Detection and Characterization of Enterovirus B73 from a Child in Brazil
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Information
2.2. Sample Processing
2.3. Typing and Genomic Analysis
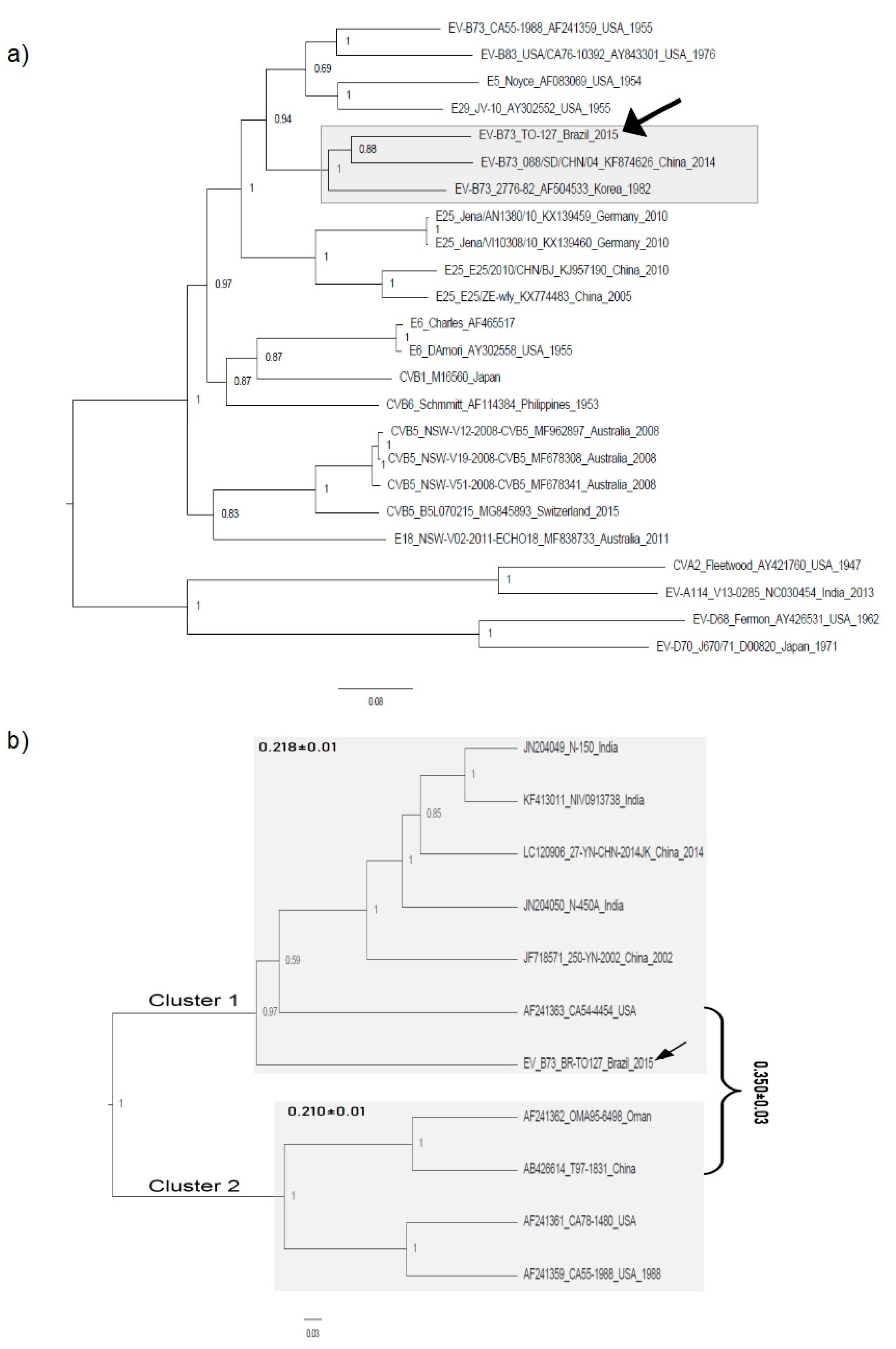
2.4. Phylogenetic Analysis
3. Results
3.1. Typing and Genomic Analysis
3.2. Phylogenetic Analysis
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zell, R.; Delwart, E.; Gorbalenya, A.E.; Hovi, T.; King, A.M.Q.; Knowles, N.J.; Lindberg, A.M.; Pallansch, M.A.; Palmenberg, A.C.; Reuter, G.; et al. ICTV virus taxonomy profile: Picornaviridae. J. Gen. Virol. 2017, 98, 2421–2422. [Google Scholar] [CrossRef] [PubMed]
- Pallansch, M.A.; Oberste, M.S.; Whitton, J.L. Enteroviruses: Polioviruses, Coxsackieviruses, Echoviruses, and Newer Enteroviruses. In Fields Virology; Knipe, D.M., Howley, P.M., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2013; ISBN 9781451105636. [Google Scholar]
- Oberste, M.S.; Schnurr, D.; Maher, K.; Al-Busaidy, S.; Pallansch, M.A. Molecular identification of new picornaviruses and characterization of a proposed enterovirus 73 serotype. J. Gen. Virol. 2001, 82, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, A.C.; Thézé, J.; Komninakis, S.C.V.; Sanz-Duro, R.L.; Felinto, M.R.L.; Moura, L.C.C.; de Oliveira Barroso, I.M.; Santos, L.E.C.; de Lemos Nunes, M.A.; Moura, A.A. Spread of Chikungunya Virus East/Central/South African Genotype in Northeast Brazil. Emerg. Infect. Dis. 2017, 23, 1742. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Naccache, S.N.; Ng, T.; Federman, S.; Li, L.; Chiu, C.Y.; Delwart, E.L. An ensemble strategy that significantly improves de novo assembly of microbial genomes from metagenomic next-generation sequencing data. Nucleic Acids Res. 2015, 43, e46. [Google Scholar] [CrossRef] [PubMed]
- Kroneman, A.; Vennema, H.; Deforche, K.; Avoort, H.V.D.; Penaranda, S.; Oberste, M.S.; Vinjé, J.; Koopmans, M. An automated genotyping tool for enteroviruses and noroviruses. J. Clin. Virol. 2011, 51, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Hall, T.A. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Katoh, K.; Toh, H. Recent developments in the MAFFT multiple sequence alignment program. Brief. Bioinform. 2008, 9, 286–298. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across Computing Platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef] [PubMed]
- Norder, H.; Bjerregaard, L.; Magnius, L.O. Open reading frame sequence of an Asian enterovirus 73 strain reveals that the prototype from California is recombinant. J. Gen. Virol. 2002, 83, 1721–1728. [Google Scholar] [CrossRef] [PubMed]
- Oberste, M.S.; Maher, K.; Kilpatrick, D.R.; Pallansch, M.A. Molecular evolution of the human enteroviruses: Correlation of serotype with VP1 sequence and application to picornavirus classification. J. Virol. 1999, 73, 1941–1948. [Google Scholar] [PubMed]
- Ding, Z.R.; Tang, J.J.; Tian, B.J.; Zhang, J.; Li, L.Q.; Zhao, Z.X.; He, L.F. Status of enterovirus infection and molecular identification among healthy children at the areas bordering Myanmar, in Yunnan province, China. Zhonghua Liu Xing Bing Xue Za Zhi 2010, 31, 185–188. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Garcia, M.D.; Kebe, O.; Fall, A.D.; Ndiaye, K. Identification and molecular characterization of non-polio enteroviruses from children with acute flaccid paralysis in West Africa, 2013–2014. Sci. Rep. 2017, 7, 3808. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Yoshida, H.; Ding, Z.; Tao, Z.; Zhang, J.; Tian, B.; Zhao, Z.; Zhang, L. Molecular epidemiology and recombination of human enteroviruses from AFP surveillance in Yunnan, China from 2006 to 2010. Sci. Rep. 2014, 4, 6058. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Tao, Z.; Wang, H.; Lin, X.; Song, L.; Li, Y.; Zhang, L.; Wang, S.; Cui, N.; Xu, A. Complete genome analysis of human enterovirus B73 isolated from an acute flaccid paralysis patient in Shandong, China. Virus Genes 2014, 49, 38–44. [Google Scholar] [CrossRef] [PubMed]
| Region | Position 1 | TO-127 versus EV-B73 2 | TO-127 versus EV-B 3 | ||
|---|---|---|---|---|---|
| Nucleotide | Amino Acid | Nucleotide | Amino Acid | ||
| Partial 5′ UTR | 1–327 | 76–82 | Non coding | 81–83 | Non coding |
| VP4 | 328–528 | 72–78 | 82–95 | 70–77 | 81–87 |
| VP2 | 529–1311 | 77–81 | 91–97 | 66–72 | 76–86 |
| VP3 | 1312–2022 | 76–82 | 93–98 | 66–71 | 74–81 |
| VP1 | 2023–2889 | 76–82 | 89–96 | 58–65 | 61–70 |
| 2A | 2890–3339 | 72–78 | 87–93 | 78–80 | 92–95 |
| 2B | 3340–3636 | 69–79 | 82–95 | 79–82 | 83–98 |
| 2C | 3637–4623 | 79–82 | 95–97 | 80–82 | 97–98 |
| 3A | 4624–4890 | 73–78 | 94–95 | 77–78 | 93–95 |
| 3B | 4891–4956 | 72–84 | 90–95 | 80–85 | 94–95 |
| 3C | 4957–5505 | 79–82 | 95–97 | 81–82 | 96–97 |
| 3D | 5506–6891 | 76–81 | 90–97 | 79–82 | 96–97 |
| 3’ UTR | 6895–6993 | 88–89 | Non coding | 88–91 | Non coding |
| CDS | 328–6891 | 79–80 | 94–96 | 74–76 | 87–90 |
| Whole Genome | 01–6993 | 79–81 | Non coding | 75–77 | Non coding |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ribeiro, G.d.O.; Luchs, A.; Milagres, F.A.d.P.; Komninakis, S.V.; Gill, D.E.; Lobato, M.C.A.B.S.; Brustulin, R.; Chagas, R.T.d.; Abrão, M.d.F.N.d.S.; Soares, C.V.d.D.A.; et al. Detection and Characterization of Enterovirus B73 from a Child in Brazil. Viruses 2019, 11, 16. https://doi.org/10.3390/v11010016
Ribeiro GdO, Luchs A, Milagres FAdP, Komninakis SV, Gill DE, Lobato MCABS, Brustulin R, Chagas RTd, Abrão MdFNdS, Soares CVdDA, et al. Detection and Characterization of Enterovirus B73 from a Child in Brazil. Viruses. 2019; 11(1):16. https://doi.org/10.3390/v11010016
Chicago/Turabian StyleRibeiro, Geovani de Oliveira, Adriana Luchs, Flávio Augusto de Pádua Milagres, Shirley Vasconcelos Komninakis, Danielle Elise Gill, Márcia Cristina Alves Brito Sayão Lobato, Rafael Brustulin, Rogério Togisaki das Chagas, Maria de Fátima Neves dos Santos Abrão, Cassia Vitória de Deus Alves Soares, and et al. 2019. "Detection and Characterization of Enterovirus B73 from a Child in Brazil" Viruses 11, no. 1: 16. https://doi.org/10.3390/v11010016
APA StyleRibeiro, G. d. O., Luchs, A., Milagres, F. A. d. P., Komninakis, S. V., Gill, D. E., Lobato, M. C. A. B. S., Brustulin, R., Chagas, R. T. d., Abrão, M. d. F. N. d. S., Soares, C. V. d. D. A., Witkin, S. S., Villanova, F., Deng, X., Sabino, E. C., Delwart, E., Da Costa, A. C., & Leal, É. (2019). Detection and Characterization of Enterovirus B73 from a Child in Brazil. Viruses, 11(1), 16. https://doi.org/10.3390/v11010016









