Distinct Lineages of Feline Parvovirus Associated with Epizootic Outbreaks in Australia, New Zealand and the United Arab Emirates
Abstract
1. Introduction
2. Materials and Methods
2.1. Retrospective Case Data Analysis
2.2. Prospective Sample Collection
2.3. DNA Extraction and PCR
2.4. Sequence Analysis
2.5. FPV Viral Load Determination by qPCR and Multiplex PCR for Faecal Co-Pathogens
2.6. Samples for Histological Examination
3. Results
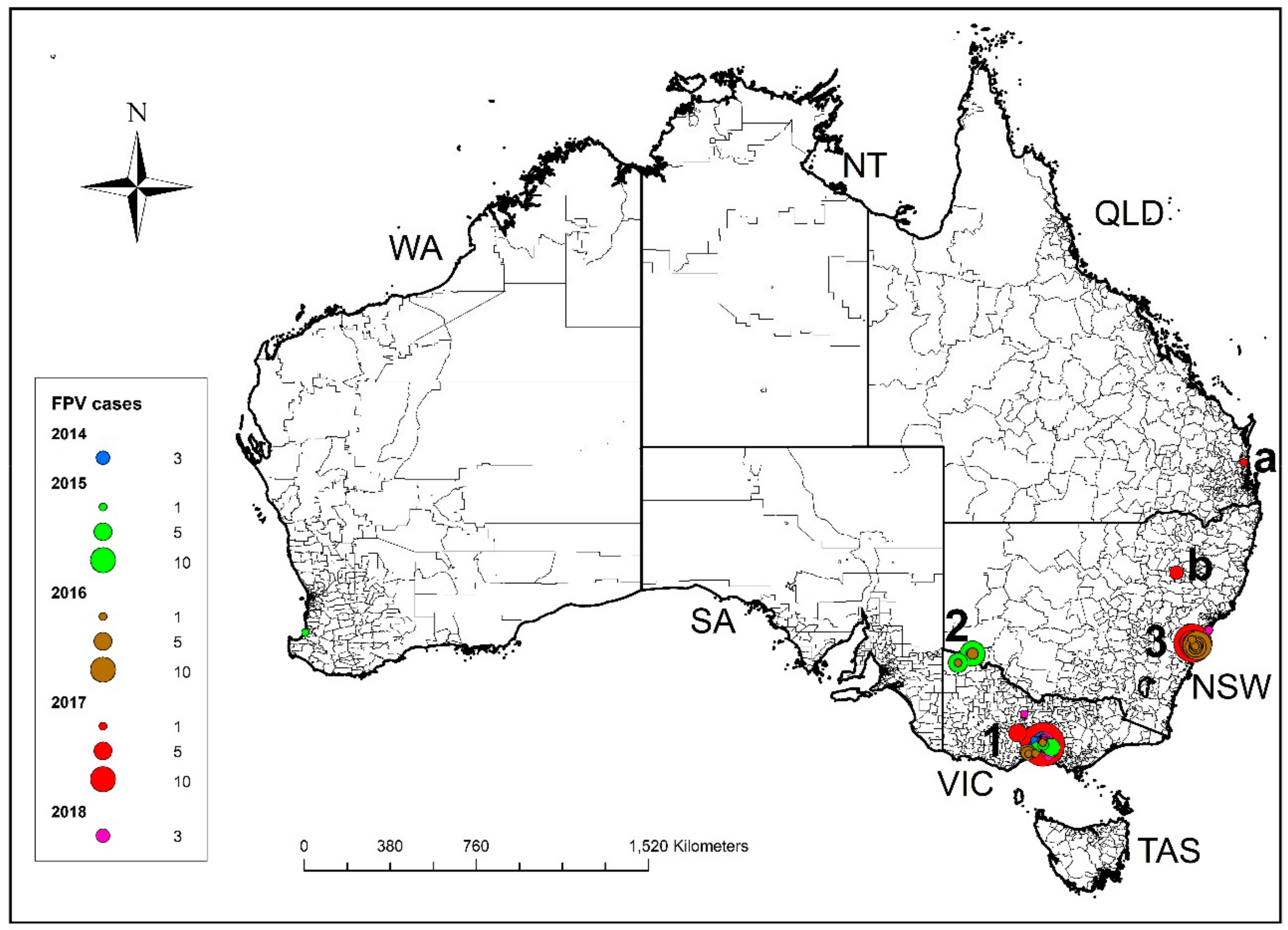
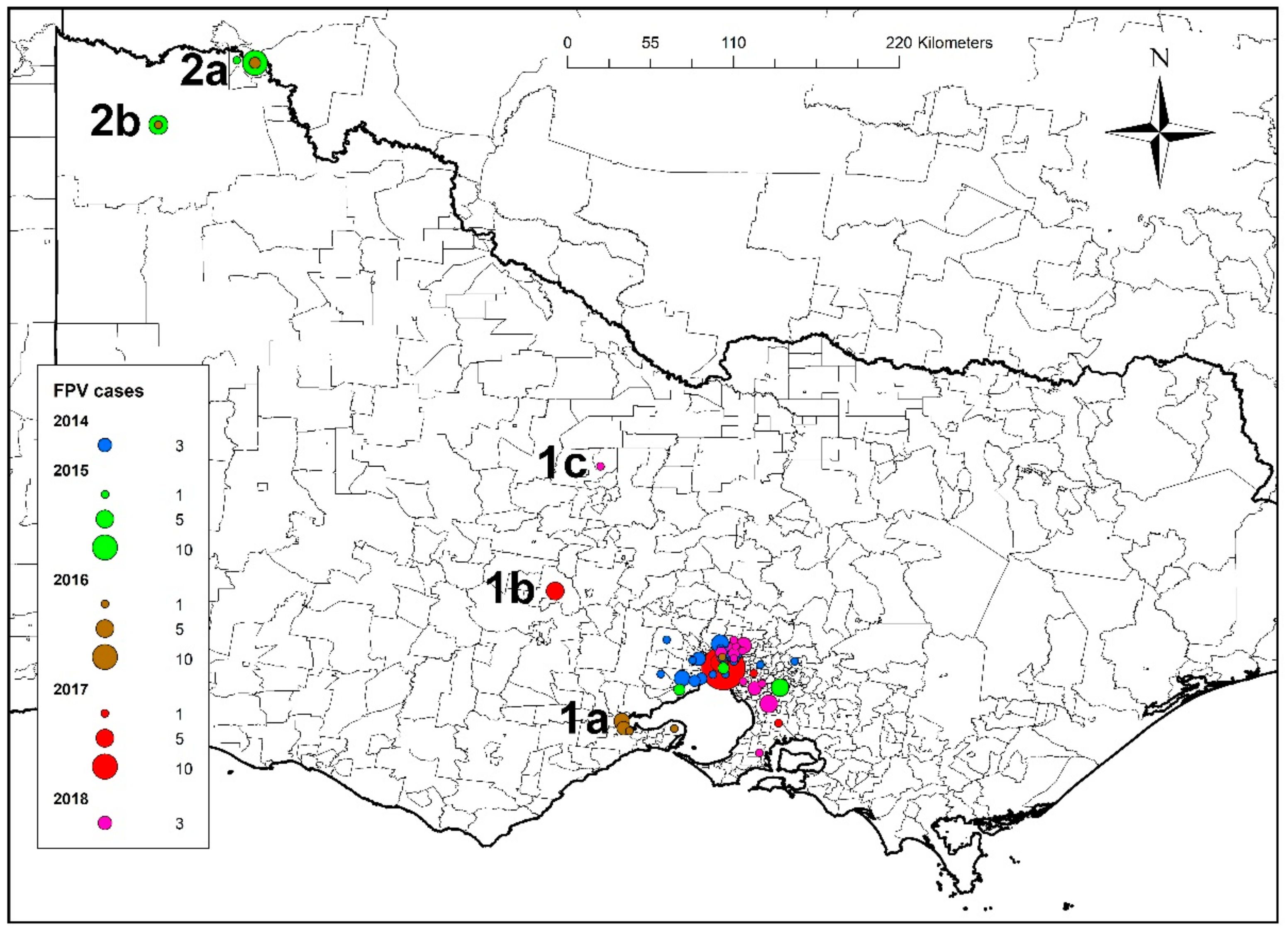
3.1. Australian Cases
3.1.1. Australian Cases—Outbreak Sites, Case Numbers and Distribution
3.1.2. Australian Cases—Vaccination Protocols and Coverage
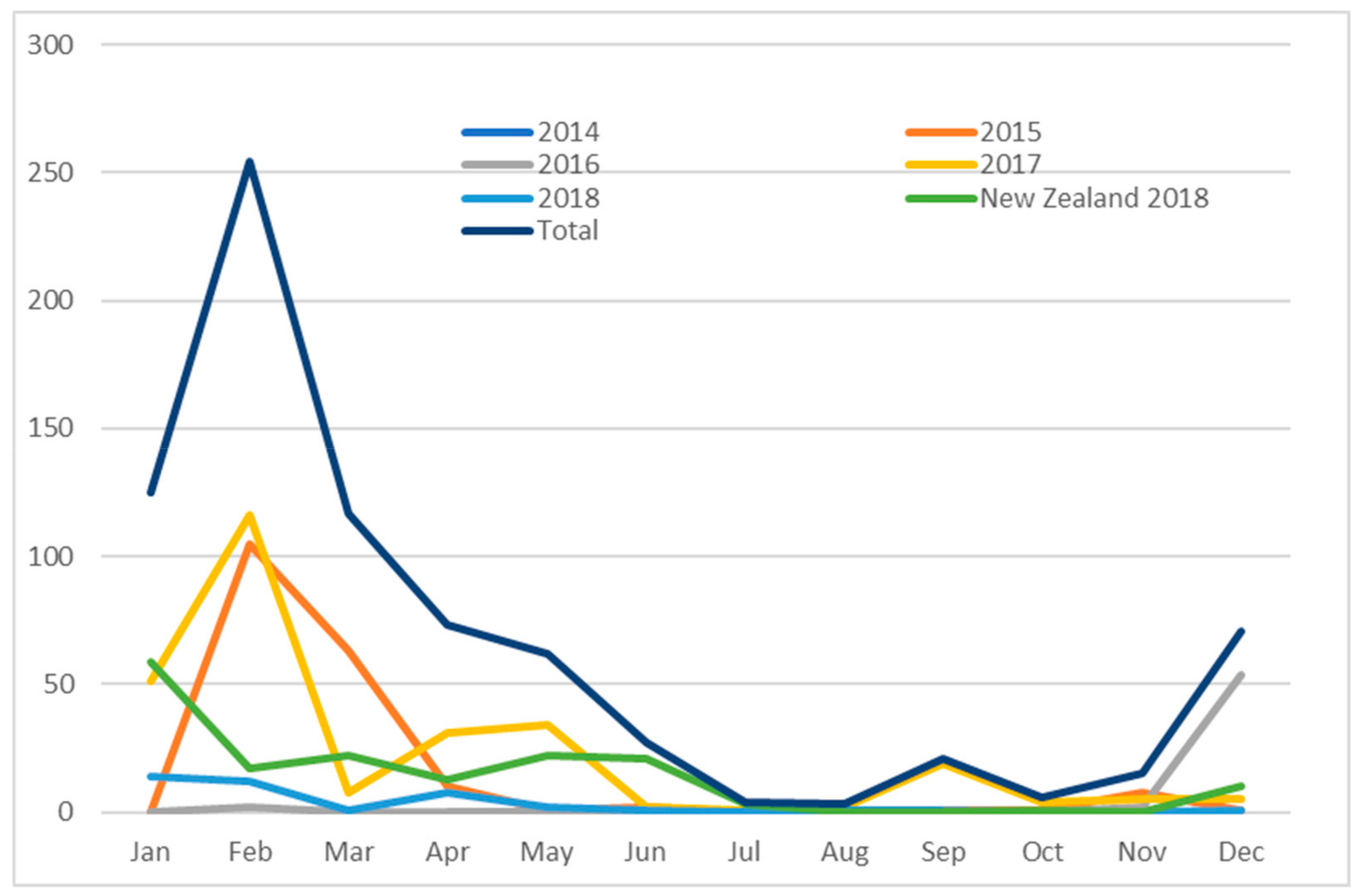
3.2. Case data—New Zealand Cases
3.3. Case Data—UAE Cases
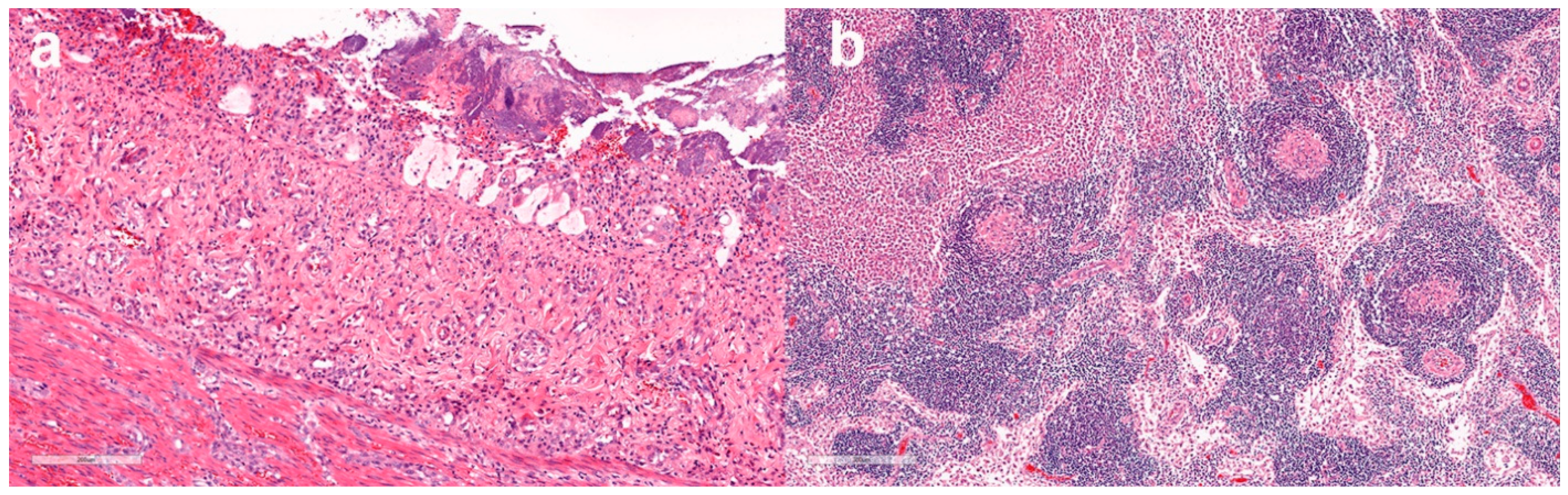
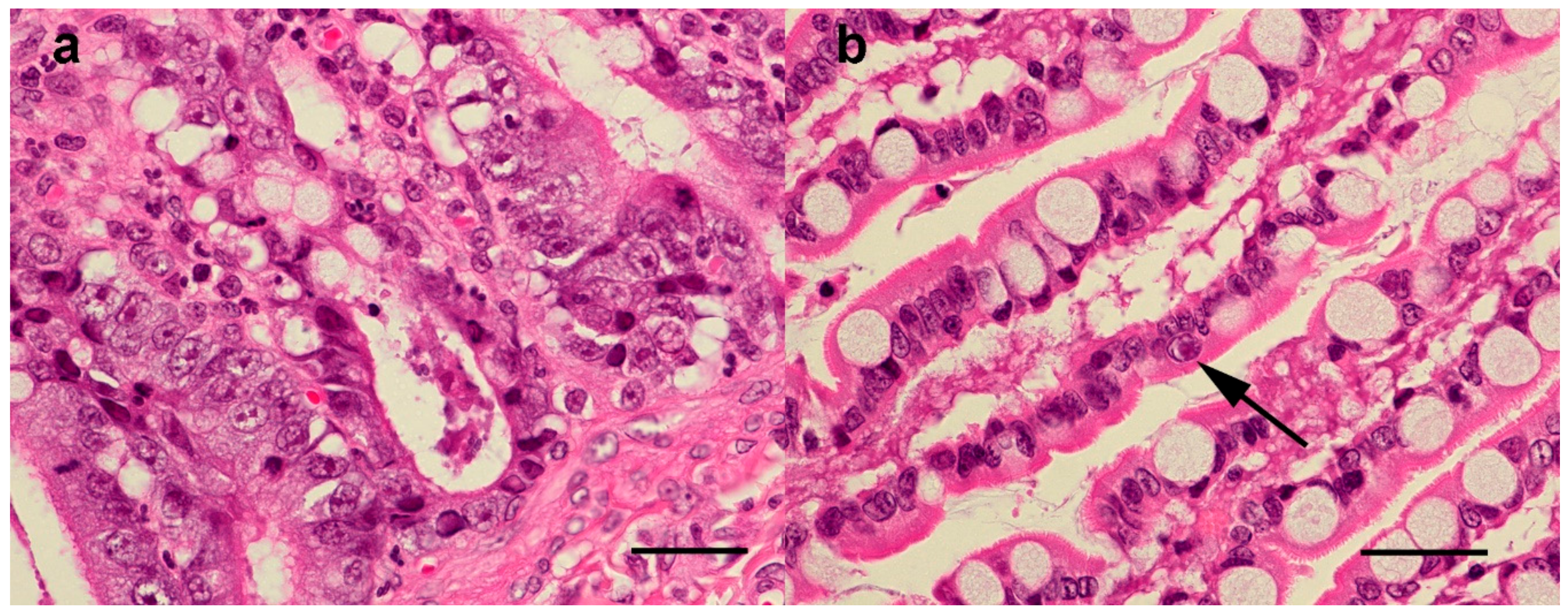
3.4. Histopathological Findings
3.4.1. Histopathological Findings—Australian Cases
3.4.2. Histopathological findings—New Zealand cases
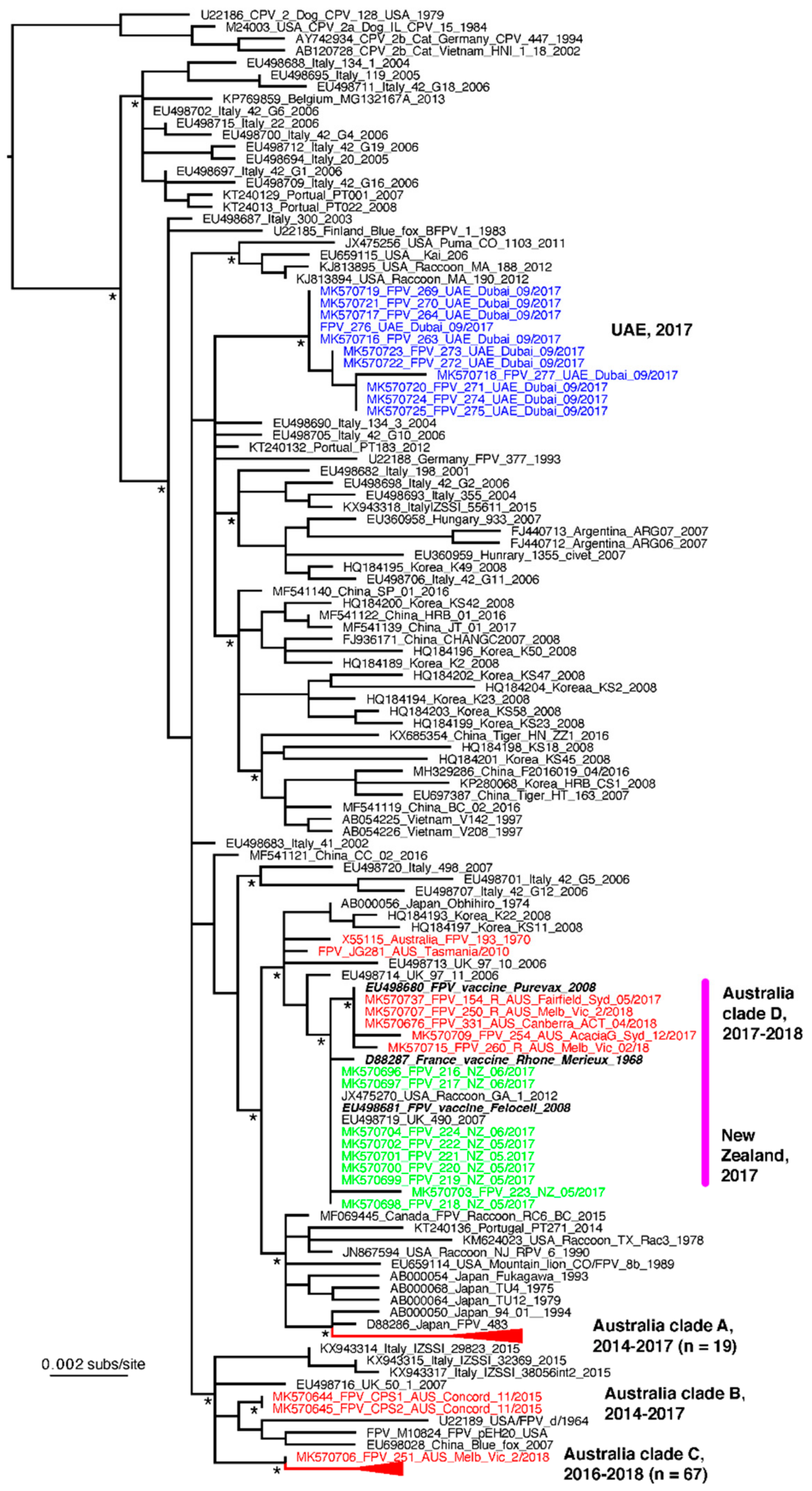
3.5. VP2 Sequencing and Phylogenetic Analysis
3.6. FPV Viral Load and Concurrent Faecal Pathogens
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Barrs, V.R. Feline Panleukopenia: A Re-emergent Disease. Vet. Clin. North. Am. Small Anim. Pract. 2019, 49, 651–670. [Google Scholar] [CrossRef] [PubMed]
- Cotmore, S.F.; Agbandje-McKenna, M.; Chiorini, J.A.; Mukha, D.V.; Pintel, D.J.; Qiu, J.; Soderlund-Venermo, M.; Tattersall, P.; Tijssen, P.; Gatherer, D.; et al. The family Parvoviridae. Arch. Virol. 2014, 159, 1239–1247. [Google Scholar] [CrossRef] [PubMed]
- Hueffer, K.; Govindasamy, L.; Agbandje-McKenna, M.; Parrish, C.R. Combinations of two capsid regions controlling canine host range determine canine transferrin receptor binding by canine and feline parvoviruses. J. Virol. 2003, 77, 10099–10105. [Google Scholar] [CrossRef] [PubMed]
- Goodman, L.B.; Lyi, S.M.; Johnson, N.C.; Cifuente, J.O.; Hafenstein, S.L.; Parrish, C.R. Binding site on the transferrin receptor for the parvovirus capsid and effects of altered affinity on cell uptake and infection. J. Virol. 2010, 84, 4969–4978. [Google Scholar] [CrossRef]
- Mochizuki, M.; Horiuchi, M.; Hiragi, H.; San Gabriel, M.C.; Yasuda, N.; Uno, T. Isolation of canine parvovirus from a cat manifesting clinical signs of feline panleukopenia. J. Clin. Microbiol. 1996, 34, 2101–2105. [Google Scholar]
- Ikeda, Y.; Nakamura, K.; Miyazawa, T.; Takahashi, E.; Mochizuki, M. Feline host range of canine parvovirus: Recent emergence of new antigenic types in cats. Emerg. Infect. Dis. 2002, 8, 341–346. [Google Scholar] [CrossRef]
- Gamoh, K.; Shimazaki, Y.; Makie, H.; Senda, M.; Itoh, O.; Inoue, Y. The pathogenicity of canine parvovirus type-2b, FP84 strain isolated from a domestic cat, in domestic cats. J. Vet. Med. Sci. 2003, 65, 1027–1029. [Google Scholar] [CrossRef]
- Decaro, N.; Desario, C.; Amorisco, F.; Losurdo, M.; Colaianni, M.L.; Greco, M.F.; Buonavoglia, C. Canine parvovirus type 2c infection in a kitten associated with intracranial abscess and convulsions. J. Feline Med. Surg. 2011, 13, 231–236. [Google Scholar] [CrossRef]
- Kelman, M.; Ward, M.P.; Barrs, V.R.; Norris, J.M. The geographic distribution and financial impact of canine parvovirus in Australia. Transbound. Emerg. Dis. 2019, 66, 299–311. [Google Scholar] [CrossRef]
- Ward, M.P.; Kelman, M. Companion animal disease surveillance: A new solution to an old problem? Spat. Spatio-Temporal Epidemiol. 2011, 2, 147–157. [Google Scholar] [CrossRef]
- Decaro, N.; Elia, G.; Desario, C.; Roperto, S.; Martella, V.; Campolo, M.; Lorusso, A.; Cavalli, A.; Buonavoglia, C. A minor groove binder probe real-time PCR assay for discrimination between type 2-based vaccines and field strains of canine parvovirus. J. Virol. Methods 2006, 136, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Decaro, N.; Desario, C.; Miccolupo, A.; Campolo, M.; Parisi, A.; Martella, V.; Amorisco, F.; Lucente, M.S.; Lavazza, A.; Buonavoglia, C. Genetic analysis of feline panleukopenia viruses from cats with gastroenteritis. J. Gen. Virol. 2008, 89, 2290–2298. [Google Scholar] [CrossRef] [PubMed]
- Guindon, S.; Dufayard, J.F.; Lefort, V.; Anisimova, M.; Hordijk, W.; Gascuel, O. New algorithms and methods to estimate maximum-likelihood phylogenies: Assessing the performance of PhyML 3.0. Syst. Biol. 2010, 59, 307–321. [Google Scholar] [CrossRef] [PubMed]
- Decaro, N.; Elia, G.; Martella, V.; Desario, C.; Campolo, M.; Trani, L.D.; Tarsitano, E.; Tempesta, M.; Buonavoglia, C. A real-time PCR assay for rapid detection and quantitation of canine parvovirus type 2 in the feces of dogs. Vet. Microbiol. 2005, 105, 19–28. [Google Scholar] [CrossRef]
- Decaro, N.; Buonavoglia, D.; Desario, C.; Amorisco, F.; Colaianni, M.L.; Parisi, A.; Terio, V.; Elia, G.; Lucente, M.S.; Cavalli, A.; et al. Characterisation of canine parvovirus strains isolated from cats with feline panleukopenia. Res. Vet. Sci. 2010, 89, 275–278. [Google Scholar] [CrossRef]
- Miranda, C.; Parrish, C.R.; Thompson, G. Canine parvovirus 2c infection in a cat with severe clinical disease. J. Vet. Diagn. Investig. 2014, 26, 462–464. [Google Scholar] [CrossRef]
- Csiza, C.K.; Scott, F.W.; de Lahunta, A.; Gillespie, J.H. Pathogenesis of feline panleukopenia virus in susceptible newborn kittens I. Clinical signs, hematology, serology, and virology. Infect. Immun. 1971, 3, 833–837. [Google Scholar]
- Johnson, R.H. Feline panleucopaenia virus. III. Some properties compared to a feline herpes virus. Res. Vet. Sci. 1966, 7, 112. [Google Scholar] [CrossRef]
- Parrish, C.R. Host range relationships and the evolution of canine parvovirus. Vet. Microbiol. 1999, 69, 29–40. [Google Scholar] [CrossRef]
- Studdert, M.J.; Peterson, J.E. Some properties of feline panleukopenia virus. Arch. Fur Die Gesamte Virusforsch. 1973, 42, 346–354. [Google Scholar] [CrossRef]
- Martyn, J.C.; Davidson, B.E.; Studdert, M.J. Nucleotide sequence of feline panleukopenia virus: Comparison with canine parvovirus identifies host-specific differences. J. Gen. Virol. 1990, 71, 2747–2753. [Google Scholar] [CrossRef] [PubMed]
- Patterson, E.V.; Reese, M.J.; Tucker, S.J.; Dubovi, E.J.; Crawford, P.C.; Levy, J.K. Effect of vaccination on parvovirus antigen testing in kittens. J. Am. Vet. Med. Assoc. 2007, 230, 359–363. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, M.; Schwertler, S.; Speck, S.; Truyen, U.; Reese, S.; Hartmann, K. Faecal shedding of parvovirus deoxyribonucleic acid following modified live feline panleucopenia virus vaccination in healthy cats. Vet. Rec. 2019, 185, 83. [Google Scholar] [CrossRef] [PubMed]
- Freisl, M.; Speck, S.; Truyen, U.; Reese, S.; Proksch, A.L.; Hartmann, K. Faecal shedding of canine parvovirus after modified-live vaccination in healthy adult dogs. Vet. J. 2017, 219, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Haynes, S.M.; Holloway, S.A. Identification of parvovirus in the bone marrow of eight cats. Aust. Vet. J. 2012, 90, 136–139. [Google Scholar] [CrossRef]
- Pets in Australia: A Survey of Pets and People. Available online: https://animalmedicinesaustralia.org.au/resources-media/ (accessed on 17 November 2019).
- Legge, S.; Murphy, B.G.; McGregor, H.; Woinarski, J.C.Z.; Augusteyn, J.; Ballard, G.; Baseler, M.; Buckmaster, T.; Dickman, C.R.; Doherty, T.; et al. Enumerating a continental-scale threat: How many feral cats are in Australia? Biol. Conserv. 2017, 206, 293–303. [Google Scholar] [CrossRef]
- Coman, B.J.; Jones, E.H.; Westbury, H.A. Protozoan and viral infections of feral cats. Aust. Vet. J. 1981, 57, 319–323. [Google Scholar] [CrossRef]
- Meason-Smith, C.; Diesel, A.; Patterson, A.P.; Older, C.E.; Johnson, T.J.; Mansell, J.M.; Suchodolski, J.S.; Rodrigues Hoffmann, A. Characterization of the cutaneous mycobiota in healthy and allergic cats using next generation sequencing. Vet. Dermatol. 2017, 28, 71-e17. [Google Scholar] [CrossRef]
- Decaro, N.; Buonavoglia, C. Canine parvovirus—A review of epidemiological and diagnostic aspects, with emphasis on type 2c. Vet. Microbiol. 2012, 155, 1–12. [Google Scholar] [CrossRef]
- Decaro, N.; Crescenzo, G.; Desario, C.; Cavalli, A.; Losurdo, M.; Colaianni, M.L.; Ventrella, G.; Rizzi, S.; Aulicino, S.; Lucente, M.S.; et al. Long-term viremia and fecal shedding in pups after modified-live canine parvovirus vaccination. Vaccine 2014, 32, 3850–3853. [Google Scholar] [CrossRef]
- Decaro, N.; Desario, C.; Billi, M.; Lorusso, E.; Colaianni, M.L.; Colao, V.; Elia, G.; Ventrella, G.; Kusi, I.; Bo, S.; et al. Evaluation of an in-clinic assay for the diagnosis of canine parvovirus. Vet. J. 2013, 198, 504–507. [Google Scholar] [CrossRef]
- Decaro, N.; Desario, C.; Elia, G.; Campolo, M.; Lorusso, A.; Mari, V.; Martella, V.; Buonavoglia, C. Occurrence of severe gastroenteritis in pups after canine parvovirus vaccine administration: A clinical and laboratory diagnostic dilemma. Vaccine 2007, 25, 1161–1166. [Google Scholar] [CrossRef]
- Pesavento, P.A.; Murphy, B.G. Common and emerging infectious diseases in the animal shelter. Vet. Pathol. 2014, 51, 478–491. [Google Scholar] [CrossRef]
- Bergmann, M.; Schwertler, S.; Reese, S.; Speck, S.; Truyen, U.; Hartmann, K. Antibody response to feline panleukopenia virus vaccination in healthy adult cats. J. Feline Med. Surg. 2017. [Google Scholar] [CrossRef]
- Lappin, M.R.; Andrews, J.; Simpson, D.; Jensen, W.A. Use of serologic tests to predict resistance to feline herpesvirus 1, feline calicivirus, and feline parvovirus infection in cats. J. Am. Vet. Med. Assoc. 2002, 220, 38–42. [Google Scholar] [CrossRef]
- Day, M.J.; Horzinek, M.C.; Schultz, R.D.; Squires, R.A. WSAVA Guidelines for the vaccination of dogs and cats. J. Small Anim. Pract. 2016, 57, E1-e45. [Google Scholar] [CrossRef]
- Jakel, V.; Cussler, K.; Hanschmann, K.M.; Truyen, U.; Konig, M.; Kamphuis, E.; Duchow, K. Vaccination against Feline Panleukopenia: Implications from a field study in kittens. BMC Vet. Res. 2012, 8, 62. [Google Scholar] [CrossRef]
- Dinnage, J.D.; Scarlett, J.M.; Richards, J.R. Descriptive epidemiology of feline upper respiratory tract disease in an animal shelter. J. Feline Med. Surg. 2009, 11, 816–825. [Google Scholar] [CrossRef]
- RSPCA Australia National Statistics 1999–2000. Available online: https://www.rspca.org.au/sites/default/files/website/The-facts/Statistics/RSPCA%20Australia%20National%20Statistics1999-2000.pdf (accessed on 29 October 19).
- RSPCA Australia National Statistics 2017–2018. Available online: https://www.rspca.org.au/sites/default/files/RSPCA%20Australia%20Annual%20Statistics%202017-2018.pdf (accessed on 29 October 2019).
- Janke, N.; Berke, O.; Flockhart, T.; Bateman, S.; Coe, J.B. Risk factors affecting length of stay of cats in an animal shelter: A case study at the Guelph Humane Society, 2011–2016. Prev. Vet. Med. 2017, 148, 44–48. [Google Scholar] [CrossRef]
- Reif, J.S. Seasonality, natality and herd immunity in feline panleukopenia. Am. J. Epidemiol. 1976, 103, 81–87. [Google Scholar] [CrossRef]
- Decaro, N.; Desario, C.; Campolo, M.; Elia, G.; Martella, V.; Ricci, D.; Lorusso, E.; Buonavoglia, C. Clinical and virological findings in pups naturally infected by canine parvovirus type 2 Glu-426 mutant. J. Vet. Diagn. Investig. 2005, 17, 133–138. [Google Scholar] [CrossRef]
- Decaro, N.; Campolo, M.; Desario, C.; Elia, G.; Martella, V.; Lorusso, E.; Buonavoglia, C. Maternally-derived antibodies in pups and protection from canine parvovirus infection. Biologicals 2005, 33, 261–267. [Google Scholar] [CrossRef]
- Decaro, N.; Buonavoglia, C. Canine parvovirus post-vaccination shedding: Interference with diagnostic assays and correlation with host immune status. Vet. J. 2017, 221, 23–24. [Google Scholar] [CrossRef]
| Primer | Sequence 5′–3′ | Fragment Size (bp) |
|---|---|---|
| CPV2679-F | CCAGATCATCCATCAACATCA | 853 |
| CPV3511-R | TGAACATCATCTGGATCTGTACC | |
| CPV3381-F | CCATGGAAACCAACCATACC | 736 |
| CPV4116-R | AGTTAATTCCTGTTTTACCTCCAA | |
| 555-F | CAGGAAGATATCCAGAAGGA | 583 |
| 555-R | GGTGCTAGTTGATATGTAATAAACA |
| Year | NSW 1 | QLD 2 | VIC 3 | WA 4 | Total |
|---|---|---|---|---|---|
| 2014 | 0 | 0 | 40 | 0 | 40 |
| 2015 | 3 | 0 | 188 | 1 | 192 |
| 2016 | 38 | 0 | 21 | 0 | 59 |
| 2017 | 211 | 1 | 66 | 0 | 278 |
| 2018 | 3 | 0 | 38 | 0 | 41 |
| Total | 255 | 1 | 353 | 1 | 610 |
| Cumulative | ||||
|---|---|---|---|---|
| Age | Frequency | Percent | Frequency | Percent |
| 0–5 weeks | 51 | 13 | 51 | 13 |
| 6–10 weeks | 155 | 39 | 206 | 52 |
| 11–14 weeks | 39 | 10 | 245 | 61 |
| 15–18 weeks | 47 | 12 | 292 | 73 |
| 19–24 weeks | 22 | 6 | 314 | 79 |
| 6–12 months | 22 | 6 | 336 | 84 |
| 1–2 years | 46 | 12 | 382 | 96 |
| >2 years | 17 | 4 | 399 | 100 |
| Case | Age (Weeks) | N 1 | D 2 | Clinical Signs | FPV Viral Copies/mg of Lymph Node | FPV Viral Copies/mg of Faeces | Faecal Co-Pathogens Detected on Multiplex PCR/Giardia Faecal Antigen Tests | Histological Findings—Small Intestine |
|---|---|---|---|---|---|---|---|---|
| 216 | 10.5 | 2 | 15 | Weight loss, diarrhoea | 2.48 × 108 | 4.12 × 103 | Feline coronavirus | NT |
| 217 | 11 | 2 | 15 | Diarrhoea | 9.63 × 106 | 6.79 × 103 | NT | NT |
| 218 | 10.5 | 1 | 14 | Dehydration, diarrhoea | 2.87 × 107 | 4.12 × 103 | Giardia spp. | Enteritis, acute, neutrophlic, mild |
| 219 | 9.5 | 1 | 11 | Diarrhoea | 1.77 × 103 | 3.4 × 103 | Feline coronavirus | NT, CBC WNL |
| 220 | 13 | 1 | 7 | Diarrhoea | 8.22 × 109 | 6.48 × 104 | Feline coronavirus | Mild diffuse fibrosis of lamina propria, CBC: mild lymphopenia |
| 221 | 7 | 1 | 7 | Conjunctivitis, weight loss | 1.08 × 1010 | 1.27 × 108 | Clostridium perfringens | Multifocal crypt necrosis, crypt abscesses, lymphoid depletion, viral inclusion bodies |
| 222 | 10 | 1 | 6 | Diarrhoea | 1.17 × 1010 | 1.32 × 106 | None | Enteritis, neutrophilic and eosinophilic, mild, acute |
| 223 | 10 | 1 | 6 | Diarrhoea | 1.32 × 1010 | NT | NT | Enteritis, plasmacytic, mild |
| 224 | 18 | 2 | 8 | Diarrhoea, sneezing | 1.79 × 108 | 8.02 | Feline coronavirus | NT |
| 88153 | 6 | U | - | Vomiting, dehydration | NT | NT | Giardia spp. | Similar but more severe lesions, to case 221 |
| Accession No. | Variant | Year | Amino Acid VP2 Location | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 5 | 80 | 87 | 93 | 101 | 103 | 145 | 204 | 232 | 297 | 300 | 305 | 323 | 373 | 375 | 426 | 445 | 462 | 555 | 562 | 564 | 568 | |||
| EU659111 1 | FPV ref | 1967 | A | K | M | K | T | V | I | I | V | S | A | D | D | D | D | N | T | P | V | V | N | A |
| MK570663 | This study | 2016–2018 | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • |
| EU498681 2 | Felocell * | 2008 | • | • | • | • | • | • | • | • | I | • | • | • | • | • | • | • | • | • | • | L | • | • |
| EU498680 3 | Purevax * | 2008 | • | • | • | • | • | • | • | • | I | • | • | • | • | • | • | • | • | • | • | L | • | • |
| MK570709 | This study * | 2017 | • | • | • | • | M | • | L | • | I | • | • | • | • | • | • | • | • | • | • | L | • | • |
| MK570715 | This study * | 2018 | • | • | • | • | V | • | • | • | I | • | • | • | • | • | • | • | • | • | • | L | • | • |
| MK570637 4 | This study | 2015–2016 | T | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • |
| MK570646 | This study | 2015 | T | • | • | • | • | • | • | • | • | • | • | • | • | • | • | • | I | • | • | • | • | • |
| MK570644 5 | This study | 2015 | • | • | • | • | I | • | • | • | I | • | • | • | • | • | • | • | • | L | • | • | • | • |
| MK570720 6 | This study | 2017 | • | • | • | • | • | • | • | V | • | • | • | • | • | • | • | • | • | • | • | • | • | • |
| MK570654 7 | This study | 2017–2018 | • | • | • | • | • | • | • | • | I | • | • | • | • | • | • | • | • | • | • | • | • | • |
| MK570710 | This study | 2017 | • | • | • | • | • | • | • | • | I | • | • | • | • | N | • | • | • | • | • | • | • | • |
| CPV Faecal Antigen Test 1 | Parvovirus Detection by Faecal qPCR 2 | ||||
|---|---|---|---|---|---|
| 21.11.2016 | 07.01.2017 | 04.02.2017 | 07.03.2017 | 11.04.2017 | |
| Result | positive | positive | positive | positive | negative |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Van Brussel, K.; Carrai, M.; Lin, C.; Kelman, M.; Setyo, L.; Aberdein, D.; Brailey, J.; Lawler, M.; Maher, S.; Plaganyi, I.; et al. Distinct Lineages of Feline Parvovirus Associated with Epizootic Outbreaks in Australia, New Zealand and the United Arab Emirates. Viruses 2019, 11, 1155. https://doi.org/10.3390/v11121155
Van Brussel K, Carrai M, Lin C, Kelman M, Setyo L, Aberdein D, Brailey J, Lawler M, Maher S, Plaganyi I, et al. Distinct Lineages of Feline Parvovirus Associated with Epizootic Outbreaks in Australia, New Zealand and the United Arab Emirates. Viruses. 2019; 11(12):1155. https://doi.org/10.3390/v11121155
Chicago/Turabian StyleVan Brussel, Kate, Maura Carrai, Carrie Lin, Mark Kelman, Laura Setyo, Danielle Aberdein, Juliana Brailey, Michelle Lawler, Simone Maher, Ildiko Plaganyi, and et al. 2019. "Distinct Lineages of Feline Parvovirus Associated with Epizootic Outbreaks in Australia, New Zealand and the United Arab Emirates" Viruses 11, no. 12: 1155. https://doi.org/10.3390/v11121155
APA StyleVan Brussel, K., Carrai, M., Lin, C., Kelman, M., Setyo, L., Aberdein, D., Brailey, J., Lawler, M., Maher, S., Plaganyi, I., Lewis, E., Hawkswell, A., Allison, A. B., Meers, J., Martella, V., Beatty, J. A., Holmes, E. C., Decaro, N., & Barrs, V. R. (2019). Distinct Lineages of Feline Parvovirus Associated with Epizootic Outbreaks in Australia, New Zealand and the United Arab Emirates. Viruses, 11(12), 1155. https://doi.org/10.3390/v11121155








