MicroRNA Expression Profile in Peripheral Blood Lymphocytes of Sheep Vaccinated with Nigeria 75/1 Peste Des Petits Ruminants Virus
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement and Animal Experiment
2.2. Primary Sheep Testicular Cells Isolation and Virus Inoculation
2.3. Library Construction and Small RNA Sequencing
2.4. Analysis of Small RNA Deep Sequencing Data
2.5. Target Prediction of miRNAs
2.6. Validation Using RT-qPCR
3. Results
3.1. Confirmation of PPR Vaccine Virus Inoculation in Sheep PBMC and ST Cells
3.2. Construction and Sequencing of sRNA Libraries
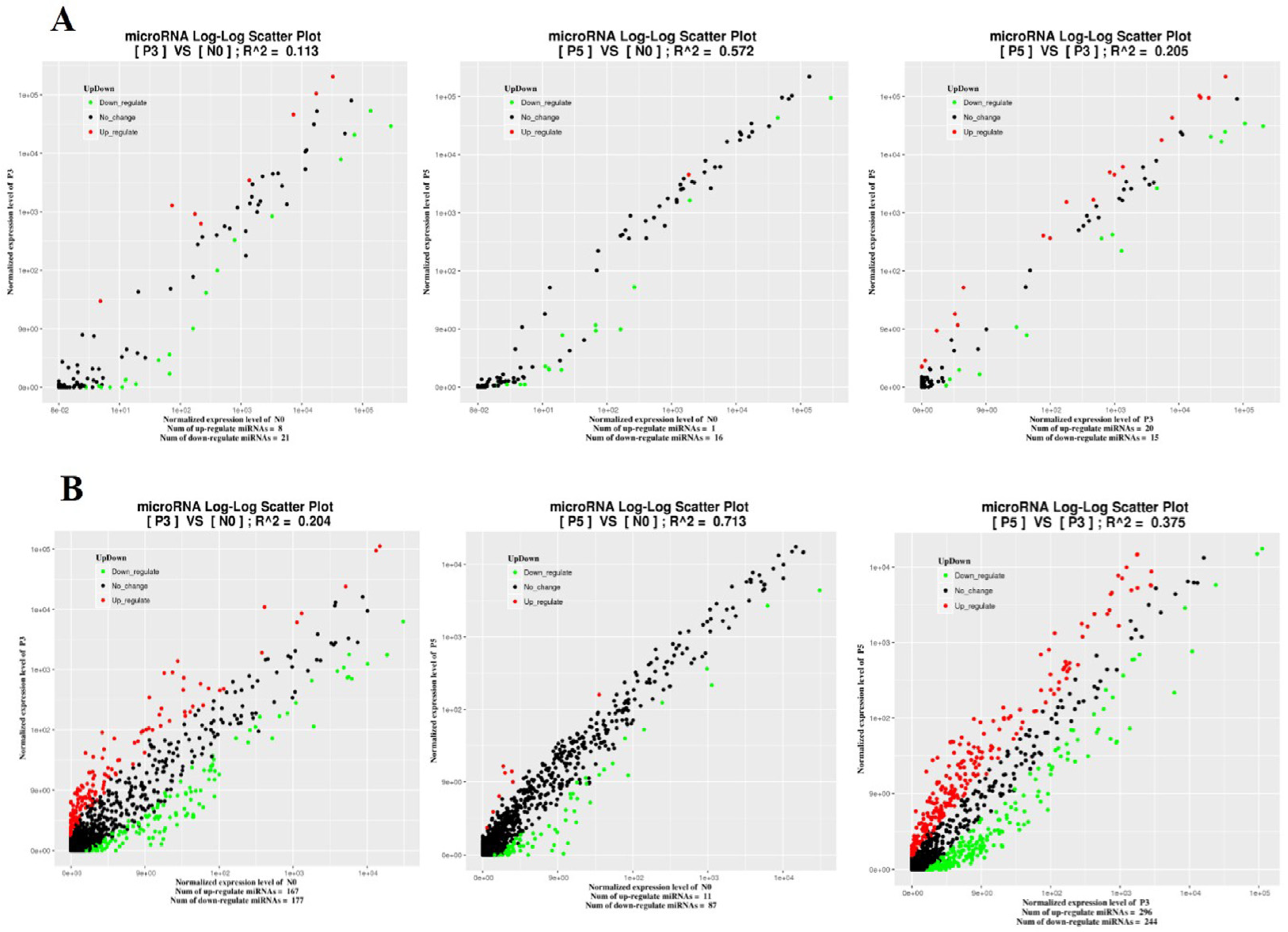
3.3. Expression Analysis of miRNA upon PPR Vaccine Virus Inoculation at Different Time Points
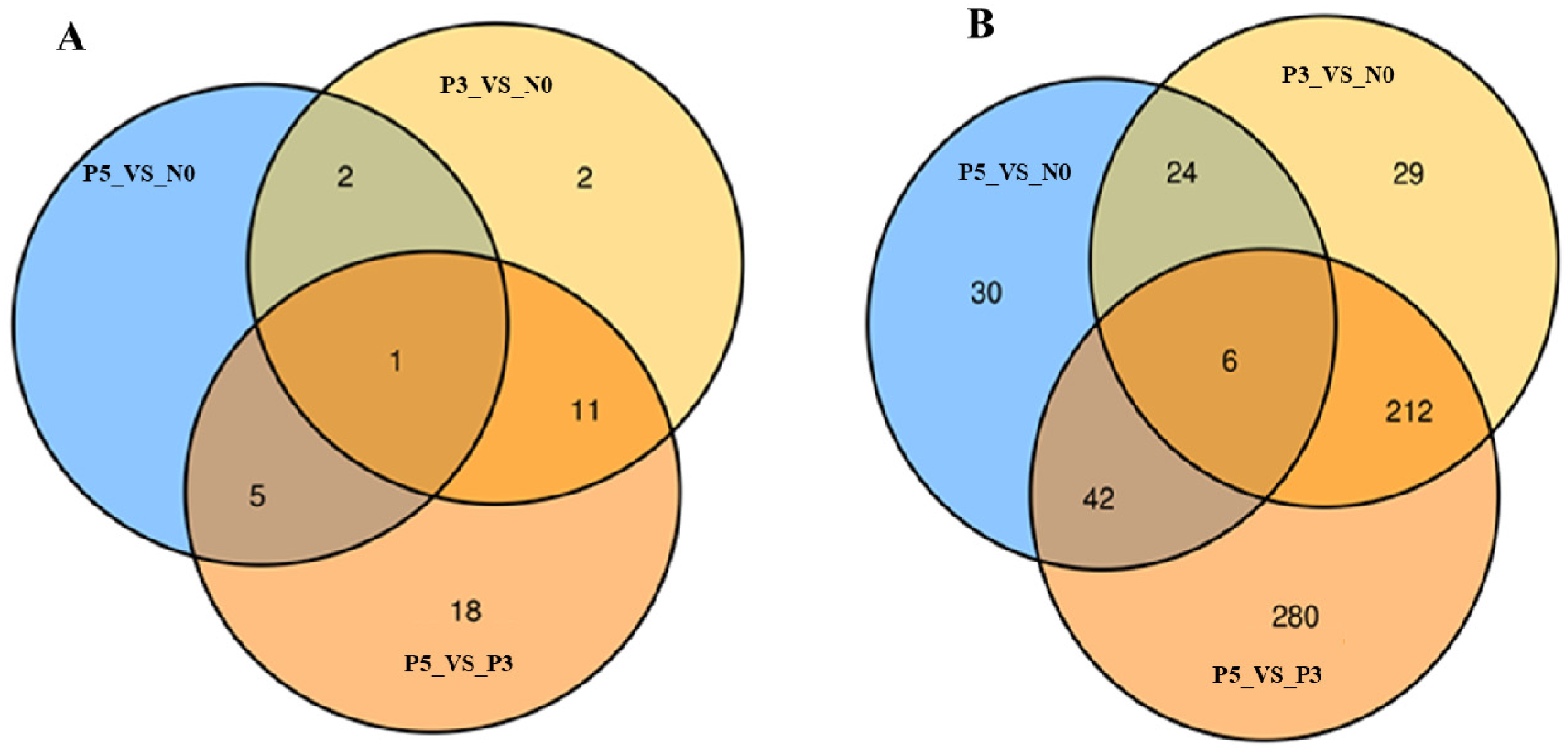
3.4. Identification of Time Point Specific Different Expression miRNAs
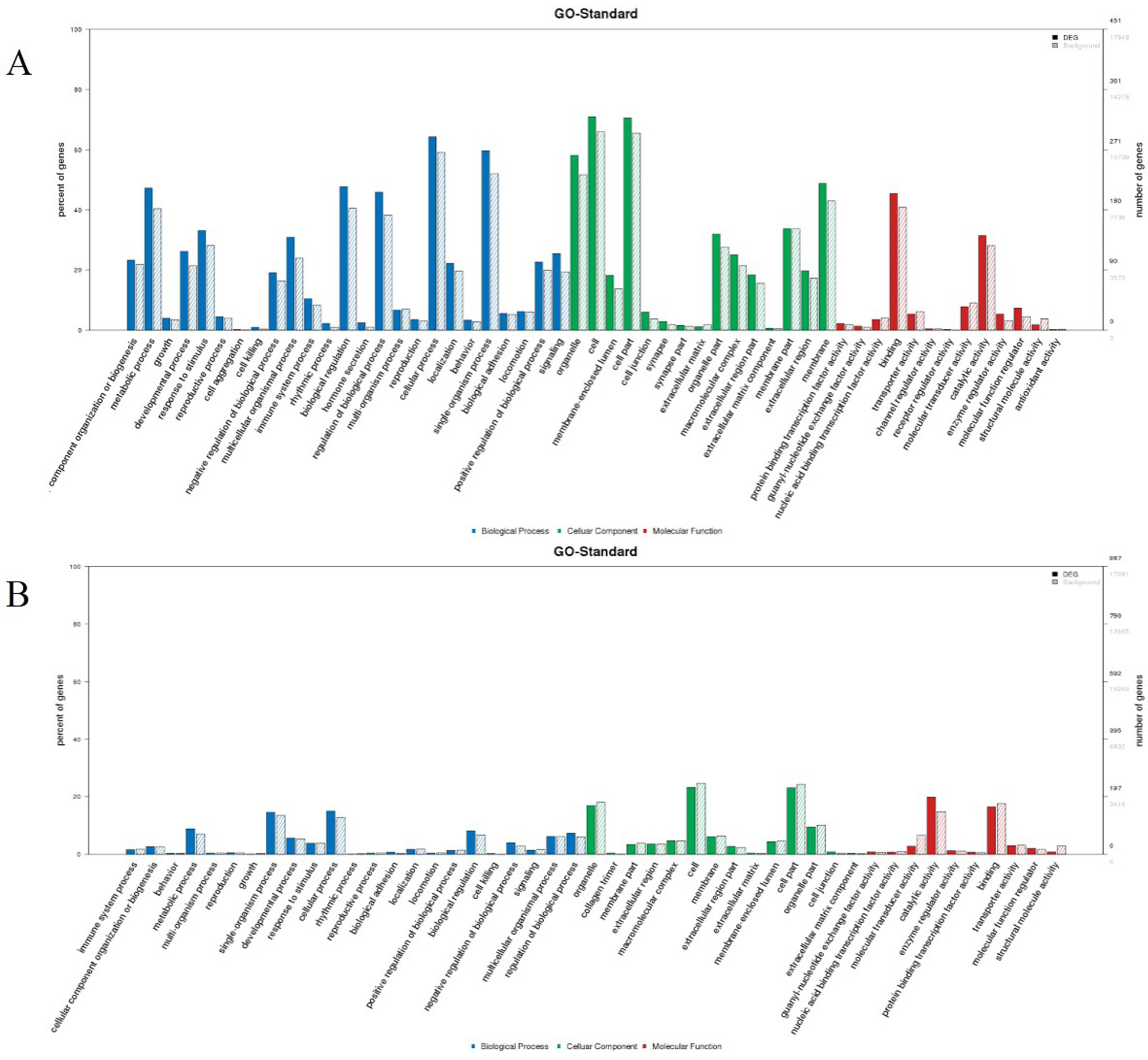
3.5. Target Gene Prediction, and GO and KEGG Analyses
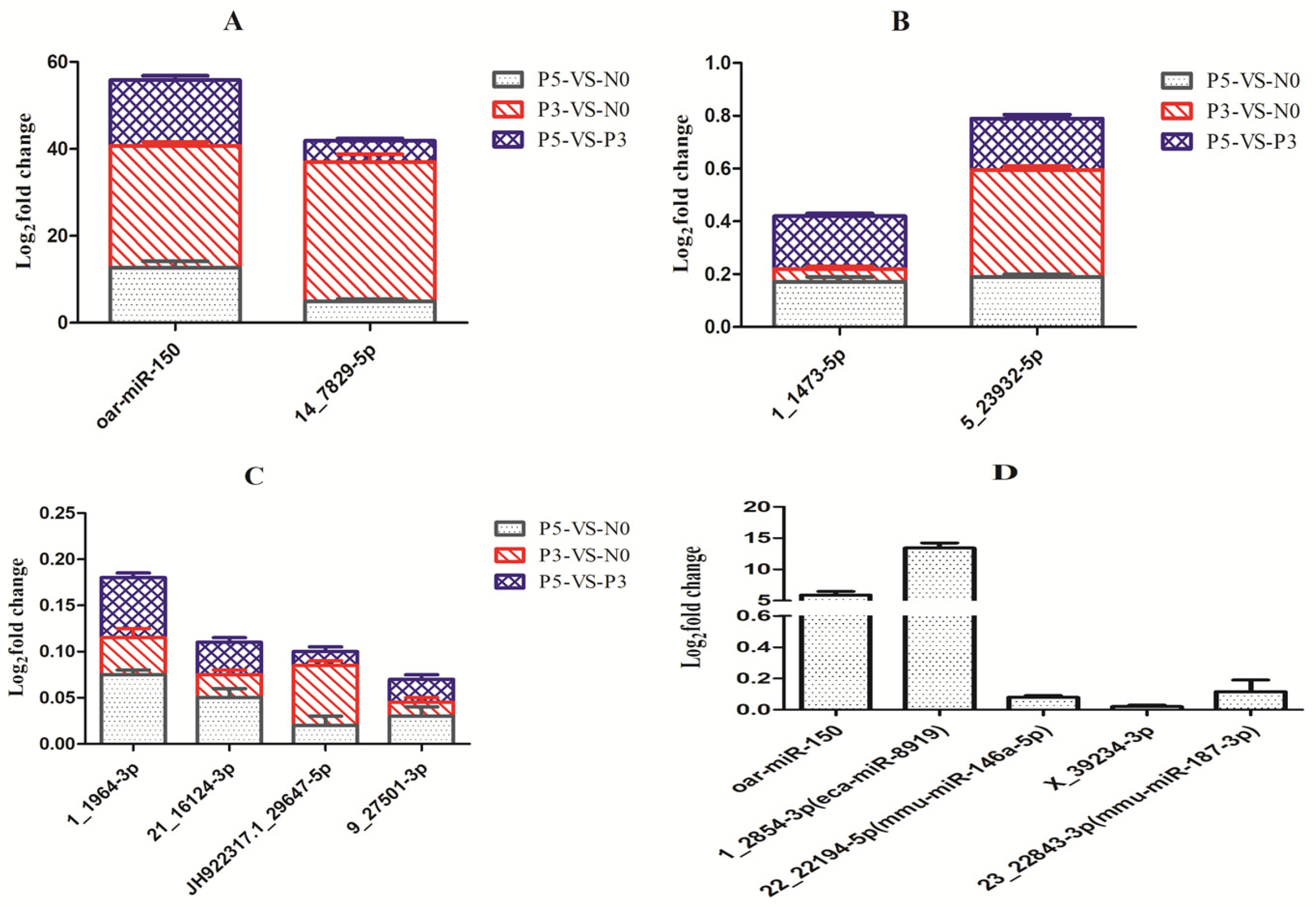
3.6. Validation of Known and Novel miRNAs by Stem-Loop RT-qPCR
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Axtell, M.J.; Westholm, J.O.; Lai, E.C. Vive la difference: Biogenesis and evolution of microRNAs in plants and animals. Genome Biol. 2011, 12, 221. [Google Scholar] [CrossRef]
- Bartel, D.P. MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell 2004, 116, 281–297. [Google Scholar] [CrossRef]
- Bartel, D.P. MicroRNAs: Target recognition and regulatory functions. Cell 2009, 136, 215–233. [Google Scholar] [CrossRef]
- Kloosterman, W.P.; Plasterk, R.H. The diverse functions of microRNAs in animal development and disease. Dev. Cell 2006, 11, 441–450. [Google Scholar] [CrossRef]
- Xing, S.; Du, J.; Gao, S.; Tian, Z.; Zheng, Y.; Liu, G.; Luo, J.; Yin, H. Analysis of the miRNA expression profile in an Aedes albopictus cell line in response to bluetongue virus infection. Infect. Genet. Evol. 2016, 39, 74–84. [Google Scholar] [CrossRef]
- Skovgaard, K.; Cirera, S.; Vasby, D.; Podolska, A.; Breum, S.O.; Durrwald, R.; Schlegel, M.; Heegaard, P.M. Expression of innate immune genes, proteins and microRNAs in lung tissue of pigs infected experimentally with influenza virus (H1N2). Innate. Immun. 2013, 19, 531–544. [Google Scholar] [CrossRef]
- Zhang, Y.; Jing, J.; Li, X.; Wang, J.; Feng, X.; Cao, R.; Chen, P. Integration analysis of miRNA and mRNA expression profiles in swine testis cells infected with Japanese encephalitis virus. Infect. Genet. Evol. 2015, 32, 342–347. [Google Scholar] [CrossRef]
- Xu, X.; Ye, Q. Regulation of viral oncogenesis by microRNAs. Mol. Cell. Oncol. 2014, 1, e29910. [Google Scholar] [CrossRef]
- Shrinet, J.; Jain, S.; Jain, J.; Bhatnagar, R.K.; Sunil, S. Next generation sequencing reveals regulation of distinct Aedes microRNAs during chikungunya virus development. PLoS Negl. Trop. Dis. 2014, 8, e2616. [Google Scholar] [CrossRef]
- Ghosh, Z.; Mallick, B.; Chakrabarti, J. Cellular versus viral microRNAs in host-virus interaction. Nucleic Acids. Res. 2009, 37, 1035–1048. [Google Scholar] [CrossRef]
- Chang, J.; Guo, J.T.; Jiang, D.; Guo, H.; Taylor, J.M.; Block, T.M. Liver-specific microRNA miR-122 enhances the replication of hepatitis C virus in nonhepatic cells. J. Virol. 2008, 82, 8215–8223. [Google Scholar] [CrossRef]
- Chang, S.T.; Thomas, M.J.; Sova, P.; Green, R.R.; Palermo, R.E.; Katze, M.G. Next-generation sequencing of small RNAs from HIV-infected cells identifies phased microrna expression patterns and candidate novel microRNAs differentially expressed upon infection. mBio 2013, 4, e00549-12. [Google Scholar] [CrossRef]
- Wang, Y.; Brahmakshatriya, V.; Zhu, H.; Lupiani, B.; Reddy, S.M.; Yoon, B.J.; Gunaratne, P.H.; Kim, J.H.; Chen, R.; Wang, J.; et al. Identification of differentially expressed miRNAs in chicken lung and trachea with avian influenza virus infection by a deep sequencing approach. BMC Genom. 2009, 10, 512. [Google Scholar] [CrossRef]
- Cui, L.; Guo, X.; Qi, Y.; Qi, X.; Ge, Y.; Shi, Z.; Wu, T.; Shan, J.; Shan, Y.; Zhu, Z.; et al. Identification of microRNAs involved in the host response to enterovirus 71 infection by a deep sequencing approach. J. Biomed. Biotechnol. 2010, 2010, 425939. [Google Scholar] [CrossRef]
- Diallo, A.; Minet, C.; Le Goff, C.; Berhe, G.; Albina, E.; Libeau, G.; Barrett, T. The threat of peste des petits ruminants: Progress in vaccine development for disease control. Vaccine 2007, 25, 5591–5597. [Google Scholar] [CrossRef]
- Shaila, M.S.; Shamaki, D.; Forsyth, M.A.; Diallo, A.; Goatley, L.; Kitching, R.P.; Barrett, T. Geographic distribution and epidemiology of peste des petits ruminants virus. Virus Res. 1996, 43, 149–153. [Google Scholar] [CrossRef]
- Lefevre, P.C.; Diallo, A. Peste des petits ruminants. Rev. Sci. Tech. 1990, 9, 935–981. [Google Scholar] [CrossRef]
- Nanda, Y.P.; Chatterjee, A.; Purohit, A.K.; Diallo, A.; Innui, K.; Sharma, R.N.; Libeau, G.; Thevasagayam, J.A.; Bruning, A.; Kitching, R.P.; et al. The isolation of peste des petits ruminants virus from northern India. Vet. Microbiol. 1996, 51, 207–216. [Google Scholar] [CrossRef]
- Batten, C.A.; Banyard, A.C.; King, D.P.; Henstock, M.R.; Edwards, L.; Sanders, A.; Buczkowski, H.; Oura, C.C.; Barrett, T. A real time RT-PCR assay for the specific detection of Peste des petits ruminants virus. J. Virol. Methods 2011, 171, 401–404. [Google Scholar] [CrossRef]
- Bailey, D.; Banyard, A.; Dash, P.; Ozkul, A.; Barrett, T. Full genome sequence of peste des petits ruminants virus, a member of the Morbillivirus genus. Virus Res. 2005, 110, 119–124. [Google Scholar] [CrossRef]
- Abera, T.; Thangavelu, A.; Joy Chandran, N.D.; Raja, A. A SYBR Green I based real time RT-PCR assay for specific detection and quantitation of Peste des petits ruminants virus. BMC Vet. Res. 2014, 10, 22. [Google Scholar] [CrossRef]
- Avota, E.; Gassert, E.; Schneider-Schaulies, S. Measles virus-induced immunosuppression: From effectors to mechanisms. Med. Microbiol. Immunol. 2010, 199, 227–237. [Google Scholar] [CrossRef]
- Schneider-Schaulies, S.; Niewiesk, S.; Schneider-Schaulies, J.; ter Meulen, V. Measles virus induced immunosuppression: Targets and effector mechanisms. Curr. Mol. Med. 2001, 1, 163–181. [Google Scholar] [CrossRef]
- Iwasa, T.; Suga, S.; Qi, L.; Komada, Y. Apoptosis of human peripheral blood mononuclear cells by wild-type measles virus infection is induced by interaction of hemagglutinin protein and cellular receptor, SLAM via caspase-dependent pathway. Microbiol. Immunol. 2010, 54, 405–416. [Google Scholar] [CrossRef]
- Manjunath, S.; Kumar, G.R.; Mishra, B.P.; Mishra, B.; Sahoo, A.P.; Joshi, C.G.; Tiwari, A.K.; Rajak, K.K.; Janga, S.C. Genomic analysis of host - Peste des petits ruminants vaccine viral transcriptome uncovers transcription factors modulating immune regulatory pathways. Vet. Res. 2015, 46, 15. [Google Scholar] [CrossRef]
- Bolt, G.; Berg, K.; Blixenkrone-Moller, M. Measles virus-induced modulation of host-cell gene expression. J. Gen. Virol. 2002, 83, 1157–1165. [Google Scholar] [CrossRef]
- Pandey, A.; Sahu, A.R.; Wani, S.A.; Saxena, S.; Kanchan, S.; Sah, V.; Rajak, K.K.; Khanduri, A.; Sahoo, A.P.; Tiwari, A.K.; et al. Modulation of Host miRNAs Transcriptome in Lung and Spleen of Peste des Petits Ruminants Virus Infected Sheep and Goats. Front. Microbiol. 2017, 8, 1146. [Google Scholar] [CrossRef]
- Qi, X.; Wang, T.; Xue, Q.; Li, Z.; Yang, B.; Wang, J. MicroRNA expression profiling of goat peripheral blood mononuclear cells in response to peste des petits ruminants virus infection. Vet. Res. 2018, 49, 62. [Google Scholar] [CrossRef]
- Khanduri, A.; Sahu, A.R.; Wani, S.A.; Khan, R.I.N.; Pandey, A.; Saxena, S.; Malla, W.A.; Mondal, P.; Rajak, K.K.; Muthuchelvan, D.; et al. Dysregulated miRNAome and Proteome of PPRV Infected Goat PBMCs Reveal a Coordinated Immune Response. Front. Immunol. 2018, 9, 2631. [Google Scholar] [CrossRef]
- Wang, L.; Feng, Z.; Wang, X.; Zhang, X. DEGseq: An R package for identifying differentially expressed genes from RNA-seq data. Bioinformatics 2010, 26, 136–138. [Google Scholar] [CrossRef]
- Kukurba, K.R.; Montgomery, S.B. RNA Sequencing and Analysis. Cold Spring Harb. Protoc. 2015, 2015, 951–969. [Google Scholar] [CrossRef]
- Schmittgen, T.D.; Lee, E.J.; Jiang, J.; Sarkar, A.; Yang, L.; Elton, T.S.; Chen, C. Real-time PCR quantification of precursor and mature microRNA. Methods 2008, 44, 31–38. [Google Scholar] [CrossRef]
- Liu, Y.; Zhou, Y.; Wu, J.; Zheng, P.; Li, Y.; Zheng, X.; Puthiyakunnon, S.; Tu, Z.; Chen, X.G. The expression profile of Aedes albopictus miRNAs is altered by dengue virus serotype-2 infection. Cell Biosci. 2015, 5, 16. [Google Scholar] [CrossRef]
- Guan, Y.; Liang, G.; Hawken, P.A.; Malecki, I.A.; Cozens, G.; Vercoe, P.E.; Martin, G.B.; Guan le, L. Roles of small RNAs in the effects of nutrition on apoptosis and spermatogenesis in the adult testis. Sci. Rep. 2015, 5, 10372. [Google Scholar] [CrossRef]
- Miao, X.; Luo, Q.; Qin, X.; Guo, Y. Genome-wide analysis of microRNAs identifies the lipid metabolism pathway to be a defining factor in adipose tissue from different sheep. Sci. Rep. 2015, 5, 18470. [Google Scholar] [CrossRef]
- Lagana, A.; Veneziano, D.; Spata, T.; Tang, R.; Zhu, H.; Mohler, P.J.; Kilic, A. Identification of General and Heart-Specific miRNAs in Sheep (Ovis aries). PLoS One. 2015, 10, e0143313. [Google Scholar] [CrossRef]
- Sghaier, S.; Cosseddu, G.M.; Ben Hassen, S.; Hammami, S.; Ammar, H.H.; Petrini, A.; Monaco, F. Peste des petits ruminants virus, Tunisia, 2012-2013. Emerg. Infect. Dis. 2014, 20, 2184–2186. [Google Scholar] [CrossRef]
- Skalsky, R.L.; Cullen, B.R. Viruses, microRNAs, and host interactions. Annu. Rev. Microbiol. 2010, 64, 123–141. [Google Scholar] [CrossRef]
- Du, J.; Gao, S.; Tian, Z.; Xing, S.; Huang, D.; Zhang, G.; Zheng, Y.; Liu, G.; Luo, J.; Chang, H.; et al. MicroRNA expression profiling of primary sheep testicular cells in response to bluetongue virus infection. Infect. Genet. Evol. 2017, 49, 256–267. [Google Scholar] [CrossRef]
- Miao, X.; Luo, Q.; Zhao, H.; Qin, X. Genome-wide analysis of miRNAs in the ovaries of Jining Grey and Laiwu Black goats to explore the regulation of fecundity. Sci. Rep. 2016, 6, 37983. [Google Scholar] [CrossRef]
- Kumari, B.; Jain, P.; Das, S.; Ghosal, S.; Hazra, B.; Trivedi, A.C.; Basu, A.; Chakrabarti, J.; Vrati, S.; Banerjee, A. Dynamic changes in global microRNAome and transcriptome reveal complex miRNA-mRNA regulated host response to Japanese Encephalitis Virus in microglial cells. Sci. Rep. 2016, 6, 20263. [Google Scholar] [CrossRef]
- Jiang, L.; Sun, Q. The expression profile of human peripheral blood mononuclear cell miRNA is altered by antibody-dependent enhancement of infection with dengue virus serotype 3. Virol. J. 2018, 15, 50. [Google Scholar] [CrossRef]
- Rashad, N.M.; El-Shal, A.S.; Shalaby, S.M.; Mohamed, S.Y. Serum miRNA-27a and miRNA-18b as potential predictive biomarkers of hepatitis C virus-associated hepatocellular carcinoma. Mol. Cell. Biochem. 2018, 447, 125–136. [Google Scholar] [CrossRef]
- Keshavarz, M.; Dianat-Moghadam, H.; Sofiani, V.H.; Karimzadeh, M.; Zargar, M.; Moghoofei, M.; Biglari, H.; Ghorbani, S.; Nahand, J.S.; Mirzaei, H. miRNA-based strategy for modulation of influenza A virus infection. Epigenomics 2018, 10, 829–844. [Google Scholar] [CrossRef]
- Slonchak, A.; Hussain, M.; Torres, S.; Asgari, S.; Khromykh, A.A. Expression of mosquito microRNA Aae-miR-2940-5p is down-regulated in response to West Nile virus infection to restrict viral replication. J. Virol. 2014, 88, 8457–8467. [Google Scholar] [CrossRef]
- Norman, K.L.; Sarnow, P. Modulation of hepatitis C virus RNA abundance and the isoprenoid biosynthesis pathway by microRNA miR-122 involves distinct mechanisms. J. Virol. 2010, 84, 666–670. [Google Scholar] [CrossRef]
- Manjunath, S.; Mishra, B.P.; Mishra, B.; Sahoo, A.P.; Tiwari, A.K.; Rajak, K.K.; Muthuchelvan, D.; Saxena, S.; Santra, L.; Sahu, A.R.; et al. Comparative and temporal transcriptome analysis of peste des petits ruminants virus infected goat peripheral blood mononuclear cells. Virus Res. 2017, 229, 28–40. [Google Scholar] [CrossRef]
- Pawar, R.M.; Dhinakar Raj, G.; Balachandran, C. Relationship between the level of signaling lymphocyte activation molecule mRNA and replication of Peste-des-petits-ruminants virus in peripheral blood mononuclear cells of host animals. Acta Virol. 2008, 52, 231–236. [Google Scholar]
- Ambros, V. The functions of animal microRNAs. Nature 2004, 431, 350–355. [Google Scholar] [CrossRef]
- Zhou, R.; Miao, Y.; Li, Y.; Li, X.; Xi, J.; Zhang, Z. MicroRNA-150 promote apoptosis of ovine ovarian granulosa cells by targeting STAR gene. Theriogenology 2019, 127, 66–71. [Google Scholar] [CrossRef]
- Huang, L.; Liu, X. microRNA-370 Promotes Cell Growth by Targeting WNK2 in Breast Cancer. DNA Cell Biol. 2019, 38, 501–509. [Google Scholar] [CrossRef] [PubMed]
- Gilles, M.E.; Slack, F.J. Let-7 microRNA as a potential therapeutic target with implications for immunotherapy. Expert. Opin. Ther. Targets 2018, 22, 929–939. [Google Scholar] [CrossRef]
- Brennan, E.; Wang, B.; McClelland, A.; Mohan, M.; Marai, M.; Beuscart, O.; Derouiche, S.; Gray, S.; Pickering, R.; Tikellis, C.; et al. Protective Effect of let-7 miRNA Family in Regulating Inflammation in Diabetes-Associated Atherosclerosis. Diabetes 2017, 66, 2266–2277. [Google Scholar] [CrossRef]
- Pfeffer, S.R.; Yang, C.H.; Pfeffer, L.M. The Role of miR-21 in Cancer. Drug Dev. Res. 2015, 76, 270–277. [Google Scholar] [CrossRef]
- Chen, Y.; Chen, J.; Wang, H.; Shi, J.; Wu, K.; Liu, S.; Liu, Y.; Wu, J. HCV-induced miR-21 contributes to evasion of host immune system by targeting MyD88 and IRAK1. PLoS Pathog. 2013, 9, e1003248. [Google Scholar] [CrossRef]
- Kanokudom, S.; Vilaivan, T.; Wikan, N.; Thepparit, C.; Smith, D.R.; Assavalapsakul, W. miR-21 promotes dengue virus serotype 2 replication in HepG2 cells. Antiviral Res. 2017, 142, 169–177. [Google Scholar] [CrossRef]
- Parikh, V.N.; Park, J.; Nikolic, I.; Channick, R.; Yu, P.B.; De Marco, T.; Hsue, P.Y.; Chan, S.Y. Brief Report: Coordinated Modulation of Circulating miR-21 in HIV, HIV-Associated Pulmonary Arterial Hypertension, and HIV/Hepatitis C Virus Coinfection. J. Acquir. Immune. Defic. Syndr. 2015, 70, 236–241. [Google Scholar] [CrossRef]
- Yang, G.D.; Huang, T.J.; Peng, L.X.; Yang, C.F.; Liu, R.Y.; Huang, H.B.; Chu, Q.Q.; Yang, H.J.; Huang, J.L.; Zhu, Z.Y.; et al. Epstein-Barr Virus_Encoded LMP1 upregulates microRNA-21 to promote the resistance of nasopharyngeal carcinoma cells to cisplatin-induced Apoptosis by suppressing PDCD4 and Fas-L. PLoS ONE 2013, 8, e78355. [Google Scholar] [CrossRef] [PubMed]
- Anastasiadou, E.; Garg, N.; Bigi, R.; Yadav, S.; Campese, A.F.; Lapenta, C.; Spada, M.; Cuomo, L.; Botta, A.; Belardelli, F.; et al. Epstein-Barr virus infection induces miR-21 in terminally differentiated malignant B cells. Int. J. Cancer 2015, 137, 1491–1497. [Google Scholar] [CrossRef]
- He, S.; Li, L.; Sun, S.; Zeng, Z.; Lu, J.; Xie, L. A Novel Murine Chronic Obstructive Pulmonary Disease Model and the Pathogenic Role of MicroRNA-21. Front. Physiol. 2018, 9, 503. [Google Scholar] [CrossRef]
- Scheel, T.K.; Luna, J.M.; Liniger, M.; Nishiuchi, E.; Rozen-Gagnon, K.; Shlomai, A.; Auray, G.; Gerber, M.; Fak, J.; Keller, I.; et al. A Broad RNA Virus Survey Reveals Both miRNA Dependence and Functional Sequestration. Cell Host Microbe. 2016, 19, 409–423. [Google Scholar] [CrossRef] [PubMed]
- Bai, M.; Sun, L.; Jia, C.; Li, J.; Han, Y.; Liu, H.; Chen, Y.; Jiang, H. Integrated Analysis of miRNA and mRNA Expression Profiles Reveals Functional miRNA-Targets in Development Testes of Small Tail Han Sheep. G3 (Bethesda) 2019, 9, 523–533. [Google Scholar] [CrossRef] [PubMed]
- Mondal, B.; Sreenivasa, B.P.; Dhar, P.; Singh, R.P.; Bandyopadhyay, S.K. Apoptosis induced by peste des petits ruminants virus in goat peripheral blood mononuclear cells. Virus Res. 2001, 73, 113–119. [Google Scholar] [CrossRef]
- Braicu, C.; Gulei, D.; Cojocneanu, R.; Raduly, L.; Jurj, A.; Knutsen, E.; Calin, G.A.; Berindan-Neagoe, I. miR-181a/b therapy in lung cancer: Reality or myth? Mol. Oncol. 2019, 13, 9–25. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.Q.; Gao, B.W.; Guo, H.X.; Ren, Q.L.; Wang, X.W.; Chen, J.F.; Wang, J.; Zhang, Z.J.; Ma, Q.; Xing, B.S. miR-181a promotes porcine granulosa cell apoptosis by targeting TGFBR1 via the activin signaling pathway. Mol. Cell. Endocrinol. 2019, 499, 110603. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Zhang, X.; Zheng, Y.; Li, Y.; Wang, X.; Hu, N.; Zhou, P.; Wang, K. Propofol Protects Rat Cardiomyocytes from Anthracycline-Induced Apoptosis by Regulating MicroRNA-181a In Vitro and In Vivo. Oxid. Med. Cell. Longev. 2018, 2018, 2109216. [Google Scholar] [CrossRef]
- Sun, Y.; Shi, X.; Peng, X.; Li, Y.; Ma, H.; Li, D.; Cao, X. MicroRNA-181a exerts anti-inflammatory effects via inhibition of the ERK pathway in mice with intervertebral disc degeneration. J. Cell. Physiol. 2019. [Google Scholar] [CrossRef]
- Zhou, Y.; Li, G.Y.; Ren, J.P.; Wang, L.; Zhao, J.; Ning, S.B.; Zhang, Y.; Lian, J.Q.; Huang, C.X.; Jia, Z.S.; et al. Protection of CD4+ T cells from hepatitis C virus infection-associated senescence via DeltaNp63-miR-181a-Sirt1 pathway. J. Leukoc. Biol. 2016, 100, 1201–1211. [Google Scholar] [CrossRef]
- Huang, G.L.; Sun, J.; Lu, Y.; Liu, Y.; Cao, H.; Zhang, H.; Calin, G.A. MiR-200 family and cancer: From a meta-analysis view. Mol. Aspects Med. 2019. [Google Scholar] [CrossRef]
- Liu, C.; Hu, W.; Li, L.L.; Wang, Y.X.; Zhou, Q.; Zhang, F.; Song-Yang, Y.Y.; Zhu, W.; Sun, C.C.; Li, D.J. Roles of miR-200 family members in lung cancer: More than tumor suppressors. Future Oncol. 2018, 14, 2875–2886. [Google Scholar] [CrossRef]
- Tian, H.; He, Z. miR-200c targets nuclear factor IA to suppress HBV replication and gene expression via repressing HBV Enhancer I activity. Biomed. Pharmacother. 2018, 99, 774–780. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Lan, P.; Han, Q.; Huang, M.; Zhang, Z.; Xu, G.; Song, J.; Wang, J.; Wei, H.; Zhang, J.; et al. Oncofetal gene SALL4 reactivation by hepatitis B virus counteracts miR-200c in PD-L1-induced T cell exhaustion. Nat. Commun. 2018, 9, 1241. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Kang, R.; Liu, W.; Yang, Y.; Ding, R.; Huang, Q.; Meng, J.; Xiong, L.; Guo, Z. Identification and Analysis of P53-Mediated Competing Endogenous RNA Network in Human Hepatocellular Carcinoma. Int. J. Biol. Sci. 2017, 13, 1213–1221. [Google Scholar] [CrossRef] [PubMed]
- de Queiroz, K.B.; Dos Santos Fontes Pereira, T.; Araujo, M.S.S.; Gomez, R.S.; Coimbra, R.S. Resveratrol Acts Anti-Inflammatory and Neuroprotective in an Infant Rat Model of Pneumococcal Meningitis by Modulating the Hippocampal miRNome. Mol. Neurobiol. 2018, 55, 8869–8884. [Google Scholar] [CrossRef]
- Ge, X.L.; Wang, J.L.; Liu, X.; Zhang, J.; Liu, C.; Guo, L. Inhibition of miR-19a protects neurons against ischemic stroke through modulating glucose metabolism and neuronal apoptosis. Cell Mol. Biol. Lett. 2019, 24, 37. [Google Scholar] [CrossRef]
- Zou, M.; Wang, F.; Gao, R.; Wu, J.; Ou, Y.; Chen, X.; Wang, T.; Zhou, X.; Zhu, W.; Li, P.; et al. Autophagy inhibition of hsa-miR-19a-3p/19b-3p by targeting TGF-beta R II during TGF-beta1-induced fibrogenesis in human cardiac fibroblasts. Sci. Rep. 2016, 6, 24747. [Google Scholar] [CrossRef]
- Bott, A.; Erdem, N.; Lerrer, S.; Hotz-Wagenblatt, A.; Breunig, C.; Abnaof, K.; Worner, A.; Wilhelm, H.; Munstermann, E.; Ben-Baruch, A.; et al. miRNA-1246 induces pro-inflammatory responses in mesenchymal stem/stromal cells by regulating PKA and PP2A. Oncotarget 2017, 8, 43897–43914. [Google Scholar] [CrossRef]
- Samir, M.; Vaas, L.A.; Pessler, F. MicroRNAs in the Host Response to Viral Infections of Veterinary Importance. Front. Vet. Sci. 2016, 3, 86. [Google Scholar] [CrossRef]
- Lu, L.F.; Gasteiger, G.; Yu, I.S.; Chaudhry, A.; Hsin, J.P.; Lu, Y.; Bos, P.D.; Lin, L.L.; Zawislak, C.L.; Cho, S.; et al. A Single miRNA-mRNA Interaction Affects the Immune Response in a Context- and Cell-Type-Specific Manner. Immunity 2015, 43, 52–64. [Google Scholar] [CrossRef]
- Zhang, W.; Liu, K.; Liu, S.; Ji, B.; Liu, Y. MicroRNA744 inhibits migration and invasion of hepatocellular carcinoma cells by targeting SOX12. Oncol. Rep. 2018, 40, 3585–3592. [Google Scholar] [CrossRef]
- Xu, A.J.; Fu, L.N.; Wu, H.X.; Yao, X.L.; Meng, R. MicroRNA744 inhibits tumor cell proliferation and invasion of gastric cancer via targeting brainderived neurotrophic factor. Mol. Med. Rep. 2017, 16, 5055–5061. [Google Scholar] [CrossRef] [PubMed]
- Mao, Y.; Wei, F.; Wei, C. microRNA574 inhibits cell proliferation and invasion in glioblastoma multiforme by directly targeting zinc finger Eboxbinding homeobox 1. Mol. Med. Rep. 2018, 18, 1826–1834. [Google Scholar] [CrossRef] [PubMed]
- Lai, X.; Guo, Y.; Guo, Z.; Liu, R.; Wang, X.; Wang, F. Downregulation of microRNA574 in cancer stem cells causes recurrence of prostate cancer via targeting REL. Oncol. Rep. 2016, 36, 3651–3656. [Google Scholar] [CrossRef] [PubMed]
| miRNA Name | Primer | Sequences |
|---|---|---|
| oar-miR-150 | RT | GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACcactgg |
| AS | TCTCCCAACCCTTGTACCAGT | |
| 14_7829-5p | RT | GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACactaca |
| AS | ATTTCCCTGTCTTCAATCCTGTAGT | |
| 1_1473-5p | RT | GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACagccca |
| AS | CGCAAAGAATTCTCCTTTTGG | |
| 1_1964-3p | RT | GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACccttag |
| AS | TAGTCAGGATGGCCGAGCG | |
| 21_16124-3p | RT | GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACgctcag |
| AS | GATCAGTGATGAAAGTAGCCAAATC | |
| 5_23932-5p | RT | GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACaaccta |
| AS | GTCGCTGAGAACTGAATTCCATAG | |
| JH922317.1_29647-5p | RT | GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACactcag |
| AS | CCGAACAATATCCTGGTGCTG | |
| 9_27501-3p | RT | GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACcctcaa |
| AS | GCTGTCTAGACTGAAGCTCCTTG | |
| 1_2854-3p | RT | GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACgcctta |
| AS | GTCAGGATGGCCGAGCGGT | |
| 22_22194-5p | RT | GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACacagcc |
| AS | CGTGAGAACTGAATTCCATAGGCT | |
| X_39234-3p | RT | GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACtatagc |
| AS | GTAAAACGTGAGGCGCTGCT | |
| 23_22843-3p | RT | GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACccggct |
| AS | TCGTGTCTTGTGTTGCAGCC | |
| SNORA58 | F | CACCTTGCTATGTCTTGGCTTG |
| R | ATTCTGCTGGCTGCATCTGAC |
| #of miRNA Changed after PPR Vaccine virus Inoculation | |||||||
|---|---|---|---|---|---|---|---|
| Up-Regulated | Down-Regulated | ||||||
| 3dpi vs. 0dpi | 5dpi vs. 0dpi | 5dpi vs. 3dpi | 3dpi vs. 0dpi | 5dpi vs. 0dpi | 5dpi vs. 3dpi | ||
| PBMC | Known miRNA | 8 | 1 | 20 | 21 | 16 | 15 |
| Novel miRNA | 167 | 11 | 296 | 177 | 87 | 244 | |
| ST | Known miRNA | 1 | 2 | ||||
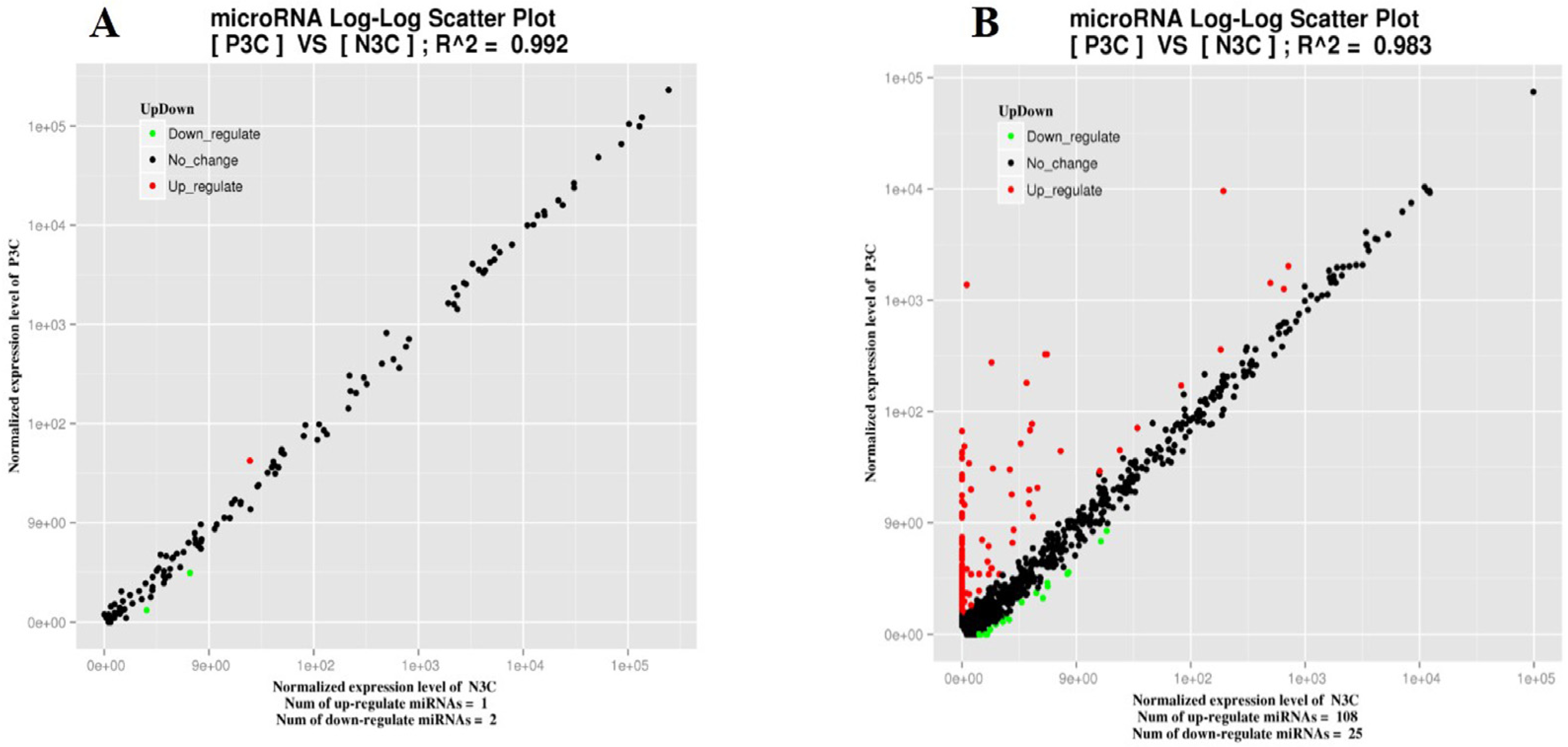
| Novel miRNA | 108 | 25 | |||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, Y.; Qin, X.; Meng, X.; Zhu, X.; Zhang, X.; Li, Y.; Zhang, Z. MicroRNA Expression Profile in Peripheral Blood Lymphocytes of Sheep Vaccinated with Nigeria 75/1 Peste Des Petits Ruminants Virus. Viruses 2019, 11, 1025. https://doi.org/10.3390/v11111025
Yang Y, Qin X, Meng X, Zhu X, Zhang X, Li Y, Zhang Z. MicroRNA Expression Profile in Peripheral Blood Lymphocytes of Sheep Vaccinated with Nigeria 75/1 Peste Des Petits Ruminants Virus. Viruses. 2019; 11(11):1025. https://doi.org/10.3390/v11111025
Chicago/Turabian StyleYang, Yang, Xiaodong Qin, Xuelian Meng, Xueliang Zhu, Xiangle Zhang, Yanmin Li, and Zhidong Zhang. 2019. "MicroRNA Expression Profile in Peripheral Blood Lymphocytes of Sheep Vaccinated with Nigeria 75/1 Peste Des Petits Ruminants Virus" Viruses 11, no. 11: 1025. https://doi.org/10.3390/v11111025
APA StyleYang, Y., Qin, X., Meng, X., Zhu, X., Zhang, X., Li, Y., & Zhang, Z. (2019). MicroRNA Expression Profile in Peripheral Blood Lymphocytes of Sheep Vaccinated with Nigeria 75/1 Peste Des Petits Ruminants Virus. Viruses, 11(11), 1025. https://doi.org/10.3390/v11111025






