Experimental Vertical Transmission of Chikungunya Virus by Brazilian and Florida Aedes Albopictus Populations
Abstract
:1. Introduction
2. Materials and Methods
2.1. Ethics Statement
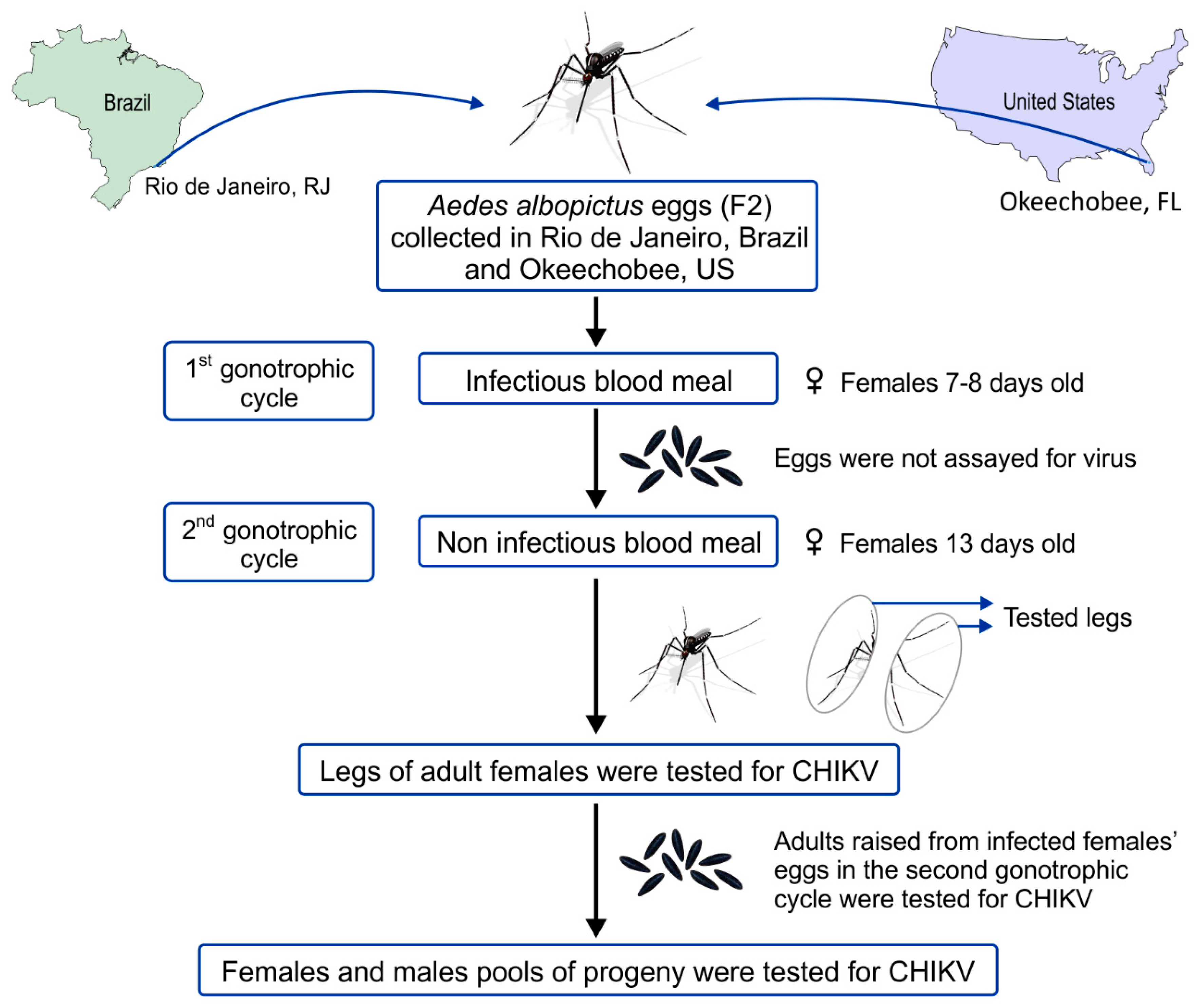
2.2. Mosquito Populations
2.3. Mosquito Rearing
2.4. Virus and Mosquito Oral Infection
2.5. Maintenance of Adults
2.6. Determination of Progeny Infectious Status
2.7. Statistical Analyses
3. Results
3.1. Parental Female Feeding and Oviposition
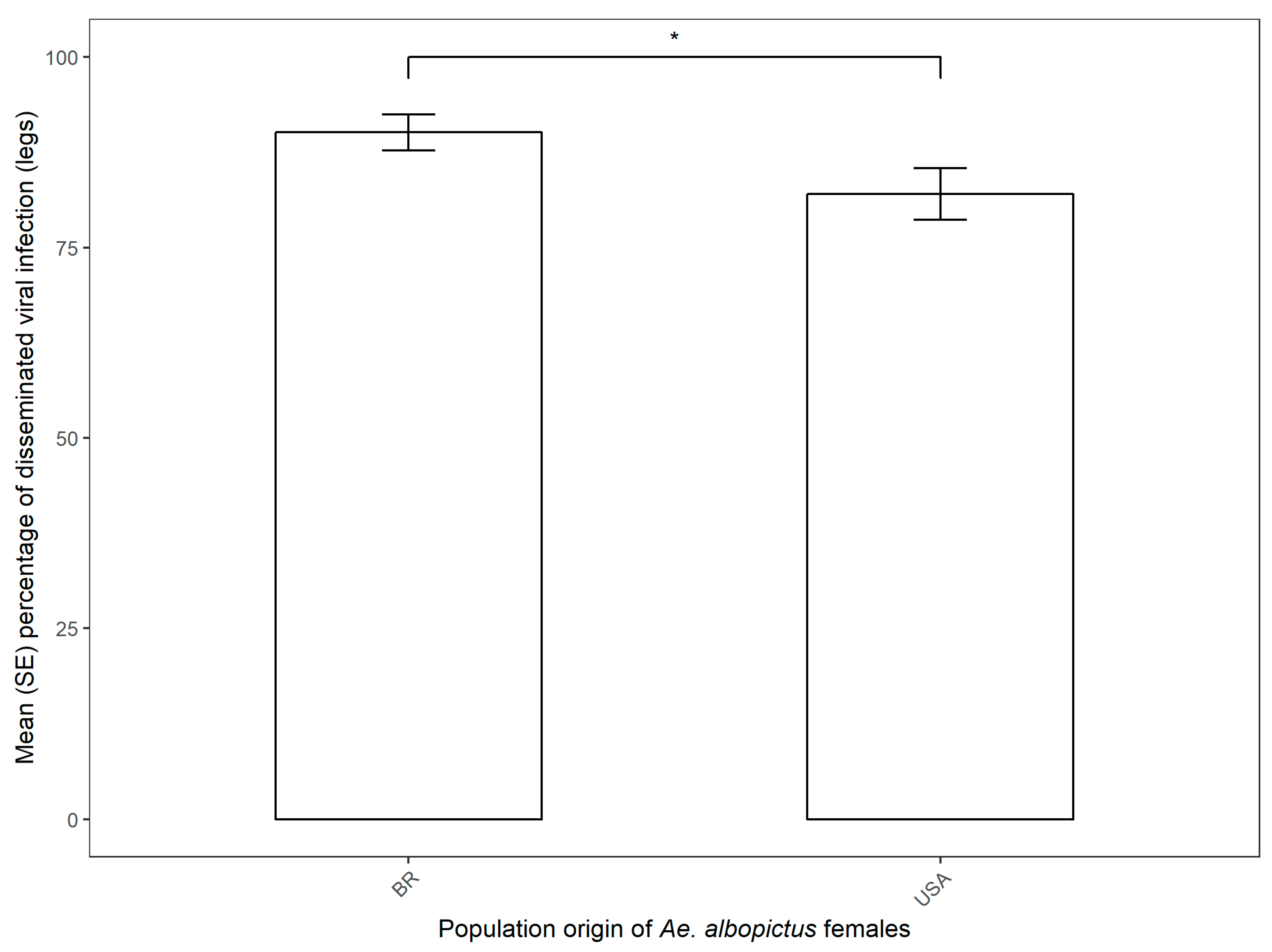
3.2. CHIKV Dissemination in Parental Females and Vertical Transmission
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Honório, N.A.; Câmara, D.C.P.; Calvet, G.A.; Brasil, P. Chikungunya: An arbovirus infection in the process of establishment and expansion in Brazil. Cad. Saúde Públ. 2015, 31, 906–908. [Google Scholar] [CrossRef] [PubMed]
- Lowe, R.; Barcellos, C.; Brasil, P.; Cruz, O.G.; Honório, N.A.; Kuper, H.; Carvalho, M.S. The Zika Virus Epidemic in Brazil: From Discovery to Future Implications. Int. J. Environ. Res. Public Health 2018, 15, 96. [Google Scholar] [CrossRef] [PubMed]
- Jupp, P.G.; McIntosh, B.M. Chikungunya virus disease. In The Arboviruses: Epidemiology and Ecology Vol. II; Monath, T.P., Ed.; CRC Press: Boca Raton, FL, USA; Volume 2, pp. 137–157.
- Higgs, S.; Vanlandingham, D. Chikungunya virus and its mosquito vectors. Vector Borne Zoonotic Dis. 2015, 15, 231–240. [Google Scholar] [CrossRef] [PubMed]
- Weaver, S.C. Chikungunya in the New World: Prospects for spread and health impact. PLoS Negl. Trop. Dis. 2014, 8, e2921. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Chikungunya Virus. Geographic Distribution. Available online: https://www.cdc.gov/chikungunya/geo/index.html (accessed on 11 March 2019).
- Ministério da Saúde. Secretaria de Vigilância em Saúde. Boletins epidemiológicos. Available online: http://portalms.saude.gov.br/boletins-epidemiologicos (accessed on 11 March 2019).
- Centers for Disease Control and Prevention. Chikungunya Virus. 2018 Provisional Data for the United States. Available online: https://www.cdc.gov/chikungunya/geo/united-states-2018.html (accessed on 11 March 2019).
- Lounibos, L.P.; Kramer, L.D. Invasiveness of Aedes aegypti and Aedes albopictus and Vectorial Capacity for Chikungunya Virus. J. Infect. Dis. 2016, 214, S453–S458. [Google Scholar] [CrossRef]
- Kraemer, M.U.; Sinka, M.E.; Duda, K.A.; Mylne, A.Q.; Shearer, F.M.; Barker, C.M.; Moore, C.G.; Carvalho, R.G.; Coelho, G.E.; Van Bortel, W.; et al. The global distribution of the arbovirus vectors Aedes aegypti and Ae. albopictus. eLife 2015, 4, e08347. [Google Scholar] [CrossRef]
- Honório, N.A.; Castro, M.G.; Barros, F.S.; Magalhães, M.A.; Sabroza, P.C. The spatial distribution of Aedes aegypti and Aedes albopictus in a transition zone, Rio de Janeiro, Brazil. Cad. Saude Publ. 2009, 25, 1203–1214. [Google Scholar] [CrossRef]
- Benedict, M.Q.; Levine, R.S.; Hawley, W.A.; Lounibos, L.P. Spread of the tiger: Global risk of invasion by the mosquito Aedes albopictus. Vector Borne Zoonotic Dis. 2007, 7, 76–85. [Google Scholar] [CrossRef] [PubMed]
- Ayllón, T.; Câmara, D.C.P.; Morone, F.C.; Gonçalves, L.S.; Barros, F.S.M.; Brasil, P.; Carvalho, M.S.; Honório, N.A. Dispersion and oviposition of Aedes albopictus in a Brazilian slum: Initial evidence of Asian tiger mosquito domiciliation in urban environments. PLoS ONE 2018, 13, e0195014. [Google Scholar] [CrossRef] [PubMed]
- Lima-Camara, T.N.; Honório, N.A.; Lourenço-de-Oliveira, R. Frequency and spatial distribution of Aedes aegypti and Aedes albopictus (Diptera, Culicidae) in Rio de Janeiro, Brazil. Cad. Saude Publ. 2006, 22, 2079–2084. [Google Scholar] [CrossRef]
- Honório, N.A.; Wiggins, K.; Câmara, D.C.P.; Eastmond, B.; Alto, B.W. Chikungunya virus vector competency of Brazilian and Florida mosquito vectors. PLoS Negl. Trop. Dis. 2018, 12, e0006521. [Google Scholar] [CrossRef] [PubMed]
- Vega-Rúa, A.; Zouache, K.; Girod, R.; Failloux, A.B.; Lourenço-de-Oliveira, R. High level of vector competence of Aedes aegypti and Aedes albopictus from ten American countries as a crucial factor in the spread of Chikungunya virus. J. Virol. 2014, 88, 6294–6306. [Google Scholar] [CrossRef] [PubMed]
- Alto, B.W.; Wiggins, K.; Eastmond, B.; Velez, D.; Lounibos, L.P.; Lord, C.C. Transmission risk of two chikungunya lineages by invasive mosquito vectors from Florida and the Dominican Republic. PLoS Negl. Trop. Dis. 2017, 11, e0005724. [Google Scholar] [CrossRef] [PubMed]
- Costa-da-Silva, A.L.; Ioshino, R.S.; Petersen, V.; Lima, A.F.; dos Passos Cunha, M.; Wiley, M.R.; Ladner, J.T.; Prieto, K.; Palacios, G.; Costa, D.D.; et al. First report of naturally infected Aedes aegypti with chikungunya virus genotype ECSA in the Americas. PLoS Negl. Trop. Dis. 2017, 11, e0005630. [Google Scholar] [CrossRef] [PubMed]
- Clements, A.N. The biology of mosquitoes. In Transmission of Viruses and Interactions with Bacteria; CABI: Wallingford, UK, 2012; Volume 3, 571p. [Google Scholar]
- Buckner, E.A.; Alto, B.W.; Lounibos, L.P. Vertical transmission of Key West dengue-1 virus by Aedes aegypti and Aedes albopictus (Diptera:Culicidae) mosquitoes from Florida. J. Med. Entomol. 2013, 50, 1291–1297. [Google Scholar] [CrossRef] [PubMed]
- Lequime, S.; Lambrechts, L. Vertical transmission of arboviruses in mosquitoes: A historical perspective. Infect. Genet. Evol. 2014, 28, 681–690. [Google Scholar] [CrossRef] [PubMed]
- Thenmozhi, V.; Tewari, S.C.; Manavalan, R.; Balasubramanian, A.; Gajanana, A. Natural vertical transmission of dengue viruses in Aedes aegypti in southern India. Trans. R. Soc. Trop. Med. Hyg. 2000, 94, 507. [Google Scholar] [CrossRef]
- Angel, B.; Joshi, V. Distribution and seasonality of vertically transmitted dengue viruses in Aedes mosquitoes in arid and semi-arid areas of Rajasthan, India. J. Vector Borne Dis. 2008, 45, 56–59. [Google Scholar] [PubMed]
- Delatte, H.; Paupy, C.; Dehecq, J.S.; Thiria, J.; Failloux, A.B.; Fontenille, D. Aedes albopictus, vector of chikungunya and dengue viruses in Reunion Island: Biology and control. Parasite 2008, 15, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Ratsitorahina, M.; Harisoa, J.; Ratovonjato, J.; Biacabe, S.; Reynes, J.M.; Zeller, H.; Raoelina, Y.; Talarmin, A.; Richard, V.; Soares, J.L. Outbreak of dengue and Chikungunya fevers, Toamasina, Madagascar, 2006. Emerg. Infect. Dis. 2008, 14, 1135–1137. [Google Scholar] [CrossRef] [PubMed]
- Thavara, U.; Tawatsin, A.; Pengsakul, T.; Bhakdeenuan, P.; Chanama, S.; Anantapreecha, S.; Molito, C.; Chompoosri, J.; Thammapalo, S.; Sawanpanyalert, P.; et al. Outbreak of Chikungunya fever in Thailand and virus detection in field population of vector mosquitoes, Aedes aegypti (L.) and Aedes albopictus Skuse (Diptera: Culicidae). Southeast Asian J. Trop. Med. Public Health 2009, 40, 951–962. [Google Scholar] [PubMed]
- Hailin, Z.; Yunzhi, Z.; Zhuqing, M. Transovarial transmission of Chikungunya virus in Aedes albopictus and Aedes aegypti mosquitoes. Chin. J. Virol. 1993, 9, 222–227. [Google Scholar]
- Chompoosri, J.; Thavara, U.; Tawatsin, A.; Boonserm, R.; Phumee, A.; Sangkitporn, S.; Siriyasatien, P. Vertical transmission of Indian Ocean Lineage of chikungunya virus in Aedes aegypti and Aedes albopictus mosquitoes. Parasit. Vectors 2016, 9, 227. [Google Scholar] [CrossRef]
- Agarwal, A.; Dash, P.K.; Singh, A.K.; Sharma, S.; Gopalan, N.; Rao, P.V.; Parida, M.M.; Reiter, P. Evidence of experimental vertical transmission of emerging novel ECSA genotype of Chikungunya Virus in Aedes aegypti. PLoS Negl. Trop. Dis. 2014, 8, e2990. [Google Scholar] [CrossRef] [PubMed]
- Mourya, D.T. Absence of transovarial transmission of Chikungunya virus in Aedes aegypti & Ae. albopictus mosquitoes. Indian J. Med. Res. 1987, 85, 593–595. [Google Scholar] [PubMed]
- Vazeille, M.; Mousson, L.; Failloux, A.B. Failure to demonstrate experimental vertical transmission of the epidemic strain of Chikungunya virus in Aedes albopictus from La Reunion Island, Indian Ocean. Mem. Inst. Oswaldo Cruz 2009, 104, 632–635. [Google Scholar] [CrossRef] [PubMed]
- Jupp, P.G.; McIntosh, B.M.; Dos Santos, I. Laboratory vector studies on six mosquito and one tick species with chikungunya virus. Trans. R. Soc. Trop. Med. 1981, 75, 15–19. [Google Scholar] [CrossRef]
- Kaur, P.; Lee, R.C.H.; Chu, J.J.H. Infectious viral quantification of chikungunya virus-virus plaque assay. In Chikungunya Virus: Methods in Molecular Biology; Chu, J.J.H., Ang, S.K., Eds.; Springer: New York, NY, USA, 2016; pp. 93–103. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing. Available online: https://www.r-project.org/ (accessed on 1 February 2018).
- RStudio Team. RStudio: Integrated Development for R. Available online: https://www.rstudio.com/ (accessed on 1 February 2018).
- Venables, W.N.; Ripley, B.D. Modern Applied Statistics with S, 4th ed.; Springer: New York, NY, USA, 2003; 498p. [Google Scholar]
- Simon, F.; Javelle, E.; Gasque, P. Chikungunya virus infections. N. Engl. J. Med. 2015, 373, 93–94. [Google Scholar] [CrossRef] [PubMed]
- Olowokure, B.; Francis, L.; Polson-Edwards, K.; Nasci, R.; Quenel, P.; Aldighieri, S.; Rousset, D.; Gutierrez, C.; Ramon-Pardo, P.; Santos, T.D.; et al. The Caribbean response to chikungunya. Lancet Infect. Dis. 2014, 14, 1039–1040. [Google Scholar] [CrossRef]
- Nunes, M.R.; Faria, N.R.; Vasconcelos, J.M.; Golding, N.; Kraemer, M.U.; Oliveira, L.F.; Azevedo, R.S.; Silva, D.E.; Silva, E.V.; Silva, S.P.; et al. Emergence and potential for spread of chikungunya virus in Brazil. BMC Med. 2015, 13, 102. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, M.G.; Andrade, A.M.; Costa, M.C.; Castro, J.N.; Oliveira, F.L.; Goes, C.S.; Maia, M.; Santana, E.B.; Nunes, B.T.; Vasconcelos, P.F. East/central/south African genotype chikungunya virus, Brazil, 2014. Emerg. Infect. Dis. 2015, 21, 906–907. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Chikungunya Virus. 2014 Final Data for the United States. Available online: https://www.cdc.gov/chikungunya/geo/united-states-2014.html (accessed on 11 March 2019).
- Adams, B.; Boots, M. How important is vertical transmission in mosquitoes for the persistence of dengue? Insights from a mathematical model. Epidemics 2010, 2, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Chikungunya Virus. Mosquito Surveillance Software. Available online: https://www.cdc.gov/westnile/resourcepages/mosqSurvSoft.html (accessed on 19 March 2019).
- Bellini, R.; Medici, A.; Calzolari, M.; Bonilauri, P.; Cavrini, F.; Sambri, V.; Angelini, P.; Dottori, M. Impact of chikungunya virus on Aedes albopictus females and possibility of vertical transmission using the actors of the 2007 outbreak in Italy. PLoS ONE 2012, 7, e28360. [Google Scholar] [CrossRef] [PubMed]
- Vazeille, M.; Moutailler, S.; Coudrier, D.; Rousseaux, C.; Khun, H.; Huerre, M.; Thiria, J.; Dehecq, J.S.; Fontenille, D.; Schuffenecker, I.; et al. Two Chikungunya isolates from the outbreak of La Reunion (Indian Ocean) exhibit different patterns of infection in the mosquito Aedes albopictus. PLoS ONE 2007, 2, e1168. [Google Scholar] [CrossRef] [PubMed]
- Niyas, K.P.; Abraham, R.; Unnikrishnan, R.N.; Mathew, T.; Nair, S.; Manakkadan, A.; Issac, A.; Sreekumar, E. Molecular characterization of Chikungunya virus isolates from clinical samples and adult Aedes albopictus mosquitoes emerged from larvae from Kerala, South India. Virol. J. 2010, 7, 189. [Google Scholar] [CrossRef] [PubMed]
- Ferreira-de-Lima, V.H.; Lima-Camara, T.N. Natural vertical transmission of dengue virus in Aedes aegypti and Aedes albopictus: A systematic review. Parasit. Vectors 2018, 11, 77. [Google Scholar] [CrossRef] [PubMed]

| Mosquito Stages | Descriptive Statistics | Ae. albopictus Population | |
|---|---|---|---|
| Rio de Janeiro/BR | Okeechobee/US | ||
| Eggs | Total | 5299 | 3384 |
| Mean (SD) | 23.14 (25.80) | 26.23 (25.86) | |
| Minimum–Maximum | 0–119 | 0–121 | |
| Pupae | Total | 2818 | 1885 |
| Mean (SD) | 20.13 (16.82) | 20.71 (20.85) | |
| Minimum-Maximum | 0–86 | 1–107 | |
| Adult females | Total | 1309 | 866 |
| Mean (SD) | 9.49 (8.06) | 9.52 (9.75) | |
| Minimum–Maximum | 0–43 | 0–46 | |
| Positive/Total Female Pools | 1/138 | 0/94 | |
| Minimum Infection Rate | 0.76 | 0 | |
| Adult males | Total | 1399 | 942 |
| Mean (SD) | 10.14 (8.67) | 10.35 (10.76) | |
| Minimum–Maximum | 0–38 | 0–60 | |
| Positive/Total Male Pools | 4/147 | 1/102 | |
| Minimum Infection Rate | 2.86 | 1.06 | |
| Response | Model | Effect | Estimate | Standard Error | 95% CI | |
|---|---|---|---|---|---|---|
| Lower Bound | Upper Bound | |||||
| Viral dissemination (parental females) | #1 | (Intercept) | 9.125 | 0.263 | 5.625 | 15.904 |
| Population origin: USA | 0.500 | 0.350 | 0.248 | 0.987 | ||
| #2 | (Intercept) | 6.312 | 0.122 | 5.007 | 8.067 | |
| Vertical transmission: positive | 2.376 | 1.566 | NA | NA | ||
| Viral titer (parental females) | #3 | (Intercept) | 12.607 | 0.426 | 11.773 | 13.441 |
| Population origin: USA | −2.913 | 0.642 | −4.172 | −1.654 | ||
| #4 | (Intercept) | 11.332 | 0.334 | 10.678 | 11.987 | |
| Vertical transmission: positive | −0.143 | 2.274 | −4.599 | 4.314 | ||
| Viral titer (progeny) | #5 | (Intercept) | 1.306 | 0.033 | 1.241 | 1.372 |
| Population origin: USA | −0.006 | 0.053 | −0.111 | 0.099 | ||
| #6 | (Intercept) | 2.462 | 0.099 | 2.268 | 2.656 | |
| Vertical transmission: positive | −0.580 | 0.886 | −2.316 | 1.155 | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Honório, N.A.; Wiggins, K.; Eastmond, B.; Câmara, D.C.P.; Alto, B.W. Experimental Vertical Transmission of Chikungunya Virus by Brazilian and Florida Aedes Albopictus Populations. Viruses 2019, 11, 353. https://doi.org/10.3390/v11040353
Honório NA, Wiggins K, Eastmond B, Câmara DCP, Alto BW. Experimental Vertical Transmission of Chikungunya Virus by Brazilian and Florida Aedes Albopictus Populations. Viruses. 2019; 11(4):353. https://doi.org/10.3390/v11040353
Chicago/Turabian StyleHonório, Nildimar Alves, Keenan Wiggins, Bradley Eastmond, Daniel Cardoso Portela Câmara, and Barry W. Alto. 2019. "Experimental Vertical Transmission of Chikungunya Virus by Brazilian and Florida Aedes Albopictus Populations" Viruses 11, no. 4: 353. https://doi.org/10.3390/v11040353
APA StyleHonório, N. A., Wiggins, K., Eastmond, B., Câmara, D. C. P., & Alto, B. W. (2019). Experimental Vertical Transmission of Chikungunya Virus by Brazilian and Florida Aedes Albopictus Populations. Viruses, 11(4), 353. https://doi.org/10.3390/v11040353





