Detection of Usutu, Sindbis, and Batai Viruses in Mosquitoes (Diptera: Culicidae) Collected in Germany, 2011–2016
Abstract
1. Introduction
2. Materials and Methods
2.1. Mosquito Collection and Identification
2.2. DNA/RNA Extraction and Pathogen Screening
2.3. Phylogenetic Analyses
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Medlock, J.M.; Hansford, K.M.; Versteirt, V.; Cull, B.; Kampen, H.; Fontenille, D.; Hendrickx, G.; Zeller, H.; van Bortel, W.; Schaffner, F. An entomological review of invasive mosquitoes in Europe. Bull. Entomol. Res. 2015, 105, 637–663. [Google Scholar] [CrossRef] [PubMed]
- Tomasello, D.; Schlagenhauf, P. Chikungunya and dengue autochthonous cases in Europe, 2007–2012. Travel Med. Infect. Dis. 2013, 11, 274–284. [Google Scholar] [CrossRef] [PubMed]
- Becker, N.; Krüger, A.; Kuhn, C.; Plenge-Bönig, A.; Thomas, S.M.; Schmidt-Chanasit, J.; Tannich, E. Stechmücken als Überträger exotischer Krankheitserreger in Deutschland. Bundesgesundheitsblatt 2014, 57, 531–540. [Google Scholar] [CrossRef] [PubMed]
- Lundström, J.O. Mosquito-borne viruses in western Europe: A review. J. Soc. Vector Ecol. 1999, 24, 1–39. [Google Scholar]
- Hubálek, Z. Mosquito-borne viruses in Europe. Parasitol. Res. 2008, 103, 29–43. [Google Scholar] [CrossRef] [PubMed]
- Colpitts, T.M.; Conway, M.J.; Montgomery, R.R.; Fikrig, E. West Nile virus: Biology, transmission, and human infection. Clin. Microbiol. Rev. 2012, 25, 635–648. [Google Scholar] [CrossRef] [PubMed]
- Staples, E.; Breiman, R.F.; Powers, A.M. Chikungunya fever: An epidemiological review of a re-emerging infectious disease. Clin. Infect. Dis. 2009, 49, 942–948. [Google Scholar] [CrossRef] [PubMed]
- Gubler, D.J. Dengue and dengue hemorrhagic fever. Clin. Microbiol. Rev. 1998, 11, 480–496. [Google Scholar] [PubMed]
- Rezza, G. Dengue and chikungunya: Long-distance spread and outbreaks in naïve areas. Pathog. Glob. Health 2014, 8, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Becker, N.; Jöst, H.; Ziegler, U.; Eiden, M.; Höper, D.; Emmerich, P.; Fichet-Calvet, E.; Ehichioya, D.U.; Czajka, C.; Gabriel, M.; et al. Epizootic emergence of Usutu virus in wild and captive birds in Germany. PLoS ONE 2012, 7, e32604. [Google Scholar] [CrossRef]
- Sieg, M.; Schmidt, V.; Ziegler, U.; Keller, M.; Höper, D.; Heenemann, K.; Ruckner, A.; Nieper, H.; Muluneh, A.; Groschup, M.H.; et al. Outbreak and cocirculation of three different Usutu virus strains in eastern Germany. Vector Borne Zoonotic Dis. 2017, 17, 662–664. [Google Scholar] [CrossRef] [PubMed]
- Cadar, D.; Maier, P.; Müller, S.; Kress, J.; Chudy, M.; Bialonski, A.; Schlaphof, A.; Jansen, S.; Jöst, H.; Tannich, E.; et al. Blood donor screening for West Nile virus (WNV) revealed acute Usutu virus (USUV) infection, Germany, September 2016. Eurosurveillance 2017, 22, 30501. [Google Scholar] [CrossRef] [PubMed]
- Santini, M.; Vilibic-Cavlek, T.; Barsic, B.; Barbic, L.; Savic, V.; Stevanovic, V.; Listes, E.; di Gennaro, A.; Savini, G. First cases of human Usutu virus neuroinvasive infection in Croatia, August-September 2013: Clinical and laboratory features. J. Neurovirol. 2015, 121, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Simonin, Y.; Sillam, O.; Carles, M.J.; Gutierrez, S.; Gil, P.; Constant, O.; Martin, M.F.; Girard, G.; Van de Perre, P.; Salinas, S.; et al. Human Usutu virus infection with atypical neurologic presentation, Montpellier, France, 2016. Emerg. Infect. Dis. 2018, 24, 875–878. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, U. (Friedrich-Loeffler-Institut, D-17493 Greifswald, Germany). personal communication, 2018.
- Eiden, M.; Ziegler, U.; Keller, M.; Müller, K.; Granzow, H.; Jöst, H.; Schmidt-Chanasit, J.; Groschup, M.H. Isolation of Sindbis virus from a hooded crow in Germany. Vector Borne Zoonotic Dis. 2014, 14, 220–222. [Google Scholar] [CrossRef] [PubMed]
- Jöst, H.; Burck-Kammerer, S.; Hutter, G.; Lattwein, E.; Lederer, S.; Litzba, N.; Bock-Hensley, O.; Emmerich, P.; Günther, S.; Becker, N.; et al. Medical importance of Sindbis virus in Southwest Germany. J. Clin. Virol. 2011, 52, 278–279. [Google Scholar] [CrossRef] [PubMed]
- Sluka, F. The clinical picture of the Čalovo virus infection. In Arboviruses of the California Complex and the Bunyamwera Group; Bárdoš, V., Blaskovic, D., Libikova, H., Eds.; SAS: Bratislava, Slovakia, 1969; pp. 337–339. [Google Scholar]
- Adouchief, S.; Smura, T.; Sane, J.; Vapalahti, O.; Kurkela, S. Sindbis virus as a human pathogen-epidemiology, clinical picture and pathogenesis. Rev. Med. Virol. 2016, 26, 221–241. [Google Scholar] [CrossRef] [PubMed]
- Ackermann, R.; Spithaler, W.; Profitlich, W.; Spieckermann, D. Über die Verbreitung von Viren der California-Enzephalitis-Gruppe in der Bundesrepublik Deutschland. DMW-Deutsche Medizinische Wochenschrift 1970, 95, 1507–1513. [Google Scholar] [CrossRef] [PubMed]
- Spieckermann, D.; Ackermann, R. Isolierung von Viren der California-Enzephalitis-Gruppe aus Stechmücken in Nordbayern. Zentralb Bakteriol Parasitenk Infektionskr Hyg Abt I Orig A 1972, 221, 283–295. [Google Scholar]
- Pilaski, J. Contributions to the ecology of Tahyna virus in Central Europe. Bull. Soc. Vector Ecol. 1987, 12, 544–553. [Google Scholar]
- Schüssler, E. Tahyna-Virus: Untersuchungen zum Vorkommen am Oberrhein und Sequenzvergleiche des M-Segments bei zehn Virusisolaten. Ph.D. Thesis, University of Heidelberg, Heidelberg, Germany, 2000. [Google Scholar]
- Sluka, F. Zur Erkenntnis der klinischen Formen des Valtice-Fiebers. Wiener Medizinische Wochenschrift 1969, 45, 765–769. [Google Scholar]
- Bárdos, V. Zur Ökologie und medizinischen Bedeutung des Tahyna-Virus. Münchener Medizinische Wochenschrift 1976, 118, 1617–1620. [Google Scholar] [PubMed]
- Schaffner, F.; Angel, G.; Geoffroy, B.; Hervy, J.P.; Rhaiem, A.; Brunhes, J. The Mosquitoes of Europe (CD-Rom); IRD Éditions& EID Méditerrannée: Montpellier, France, 2001. [Google Scholar]
- Becker, N.; Petrić, D.; Boase, C.; Lane, J.; Zgomba, M.; Dahl, C.; Kaiser, A. Mosquitoes and Their Control; Springer: New York, NY, USA, 2003. [Google Scholar]
- Kronefeld, M.; Werner, D.; Kampen, H. PCR identification and distribution of Anopheles daciae (Diptera: Culicidae) in Germany. Parasitol. Res. 2014, 113, 2079–2086. [Google Scholar] [CrossRef] [PubMed]
- Rudolf, M.; Czajka, C.; Börstler, J.; Melaun, C.; Jöst, H.; von Thien, H.; Badusche, M.; Becker, N.; Schmidt-Chanasit, J.; Krüger, A.; et al. First nationwide surveillance of Culex pipiens complex and Culex torrentium mosquitoes demonstrated the presence of Culex pipiens biotype pipiens/molestus hybrids in Germany. PLoS ONE 2013, 8, e71832. [Google Scholar] [CrossRef] [PubMed]
- Chao, D.Y.; Davis, B.S.; Chang, G.J. Development of multiplex real-time reverse transcriptase PCR assays for detecting eight medically important flaviviruses in mosquitoes. J. Clin. Microbiol. 2007, 45, 584–589. [Google Scholar] [CrossRef] [PubMed]
- Vina-Rodriguez, A.; Sachse, K.; Ziegler, U.; Chaintoutis, S.C.; Keller, M.; Groschup, M.H.; Eiden, M. A novel pan-flavivirus detection and identification assay based on RT-qPCR and microarray. BioMed Res. Int. 2017, 2017, e4248756. [Google Scholar] [CrossRef] [PubMed]
- Jöst, H.; Bialonski, A.; Schmetz, C.; Günther, S.; Becker, N.; Schmidt-Chanasit, J. Isolation and phylogenetic analysis of Batai virus, Germany. Am. J. Trop. Med. Hyg. 2011, 84, 241–243. [Google Scholar] [CrossRef] [PubMed]
- Rezza, G.; Nicoletti, L.; Angelini, R.; Romi, R.; Finarelli, A.C.; Panning, M.; Cordioli, P.; Fortuna, C.; Boros, S.; Magurano, F.; et al. Infection with chikungunya virus in Italy: An outbreak in a temperate region. Lancet 2007, 370, 1840–1846. [Google Scholar] [CrossRef]
- Weidmann, M.; Rudaz, V.; Nunes, M.R.T.; Vasconcelos, P.F.C.; Hufert, F.T. Rapid detection of human pathogenic orthobunyaviruses. J. Clin. Microbiol. 2003, 41, 3299–3305. [Google Scholar] [CrossRef] [PubMed]
- Jöst, H.; Bialonski, A.; Storch, V.; Günther, S.; Becker, N.; Schmidt-Chanasit, J. Isolation and phylogenetic analysis of Sindbis viruses from mosquitoes in Germany. J. Clin. Microbiol. 2010, 48, 1900–1903. [Google Scholar] [CrossRef] [PubMed]
- Bakonyi, T.; Gould, E.A.; Kolodziejek, J.; Weissenböck, H.; Nowotny, N. Complete genome analysis and molecular characterization of Usutu virus that emerged in Austria in 2001: Comparison with the South African strain SAAR-1776 and other flaviviruses. Virology 2004, 328, 301–310. [Google Scholar] [CrossRef] [PubMed]
- Huhtamo, E.; Lambert, A.J.; Costantino, S.; Servino, L.; Krizmancic, L.; Boldorini, R.; Allegrini, S.; Grasso, I.; Korhonen, E.M.; Valpalahti, O.; et al. Isolation and full genomic characterization of Batai virus from mosquitoes, Italy 2009. J. Gen. Virol. 2013, 94, 1242–1248. [Google Scholar] [CrossRef] [PubMed]
- Kearse, M.; Moir, R.; Wilson, A.; Stones-Havas, S.; Cheung, M.; Sturrock, S.; Buxton, S.; Cooper, A.; Markowitz, S.; Duran, C.; et al. Geneious basic: An integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 2012, 28, 1647–1649. [Google Scholar] [CrossRef] [PubMed]
- Ziegler, U.; Jöst, H.; Müller, K.; Fischer, D.; Rinder, M.; Tietze, D.T.; Danner, K.J.; Becker, N.; Skuballa, J.; Hamann, H.P.; et al. Epidemic spread of Usutu virus in Southwest Germany in 2011 to 2013 and monitoring of wild birds for Usutu and West Nile viruses. Vector Borne Zoonotic Dis. 2015, 15, 481–488. [Google Scholar] [CrossRef] [PubMed]
- Cadar, D.; Lühken, R.; van der Jeugd, H.; Garigliany, M.; Ziegler, U.; Keller, M.; Lahoreau, J.; Lachmann, L.; Becker, N.; Kik, M.; et al. Widespread activity of multiple lineages of Usutu virus, western Europe, 2016. Eurosurveillance 2017, 22, 30452. [Google Scholar] [CrossRef] [PubMed]
- Lühken, R.; Jöst, H.; Cadar, D.; Thomas, S.M.; Bosch, S.; Tannich, E.; Becker, N.; Ziegler, U.; Lachmann, L.; Schmidt-Chanasit, J. Distribution of Usutu virus in Germany and its effect on breeding bird populations. Emerg. Infect. Dis. 2017, 23, 1994–2001. [Google Scholar] [CrossRef] [PubMed]
- Liang, G.D.; Li, L.; Zhou, G.L.; Fu, S.H.; Li, Q.P.; Li, F.S.; He, H.H.; Jin, Q.; He, Y.; Chen, B.Q.; et al. Isolation and complete nucleotide sequence of a Chinese Sindbis-like virus. J. Gen. Virol. 2000, 81, 1347–1351. [Google Scholar] [CrossRef] [PubMed]
- Sammels, L.M.; Lindsay, M.D.; Poidinger, M.; Coelen, R.J.; Mackenzie, J.S. Geographic distribution and evolution of Sindbis virus in Australia. J. Gen. Virol. 1999, 80, 739–748. [Google Scholar] [CrossRef] [PubMed]
- Jöst, H.; Bialonski, A.; Maus, D.; Sambri, V.; Eiden, M.; Groschup, M.H.; Günther, S.; Becker, N.; Schmidt-Chanasit, J. Isolation ofUsutuvirus in Germany. Am. J. Trop. Med. Hyg. 2011, 85, 551–553. [Google Scholar] [CrossRef] [PubMed]
- Kampen, H.; Walther, D. Vector potential of mosquito species (Diptera: Culicidae) occurring in central Europe. Parasitol. Res. Monogr. 2018, 10. in press. [Google Scholar]
- Lanciotti, R.S.; Roehrig, J.T.; Deubel, V.; Smith, J.; Parker, M.; Steele, K.; Crise, B.; Volpe, K.E.; Crabtree, M.B.; Scherret, J.H.; et al. Origin of the West Nile virus responsible for an outbreak of encephalitis in the northeastern United States. Science 1999, 286, 2333–2337. [Google Scholar] [CrossRef] [PubMed]
- Leggewie, M.; Badusche, M.; Rudolf, M.; Jansen, S.; Börstler, J.; Krumkamp, R.; Huber, K.; Krüger, A.; Schmidt-Chanasit, J.; Tannich, E.; et al. Culex pipiens and Culex torrentium populations from Central Europe are susceptible to West Nile virus infection. One Health 2016, 2, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Ndiaye el, H.; Fall, G.; Gaye, A.; Bob, N.S.; Talla, C.; Diagne, C.T.; Diallo, D.; Ba, Y.; Dia, I.; Kohl, A.; et al. Vector competence of Aedes vexans (Meigen), Culex poicilipes (Theobald) and Cx. quinquefasciatus Say from Senegal for West and East African lineages of Rift Valley fever virus. Parasit. Vectors 2016, 9, e94. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, K.L.; Bixby, M.A.; Morin, K.J.; Bradley, D.S.; Vaughan, J.A. Potential of a northern population of Aedes vexans (Diptera: Culicidae) to transmit Zika virus. J. Med. Entomol. 2017, 54, 1354–1359. [Google Scholar] [CrossRef] [PubMed]
- Kampen, H.; Werner, D. Out of the bush: The Asian bush mosquito becomes invasive. Parasit. Vectors 2014, 7, 59. [Google Scholar] [CrossRef] [PubMed]
- Walther, D.; Scheuch, D.E.; Kampen, H. The invasive Asian tiger mosquito Aedes albopictus (Diptera: Culicidae) in Germany: Local reproduction and overwintering. Acta Trop. 2017, 166, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Werner, D.; Zielke, D.; Kampen, H. First record of Aedes koreicus (Diptera: Culicidae) in Germany. Parasitol. Res. 2016, 115, 1331–1334. [Google Scholar] [CrossRef] [PubMed]
- EASAC (European Academies’ Science Advisory Council). Extreme weather events in Europe—Statement. Available online: https://easac.eu/fileadmin/PDF_s/reports_statements/Extreme_Weather/EASAC_Extreme_Weather_2018_web_23March.pdf (accessed on 22 July 2018).
- Rudolf, I.; Betášová, L.; Blažejová, H.; Venclíková, K.; Straková, P.; Šebesta, O.; Mendel, J.; Bakonyi, T.; Schaffner, F.; Nowotny, N.; Hubálek, Z. West Nile virus in overwintering mosquitoes, Central Europe. Parasit. Vectors 2017, 10, e452. [Google Scholar] [CrossRef] [PubMed]
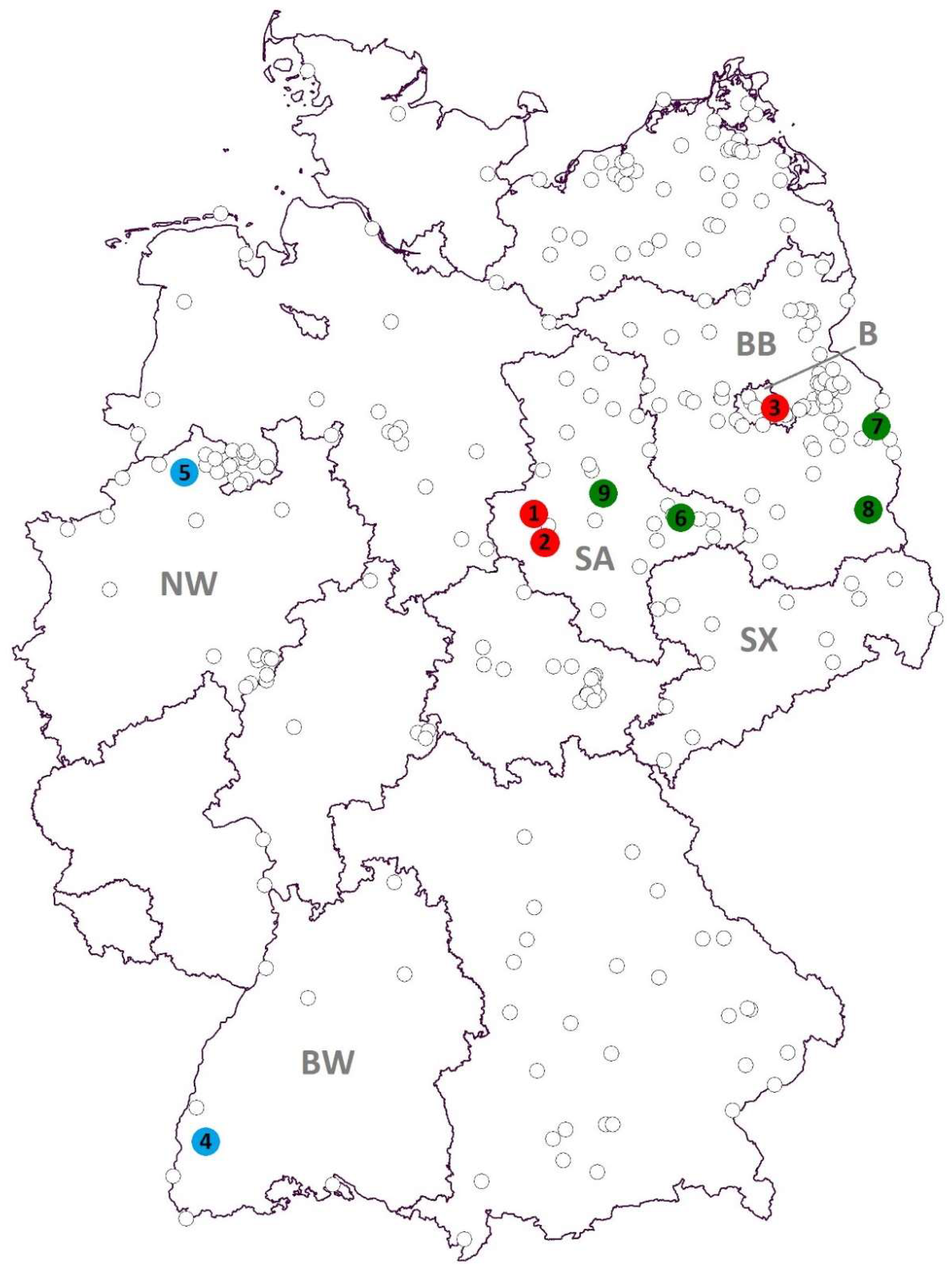
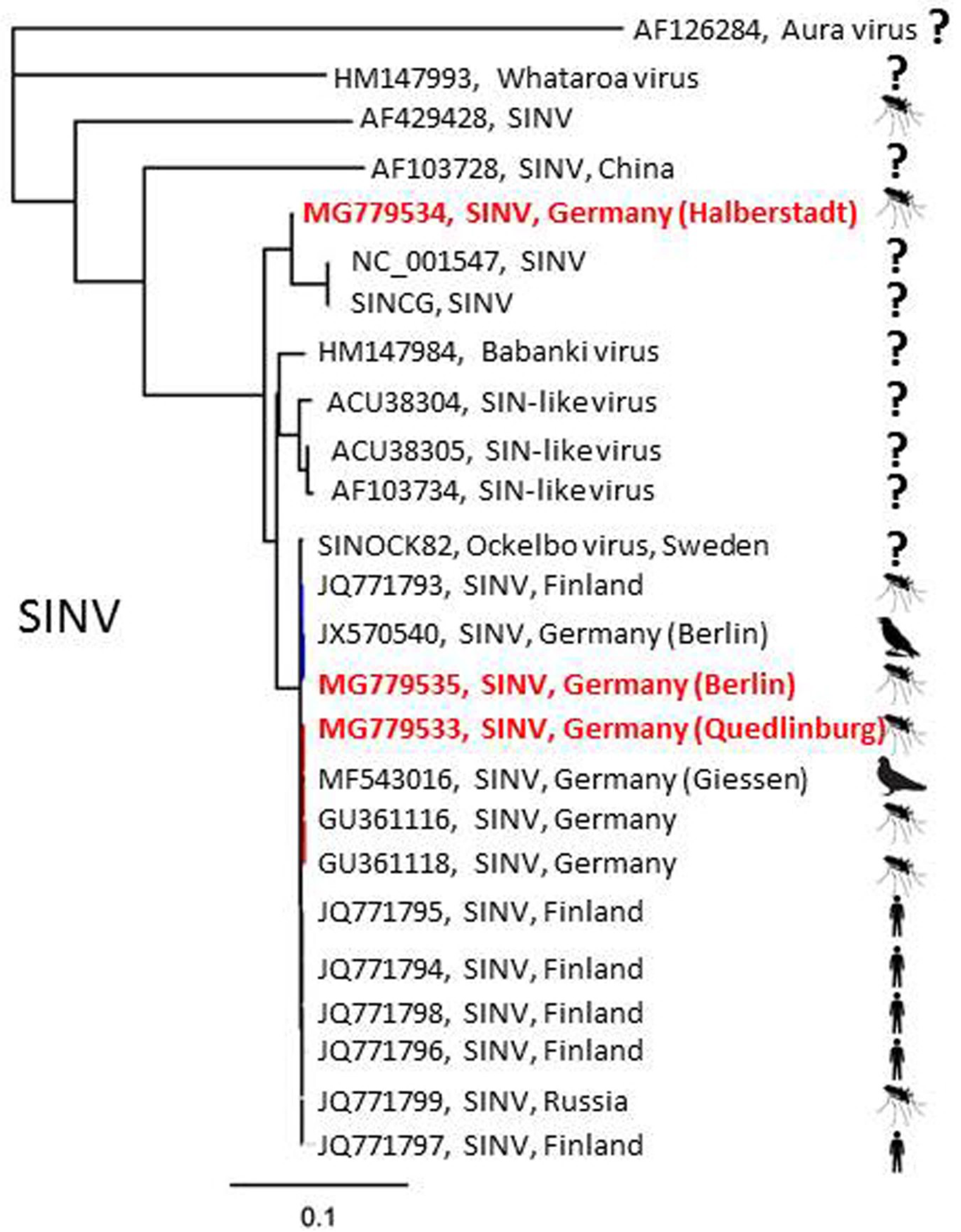

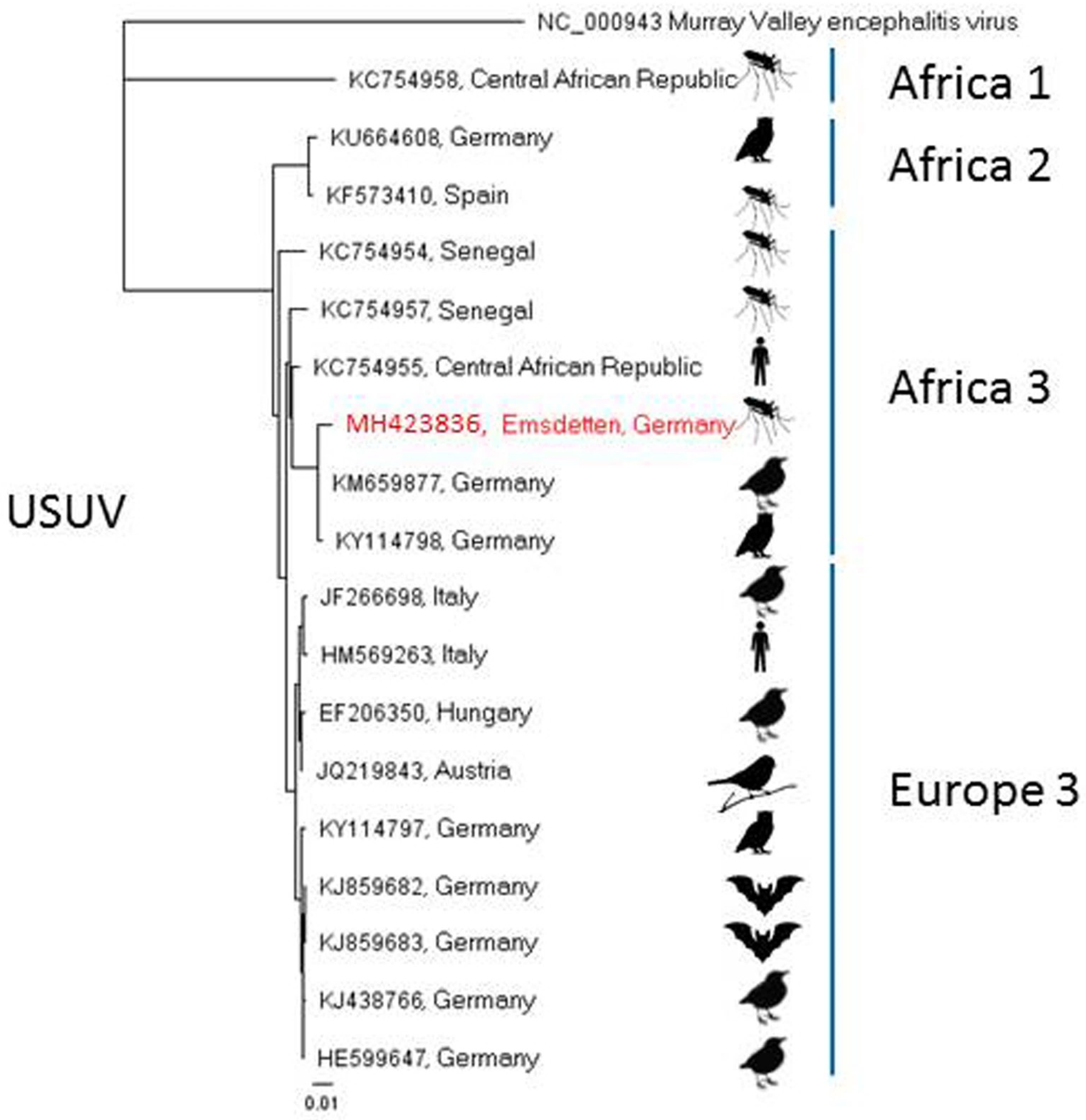
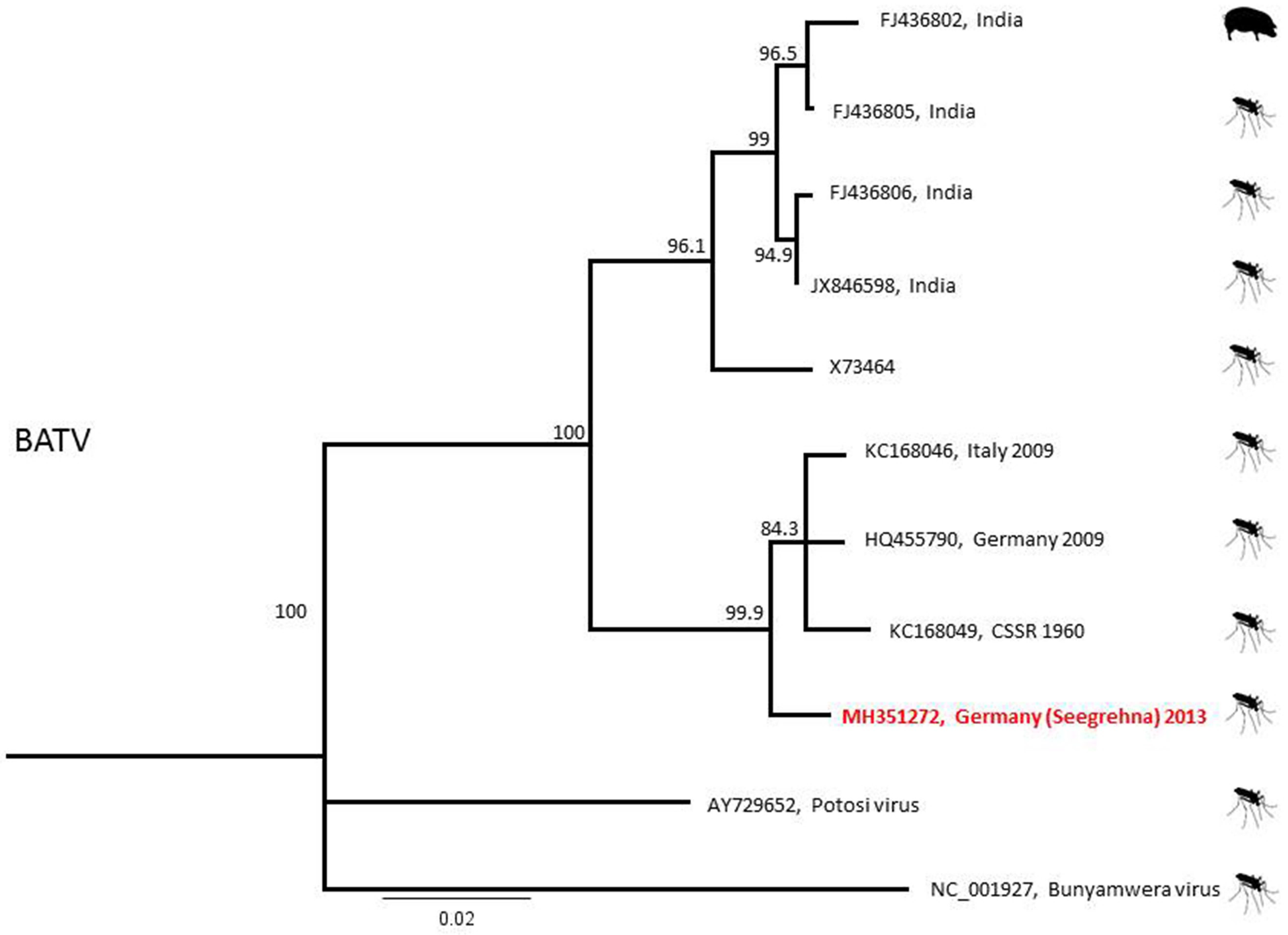
| Detected Virus | Collection Site (Number in Figure 1) | Collection Date | Mosquito Species | Number of Pools (Pool Size) |
|---|---|---|---|---|
| SINV | Halberstadt (1) | 28 July 2013 | Culex pipiens s.l. | 1 (30) |
| Quedlinburg (2) | 29 July 2015 | Culex torrentium | 1 (1) | |
| Berlin (3) | 25 July 2016 | Culex pipiens s.l. | 1 (12) | |
| USUV | Freiburg (4) | 4 September 2014 | Culex pipiens s.l. | 1 (32) |
| Emsdetten (5) | 15 July 2016 | Culex pipiens s.l. | 1 (50) | |
| BATV | Seegrehna (6) | 4 July 2013 | Anopheles messeae | 1 (1) |
| 17 July 2013 | Anopheles daciae | 1 (1) | ||
| Anopheles messeae | 1 (1) | |||
| 21 August 2013 | Anopheles messeae | 1 (1) | ||
| Culex pipiens s.l. | 1 (25) | |||
| Frankfurt/Oder (7) | 13 January 2012 | Culiseta sp. | 1 (15) | |
| Maust (8) | 28 July 2013 | Culex pipiens s.l. | 1 (20) | |
| Schönebeck/Elbe (9) | 1 August 2013 | Anopheles messeae | 6 (1) | |
| Anopheles daciae | 7 (1) | |||
| Aedes vexans | 1 (24) | |||
| Aedes vexans | 1 (33) | |||
| Culex modestus | 1 (44) | |||
| Culex pipiens s.l. | 1 (27) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scheuch, D.E.; Schäfer, M.; Eiden, M.; Heym, E.C.; Ziegler, U.; Walther, D.; Schmidt-Chanasit, J.; Keller, M.; Groschup, M.H.; Kampen, H. Detection of Usutu, Sindbis, and Batai Viruses in Mosquitoes (Diptera: Culicidae) Collected in Germany, 2011–2016. Viruses 2018, 10, 389. https://doi.org/10.3390/v10070389
Scheuch DE, Schäfer M, Eiden M, Heym EC, Ziegler U, Walther D, Schmidt-Chanasit J, Keller M, Groschup MH, Kampen H. Detection of Usutu, Sindbis, and Batai Viruses in Mosquitoes (Diptera: Culicidae) Collected in Germany, 2011–2016. Viruses. 2018; 10(7):389. https://doi.org/10.3390/v10070389
Chicago/Turabian StyleScheuch, Dorothee E., Mandy Schäfer, Martin Eiden, Eva C. Heym, Ute Ziegler, Doreen Walther, Jonas Schmidt-Chanasit, Markus Keller, Martin H. Groschup, and Helge Kampen. 2018. "Detection of Usutu, Sindbis, and Batai Viruses in Mosquitoes (Diptera: Culicidae) Collected in Germany, 2011–2016" Viruses 10, no. 7: 389. https://doi.org/10.3390/v10070389
APA StyleScheuch, D. E., Schäfer, M., Eiden, M., Heym, E. C., Ziegler, U., Walther, D., Schmidt-Chanasit, J., Keller, M., Groschup, M. H., & Kampen, H. (2018). Detection of Usutu, Sindbis, and Batai Viruses in Mosquitoes (Diptera: Culicidae) Collected in Germany, 2011–2016. Viruses, 10(7), 389. https://doi.org/10.3390/v10070389





