Drought Differentially Affects Growth, Transpiration, and Water Use Efficiency of Mixed and Monospecific Planted Forests
Abstract
:1. Introduction
2. Materials and Methods
2.1. Site Descriptions
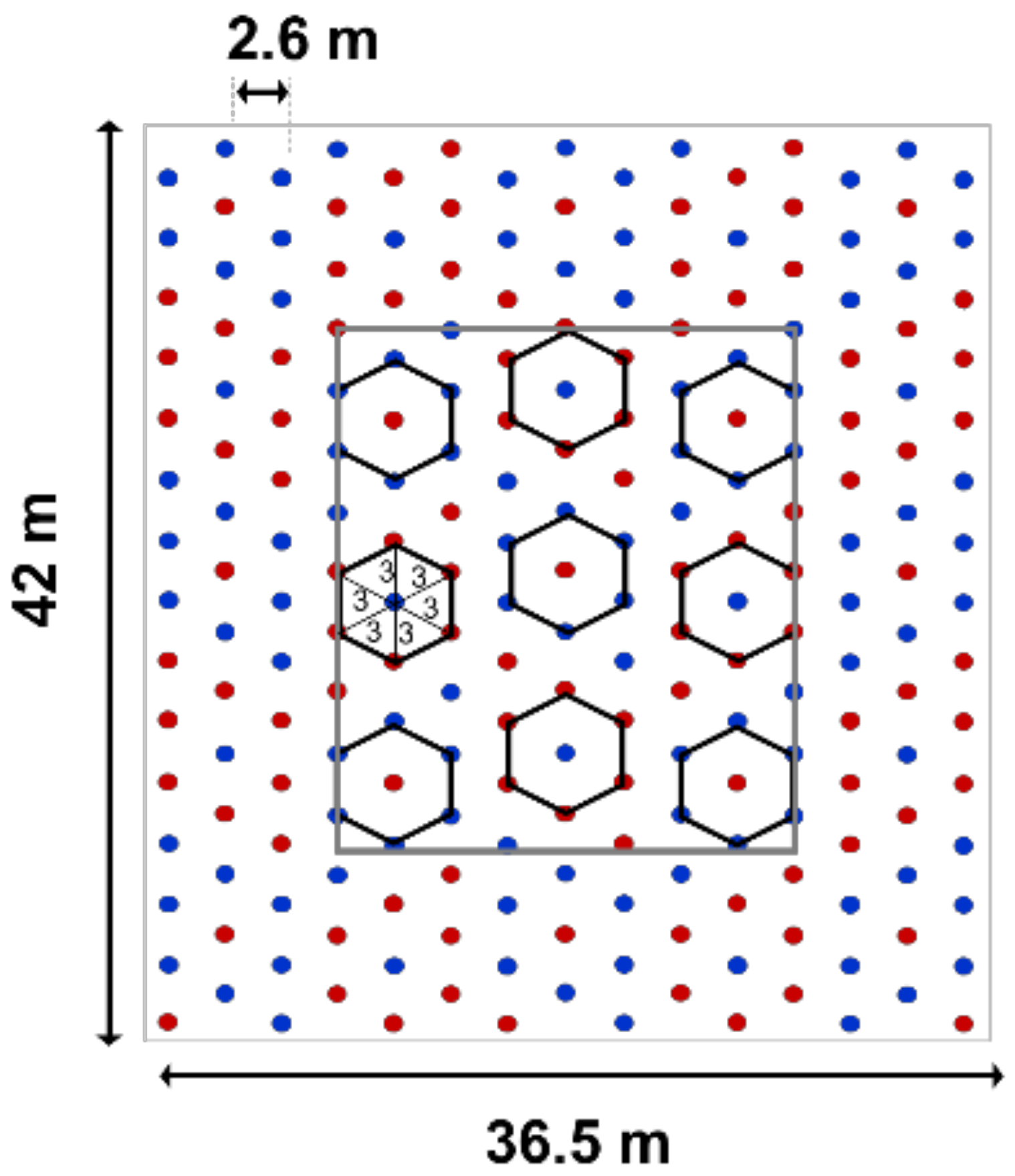
2.2. Sampling Design
2.3. Micrometeorogical and Soil Moisture Data
2.4. Basal Area Growth and Biomass Measurements
2.5. Tree and Stand Transpiration and Water Use Efficiency
2.6. Statistical Analyses
3. Results
3.1. Normal Year and Drought Year Meteorological Changes
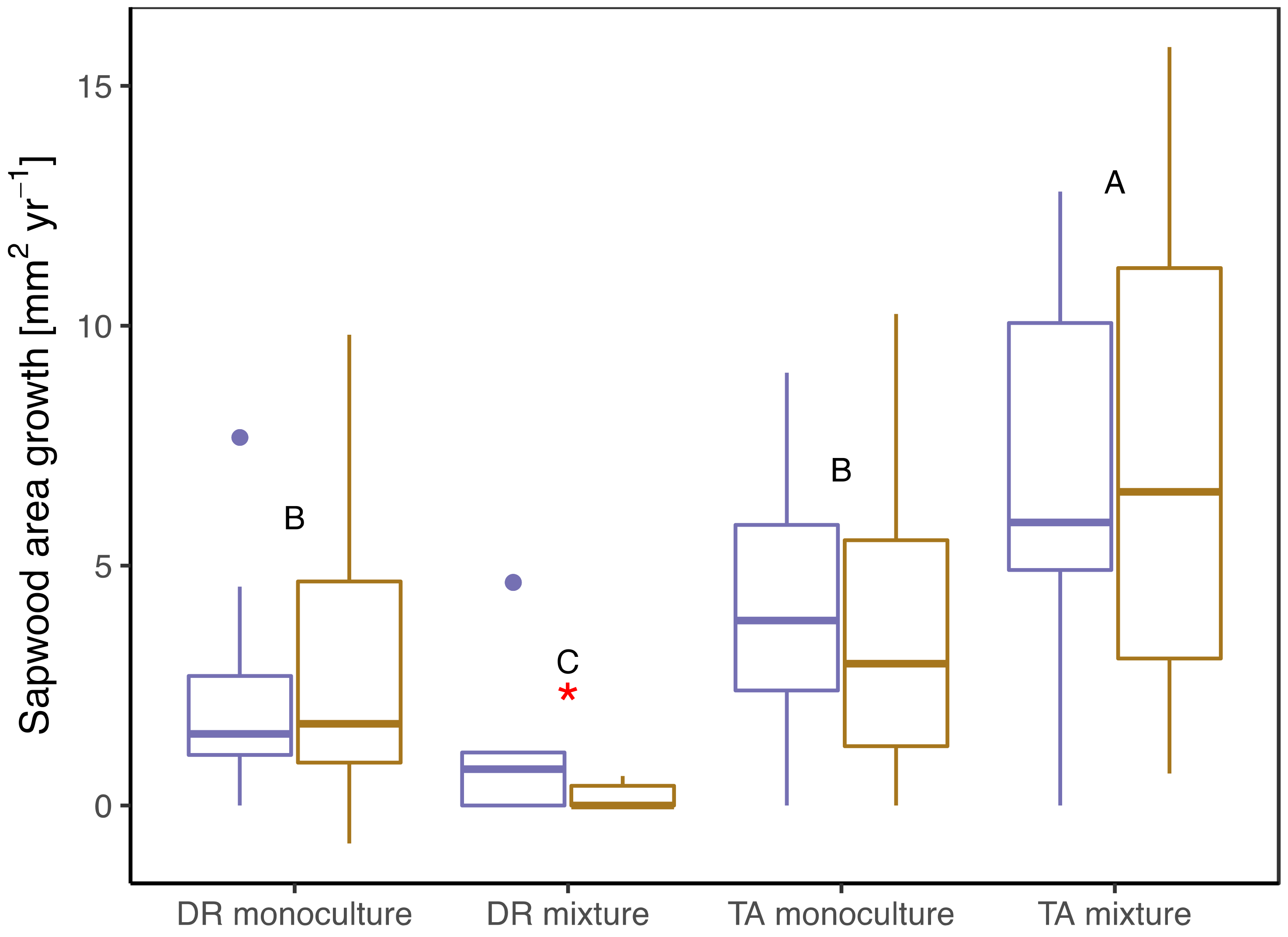
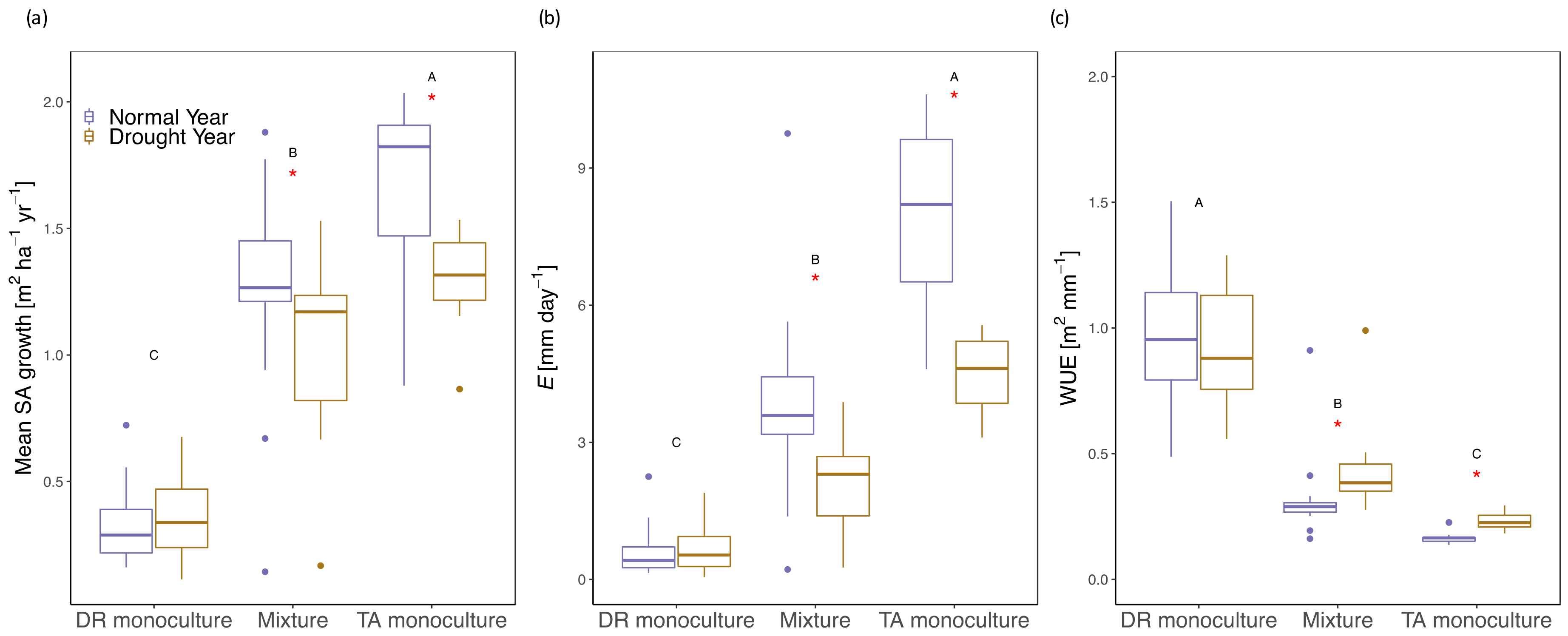
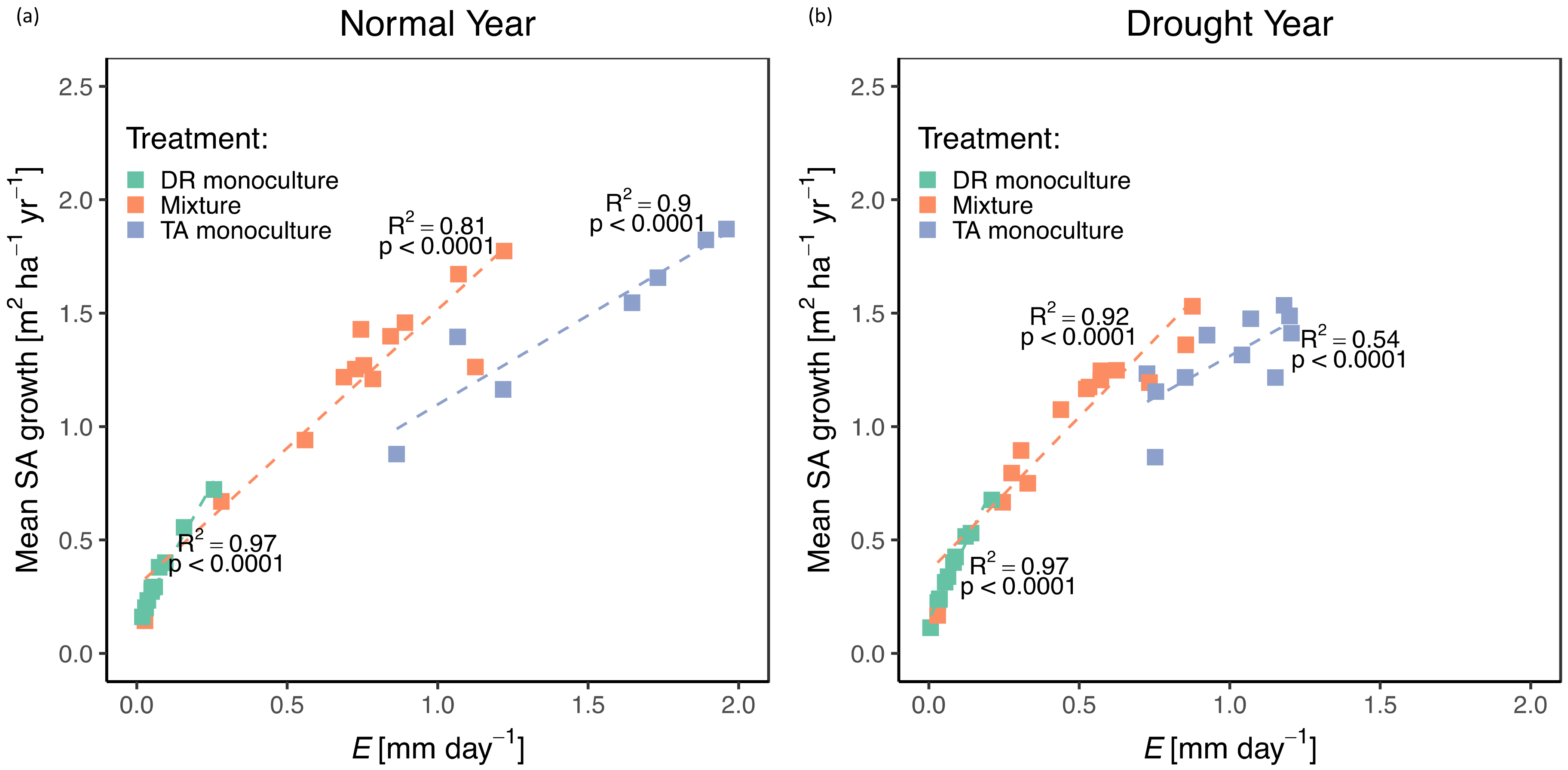
3.2. Growth/Productivity
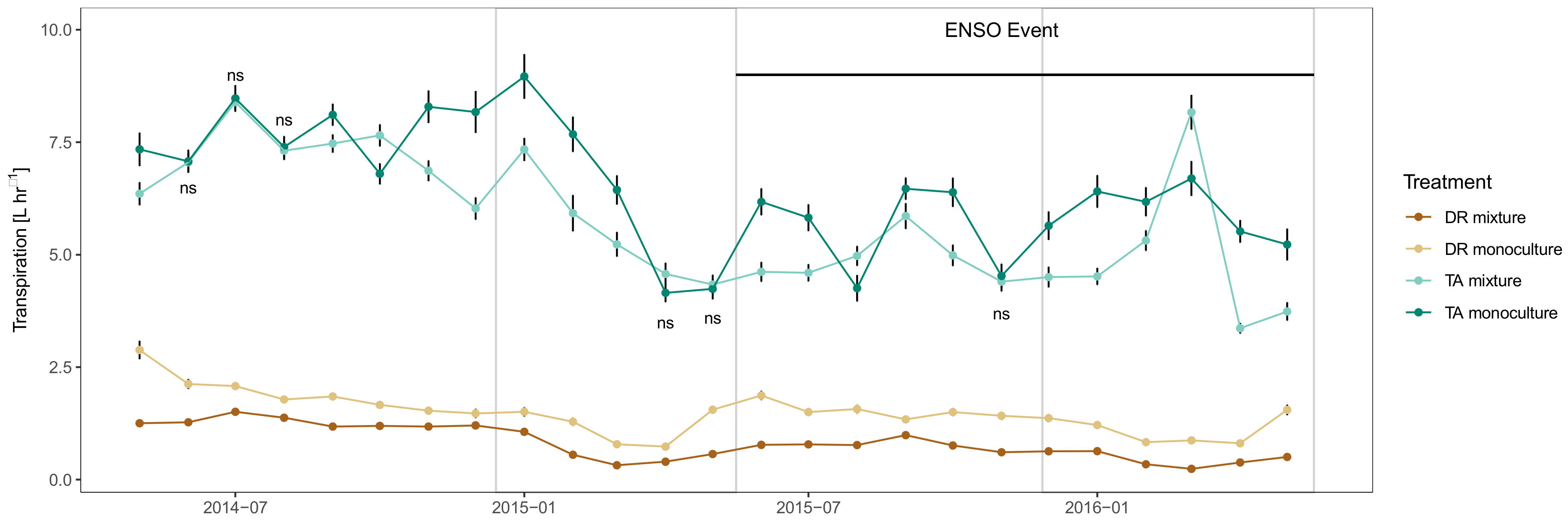
3.3. Tree- and Stand- Transpiration
3.4. Monthly Differences in Whole Tree Transpiration
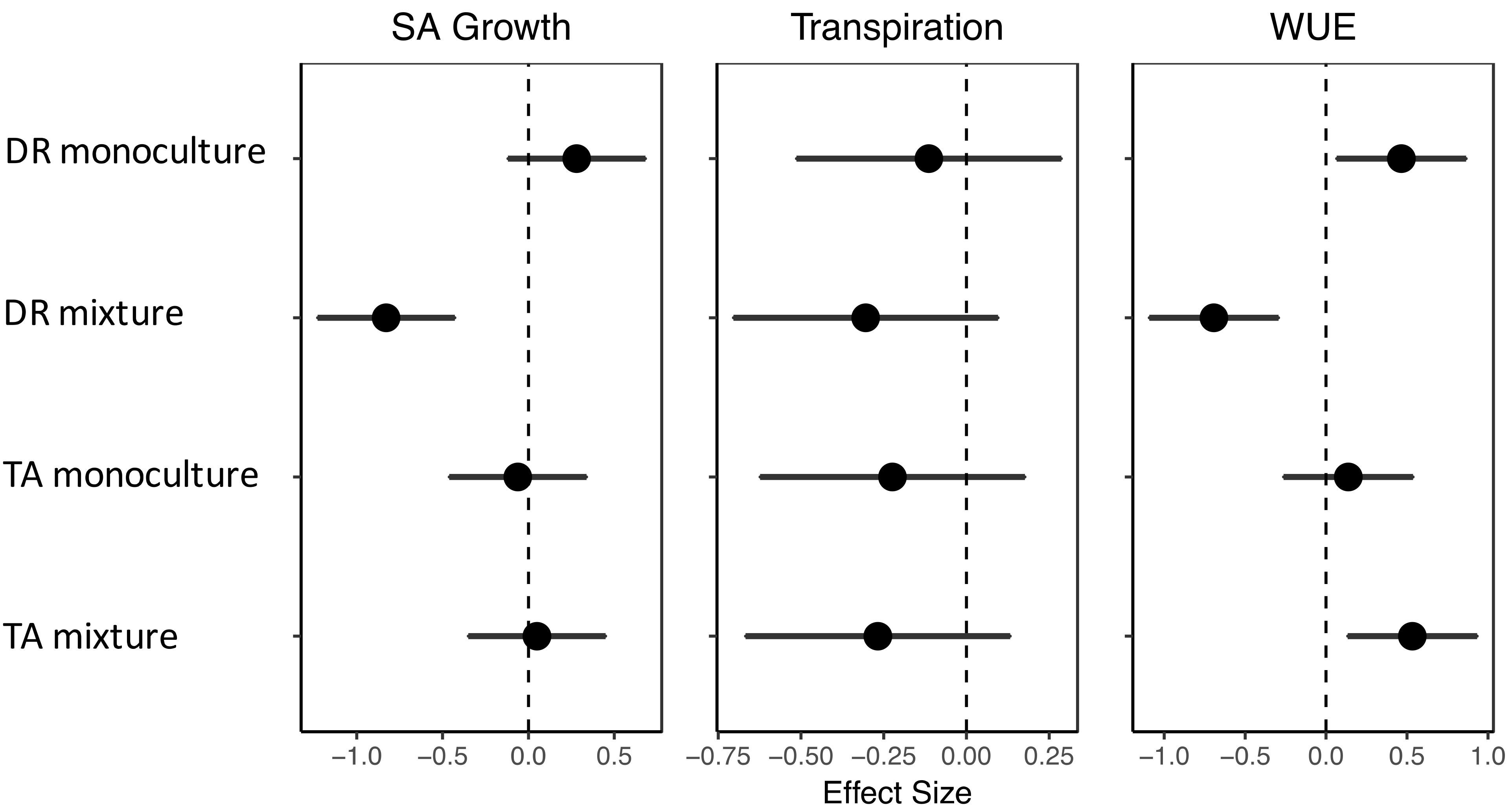
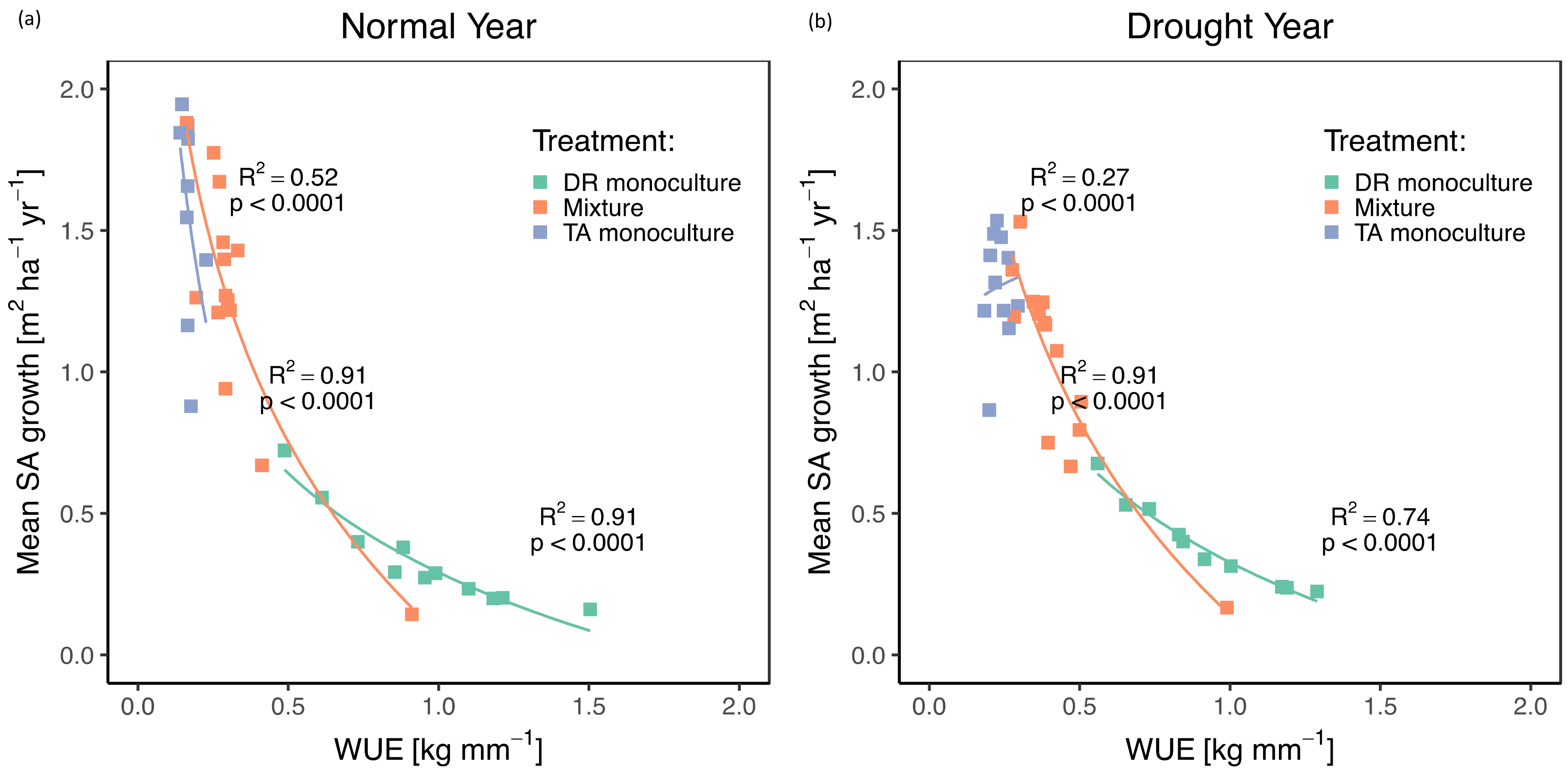
3.5. Water-Use Efficiency
4. Discussion
4.1. Role of Complementary Interactions in Mixtures During a “Normal” Year
4.2. Effects of Drought on Growth, Transpiration, and WUE
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Food and Agriculture Organization Global Forest Products: Facts and Figures. 2016, p. 16. Available online: http://120.52.51.13/www.fao.org/3/I7034EN/i7034en.pdf (accessed on 3 May 2018).
- Chazdon, R.L. Tropical forest recovery: Legacies of human impact and natural disturbances. Perspect. Plant. Ecol. Evol. Syst. 2003, 6, 51–71. [Google Scholar] [CrossRef]
- Paquette, A.; Hector, A.; Castagneyrol, B.; Vanhellemont, M.; Koricheva, J.; Scherer-Lorenzen, M.; Verheyen, K. A million and more trees for science. Nat. Ecol. Evol. 2018, 2, 763–766. [Google Scholar] [CrossRef] [PubMed]
- Paul, C.; Griess, V.C.; Havardi-Burger, N.; Weber, M. Timber-based agrisilviculture improves financial viability of hardwood plantations: A case study from Panama. Agrofor. Syst. 2015, 89, 217–235. [Google Scholar] [CrossRef]
- Piotto, D. A meta-analysis comparing tree growth in monocultures and mixed plantations. For. Ecol. Manag. 2008, 255, 781–786. [Google Scholar] [CrossRef]
- Montagnini, F. Accumulation in above-ground biomass and soil storage of mineral nutrients in pure and mixed plantations in a humid tropical lowland. For. Ecol. Manag. 2000, 134, 257–270. [Google Scholar] [CrossRef]
- Law, B.E.; Falge, E.; Gu, L.; Baldocchi, D.D.; Bakwin, P.; Berbigier, P.; Davis, K.; Dolman, A.J.; Falk, M.; Fuentes, J.D.; et al. Environmental controls over carbon dioxide and water vap exchange of terrestrial vegetation. Agric. For. Meteorol. 2002, 113, 97–120. [Google Scholar] [CrossRef]
- Zhang, L.; Hicken, K.; Dawes, W.R.; Chiew, F.H.; Western, A.W.; Briggs, P. A rational function approach for estimating mean annual evapotranspiration. Water Resour. Res. 2004, 40, 1–14. [Google Scholar] [CrossRef]
- Bruijnzeel, L.A. Hydrological functions of tropical forests: Not seeing the soil for the trees? Argic. Ecosyst. Environ. 2004, 4, 185–228. [Google Scholar] [CrossRef]
- Jackson, R.B.; Avissar, R.; Jackson, R.B.; Jobba, E.G.; Roy, S.B.; Barrett, D.J.; Cook, C.W.; Farley, K.A.; Maitre, D.C.; Mccarl, B.A.; et al. Trading Water for Carbon with Biological Carbon Sequestration. Science 2009, 310, 1944–1947. [Google Scholar] [CrossRef]
- Bennett, A.C.; Mcdowell, N.G.; Allen, C.D.; Anderson-Teixeira, K.J. Larger trees suffer most during drought in forests worldwide. Nat. Plants 2015, 1, 1–5. [Google Scholar] [CrossRef]
- Asbjornsen, H.; Campbell, J.L.; Jennings, K.A.; Vadeboncoeur, M.A.; McIntire, C.; Templer, P.H.; Phillips, R.P.; Bauerle, T.L.; Dietze, M.C.; Frey, S.D. Guidelines and considerations for designing field experiments simulating precipitation extremes in forest ecosystems. Methods Ecol. Evol. 2018, 9, 2310–2325. [Google Scholar] [CrossRef]
- Alvarado-Barrientos, M.S.; Holwerda, F.; Asbjornsen, H.; Dawson, T.E.; Bruijnzeel, L.A. Suppression of transpiration due to cloud immersion in a seasonally dry Mexican weeping pine plantation. Agric. For. Meteorol. 2014, 186, 12–25. [Google Scholar] [CrossRef]
- Bretfeld, M.; Ewers, B.E.; Hall, J.S. Plant water use responses along secondary forest succession during the 2015-2016 El Niño drought in Panama. New Phytol. 2018, 219, 885–899. [Google Scholar] [CrossRef] [PubMed]
- Zeppel, M.J.B.; Murray, B.R.; Barton, C.; Eamus, D. Seasonal responses of xylem sap velocity to VPD and solar radiation during drought in a stand of native trees in temperate Australia. Funct. Plant. Biol. 2004, 31, 461–470. [Google Scholar] [CrossRef]
- Allen, K.; Dupuy, J.M.; Gei, M.G.; Hulshof, C.; Medvigy, D.; Pizano, C.; Salgado-Negret, B.; Smith, C.M.; Trierweiler, A.; Van Bloem, S.J.; et al. Will seasonally dry tropical forests be sensitive or resistant to future changes in rainfall regimes? Environ. Res. Lett. 2017, 12, 023001. [Google Scholar] [CrossRef]
- McDowell, N.; Pockman, W.T.; Allen, C.D.; Breshears, D.D.; Cobb, N.; Kolb, T.; Plaut, J.; Sperry, J.; West, A.; Williams, D.G.; et al. Mechanisms of plant survival and mortality during drought: Why do some plants survive while others succumb to drought? New Phytol. 2008, 178, 719–739. [Google Scholar] [CrossRef] [PubMed]
- Binkley, D.; Stape, J.; Ryan, M. Thinking about efficiency of resource use in forests. For. Ecol. Manag. 2004, 233, 195–204. [Google Scholar] [CrossRef]
- Bellard, C.; Bertelsmeier, C.; Leadley, P.; Thuiller, W.; Courchamp, F. Impacts of climate change on the future of biodiversity. Ecol. Lett. 2012, 15, 365–377. [Google Scholar] [CrossRef]
- Pretzsch, H.; Schütze, G.; Uhl, E. Resistance of European tree species to drought stress in mixed versus pure forests: Evidence of stress release by inter-specific facilitation. Plant. Biol. (Stuttg). 2013, 15, 483–495. [Google Scholar] [CrossRef]
- Forrester, D.I.; Theiveyanathan, S.; Collopy, J.J.; Marcar, N.E. Enhanced water use efficiency in a mixed Eucalyptus globulus and Acacia mearnsii plantation. For. Ecol. Manag. 2010, 259, 1761–1770. [Google Scholar] [CrossRef]
- Kunert, N.; Schwendenmann, L.; Potvin, C.; Hölscher, D. Tree diversity enhances tree transpiration in a Panamanian forest plantation. J. Appl. Ecol. 2012, 49, 135–144. [Google Scholar] [CrossRef]
- Jucker, T.; Bouriaud, O.; Avacaritei, D.; Dǎnilǎ, I.; Duduman, G.; Valladares, F.; Coomes, D.A. Competition for light and water play contrasting roles in driving diversity-productivity relationships in Iberian forests. J. Ecol. 2014, 102, 1202–1213. [Google Scholar] [CrossRef]
- Lebourgeois, F.; Gomez, N.; Pinto, P.; Mérian, P. Mixed stands reduce Abies alba tree-ring sensitivity to summer drought in the Vosges mountains, western Europe. For. Ecol. Manag. 2013, 303, 61–71. [Google Scholar] [CrossRef]
- Ewel, J.J.; Celis, G.; Schreeg, L. Steeply increasing growth differential between mixture and monocultures of tropical trees. Biotropica 2015, 47, 162–171. [Google Scholar] [CrossRef]
- Forrester, D.I. Transpiration and water-use efficiency in mixed-species forests versus monocultures: Effects of tree size, stand density and season. Tree Physiol. 2015, 35, 289–304. [Google Scholar] [CrossRef] [PubMed]
- Hooper, D.U.; Dukes, J.S. Overyielding among plant functional groups in a long-term experiment. Ecol. Lett. 2004, 7, 95–105. [Google Scholar] [CrossRef]
- He, Q.; Bertness, M.D.; Altieri, A.H. Global shifts towards positive species interactions with increasing environmental stress. Ecol. Lett. 2013, 16, 695–706. [Google Scholar] [CrossRef] [PubMed]
- Forrester, D.I. The spatial and temporal dynamics of species interactions in mixed-species forests: From pattern to process. For. Ecol. Manag. 2014, 312, 282–292. [Google Scholar] [CrossRef]
- Potter, K.M.; Woodall, C.W. Does biodiversity make a difference? Relationships between species richness, evolutionary diversity, and aboveground live tree biomass across U.S. forests. For. Ecol. Manag. 2014, 321, 117–129. [Google Scholar] [CrossRef]
- Asbjornsen, H.; Mora, G.; Helmers, M. Variation in depth of water uptake in contrasting perennial and annual ecosystems in central Iowa: Application of a multi-source mass-balance isotopic approach. Agric. Ecosyst. Environ. 2007, 121, 343–356. [Google Scholar] [CrossRef]
- Hutchison, C.; Gravel, D.; Guichard, F.; Potvin, C. Effect of diversity on growth, mortality, and loss of resilience to extreme climate events in a tropical planted forest experiment. Sci. Rep. 2018, 8, 15443. [Google Scholar] [CrossRef] [PubMed]
- Mayoral, C.; van Breugel, M.; Cerezo, A.; Hall, J.S. Survival and growth of five Neotropical timber species in monocultures and mixtures. For. Ecol. Manag. 2017, 403, 1–11. [Google Scholar] [CrossRef]
- Batterman, S.A.; Hall, J.S.; Turner, B.L.; Hedin, L.O.; LaHaela Walter, J.K.; Sheldon, P.; van Breugel, M. Phosphatase activity and nitrogen fixation reflect species differences, not nutrient trading or nutrient balance, across tropical rainforest trees. Ecol. Lett. 2018, 21, 1486–1495. [Google Scholar] [CrossRef] [PubMed]
- Weber, D.; Hall, J.S. Resumen del Proyecto Agua Salud, 2009. Typescript Report. Available online: http://www.ctfs.si.edu/aguasalud/page/documents/ (accessed on 4 January 2014).
- Hassler, S.K.; Zimmermann, B.; van Breugel, M.; Hall, J.S.; Elsenbeer, H. Recovery of saturated hydraulic conductivity under secondary succession on former pasture in the humid tropics. For. Ecol. Manag. 2011, 261, 1634–1642. [Google Scholar] [CrossRef]
- Mayoral, C.; van Breugel, M.; Turner, B.; Asner, G.; Vaughn, N.R.; Hall, J.S. Predicting the effect of microsite quality and species composition on tree growth: A new modeling approach. For. Ecol. Manag. 2018, 432, 534–545. [Google Scholar] [CrossRef]
- Cernusak, L.; Aranda, J. Large variation in whole-plant water-use efficiency among tropical tree species. New Phytol. 2007, 173, 294–305. [Google Scholar] [CrossRef] [PubMed]
- Craven, D.; Hall, J.S.; Ashton, M.S.; Berlyn, G.P. Water-use efficiency and whole-plant performance of nine tropical tree species at two sites with contrasting water availability in Panama. Trees 2013, 27, 639–653. [Google Scholar] [CrossRef]
- Hernandez-Santana, V.; Hernandez-Hernandez, A.; Vadeboncoeur, M.A.; Asbjornsen, H. Scaling from single-point sap velocity measurements to stand transpiration in a multispecies deciduous forest: Uncertainty sources, stand structure effect, and future scenarios. Can. J. For. Res. 2015, 45, 1489–1497. [Google Scholar] [CrossRef]
- Allen, R.G.; Pereira, L.S.; Raes, D.; Smith, M. Crop Evapotranspiration: Guidelines for Computing Crop Water Requirements-FAO Irrigation and Drainage Paper 56; Food and Agriculture Organization of the United Nations: Rome, Italy, 1998. [Google Scholar]
- Paton, S. Meteorological and Hydrological Summary for Barro Colorado Island. 2016. Available online: https://biogeodb.stri.si.edu/physical_monitoring/pdf/BCI%20Met&Hyd%20Report%202016.pdf (accessed on 4 June 2018).
- Sinacore, K.; Hall, J.S.; Potvin, C.; Royo, A.A.; Ducey, M.J.; Ashton, M.S. Unearthing the hidden world of roots: Root biomass and architecture differ among species within the same guild. PLoS ONE 2017, 12, e0185934. [Google Scholar] [CrossRef]
- Miller, E.; Dandois, J.P.; Detto, M.; Hall, J.S. Drones as a tool for monoculture plantation assessment in the steepland tropics. Forests 2017, 8, 168. [Google Scholar] [CrossRef]
- Burgess, S.S.O.; Adams, M.A.; Turner, N.C.; Beverly, C.R.; Ong, C.K.; Khan, A.A.H.; Bleby, T.M. An improved heat pulse method to measure low and reverse rates of sap flow in woody plants. Tree Physiol. 2001, 21, 589–598. [Google Scholar] [CrossRef] [PubMed]
- Green, S.; Clothier, B.; Jardine, B. Theory and practical application of heat pulse to measure sap flow. Agron. J. 2003, 95, 1371–1379. [Google Scholar] [CrossRef]
- R Core Team. A language and environment for statistical computing 2017. Available online: https://www.R-project.org/ (accessed on 6 January 2014).
- Botta-Dukát, Z. Cautionary note on calculating standardized effect size (SES) in randomization test. Community Ecol. 2018, 19, 77–83. [Google Scholar] [CrossRef]
- Forrester, D.I.; Bauhus, J. A Review of Processes Behind Diversity—Productivity Relationships in Forests. Curr. For. Reports 2016, 45–61. [Google Scholar] [CrossRef]
- Forrester, D.I.; Smith, R.G.B. Faster growth of Eucalyptus grandis and Eucalyptus pilularis in mixed-species stands than monocultures. For. Ecol. Manag. 2012, 286, 81–86. [Google Scholar] [CrossRef]
- Loreau, M.; Hector, A. Partitioning selection and complementarity in biodiversity experiments. Nature 2001, 412, 72–76. [Google Scholar] [CrossRef] [PubMed]
- Forrester, D.I.; Bauhus, J.; Cowie, A.L.; Vanclay, J.K. Mixed-species plantations of Eucalyptus with nitrogen-fixing trees: A review. For. Ecol. Manag. 2006, 233, 211–230. [Google Scholar] [CrossRef]
- Bauhus, J.; van Winden, A.P.; Nicotra, A.B. Aboveground interactions and productivity in mixed-species plantations of Acacia mearnsii and Eucalyptus globulus. Can. J. For. Res. 2004, 694, 686–694. [Google Scholar] [CrossRef]
- Hall, J.S.; Ashton, P.M.S. Guide To Early Growth and Survival in Plantations of 64 Tree Species Native to Panama and the Neotropics; Iyer, G., Ed.; Smithsonian Tropical Research Institute: Balboa, Panama, Republic of Panama, 2016. [Google Scholar]
- Meinzer, F.C.; Goldstein, G.; Andrade, J.L. Regulation of water flux through tropical forest canopy trees: Do universal rules apply? Tree Physiol. 2001, 21, 19–26. [Google Scholar] [CrossRef]
- Stape, J.L.; Binkley, D.; Ryan, M.G. Eucalyptus production and the supply, use and efficiency of use of water, light and nitrogen across a geographic gradient in Brazil. For. Ecol. Manag. 2004, 193, 17–31. [Google Scholar] [CrossRef]
- Allen, C.D.; Macalady, A.K.; Chenchouni, H.; Bachelet, D.; McDowell, N.; Vennetier, M.; Kitzberger, T.; Rigling, A.; Breshears, D.D.; Hogg, E.H.; et al. A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. For. Ecol. Manag. 2010, 259, 660–684. [Google Scholar] [CrossRef]
- Hall, J.S.; Kirn, V.; Yanguas-Fernandez, E. (Eds.) Managing watersheds for ecosystem services in the steepland neotropics. In Inter-American Development Bank Monograph; Inter-American Development Bank: Washington, DC, USA, 2015; p. 186. [Google Scholar]
- Detto, M.; Wright, S.J.; Calderón, O.; Muller-Landau, H.C. Resource acquisition and reproductive strategies of tropical forest in response to the El Niño-Southern Oscillation. Nat. Commun. 2018, 9, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Turner, B.L.; Brenes-Arguedas, T.; Condit, R. Pervasive phosphorus limitation of tree species but not communities in tropical forests. Nature 2018, 555, 367–370. [Google Scholar] [CrossRef] [PubMed]
- Santos, F.M.; de Balieiro, F.C.; dos Santos Ataíde, D.H.; Diniz, A.R.; Chaer, G.M. Dynamics of aboveground biomass accumulation in monospecific and mixed-species plantations of Eucalyptus and Acacia on a Brazilian sandy soil. For. Ecol. Manag. 2016, 363, 86–97. [Google Scholar] [CrossRef]
| Species | Crown Phenology | a Nutrient Cycling | Root Morphology | b Water Use Efficiency | c Drought Strategy |
|---|---|---|---|---|---|
| Dalbergia retusa | Semi-deciduous | Nitrogen-fixer | Lateral roots | High | Endures & avoids |
| Terminalia amazonia | Evergreen | - | Lateral & deep roots | Low | Endures |
| T. amazonia | Mixture | D. retusa | ||||||
|---|---|---|---|---|---|---|---|---|
| Monoculture | T. amazonia | D. retusa | Monoculture | |||||
| Normal | Drought | Normal | Drought | Normal | Drought | Normal | Drought | |
| Sapwood area growth (mm2 day−1) | 4.12 (2.12) b | 3.87 (3.52) b | 6.78 (4.08) a | 7.12 (4.85) a | 1.09 (1.59) c | 0.19 (0.25) c,* | 2.20 (2.15) bc | 2.82 (2.99) bc |
| ET (L day−1) | 6.86 (3.94) a | 5.33 (3.42) a | 6.29 (3.66) a | 4.61 (3.56) a,* | 0.94 (0.50) b | 0.65 (0.29) b | 1.45 (1.22) b | 1.28 (1.09) b |
| WUE (mm2 L−1) | 0.90 (0.92) a | 1.05 (1.18) a | 1.26 (0.93) a | 1.88 (1.31) a,* | 1.32 (2.09) b | 0.23 (0.31) b | 1.78 (1.28) b | 2.85 (2.93) b |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sinacore, K.; Asbjornsen, H.; Hernandez-Santana, V.; Hall, J.S. Drought Differentially Affects Growth, Transpiration, and Water Use Efficiency of Mixed and Monospecific Planted Forests. Forests 2019, 10, 153. https://doi.org/10.3390/f10020153
Sinacore K, Asbjornsen H, Hernandez-Santana V, Hall JS. Drought Differentially Affects Growth, Transpiration, and Water Use Efficiency of Mixed and Monospecific Planted Forests. Forests. 2019; 10(2):153. https://doi.org/10.3390/f10020153
Chicago/Turabian StyleSinacore, Katherine, Heidi Asbjornsen, Virginia Hernandez-Santana, and Jefferson S. Hall. 2019. "Drought Differentially Affects Growth, Transpiration, and Water Use Efficiency of Mixed and Monospecific Planted Forests" Forests 10, no. 2: 153. https://doi.org/10.3390/f10020153
APA StyleSinacore, K., Asbjornsen, H., Hernandez-Santana, V., & Hall, J. S. (2019). Drought Differentially Affects Growth, Transpiration, and Water Use Efficiency of Mixed and Monospecific Planted Forests. Forests, 10(2), 153. https://doi.org/10.3390/f10020153






