Geometric Defects and Icosahedral Viruses
Abstract
1. Symmetry and Structure: A Double-Edged Sword
2. Evolutionary Advantages and Limitations of Icosahedral Symmetry
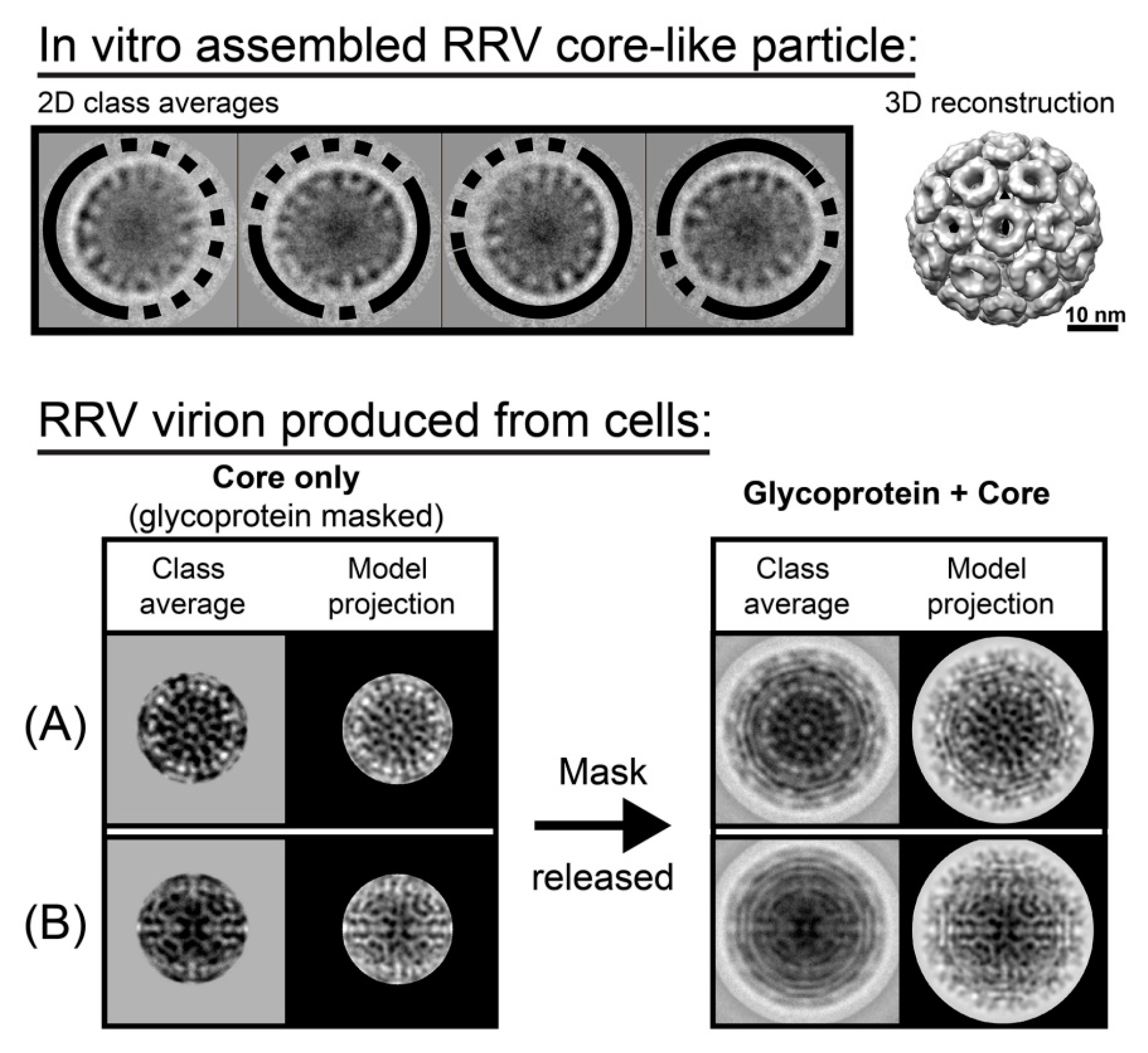
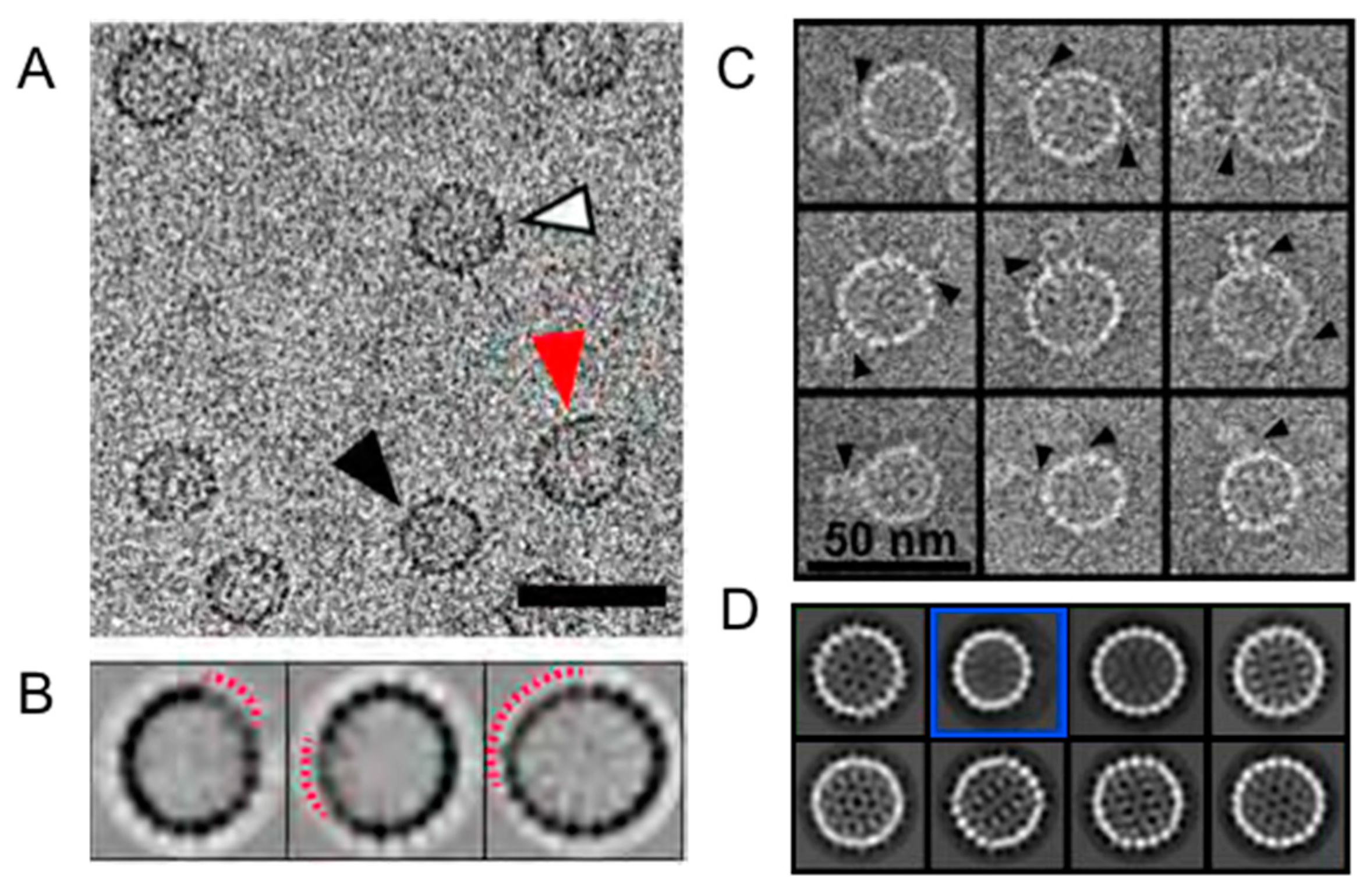
3. Symmetry Defects Are Not Rare
4. A New Perspective on Geometric Defects
Acknowledgments
Conflicts of Interest
References
- Crick, F.H.C.; Watson, J.D. The structure of small viruses. Nature 1956, 177, 473–475. [Google Scholar] [CrossRef] [PubMed]
- Caspar, D.L.D. Structure of tomato bushy stunt virus. Nature 1956, 177, 476–477. [Google Scholar] [CrossRef]
- Rossmann, M.G.; Blow, D.M. The detection of sub-units within the crystallographic asymmetric unit. Acta Crystallogr. 1962, 15, 24–31. [Google Scholar] [CrossRef]
- Baker, T.S.; Cheng, R.H. A model-based approach for determining orientations of biological macromolecules imaged by cryo-electron microscopy. J. Struct. Biol. 1996, 116, 120–130. [Google Scholar] [CrossRef] [PubMed]
- Scheres, S.H. RELION: Implementation of a Bayesian approach to cryo-EM structure determination. J. Struct. Biol. 2012, 180, 519–530. [Google Scholar] [CrossRef] [PubMed]
- Rossmann, M.G.; Johnson, J.E. Icosahedral RNA virus structure. Annu. Rev. Biochem. 1989, 58, 533–573. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.J.; Porta, C.; Chen, Z.G.; Baker, T.S.; Johnson, J.E. Identification of a Fab interaction footprint site on an icosahedral virus by cryo-electron microscopy and X-ray crystallography. Nature 1992, 355, 275–278. [Google Scholar] [CrossRef] [PubMed]
- Belnap, D.M.; Watts, N.R.; Conway, J.F.; Cheng, N.; Stahl, S.J.; Wingfield, P.T.; Steven, A.C. Diversity of core antigen epitopes of hepatitis B virus. Proc. Natl. Acad. Sci. USA 2003, 100, 10884–10889. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.H.; Baker, M.L.; Hryc, C.F.; DiMaio, F.; Jakana, J.; Wu, W.; Dougherty, M.; Haase-Pettingell, C.; Schmid, M.F.; Jiang, W.; et al. Structural basis for scaffolding-mediated assembly and maturation of a dsDNA virus. Proc. Natl. Acad. Sci. USA 2011, 108, 1355–1360. [Google Scholar] [CrossRef] [PubMed]
- Thuman-Commike, P.A.; Greene, B.; Jakana, J.; Prasad, B.V.; King, J.; Prevelige, P.E., Jr.; Chiu, W. Three-dimensional structure of scaffolding-containing phage P22 procapsids by electron cryo-microscopy. J. Mol. Biol. 1996, 260, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Wang, J.C.; Pierson, E.E.; Keifer, D.Z.; Delaleau, M.; Gallucci, L.; Cazenave, C.; Kann, M.; Jarrold, M.F.; Zlotnick, A. Importin β can bind hepatitis B virus core protein and empty core-like particles and induce structural changes. PLoS Pathog. 2016, 12, e1005802. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.C.; Newcomb, W.W. Herpesvirus capsid assembly: Insights from structural analysis. Curr. Opin. Virol. 2011, 1, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Cardone, G.; Winkler, D.C.; Trus, B.L.; Cheng, N.; Heuser, J.E.; Newcomb, W.W.; Brown, J.C.; Steven, A.C. Visualization of the herpes simplex virus portal in situ by cryo-electron tomography. Virology 2007, 361, 426–434. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.T.; Schmid, M.F.; Rixon, F.J.; Chiu, W. Electron cryotomography reveals the portal in the herpesvirus capsid. J. Virol. 2007, 81, 2065–2068. [Google Scholar] [CrossRef] [PubMed]
- Hafenstein, S.; Palermo, L.M.; Kostyuchenko, V.A.; Xiao, C.; Morais, M.C.; Nelson, C.D.; Bowman, V.D.; Battisti, A.J.; Chipman, P.R.; Parrish, C.R.; et al. Asymmetric binding of transferrin receptor to parvovirus capsids. Proc. Natl. Acad. Sci. USA 2007, 104, 6585–6589. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.C.; Nickens, D.G.; Lentz, T.B.; Loeb, D.D.; Zlotnick, A. Encapsidated hepatitis B virus reverse transcriptase is poised on an ordered RNA lattice. Proc. Natl. Acad. Sci. USA 2014, 111, 11329–11334. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Walker, S.B.; Chipman, P.R.; Nibert, M.L.; Baker, T.S. Reovirus polymerase λ3 localized by cryo-electron microscopy of virions at a resolution of 7.6 Å. Nat. Struct. Mol. Boil. 2003, 10, 1011–1018. [Google Scholar] [CrossRef] [PubMed]
- Dent, K.C.; Thompson, R.; Barker, A.M.; Hiscox, J.A.; Barr, J.N.; Stockley, P.G.; Ranson, N.A. The asymmetric structure of an icosahedral virus bound to its receptor suggests a mechanism for genome release. Structure 2013, 21, 1225–1234. [Google Scholar] [CrossRef] [PubMed]
- Dai, X.; Li, Z.; Lai, M.; Shu, S.; Du, Y.; Zhou, Z.H.; Sun, R. In situ structures of the genome and genome-delivery apparatus in a single-stranded RNA virus. Nature 2017, 541, 112–116. [Google Scholar] [CrossRef] [PubMed]
- Pierson, E.E.; Keifer, D.Z.; Selzer, L.; Lee, L.S.; Contino, N.C.; Wang, J.C.-Y.; Zlotnick, A.; Jarrold, M.F. Detection of late intermediates in virus capsid assembly by charge detection mass spectrometry. J. Am. Chem. Soc. 2014, 136, 3536–3541. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.C.-Y.; Chen, C.; Rayaprolu, V.; Mukhopadhyay, S.; Zlotnick, A. Self-assembly of an Alphavirus core-like particle is distinguished by strong intersubunit association energy and structural defects. ACS Nano 2015, 9, 8898–8906. [Google Scholar] [CrossRef] [PubMed]
- Bothner, B.; Dong, X.F.; Bibbs, L.; Johnson, J.E.; Siuzdak, G. Evidence of viral capsid dynamics using limited proteolysis and mass spectrometry. J. Biol. Chem. 1998, 273, 673–676. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Wang, J.C.-Y.; Zlotnick, A. A kinase chaperones hepatitis B virus capsid assembly and captures capsid dynamics in vitro. PLoS Pathog. 2011, 7. [Google Scholar] [CrossRef] [PubMed]
- Perilla, J.R.; Hadden, J.A.; Goh, B.C.; Mayne, C.G.; Schulten, K. All-Atom Molecular Dynamics of Virus Capsids as Drug Targets. J. Phys. Chem. Lett. 2016, 7, 1836–1844. [Google Scholar] [CrossRef] [PubMed]
- Tama, F.; Brooks, C.L. Symmetry, form, and shape: Guiding principles for robustness in macromolecular machines. Annu. Rev. Biophys. Biomol. Struct. 2006, 35, 115–133. [Google Scholar] [CrossRef] [PubMed]
- Uetrecht, C.; Watts, N.R.; Stahl, S.J.; Wingfield, P.T.; Steven, A.C.; Heck, A.J.R. Subunit exchange rates in Hepatitis B virus capsids are geometry- and temperature-dependent. Phys. Chem. Chem. Phys. 2010, 12, 13368–13371. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Zlotnick, A. Observed hysteresis of virus capsid disassembly is implicit in kinetic models of assembly. J. Biol. Chem. 2003, 278, 18249–18255. [Google Scholar] [CrossRef] [PubMed]
- Katen, S.P.; Zlotnick, A. Thermodynamics of Virus Capsid Assembly. Methods Enzymol. 2009, 455, 395–417. [Google Scholar] [PubMed]
- Veesler, D.; Johnson, J.E. Virus maturation. Annu. Rev. Biophys. 2012, 41, 473–496. [Google Scholar] [CrossRef] [PubMed]
- Prevelige, P.E.; Fane, B.A. Building the machines: Scaffolding protein functions during bacteriophage morphogenesis. Adv. Exp. Med. Boil. 2012, 726, 325–350. [Google Scholar]
- Zandi, R.; Reguera, D.; Bruinsma, R.F.; Gelbart, W.M.; Rudnick, J. Origin of icosahedral symmetry in viruses. Proc. Natl. Acad. Sci. USA 2004, 101, 15556–15560. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.D.; Brooks, C.L., 3rd. Generalized structural polymorphism in self-assembled viral particles. Nano Lett. 2008, 8, 4574–4581. [Google Scholar] [CrossRef] [PubMed]
- Paquay, S.; Kusumaatmaja, H.; Wales, D.J.; Zandi, R.; van der Schoot, P. Energetically favoured defects in dense packings of particles on spherical surfaces. Soft Matter 2016, 12, 5708–5717. [Google Scholar] [CrossRef] [PubMed]
- Briggs, J.A.; Riches, J.D.; Glass, B.; Bartonova, V.; Zanetti, G.; Kräusslich, H.G. Structure and assembly of immature HIV. Proc. Natl. Acad. Sci. USA 2009, 106, 11090–11095. [Google Scholar] [CrossRef] [PubMed]
- Schur, F.K.; Obr, M.; Hagen, W.J.; Wan, W.; Jakobi, A.J.; Kirkpatrick, J.M.; Sachse, C.; Kräusslich, H.G.; Briggs, J.A. An atomic model of HIV-1 capsid-SP1 reveals structures regulating assembly and maturation. Science 2016, 353, 506–508. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Dobro, M.J.; Woodward, C.L.; Levandovsky, A.; Danielson, C.M.; Sandrin, V.; Shi, J.; Aiken, C.; Zandi, R.; Hope, T.J.; et al. Unclosed HIV-1 capsids suggest a curled sheet model of assembly. J. Mol. Biol. 2013, 425, 112–123. [Google Scholar] [CrossRef] [PubMed]
- Mukhopadhyay, S.; Chipman, P.R.; Hong, E.M.; Kuhn, R.J.; Rossmann, M.G. In vitro-assembled alphavirus core-like particles maintain a structure similar to that of nucleocapsid cores in mature virus. J. Virol. 2002, 76, 11128–11132. [Google Scholar] [CrossRef] [PubMed]
- Kuhn, R.J.; Zhang, W.; Rossmann, M.G.; Pletnev, S.V.; Corver, J.; Lenches, E.; Jones, C.T.; Mukhopadhyay, S.; Chipman, P.R.; Strauss, E.G.; et al. Structure of dengue virus: Implications for flavivirus organization, maturation, and fusion. Cell 2002, 108, 717–725. [Google Scholar] [CrossRef]
- Mukhopadhyay, S.; Kim, B.S.; Chipman, P.R.; Rossmann, M.G.; Kuhn, R.J. Structure of West Nile virus. Science 2003, 302, 248. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Ge, P.; Yu, X.; Brannan, J.M.; Bi, G.; Zhang, Q.; Schein, S.; Zhou, Z.H. Cryo-EM structure of the mature dengue virus at 3.5-Å resolution. Nat. Struct. Mol. Biol. 2013, 20, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Sirohi, D.; Chen, Z.; Sun, L.; Klose, T.; Pierson, T.C.; Rossmann, M.G.; Kuhn, R.J. The 3.8 Å resolution cryo-EM structure of Zika virus. Science 2016, 352, 467–470. [Google Scholar] [CrossRef] [PubMed]
- Kostyuchenko, V.A.; Lim, E.X.; Zhang, S.; Fibriansah, G.; Ng, T.-S.; Ooi, J.G.; Shi, J.; Lok, S.M. Structure of the thermally stable Zika virus. Nature 2016, 533, 425–428. [Google Scholar] [CrossRef] [PubMed]
- Byrd, C.M.; Dai, D.; Grosenbach, D.W.; Berhanu, A.; Jones, K.F.; Cardwell, K.B.; Schneider, C.; Wineinger, K.A.; Page, J.M.; Harver, C.; et al. A novel inhibitor of dengue virus replication that targets the capsid protein. Antimicrob. Agents Chemother. 2013, 57, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Scaturro, P.; Trist, I.M.; Paul, D.; Kumar, A.; Acosta, E.G.; Byrd, C.M.; Jordan, R.; Brancale, A.; Bartenschlager, R. Characterization of the mode of action of a potent dengue virus capsid inhibitor. J. Virol. 2014, 88, 11540–11555. [Google Scholar] [CrossRef] [PubMed]
- Larson, S.B.; Lucas, R.W.; McPherson, A. Crystallographic structure of the T = 1 particle of brome mosaic virus. J. Mol. Biol. 2005, 346, 815–831. [Google Scholar] [CrossRef] [PubMed]
- Lucas, R.W.; Larson, S.B.; McPherson, A. The crystallographic structure of brome mosaic virus. J. Mol. Biol. 2002, 317, 95–108. [Google Scholar] [CrossRef] [PubMed]
- Bostina, M.; Levy, H.; Filman, D.J.; Hogle, J.M. Poliovirus RNA is released from the capsid near a twofold symmetry axis. J. Virol. 2011, 85, 776–783. [Google Scholar] [CrossRef] [PubMed]
- Levy, H.C.; Bostina, M.; Filman, D.J.; Hogle, J.M. Catching a virus in the act of RNA release: A novel poliovirus uncoating intermediate characterized by cryo-electron microscopy. J. Virol. 2010, 84, 4426–4441. [Google Scholar] [CrossRef] [PubMed]
- Harms, Z.D.; Selzer, L.; Zlotnick, A.; Jacobson, S.C. Monitoring assembly of virus capsids with nanofluidic devices. ACS Nano 2015, 9, 9087–9096. [Google Scholar] [CrossRef] [PubMed]
- Lutomski, C.A.; Lyktey, N.A.; Zhao, Z.; Pierson, E.E.; Zlotnick, A.; Jarrold, M.F. Hepatitis B virus capsid completion occurs through error correction. J. Am. Chem. Soc. 2017, 139, 16932–16938. [Google Scholar] [CrossRef] [PubMed]
- Pierson, E.; Keifer, D.Z.; Kukreja, A.A.; Wang, J.C.-Y.; Zlotnick, A.; Jarrold, M.J. Charge detection mass spectrometry identifies preferred non-icosahedral polymorphs in the self-assembly of Woodchuck hepatitis virus capsids. J. Mol. Biol. 2016, 428, 292–300. [Google Scholar] [CrossRef] [PubMed]
- Ning, X.; Nguyen, D.; Mentzer, L.; Adams, C.; Lee, H.; Ashley, R.; Hafenstein, S.; Hu, J. Secretion of genome-free hepatitis B virus—Single strand blocking model for virion morphogenesis of para-retrovirus. PLoS Pathog. 2011, 7. [Google Scholar] [CrossRef] [PubMed]
- Lee, L.S.; Brunk, N.; Haywood, D.G.; Keifer, D.; Pierson, E.; Kondylis, P.; Wang, J.C.; Jacobson, S.C.; Jarrold, M.F.; Zlotnick, A. A molecular breadboard: Removal and replacement of subunits in a hepatitis B virus capsid. Protein Sci. 2017, 26, 2170–2180. [Google Scholar] [CrossRef] [PubMed]
- Lander, G.C.; Tang, L.; Casjens, S.R.; Gilcrease, E.B.; Prevelige, P.; Poliakov, A.; Potter, C.S.; Carragher, B.; Johnson, J.E. The structure of an infectious P22 virion shows the signal for headful DNA packaging. Science 2006, 312, 1791–1795. [Google Scholar] [CrossRef] [PubMed]
- Bharat, T.A.; Russo, C.J.; Lowe, J.; Passmore, L.A.; Scheres, S.H. Advances in single-particle electron cryo-microscopy structure determination applied to sub-tomogram averaging. Structure 2015, 23, 1743–1753. [Google Scholar] [CrossRef] [PubMed]
- Grigorieff, N. Direct detection pays off for electron cryo-microscopy. eLife 2013, 2, e00573. [Google Scholar] [CrossRef] [PubMed]
- Grigorieff, N. FREALIGN: High-resolution refinement of single particle structures. J. Struct. Biol. 2007, 157, 117–125. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, J.C.-Y.; Mukhopadhyay, S.; Zlotnick, A. Geometric Defects and Icosahedral Viruses. Viruses 2018, 10, 25. https://doi.org/10.3390/v10010025
Wang JC-Y, Mukhopadhyay S, Zlotnick A. Geometric Defects and Icosahedral Viruses. Viruses. 2018; 10(1):25. https://doi.org/10.3390/v10010025
Chicago/Turabian StyleWang, Joseph Che-Yen, Suchetana Mukhopadhyay, and Adam Zlotnick. 2018. "Geometric Defects and Icosahedral Viruses" Viruses 10, no. 1: 25. https://doi.org/10.3390/v10010025
APA StyleWang, J. C.-Y., Mukhopadhyay, S., & Zlotnick, A. (2018). Geometric Defects and Icosahedral Viruses. Viruses, 10(1), 25. https://doi.org/10.3390/v10010025





