess Chikungunya Seroprevalence in Patients from an Endemic Region of Mexico" Viruses 11, no. 5: 407. https://doi.org/10.3390/v11050407
APA StyleKim, Y. C., López-Camacho, C., Garcia-Larragoiti, N., Cano-Mendez, A., Hernandez-Flores, K. G., Domínguez-Alemán, C. A., Antonieta Mar, M., Vivanco-Cid, H., Viveros-Sandoval, M. E., & Reyes-Sandoval, A. (2019). Development of an E2 ELISA Methodology to Assess Chikungunya Seroprevalence in Patients from an Endemic Region of Mexico. Viruses, 11(5), 407. https://doi.org/10.3390/v11050407




