Combining a Fusion Inhibitory Peptide Targeting the MERS-CoV S2 Protein HR1 Domain and a Neutralizing Antibody Specific for the S1 Protein Receptor-Binding Domain (RBD) Showed Potent Synergism against Pseudotyped MERS-CoV with or without Mutations in RBD
Abstract
1. Introduction
2. Materials and Methods
2.1. Cells, Peptides, Human mAb m336, and Plasmids
2.2. Production of Pseudoviruses
2.3. Inhibition of Pseudotyped MERS-CoV Infection
2.4. Inhibition of MERS-CoV S Protein-Mediated Cell–Cell Fusion
2.5. Inhibitory Activity of Sera from Mice Treated with m336 Alone, HR2P-M2 Alone, or m336/HR2P-M2 Combined
2.6. Inhibitor Combination Assay
2.7. Statistical Analysis
3. Results
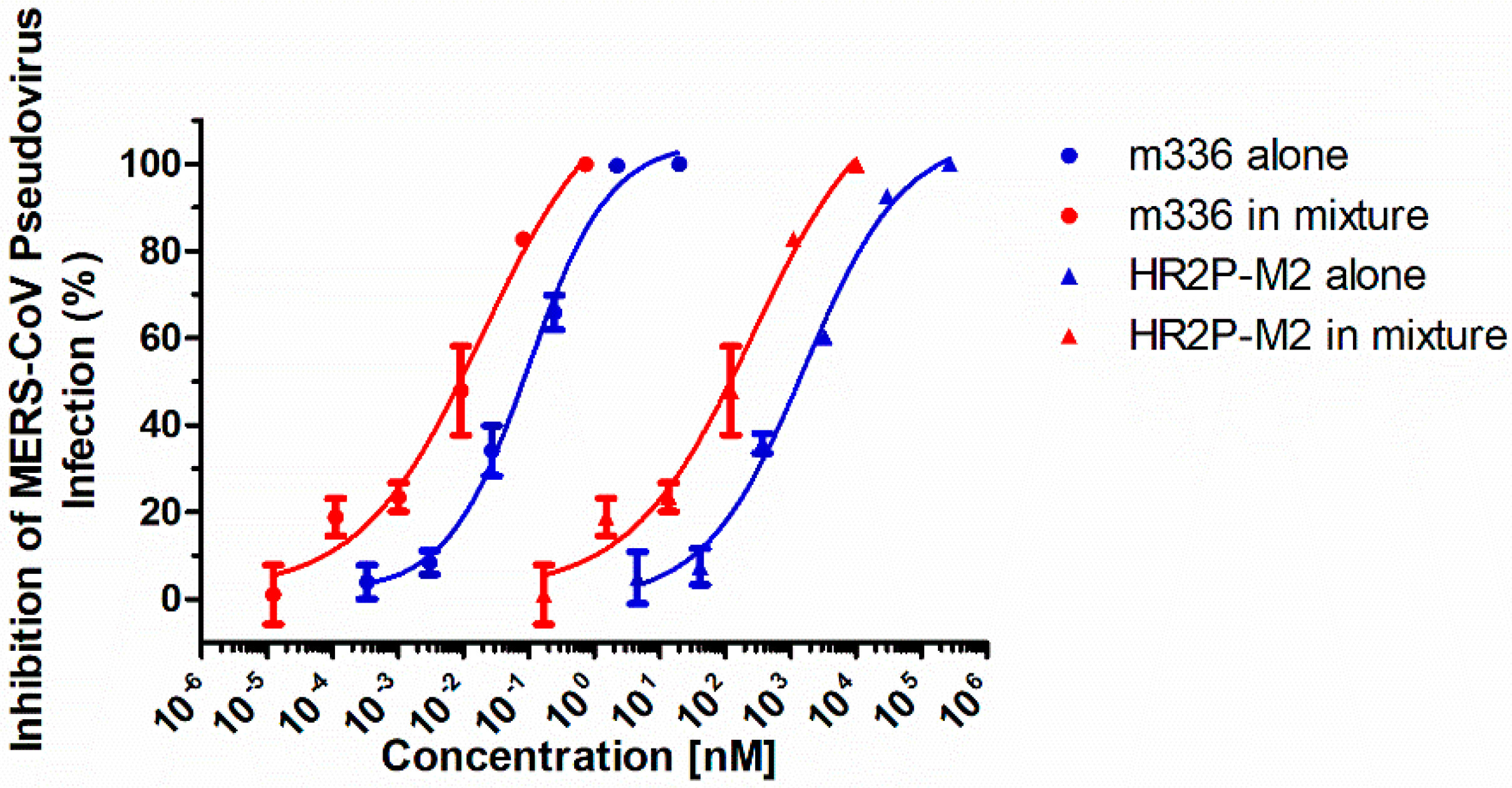
3.1. Combining HR2P-M2 with m336 Exhibited Strong Synergism against MERS-CoV Pseudovirus Infection
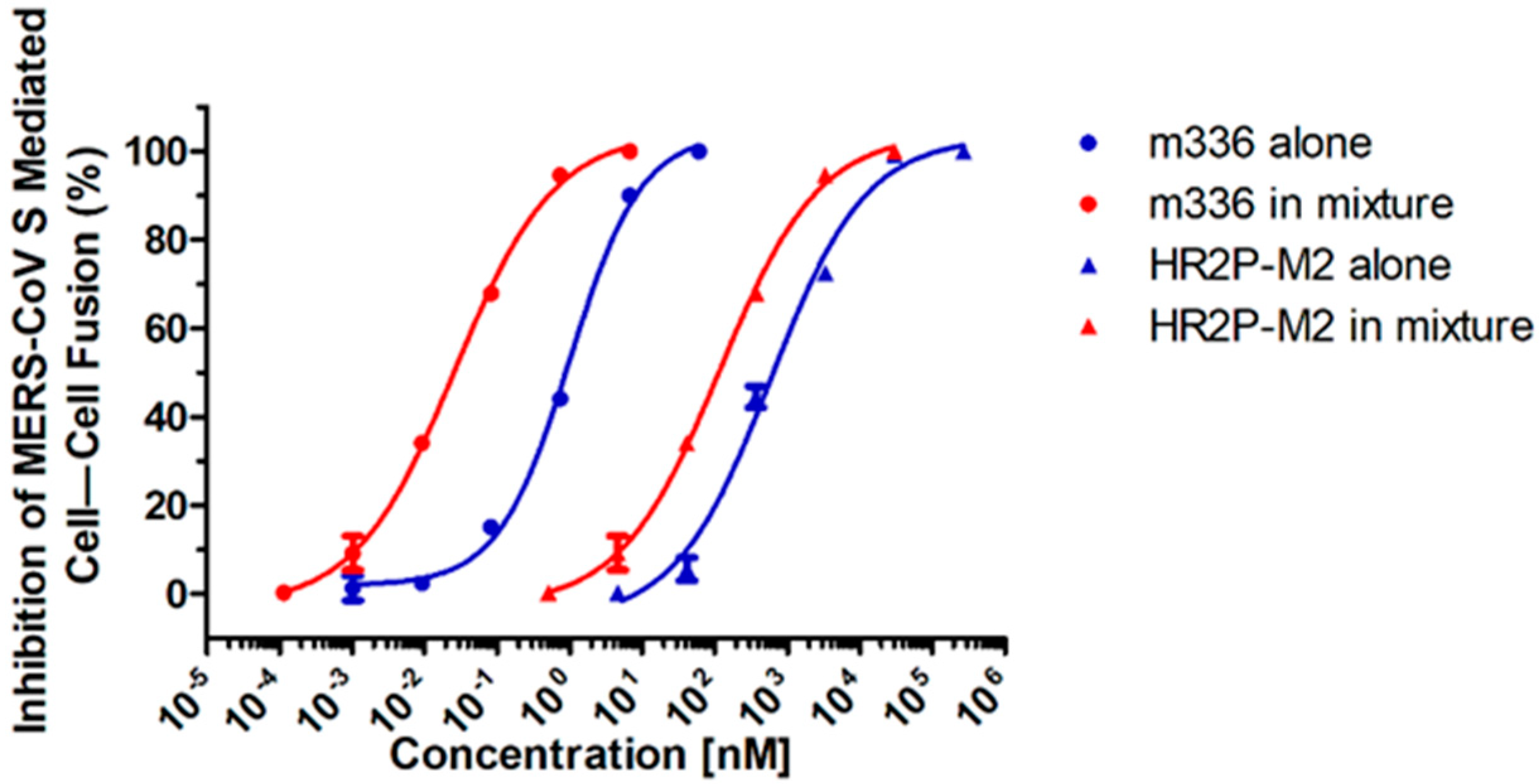
3.2. Combining HR2P-M2 with m336 Displayed Strong Synergism against MERS-CoV S Protein-Mediated Cell–Cell Fusion
3.3. MERS-CoV Pseudoviruses with Mutations in RBD Mutant of MERS-CoV Were Resistant to RBD-Specific mAb m336, While They Were Equally Sensitive to the HR1-Targeting Peptide HR2P-M2
3.4. Combining m336 with HR2P-M2 Exhibited Potent Synergism against MERS-CoV Pseudoviruses with or without Mutations in RBD or Those in the HR1 Domain
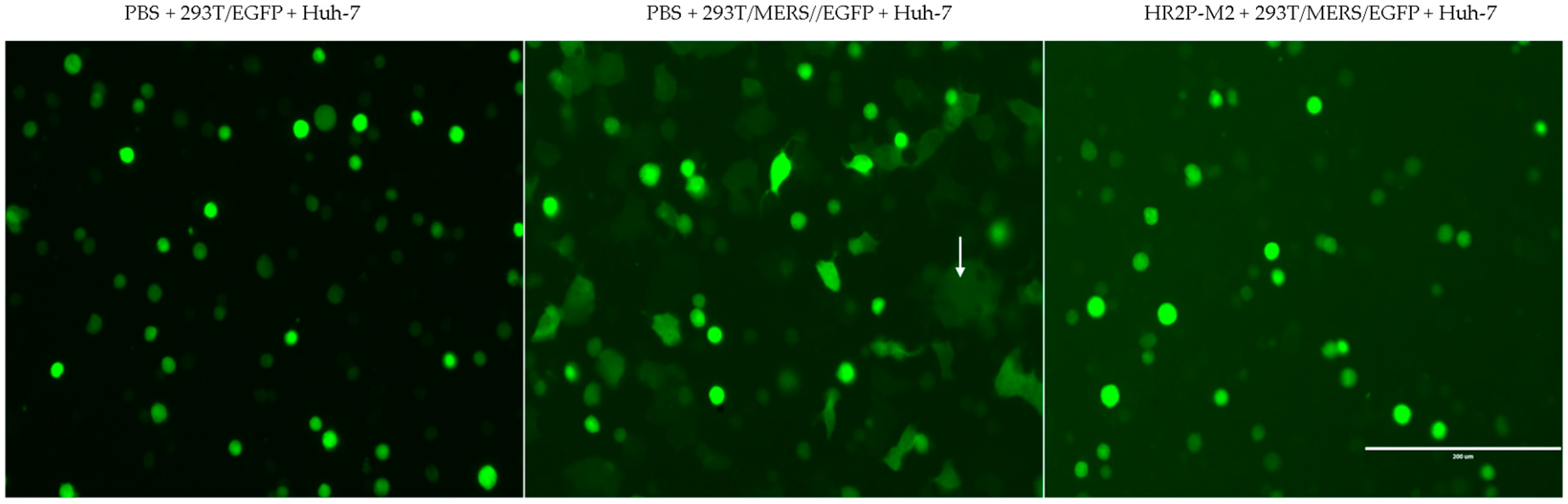
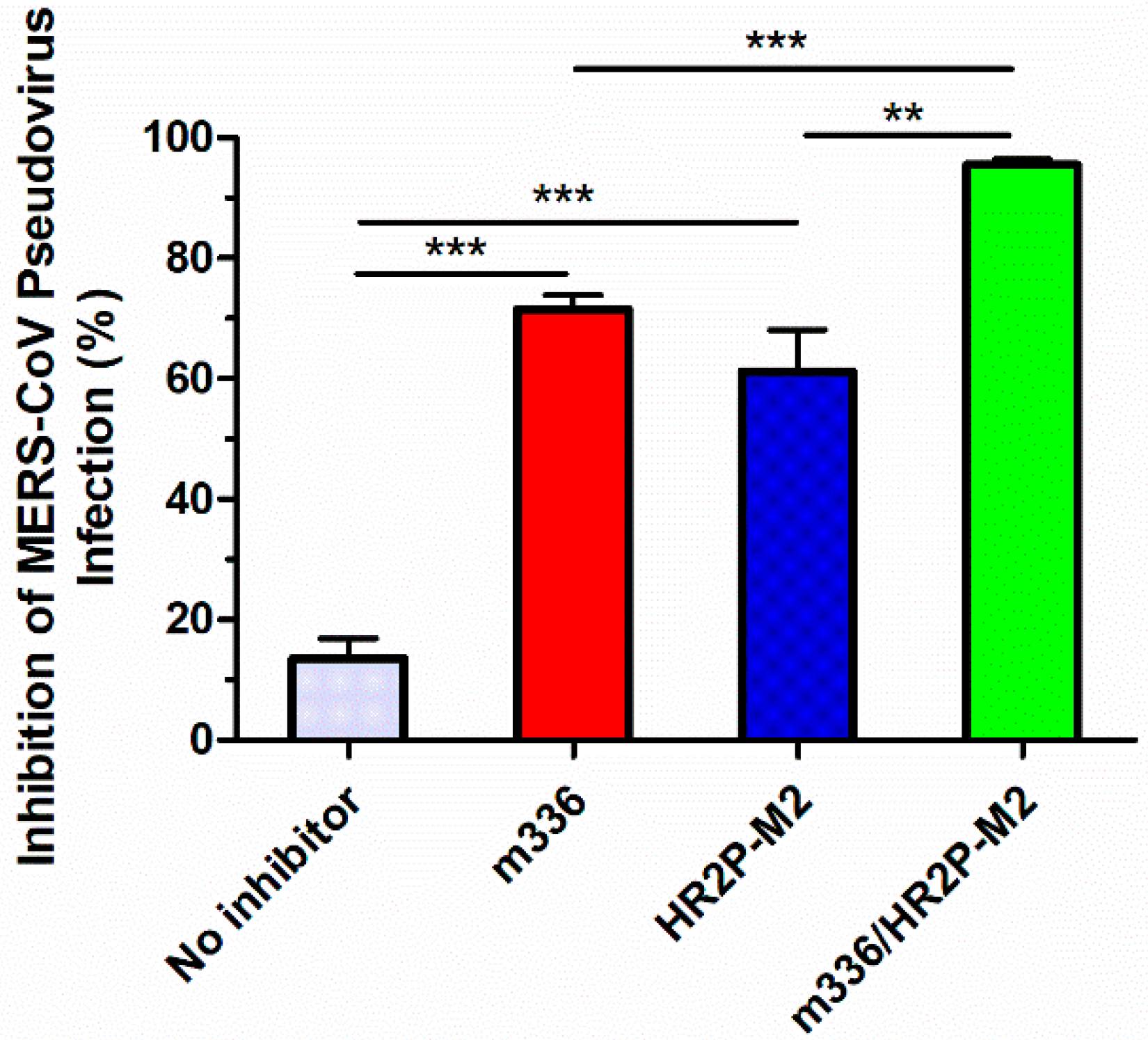
3.5. Sera from Mice Treated with the m336/HR2P-M2 Combination Showed More Efficacy in Inhibiting MERS-CoV Pseudovirus Infection than Either HR2P-M2 or m336 Alone
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zaki, A.M.; van Boheemen, S.; Bestebroer, T.M.; Osterhaus, A.D.; Fouchier, R.A. Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia. N. Engl. J. Med. 2012, 367, 1814–1820. [Google Scholar] [CrossRef] [PubMed]
- de Groot, R.J.; Baker, S.C.; Baric, R.S.; Brown, C.S.; Drosten, C.; Enjuanes, L.; Fouchier, R.A.; Galiano, M.; Gorbalenya, A.E.; Memish, Z.A.; et al. Middle East respiratory syndrome coronavirus (MERS-CoV): Announcement of the Coronavirus Study Group. J. Virol. 2013, 87, 7790–7792. [Google Scholar] [CrossRef] [PubMed]
- Belouzard, S.; Millet, J.K.; Licitra, B.N.; Whittaker, G.R. Mechanisms of coronavirus cell entry mediated by the viral spike protein. Viruses 2012, 4, 1011–1033. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Shi, X.; Jiang, L.; Zhang, S.; Wang, D.; Tong, P.; Guo, D.; Fu, L.; Cui, Y.; Liu, X.; et al. Structure of MERS-CoV spike receptor-binding domain complexed with human receptor DPP4. Cell Res. 2013, 23, 986–993. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Liu, Q.; Zhu, Y.; Chan, K.H.; Qin, L.; Li, Y.; Wang, Q.; Chan, J.F.; Du, L.; Yu, F.; et al. Structure-based discovery of Middle East respiratory syndrome coronavirus fusion inhibitor. Nat. Commun. 2014, 5, 3067. [Google Scholar] [CrossRef] [PubMed]
- Du, L.; Zhao, G.; Kou, Z.; Ma, C.; Sun, S.; Poon, V.K.; Lu, L.; Wang, L.; Debnath, A.K.; Zheng, B.J.; et al. Identification of a receptor-binding domain in the S protein of the novel human coronavirus Middle East respiratory syndrome coronavirus as an essential target for vaccine development. J. Virol. 2013, 87, 9939–9942. [Google Scholar] [CrossRef]
- Raj, V.S.; Mou, H.; Smits, S.L.; Dekkers, D.H.; Muller, M.A.; Dijkman, R.; Muth, D.; Demmers, J.A.; Zaki, A.; Fouchier, R.A.; et al. Dipeptidyl peptidase 4 is a functional receptor for the emerging human coronavirus-EMC. Nature 2013, 495, 251–254. [Google Scholar] [CrossRef]
- Gao, J.; Lu, G.; Qi, J.; Li, Y.; Wu, Y.; Deng, Y.; Geng, H.; Li, H.; Wang, Q.; Xiao, H.; et al. Structure of the fusion core and inhibition of fusion by a heptad repeat peptide derived from the S protein of Middle East respiratory syndrome coronavirus. J. Virol. 2013, 87, 13134–13140. [Google Scholar] [CrossRef]
- Tao, X.; Garron, T.; Agrawal, A.S.; Algaissi, A.; Peng, B.H.; Wakamiya, M.; Chan, T.S.; Lu, L.; Du, L.; Jiang, S.; et al. Characterization and Demonstration of the Value of a Lethal Mouse Model of Middle East Respiratory Syndrome Coronavirus Infection and Disease. J. Virol. 2016, 90, 57–67. [Google Scholar] [CrossRef]
- Ying, T.; Du, L.; Ju, T.W.; Prabakaran, P.; Lau, C.C.; Lu, L.; Liu, Q.; Wang, L.; Feng, Y.; Wang, Y.; et al. Exceptionally potent neutralization of Middle East respiratory syndrome coronavirus by human monoclonal antibodies. J. Virol. 2014, 88, 7796–7805. [Google Scholar] [CrossRef]
- Houser, K.V.; Gretebeck, L.; Ying, T.; Wang, Y.; Vogel, L.; Lamirande, E.W.; Bock, K.W.; Moore, I.N.; Dimitrov, D.S.; Subbarao, K. Prophylaxis With a Middle East Respiratory Syndrome Coronavirus (MERS-CoV)-Specific Human Monoclonal Antibody Protects Rabbits From MERS-CoV Infection. J. Infect. Dis. 2016, 213, 1557–1561. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, A.S.; Ying, T.; Tao, X.; Garron, T.; Algaissi, A.; Wang, Y.; Wang, L.; Peng, B.H.; Jiang, S.; Dimitrov, D.S.; et al. Passive Transfer of A Germline-like Neutralizing Human Monoclonal Antibody Protects Transgenic Mice Against Lethal Middle East Respiratory Syndrome Coronavirus Infection. Sci. Rep. 2016, 6, 31629. [Google Scholar] [CrossRef] [PubMed]
- van Doremalen, N.; Falzarano, D.; Ying, T.; de Wit, E.; Bushmaker, T.; Feldmann, F.; Okumura, A.; Wang, Y.; Scott, D.P.; Hanley, P.W.; et al. Efficacy of antibody-based therapies against Middle East respiratory syndrome coronavirus (MERS-CoV) in common marmosets. Antiviral Res. 2017, 143, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Ying, T.; Prabakaran, P.; Du, L.; Shi, W.; Feng, Y.; Wang, Y.; Wang, L.; Li, W.; Jiang, S.; Dimitrov, D.S.; et al. Junctional and allele-specific residues are critical for MERS-CoV neutralization by an exceptionally potent germline-like antibody. Nat. Commun. 2015, 6, 8223. [Google Scholar] [CrossRef] [PubMed]
- Du, L.; Zhao, G.; Yang, Y.; Qiu, H.; Wang, L.; Kou, Z.; Tao, X.; Yu, H.; Sun, S.; Tseng, C.T.; et al. A conformation-dependent neutralizing monoclonal antibody specifically targeting receptor-binding domain in Middle East respiratory syndrome coronavirus spike protein. J. Virol. 2014, 88, 7045–7053. [Google Scholar] [CrossRef] [PubMed]
- Tai, W.; Wang, Y.; Fett, C.A.; Zhao, G.; Li, F.; Perlman, S.; Jiang, S.; Zhou, Y.; Du, L. Recombinant Receptor-Binding Domains of Multiple Middle East Respiratory Syndrome Coronaviruses (MERS-CoVs) Induce Cross-Neutralizing Antibodies against Divergent Human and Camel MERS-CoVs and Antibody Escape Mutants. J. Virol. 2017, 91. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Cheon, S.; Min, C.K.; Sohn, K.M.; Kang, Y.J.; Cha, Y.J.; Kang, J.I.; Han, S.K.; Ha, N.Y.; Kim, G.; et al. Spread of Mutant Middle East Respiratory Syndrome Coronavirus with Reduced Affinity to Human CD26 during the South Korean Outbreak. mBio 2016, 7, e00019. [Google Scholar] [CrossRef]
- Kim, D.W.; Kim, Y.J.; Park, S.H.; Yun, M.R.; Yang, J.S.; Kang, H.J.; Han, Y.W.; Lee, H.S.; Kim, H.M.; Kim, H.; et al. Variations in Spike Glycoprotein Gene of MERS-CoV, South Korea, 2015. Emerg. Infect. Dis. 2016, 22, 100–104. [Google Scholar] [CrossRef]
- Zhao, G.; Du, L.; Ma, C.; Li, Y.; Li, L.; Poon, V.K.; Wang, L.; Yu, F.; Zheng, B.J.; Jiang, S.; et al. A safe and convenient pseudovirus-based inhibition assay to detect neutralizing antibodies and screen for viral entry inhibitors against the novel human coronavirus MERS-CoV. Virol. J. 2013, 10, 266. [Google Scholar] [CrossRef]
- Gunaratne, G.S.; Yang, Y.; Li, F.; Walseth, T.F.; Marchant, J.S. NAADP-dependent Ca(2+) signaling regulates Middle East respiratory syndrome-coronavirus pseudovirus translocation through the endolysosomal system. Cell Calcium 2018, 75, 30–41. [Google Scholar] [CrossRef]
- Wang, L.; Shi, W.; Chappell, J.D.; Joyce, M.G.; Zhang, Y.; Kanekiyo, M.; Becker, M.M.; van Doremalen, N.; Fischer, R.; Wang, N.; et al. Importance of neutralizing monoclonal antibodies targeting multiple antigenic sites on MERS-CoV Spike to avoid neutralization escape. J. Virol. 2018. [Google Scholar] [CrossRef]
- Chou, T.C. Theoretical basis, experimental design, and computerized simulation of synergism and antagonism in drug combination studies. Pharmacol. Rev. 2006, 58, 621–681. [Google Scholar] [CrossRef] [PubMed]
- Qi, Q.; Wang, Q.; Chen, W.; Yu, F.; Du, L.; Dimitrov, D.S.; Lu, L.; Jiang, S. Anti-HIV antibody and drug combinations exhibit synergistic activity against drug-resistant HIV-1 strains. J. Infect. 2017, 75, 68–71. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Wang, Q.; Yu, F.; Lu, L.; Jiang, S. Synergistic effect resulting from combinations of a bifunctional HIV-1 antagonist with antiretroviral drugs. J. Acquir. Immune Defic. Syndr. 2014, 67, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Cotten, M.; Watson, S.J.; Zumla, A.I.; Makhdoom, H.Q.; Palser, A.L.; Ong, S.H.; Al Rabeeah, A.A.; Alhakeem, R.F.; Assiri, A.; Al-Tawfiq, J.A.; et al. Spread, circulation, and evolution of the Middle East respiratory syndrome coronavirus. mBio 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Channappanavar, R.; Lu, L.; Xia, S.; Du, L.; Meyerholz, D.K.; Perlman, S.; Jiang, S. Protective Effect of Intranasal Regimens Containing Peptidic Middle East Respiratory Syndrome Coronavirus Fusion Inhibitor Against MERS-CoV Infection. J. Infect. Dis. 2015, 212, 1894–1903. [Google Scholar] [CrossRef] [PubMed]
- Zumla, A.; Hui, D.S.; Perlman, S. Middle East respiratory syndrome. Lancet 2015, 386, 995–1007. [Google Scholar] [CrossRef]
- van Boheemen, S.; de Graaf, M.; Lauber, C.; Bestebroer, T.M.; Raj, V.S.; Zaki, A.M.; Osterhaus, A.D.; Haagmans, B.L.; Gorbalenya, A.E.; Snijder, E.J.; et al. Genomic characterization of a newly discovered coronavirus associated with acute respiratory distress syndrome in humans. mBio 2012, 3. [Google Scholar] [CrossRef]
- Assiri, A.; McGeer, A.; Perl, T.M.; Price, C.S.; Al Rabeeah, A.A.; Cummings, D.A.; Alabdullatif, Z.N.; Assad, M.; Almulhim, A.; Makhdoom, H.; et al. Hospital outbreak of Middle East respiratory syndrome coronavirus. N. Engl. J. Med. 2013, 369, 407–416. [Google Scholar] [CrossRef]
- Lu, L.; Xia, S.; Ying, T.; Jiang, S. Urgent development of effective therapeutic and prophylactic agents to control the emerging threat of Middle East respiratory syndrome (MERS). Emerg. Microbes Infect. 2015, 4, e37. [Google Scholar] [CrossRef]
- Xia, S.; Liu, Q.; Wang, Q.; Sun, Z.; Su, S.; Du, L.; Ying, T.; Lu, L.; Jiang, S. Middle East respiratory syndrome coronavirus (MERS-CoV) entry inhibitors targeting spike protein. Virus Res. 2014, 194, 200–210. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; He, L.; Sun, S.; Qiu, H.; Tai, W.; Chen, J.; Li, J.; Chen, Y.; Guo, Y.; Wang, Y.; et al. A Novel Nanobody Targeting Middle East Respiratory Syndrome Coronavirus (MERS-CoV) Receptor-Binding Domain Has Potent Cross-Neutralizing Activity and Protective Efficacy against MERS-CoV. J. Virol. 2018, 92. [Google Scholar] [CrossRef] [PubMed]
- Kleine-Weber, H.; Elzayat, M.T.; Wang, L.; Graham, B.S.; Muller, M.A.; Drosten, C.; Pohlmann, S.; Hoffmann, M. Mutations in the spike protein of MERS-CoV transmitted in Korea increase resistance towards antibody-mediated neutralization. J. Virol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Ramratnam, B.; Tenner-Racz, K.; He, Y.; Vesanen, M.; Lewin, S.; Talal, A.; Racz, P.; Perelson, A.S.; Korber, B.T.; et al. Quantifying residual HIV-1 replication in patients receiving combination antiretroviral therapy. N. Engl. J. Med. 1999, 340, 1605–1613. [Google Scholar] [CrossRef] [PubMed]
- Hogg, R.S.; Rhone, S.A.; Yip, B.; Sherlock, C.; Conway, B.; Schechter, M.T.; O’Shaughnessy, M.V.; Montaner, J.S. Antiviral effect of double and triple drug combinations amongst HIV-infected adults: Lessons from the implementation of viral load-driven antiretroviral therapy. AIDS 1998, 12, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Richman, D.D.; Margolis, D.M.; Delaney, M.; Greene, W.C.; Hazuda, D.; Pomerantz, R.J. The challenge of finding a cure for HIV infection. Science 2009, 323, 1304–1307. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Lu, H.; Neurath, A.R.; Jiang, S. Combination of candidate microbicides cellulose acetate 1,2-benzenedicarboxylate and UC781 has synergistic and complementary effects against human immunodeficiency virus type 1 infection. Antimicrob. Agents Chemother. 2005, 49, 1830–1836. [Google Scholar] [CrossRef]
- Pan, C.; Cai, L.; Lu, H.; Qi, Z.; Jiang, S. Combinations of the first and next generations of human immunodeficiency virus (HIV) fusion inhibitors exhibit a highly potent synergistic effect against enfuvirtide- sensitive and -resistant HIV type 1 strains. J. Virol. 2009, 83, 7862–7872. [Google Scholar] [CrossRef]
- Pan, C.; Lu, H.; Qi, Z.; Jiang, S. Synergistic efficacy of combination of enfuvirtide and sifuvirtide, the first- and next-generation HIV-fusion inhibitors. AIDS 2009, 23, 639–641. [Google Scholar] [CrossRef]
| % Inhibition | CI | HR2P-M2 | m336 | ||||
|---|---|---|---|---|---|---|---|
| Concentration (μM) | Fold of | Concentration (nM) | Fold of | ||||
| Alone | in Mixture | Enhancement | Alone | in Mixture | Enhancement | ||
| 50 | 0.197 | 0.574 | 0.069 | 8.36 | 0.066 | 0.005 | 12.94 |
| 60 | 0.183 | 0.874 | 0.097 | 9.05 | 0.099 | 0.007 | 13.88 |
| 70 | 0.168 | 1.381 | 0.140 | 9.87 | 0.155 | 0.010 | 14.98 |
| 80 | 0.152 | 2.415 | 0.220 | 10.96 | 0.268 | 0.016 | 16.44 |
| 90 | 0.131 | 5.598 | 0.436 | 12.85 | 0.610 | 0.032 | 18.92 |
| % Inhibition | CI | HR2P-M2 | m336 | ||||
|---|---|---|---|---|---|---|---|
| Concentration (μM) | Fold of | Concentration (nM) | Fold of | ||||
| Alone | in Mixture | Enhancement | Alone | in Mixture | Enhancement | ||
| 50 | 0.271 | 0.511 | 0.110 | 4.64 | 0.440 | 0.024 | 17.96 |
| 60 | 0.274 | 0.713 | 0.156 | 4.57 | 0.625 | 0.035 | 18.04 |
| 70 | 0.278 | 1.025 | 0.228 | 4.50 | 0.918 | 0.051 | 18.12 |
| 80 | 0.282 | 1.596 | 0.362 | 4.41 | 1.466 | 0.080 | 18.22 |
| 90 | 0.288 | 3.106 | 0.726 | 4.28 | 2.965 | 0.161 | 18.38 |
| MERS-CoV Pseudovirus | IC50 (nM) of m336 | RR50 (Fold of Resistance) | p | IC50 (μM) of HR2P-M2 | RR50 (Fold of Resistance) | p |
|---|---|---|---|---|---|---|
| Wild-type | 0.055 ± 0.009 | — | — | 0.553 ± 0.056 | — | — |
| D509G | 0.116 ± 0.020 | 2.11 | <0.01 | 0.619 ± 0.079 | 1.12 | >0.05 |
| D510G | 0.450 ± 0.085 | 8.18 | <0.05 | 0.679 ± 0.144 | 1.23 | >0.05 |
| Q522H | 0.148 ± 0.051 | 2.69 | <0.01 | 0.677 ± 0.071 | 1.22 | >0.05 |
| I529T | 0.215 ± 0.055 | 3.91 | <0.01 | 0.574 ± 0.209 | 1.04 | >0.05 |
| % Inhibition | CI | HR2P-M2 | m336 | ||||
|---|---|---|---|---|---|---|---|
| Concentration (μM) | Fold of | Concentration (μM) | Fold of | ||||
| Alone | in Mixture | Enhancement | Alone | in Mixture | Enhancement | ||
| Wild type | |||||||
| 50 | 0.197 | 0.574 | 0.069 | 8.36 | 0.066 | 0.005 | 12.94 |
| 60 | 0.183 | 0.874 | 0.097 | 9.05 | 0.099 | 0.007 | 13.88 |
| 70 | 0.168 | 1.381 | 0.140 | 9.87 | 0.155 | 0.010 | 14.98 |
| 80 | 0.152 | 2.415 | 0.220 | 10.96 | 0.268 | 0.016 | 16.44 |
| 90 | 0.131 | 5.598 | 0.436 | 12.85 | 0.610 | 0.032 | 18.92 |
| D509G in RBD | |||||||
| 50 | 0.296 | 0.912 | 0.155 | 5.88 | 0.273 | 0.034 | 7.91 |
| 60 | 0.29 | 1.335 | 0.217 | 6.15 | 0.379 | 0.048 | 7.87 |
| 70 | 0.282 | 2.023 | 0.313 | 6.47 | 0.544 | 0.070 | 7.83 |
| 80 | 0.274 | 3.358 | 0.489 | 6.87 | 0.844 | 0.109 | 7.77 |
| 90 | 0.263 | 7.199 | 0.956 | 7.53 | 1.635 | 0.213 | 7.69 |
| D510G in RBD | |||||||
| 50 | 0.137 | 0.962 | 0.088 | 10.96 | 0.429 | 0.020 | 21.96 |
| 60 | 0.145 | 1.523 | 0.153 | 9.96 | 0.763 | 0.034 | 22.43 |
| 70 | 0.155 | 2.512 | 0.280 | 8.97 | 1.429 | 0.062 | 22.97 |
| 80 | 0.169 | 4.625 | 0.586 | 7.90 | 3.075 | 0.130 | 23.63 |
| 90 | 0.194 | 11.59 | 1.777 | 6.52 | 9.74 | 0.395 | 24.67 |
| Q522H in RBD | |||||||
| 50 | 0.135 | 0.799 | 0.045 | 17.59 | 0.129 | 0.010 | 12.76 |
| 60 | 0.143 | 1.243 | 0.067 | 18.67 | 0.166 | 0.015 | 11.21 |
| 70 | 0.153 | 2.012 | 0.101 | 19.93 | 0.219 | 0.022 | 9.74 |
| 80 | 0.168 | 3.619 | 0.168 | 21.57 | 0.306 | 0.037 | 8.21 |
| 90 | 0.199 | 8.757 | 0.36 | 24.30 | 0.508 | 0.08 | 6.34 |
| I529T in RBD | |||||||
| 50 | 0.256 | 0.864 | 0.098 | 8.81 | 0.153 | 0.022 | 7.03 |
| 60 | 0.242 | 1.295 | 0.139 | 9.32 | 0.23 | 0.031 | 7.45 |
| 70 | 0.227 | 2.013 | 0.203 | 9.90 | 0.358 | 0.045 | 7.92 |
| 80 | 0.211 | 3.449 | 0.323 | 10.67 | 0.614 | 0.072 | 8.55 |
| 90 | 0.188 | 7.752 | 0.649 | 11.94 | 1.383 | 0.144 | 9.58 |
| Q1020H in HR1 | |||||||
| 50 | 0.186 | 0.762 | 0.086 | 8.83 | 0.088 | 0.006 | 13.82 |
| 60 | 0.189 | 0.954 | 0.106 | 9.04 | 0.100 | 0.008 | 12.84 |
| 70 | 0.192 | 1.218 | 0.131 | 9.27 | 0.115 | 0.010 | 11.85 |
| 80 | 0.198 | 1.642 | 0.172 | 9.56 | 0.137 | 0.013 | 10.74 |
| 90 | 0.208 | 2.571 | 0.257 | 10.01 | 0.176 | 0.019 | 9.27 |
| Q1020R in HR1 | |||||||
| 50 | 0.293 | 0.69 | 0.119 | 5.79 | 0.073 | 0.009 | 8.30 |
| 60 | 0.28 | 0.905 | 0.143 | 6.32 | 0.087 | 0.011 | 8.19 |
| 70 | 0.268 | 1.216 | 0.175 | 6.95 | 0.104 | 0.013 | 8.06 |
| 80 | 0.254 | 1.743 | 0.223 | 7.80 | 0.131 | 0.017 | 7.92 |
| 90 | 0.238 | 2.997 | 0.323 | 9.29 | 0.184 | 0.024 | 7.70 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, C.; Hua, C.; Xia, S.; Li, W.; Lu, L.; Jiang, S. Combining a Fusion Inhibitory Peptide Targeting the MERS-CoV S2 Protein HR1 Domain and a Neutralizing Antibody Specific for the S1 Protein Receptor-Binding Domain (RBD) Showed Potent Synergism against Pseudotyped MERS-CoV with or without Mutations in RBD. Viruses 2019, 11, 31. https://doi.org/10.3390/v11010031
Wang C, Hua C, Xia S, Li W, Lu L, Jiang S. Combining a Fusion Inhibitory Peptide Targeting the MERS-CoV S2 Protein HR1 Domain and a Neutralizing Antibody Specific for the S1 Protein Receptor-Binding Domain (RBD) Showed Potent Synergism against Pseudotyped MERS-CoV with or without Mutations in RBD. Viruses. 2019; 11(1):31. https://doi.org/10.3390/v11010031
Chicago/Turabian StyleWang, Cong, Chen Hua, Shuai Xia, Weihua Li, Lu Lu, and Shibo Jiang. 2019. "Combining a Fusion Inhibitory Peptide Targeting the MERS-CoV S2 Protein HR1 Domain and a Neutralizing Antibody Specific for the S1 Protein Receptor-Binding Domain (RBD) Showed Potent Synergism against Pseudotyped MERS-CoV with or without Mutations in RBD" Viruses 11, no. 1: 31. https://doi.org/10.3390/v11010031
APA StyleWang, C., Hua, C., Xia, S., Li, W., Lu, L., & Jiang, S. (2019). Combining a Fusion Inhibitory Peptide Targeting the MERS-CoV S2 Protein HR1 Domain and a Neutralizing Antibody Specific for the S1 Protein Receptor-Binding Domain (RBD) Showed Potent Synergism against Pseudotyped MERS-CoV with or without Mutations in RBD. Viruses, 11(1), 31. https://doi.org/10.3390/v11010031






