Abstract
Garcinia kola Heckel (Clusiaceae), known as bitter kola, is a multipurpose tree indigenous to West and Central Africa. This highly preferred species is called “wonder plant” because all of its parts can be used as medicine. Its seeds, the most valued product of the tree, are commonly eaten to prevent/cure gastric disorders and for their typical astringent taste. There is a vast evidence that bioactive components of the seeds can serve as alternative medicine to treat/prevent severe illnesses such as malaria, hepatitis and immune-destructive diseases. Despite the species’ pharmaceutical potential and its high preference by West and Central African communities, G. kola is still at the beginning of its domestication process. Even though, there are numerous scientific articles published on species‘ biological activities, it is a difficult task to find basic information on its diversity, distribution, genetics, silvicultural management or botany. Therefore, in this very first review published on G. kola, we summarize all relevant information known about the species, target some of the challenges connected with its cultivation and propose a leading direction for future research and domestication process.
1. Introduction
From the outset of humankind, forests and especially trees have provided people with food and medicines. In 2011, the value of non-timber forest products (NTFPs) worldwide reached 88 billion USD [1]. However, traditional knowledge of these valuable tree species has been disappearing due to the pressures of modern lifestyle and effects of rampant deforestation [2]. Trees naturally produce large number of diverse bioactive compounds [3,4]. These plant-derived substances with minimal or no industrial processing started to get significant attention in global health debates. Modern medicine cannot be considered as a realistic treatment option for a substantial proportion of the world’s population, e.g., in Africa, 80 percent of population use some form of traditional herbal medicine [5].
Garcinia, a genus belonging to the diverse pantropical family Clusiaceae, consists of important fruit and medical tree species. Most of them remain in wild and semi-domesticated forms of regional importance but have been re-discovered as so-called neglected or underutilized crops [6,7]. Garcinia kola Heckel (Clusiaceae), commonly known as bitter kola, plays an important role in African ethnomedicine and traditional ceremonies. The trees are naturally found in humid tropical forests of West and Central Africa, where the local population usually harvest the fruits. However, in some regions, farmers plant and manage the trees in their homegardens or agroforests outside natural forests. Its seeds are amongst the most-traded NTFPs in West and Central Africa [4,5,6]. The species is sometimes referred to as a “wonder plant” because each of its parts can be used as medicine [8,9]. The most valued product are the seeds, commonly chewed by both rural and urban populations to avoid and treat gastric problems or simply for their typical astringent taste. The kernel contains a wide range of useful phytochemicals, e.g., high contents of tannins and flavonoids. Among these compounds, the biflavonoid kolaviron complex is the most discussed. This complex reputedly holds neuroprotective, anti-inflammatory, antimicrobial, and many other assets favorable to human health [6,7,8]. In addition, kolaviron possess anti-malarial and wound healing properties [10,11]. Therapeutic potential of kolaviron was shown in treatment of benign prostatic hyperplasia [12], neurodegenerative diseases such multiple sclerosis [13] and acquired immunodeficiency syndrome (AIDS) [14], whereas the seed extract was able to stop growth of Ebola virus in laboratory trials [15].
G. kola is listed as one of the priority species for conservation in the Sub-Saharan Forest Genetic Resources Programme (SAFORGEN) [16] and was selected as one of six preferred tree species by the World Agroforestry Centre (ICRAF) for domestication in West and Central Africa [17,18]. Mainly due to habitat loss, slow-growing seedlings, continuous felling, and overexploitation of the tree in West Africa, the species is still classified as “vulnerable” in IUCN’s Red List of Threatened Species [19].
Despite the importance, potential and popularity of G. kola in West and Central Africa, a great deal of information and basic knowledge is still missing about the species. The objective of this review was to collate all currently available information about this tree species from scientific literature. We tried to detect potential pitfalls and indicate where more investigation would be necessary. The study may also serve as a potential stepping stone for further research aimed at G. kola domestication.
2. Taxonomy
The Clusiaceae family consists mainly of woody perennials, trees, shrubs, and lianas divided into 18 genera. Among them Calophyllum, Clusia, or Garcinia are pantropically the most popular [20]. A typical morphological feature connecting all the family representatives is exudation of white-yellow colored latex from various plant parts [20,21].
Garcinia is a large genus consisting of more than 250 species of dioecious woody plants that are a common understory component of lowland tropical forests [6,22]. The genus was named after Laurent Garcin (1683–1757), a Swiss botanist within the Dutch Indies Company who published the first description of mangosteen (G. mangostana), the most popular fruit species from the Garcinia genus [7]. The genus can be divided into an Asian and African group, whereas some of the species were also introduced into South America. In the region of West and Central Africa, about 21 species of Garcinia can be found [23,24]. Among them, G. kola (bitter kola) seems to be one of the most studied species [25,26]. Additionally, there are at least two other species that are scientifically well described: G. livingstonei and G. lucida. The formerly mentioned is popular in drier parts of West to South Africa for its juicy fruit pulp and its roots that are used in traditional medicine [27,28]. G. lucida is recognized as a medicinal species of lowland forests in West and Central Africa [29,30]. Other species of the genus Garcinia include trees of local or minor importance that are mostly used as chewing sticks, e.g., G. afzelii, G. brevipediellata, G. epunctata, G. gnetoides, G. ovalifolia, G. smeathermannii, and G. staudtii [31,32,33,34].
3. Distribution and Ecology
Garcinia kola occurs naturally from Congo to Sierra Leone (Figure 1) [19,24,35]. Usually, Cameroon and Nigeria are considered as the major presence hotspots for this species. In Cameroon, Vivien and Faure [36] identified three natural stands of the species; two of them in the East region (Nki National Park, Bertoua site), and the last one situated in the Southwest region (Korup National Park).
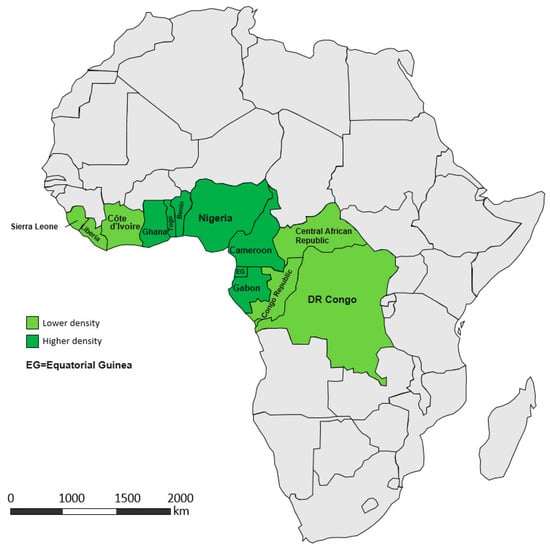
Figure 1.
Distribution of Garcinia kola among African countries. Dark green are areas with a higher abundance, light green marks a lower abundance. Source: authors’ drawing.
Although the species is reported to prevail in coastal areas and lowland plains up to 300 m a.s.l., the trees are successfully cultivated even in hilly areas about 750 m a.s.l. [24,31,37]. Typically, G. kola occurs in zones classified according to Köppen–Geiger as “tropical rainforest climate, tropical monsoon climate or tropical savannah climate” [38]. Daily temperatures usually vary between 21 °C to 31 °C, whereas the mean rainfall ranges from 1000 to 3000 mm per year in those areas. This is complemented by a relatively high air humidity of about 75% [39,40]. The species can withstand various types of soils with a slight preference for sandy loams, whereas its fine roots were found to harbor an arbuscular type of mycorrhiza [41].
4. Botanical Description
Garcinia kola is a medium-large tree naturally growing up to 30 m in height with a maximum of 100 cm in trunk diameter [21]. According to Anegbeh et al. [42], cultivated trees can reach 12 m in 12 years and usually grow below 20 m of height (Table 1). G. kola has a compact dense crown with erect, slightly drooping branches. The trunk is straight and cylindrical with smooth bark, which is dark-brown outside and pinkish inside. When wounded, the bark exudates sticky yellow water-proof latex, typical for the Clusiaceae family. Leaves are simple, opposite, obovate-elliptic with short acuminate apex. They are usually glabrous, dark green and can measure up to 20 × 6 cm (Figure 2). The inflorescence is a small terminal umbel with greenish/white flowers [32,43]. The tree is predominantly dioecious, but some flowers were reported to be bisexual. Flowering usually occurs once per year [21].

Table 1.
Comparison of selected morphological features of G. kola from three different countries; values are provided as Mean ± SD.

Figure 2.
Morphological features of Garcinia kola. (a) bark with a fresh cut; (b) seedlings; (c) leaf collection from one tree; (d) seeds obtained in Yaoundé market; (e) branches with ripening fruits; (f) trunk with typical irregular branching pattern. Source: author’s archive.
Fruits are berries of globular, sometimes slightly flattened shape with a diameter of approximately 6.5 cm and weight of about 130 g (Table 1). The exocarp is velvety, reddish-yellow and the pulp is yellow/orange releasing a slightly apricot odor. Even though the pulp is edible, its sour, resinous taste prevents it from being commonly consumed. In one season, a single full-grown tree can yield 200–1000 fruit [44]. One fruit contains about 2–4 seeds, which have a hypogeal type of germination [42,43]. The pericarp of the seed is light brown-colored when fresh but darkens with drying or age. The kernel is white with brownish-red branched lines producing red resinous globules [35]. Seed length and width are on average 3.0 × 1.5 cm, mean weight varies around 5.4 g. In West and Central Africa, the fruits are ready to be harvested from April to October [39,45], but the exact period varies within regions and climate zones. When fully ripe, the color of the fruit changes from green to reddish-yellow.
There is no official botanical descriptor yet developed for the species; however, a study by Dah-Nouvlesson et al. [46] from Benin resulted in a detection of five G. kola morphotypes. One of the options for morphological diversity evaluation is to use a descriptor developed for G. mangostana [47].
5. Use
Garcinia kola is a typical multipurpose agroforestry species regarded as one of the most important medicinal plants in Cameroon [18,32,49], Nigeria [2,12,50,51], Benin [43,46], Gabon [52], and Sierra Leone [53]. Its common name “bitter kola” reflects the typical taste of G. kola seeds, which are truly bitter/astringent. The less common expression “male kola” arose from its aphrodisiacal effect on men [54] whereas “false kola” suggests that some people consume the species’ seeds instead of cola nuts coming from Cola spp. [36,55]. G. kola offers a broad variety of products—fruits, seeds, bark, twigs, leaves, or wood can be utilized, but generally, the kernels are regarded as the most important product, whereas fruit pulp is usually discarded.
Both the seeds and bark are used in folklore remedies for treatment of gastric and liver disorders. The seeds are chewed to suppress headaches, laryngitis, bronchitis, malaria, and gonorrhea [56,57]. The seed extract is used as a cure for various types of inflammation or liver cirrhosis [35], while dried ground kernels can be mixed with honey to prepare a traditional paste against cough [44]. Pharmaceutical companies from Nigeria and Cameroon have recently started to focus on small-scale production of bitter kola syrups, eye drops, or herbal pastes. The kernels also play an important role in traditional ceremonies, e.g., celebration of a childbirth, marriage, or chieftaincy [42,58]. Offering the seeds is considered as an act of hospitality and used to welcome visitors. Finally, bitter kola nuts can be appreciated as a snack to be paired with beer or palm wine [45,59]. In the brewing industry, the seeds can successfully substitute hops [60]. Bark of G. kola is traditionally used in a more or less similar way to the seeds, mainly to cure abdominal pains and malaria [57]. Apart from its medicinal value, the bark serves in palm wine production. It is believed that the bark enhances flavor as well as alcohol content of the traditional beverage [35,61]. In countries such as Ghana, branches and roots of G. kola are sold in bundles as an essential source of chewing sticks used for dental hygiene [31]. Leaves of the species are occasionally prepared as an infusion to cure fever, and a good quality hard timber serves for tool handles or carving [62].
Due to the species’ dense crown, G. kola is also promoted and utilized as a shade tree in cocoa agroforestry systems or homegardens. Potentially, it might also be used as a windbreaker [42,54].
6. Biochemical Characterization
6.1. Nutritional Values
Even though G. kola is considered as a medicinal plant and most of the current research targets characterization of its bioactivity, the seeds are usually eaten raw, in their crude form. Therefore, it is also important to focus on their nutritional value.
Scientific literature provides quite confusing data on the species dietary properties. The published results concerning the seeds’ alimentary composition vary as follows: moisture: 7.2%–92.7%; ash: 0.33%–5.9%; crude protein: 0.58%–7.8%; crude fat: 0.19%–14.5%; crude fibre: 1.23%–20.51%; NFE: 10.85%–91.35% (Table 2). Overall, the studies agree on relatively high amounts of moisture in the seeds, around 70%, which is a crucial aspect for kernel preservation. Carbohydrates, also described as nitrogen-free extracts (NFE), form the largest part of seed proximate composition varying around 65%. On the other hand, ash content, the result of complete sample burning to inorganic substituents, is very low and in the range of only 1.5%. Mean value for crude protein is 3.5%, crude fat varies around 6.2% while crude fibre content is about 9.4%. Compared to the proximate content of cola nut (Cola spp.), also a popular masticatory stimulant in West and Central Africa, Arogba [63] revealed that bitter kola kernels contain twice the amount of protein but are twice as low in fat, whereas amounts of ash and NFE are mostly similar. More specifically, the dominant fatty acids in the seeds are represented by oleic (38 mg/kg), linoleic (36 mg/kg), and palmitic acid (32 mg/kg). The prevalent essential amino acids are lysine (2.4 g/kg), leucine (1.9 g/kg), and valine (1.7 g/kg), while glutamic acid (6.8 g/kg) and arginine (5.5 g/kg) are the predominant nonessential amino acids [64]. The seeds are low in anti-nutrients such as phytate or oxalate [65]. Regarding mineral and vitamin content of G. kola seeds, little information is available. However, relatively high amounts of vitamin C with 23.1 mg/100 g were recorded [50]. Onyekwelu et al. [65] reported an even higher value of 69 mg/100 g. To compare, other important fruit tree species indigenous to West and Central Africa showed a slightly lower values: safou (Dacryodes edulis)—24.5 mg/100 g; bush mango (Irvingia gabonensis)—55.9 mg/100 g [66]. Potassium and phosphorus are the most abundant minerals in the seeds, with values between 25–722 mg/kg for K and 3.3–720 mg/kg for P (Table 3).

Table 2.
Mean nutritional composition of G. kola seeds reported by various authors.

Table 3.
Mineral composition of G. kola seeds.
Seeds of G. kola are often peeled before consumption, and hulls are discarded as waste. Results from Eleyinmi et al. [64] proposed the feeding potential of seed coats for domestic animals due to their high protein content of 9.92 g/100 g. This is comparable to green parts of alfalfa (Medicago sativa) with a value ranging from 13.5 to 21.7 g/100 g depending on the plant maturity stage [67]. Livestock and small ruminants are generally lacking high-protein fodder in developing countries and bitter kola hulls might, according to in vitro and in vivo studies, provide a reasonable solution for this problem on a regional level.
6.2. Biological Activities and Secondary Metabolites
The most abundant phytochemicals in G. kola seeds are flavonoids. Presence of saponins, tannins, phenols, glycosides, and alkaloids has also been confirmed by various authors (Table 4). Even though anti-nutrients such as oxalate and phytate were detected, the seeds are safe for consumption and there are no reports on harmful overdosing so far [70]. Flavonoids, compounds of low molecular weight, are known as natural antioxidants, having an ability to scavenge free radicals and transform them into harmless molecules as well as to impact various aspects of immune cell activation for the human body. The compounds play a useful role in protecting the central nervous system against oxidative, excitotoxic stresses [14,57] and work as anti-tumor (benign, melanoma) agents [3]. One of the most studied and discussed components in G. kola seeds is the kolaviron biflavonoid complex (KV) (Figure 3). This complex further consists of biflavanones GB1, GB2, and kolaflavanone [70,71,72].

Table 4.
Phytochemical composition of G. kola seeds on a dry weight basis reported by various authors.
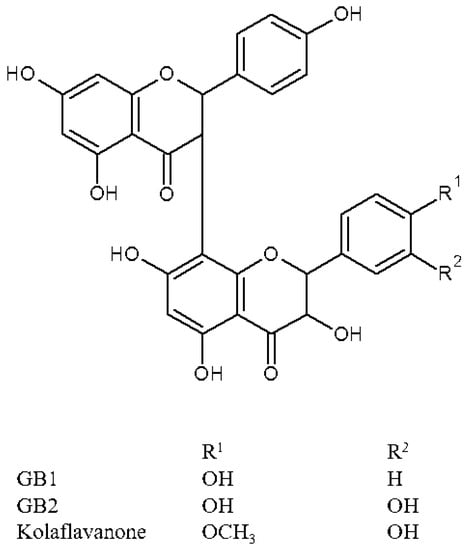
Figure 3.
Biflavonoid complex kolaviron and its components.
Kolaviron possesses antinociceptive (sedative) and anti-inflammatory activities, both centrally and peripherally, which justifies its folkloric use to relieve pain and inflammation [9,73]. The anti-inflammatory effect of KV and its components was observed in carrageenan-induced paw edema test [10,72]. Moreover, Abarikwu [74] revealed that KV can block signaling pathways implicated in lipopolysaccharide induced inflammatory gene expression in RAW 264.7 macrophage cell line. In another experiment, KV extended the lifespan of the common fruit fly (Drosophila melanogaster) by preventing oxidative stress and inflammation in the species [75]. The KV extract significantly decreased locomotion, grooming, and rearing frequencies of male Swiss mice indicating a central depressant effect of the complex. Recent findings also show that KV could prevent neuro-destructive effects of methamphetamine on hippocampal neurons, affording some protection to the hippocampus too [57]. Due to its abilities to combat oxidative and inflammatory damage induced by cuprizone, KV showed therapeutic potential against degenerative changes associated with demyelination and neurotoxicity. This finding might be later used in treatment for a multiple sclerosis [13]. Additionally, KV can be a clinically viable agent against ischemia/reperfusion injuries [76,77].
One of the previous tests of KV hepatoprotective properties demonstrated that the compound prevents liver injuries associated with tetrachloromethane [78,79] and α-amanitin and phalloidin [78] intoxication. Another study demonstrated that KV treatment (100 mg/kg) of diabetic rats might protect them against hyperglycemia-induced apoptosis, attenuate the level of lipid peroxidation, and promote survival of hepatocytes, perhaps by scavenging free radicals [80]. Alabi et al. [81] confirmed the hepatoprotective effect of KV against diclofenac-induced toxicity at low and moderate doses (100–200 mg/kg), which is comparable to commercial hepatoprotective drug (Livolin Forte) used in the treatment of liver diseases. Authors speculated that only high doses of KV (400 mg/kg) can cause liver damage. Farombi et al. [82] suggested the ability of KV to inhibit cyclooxygenase (COX-2) and inducible nitric oxide synthase (iNOS) expression through down regulation of nuclear factor kappa B (NF-κB) and activator protein-1 (AP-1) DNA binding activities could be the mechanism explaining the hepatoprotective properties of KV. These findings indicated that KV may have a protective effect against carcinogen and drug-induced oxidative and membrane damages as well as prevent any accumulation of lipid peroxidation products [14].
Apart from antioxidative, anti-inflammatory and hepatoprotective activities, KV shows high anti-malarial activities by suppressing Plasmodium berghei in infected mice [11,55]. Out of KV components, GB1 exhibited the strongest in vitro antimalarial effectivity on P. falciparum with an IC50 of 0.16 μM. In the in vivo test they confirmed that GB1 significantly increased the average life span of Plasmodium-infected mice [70]. Nworu et al. [14] discovered another promising property of KV in its immunomodulatory and immuno-restorative effects. In the future, KV complex could be harnessed for possible clinical benefits to patients fighting immune-destructive diseases such as acquired immunodeficiency syndrome (AIDS). Furthermore, KV showed potential in benign prostatic hyperplasia treatment by attenuating the infected prostatic tissue in rats, acting similarly to the regular treatment by Finasteride medication [12].
Antimicrobial properties of G. kola are attributed to benzophenones and flavanones [42,49]. These active components have been already successfully extracted in petroleum ether, ethanol, methanol and water. In a study by Indabawa & Arzai [83], methanol and water extract showed activity against Staphylococcus aureus, Klebsiella pneumoniae and Salmonella typhi. According to results of Adegboye et al. [56], the methanolic crude extract exhibited significant inhibitory action against eleven out of fifteen bacterial isolates (Bacillus, Clostridium, Corynebacterium, Escherichia, Klebsiella, Micrococcus, Pseudomonas, Staphylococcus) tested at a final concentration of 20 mg/ml. The inhibition zones exhibited by the extract against the test organisms ranged between 10 and 23 mm, similar to the zones of streptomycin and tetracycline. Poly-iso-phenyl benzophenone, called kolanone, showed great antimicrobial effect against both gram-positive and gram-negative bacteria. Its results were comparable to salicylic acid (aspirin) [84]. Interestingly, crude extract of Phomopsis sp., endophytic fungi associated with G. kola seeds, provided three cytochalasin compounds having a potential of clinically useful alternative for the treatment of cervical cancer and severe infections caused by multidrug-resistant Shigella flexneri (MIC 128 μg/mL) and Vibrio cholerae (MIC 512 μg/mL) [85]. Hydroethanolic and ethanolic extracts of G. kola leaves showed an inhibitory effect against Trypanosoma brucei brucei, a parasite causing trypanosomiasis in cattle and other domestic animals by infecting their blood plasma [86].
The typical astringent taste of G. kola kernels is caused by tannins, secondary metabolites known for their natural treatment of intestinal disorders such as diarrhea and dysentery. Apart from their microbial properties, tannins have also been reported to have a remarkable potential in cancer prevention [8,56] and together with phlobatannins they exhibit wound healing properties [71]. Finally, both cardiac glycosides and steroidal compounds were found in G. kola extracts. This coincides with the fact that the plant is traditionally used to combat chest pain or cardiac infection, while men are commonly chewing the seeds as an aphrodisiac [8,54].
Even though seeds of G. kola are frequently sold side by side with cola nuts (Cola spp.), their chemical composition is rather different. Unlike cola nuts, bitter kola seeds contain higher levels of phenolic compounds, whereas caffeine, theobromine, and catechin were not detected [87].
7. Tree Management and Cultivation
Garcinia kola seeds are still, at least partly, harvested from wild stands; therefore, the information on the tree propagation, cultivation, and sylvicultural management are relatively scarce. According to Anegbeh et al. [42] and Matig et al. [32], about 70% of bitter kola fruits in Nigeria are directly taken from wild stands in forests. Contrary to this, the tree is said to be frequently cultivated by local farmers and is rarely found scattered in the natural forest in Cameroon [23]. The tree is sometimes intentionally preserved during forest clearing and thus introduced to farmers’ compounds [32]. It is often grown in agroforestry systems together with cocoa, oil palm, and other fruit trees [44]. However, the natural regeneration of the species is said to be poor, and seedlings are slow-growing [89]. Esiegwu et al. [69] reported that seeds should be sown in a seed bed of about 3 × 4 m (12 m2) with a protection from direct sunlight and strong rains. Due to dormancy, it can take up to 18 months for the seeds to successfully germinate [24,42]. The germinated seeds are then replanted into polyethylene bags filled up to 3/4 with a mixture of black soil and sand. After 12 months, seedlings are transferred to the field, usually at the beginning of the rainy season, with a spacing of 10 × 10 m. Usually it takes about 7–15 years for the tree to start fruiting [2,44]. The time needed for tree maturation can be significantly reduced by vegetative propagation, though the techniques have not yet been fully developed or practiced in the case of G. kola. Yet, Kouakou et al. [90] discovered that the species responds well to propagation by stem cuttings. According to their results, IBA (indol-3-butyric acid) treatment promotes shoot and root production and accelerates the emergence of shoots and leaves. The best results were achieved by the cultivation of softwood cuttings with an aqueous application of IBA in a non-mist poly-propagator. Nevertheless, further research is needed to determine the best planting conditions and optimize the process of vegetative propagation.
7.1. Seed Dormancy and Germination
The major difficulty in G. kola cultivation, as for several species in genus Garcinia, is related to seed germination and embryo dormancy [91]. Literature provides contradictory information concerning the seed germination. Some publications describe the seeds as easily germinating [32], but most authors confirm that the seeds are difficult to germinate [42,60,89,91]. In that context, it is rather tricky to prescribe a standard procedure for enhancing the germination. Seeds of G. kola are recalcitrant, hence very sensitive to desiccation which may influence their viability. Matig et al. [43] revealed that the species’ germination rate decreases with lowering seed moisture content. Therefore, the authors suggested dormancy-breaking through seed coat removal and soaking in cold water. Nzegbule and Mbakwe [60] proposed another method—pre-treatment of freshly harvested seeds with cold water followed by incubation in a transparent polyethylene bag. Anegbeh et al. [42] suggested mechanical seeds scarification (nicking) before sowing as the most successful, cheapest and easiest way to enhance the germination. Kanmegne and Omokolo [91] tried to break embryo dormancy by pre-treatment of seeds with auxins, cytokinins, and gibberelins, but none of the phytohormones significantly increased germination rate, nor did they reduce the dormancy period. On the other hand, the authors revealed a regeneration potential for in vitro cultures. A treatment by NAA, BAP, and 2,4-D induced the formation of multiple roots, shoots, and callus. Also, their results showed a significant difference in seed germination rate among six studied collections, indicating that the trait may vary with accession of the species.
7.2. Harvesting and Seed Storage
Ripe fruits are usually collected from beneath the tree or harvested manually using a pole to drop down the fruits from the tree at various stages of maturation (Figure 4) [32]. Seeds are firmly attached to the fruit pulp, which makes their removal a long-termed laborious procedure. Therefore, farmers usually keep the harvested fruits in piles for 5–7 days so that the pericarp and pulpy mesocarp ferment and become soft. Once softened, fruits are pressed to release the kernels, which are then thoroughly washed and dried (Figure 5). Kernels are eaten fresh or stored for later consumption and commercialization [62]. However, seeds easily lose moisture and shrink, which negatively influences their germination rate and the market value. The quick water loss leads to a change in texture as well as in sensory and nutritional attributes of the seeds [45]. One of the most popular and easiest ways to store the seeds is to air-dry them first and to continue to store in a cool and dry place [44]. Another possibility is to wrap the nuts in leaves and store them in a wicker basket [62]. Some farmers also store the kernels in between layers of soil or in dust/ash piles [45]. In optimal conditions, seeds can be stored for about a year [37]. Apart from the fruits, bark is frequently harvested for palm wine fermentation and young branches serve for dental hygiene. Harvesting of these products is usually done in an unsustainable way that severely damages the tree. This contributes to the increasing scarcity of the species and its overall IUCN rating as “vulnerable” [19,23].

Figure 4.
Ripening stages of G. kola fruits, from immature (green) to overripe (brownish). Source: author’s archive.

Figure 5.
Softened fruit pulp for easier seeds removal. Source: author’s archive.
8. Economic Potential
Commercialization of G. kola seeds is considered as a profitable activity providing a substantial contribution to the livelihood of households, particularly those living in rural areas [62,92,93]. Nevertheless, primary producers very rarely sell bitter kola products directly to final consumers and thus different steps of value chain can be recognized. These farmers or gatherers commonly sell the seeds in bags with a weight ranging from 5 to 25 kg to retailers. Collectors sell one kg of seeds for one dollar, while the consumer final price could be almost 15 USD per kg. Price varies and fluctuates both within regions and different periods of year [46,62]. G. kola products are sold in small rural markets [94] but also in larger markets from urban areas [2,53].
In Cameroon, the seeds represent one of the most valuable NTFP with total sold volume estimated up to 50 tons annually, which represents 375 million CFA (660,000 USD) [95]. These values indicate the promising market potential of the seeds as well as unequal distribution in the value chain. In big cities such as Yaoundé or Douala, one individual seed can be sold for 50 to 100 CFA (0.09–0.17 USD) depending on season and size of the kernel (personal observation). Seeds are predominantly sold by female vendors [94] or by adolescent boys, street vendors, who sell the nuts mainly to taxi drivers at junctions of bigger cities [96].
Besides the national markets, G. kola seeds represent a prospective commodity for international trade [92]. Exports are very often realized among neighboring countries, where such supplies may compete with kernels harvested from local agro-ecosystems. As a result, seeds sold in the Benin markets may originate from Nigeria, Togo, or Ghana. The import of bitter kola seeds from Nigeria to Cameroon is also commercially important [46,53,96]. Exports outside Africa, for example to Europe or North America, have not yet been fully documented [92].
9. Domestication Status
Tree domestication is a farmer-driven and market-oriented process applied to the selection, multiplication and management of high-value but lesser-known tree species of tropical forest [97,98]. It is one of the processes that may lead towards greater sustainability and creation of more functional agro-ecosystems in agriculture. Its benefits include income diversification, improvement of local diets and health, or in saving species which are under threat of extinction from the wild due to deforestation and/or over-harvesting [99,100,101]. The process of tree domestication consists of many steps, e.g., species priority setting, selection of desired traits, superior trees selection, integration of trees into farmlands, vegetative propagation, targeted plant breeding and product commercialization [18,97,98,102,103].
According Clement et al. [102], G. kola can be categorized as incipiently domesticated. Its process of domestication is still in its early stages, yet there are obvious efforts in species cultivation and selection of the best individuals. In some West/Central African regions, people seem to already plant and manage G. kola purposefully [37,46,48], while for others fruit harvesting from the wild still prevails [2,32]. The example of the Southwest region of Cameroon clearly shows that trees are not only left on the field after forest clearance, but the farmers are also purposely planting G. kola in their gardens as valuable fruit tree species [37]. In Benin, study of Dadjo et al. [48] highlighted importance of different land management practices consideration while selecting the elite trees, land use type (home gardens, farmland) has a large impact on variation of different morphological traits.
One of the major tools to speed up the domestication process is development of methods of vegetative propagation, which shortens tree maturation period and provides an exact copy of the mother’s genotype. This is very important for farmers who demand quick results for their investment in time, money and effort [17,104]. However, these methods have not yet been fully discovered/developed by the G. kola growers and this fact significantly slows down the domestication process.
Apart from the development of vegetative propagation methods, the process of G. kola domestication cannot succeed unless solid information on species morphological, genetic and phytochemical characteristics is revealed.
10. Conclusions
Our literature review revealed that the most common and discussed topic in G. kola concerns its biological activities and secondary metabolites. This justifies the utilization of the species in various traditional phyto-remedies and shows its great potential for the worldwide pharmaceutical industry. Especially the biflavonoid kolaviron complex proved to be quite promising for future research. On the other hand, lack of data was discovered in other scientific disciplines such as genetics or silviculture management—even basic information about the tree cultivation, its ecological requirements, and methods of propagation has not yet been well documented.
To progress G. kola domestication, many knowledge gaps need to be filled. Molecular markers should be developed to evaluate the genetic diversity of different species’ populations. Morphological descriptors are still missing for the tree’s botanical characterization, and data on phytochemical composition are not ample. The differences in primary and secondary metabolite quantification published up to now shows too substantial variation. Also, long-term studies should be conducted to reveal fluctuations among secondary metabolites not only in seeds, but also in the bark of the species. With this information available, we may be able to identify and select populations possessing the pre-selected desirable traits. But what are those traits? To answer this question, we must learn more about the preferences of local people who use the species on a daily basis. Once these attributes are detected, selection of the superior tree populations may start. After the selection, the next step is to multiply the desired individuals. Therefore, field trials focusing on methods of vegetative propagation and the propagules regeneration need to be established. Finally, the biological information should be complemented by detailed economic studies focused on market/value chain of the seeds among the West and Central African markets with a prediction for future intercontinental export potential.
To summarize, G. kola is starting to be recognized as a highly valued medicinal plant, not only with importance for local populations in West and Central Africa, but also as a potential source of pharmaceuticals in developed countries. Despite this great interest in the species, a considerable amount of work must still be conducted in the field of science to fulfill the information gaps described in our review.
Funding
This research was funded by Internal Grant Agency of the Czech University of Life Sciences, Prague, grant number 20185020 and 20185004; Nadace Nadání Josefa, Marie a Zdeňky Hlávkových, and Student Mobility of the Faculty of Tropical AgriSciences, CULS Prague.
Acknowledgments
Special acknowledgment to World Agroforestry Centre West and Central Africa (WCA) regional office Yaoundé, Cameroon and Tchana family.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Sorrenti, S. Non-Wood Forest Products in International Statistical Systems; Non-Wood Forest Products Series No. 22; Food and Agriculture Organization of the United Nations: Rome, Italy, 2017. [Google Scholar]
- Onyekwelu, J.C.; Olusola, J.A.; Stimm, B.; Mosandl, R.; Agbelade, A.D. Farm-level tree growth characteristics, fruit phenotypic variation and market potential assessment of three socio-economically important forest fruit tree species. For. Trees Livelihoods 2015, 24, 27–42. [Google Scholar] [CrossRef]
- Altemimi, A.; Lakhssassi, N.; Baharlouei, A.; Watson, D.; Lightfoot, D. Phytochemicals: Extraction, isolation, and identification of bioactive compounds from plant extracts. Plants 2017, 6, 42. [Google Scholar] [CrossRef] [PubMed]
- Dranca, F.; Oroian, M. Optimization of ultrasound-assisted extraction of total monomeric anthocyanin (TMA) and total phenolic content (TPC) from eggplant (Solanum melongena L.) peel. Ultrason. Sonochem. 2016, 31, 637–646. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X. Traditional medicine: Its importance and protection. In Protecting and Promoting Traditional Knowledge: Systems, National Experiences and International Dimensions; Twarog, S., Kapoor, P., Eds.; United Nations: Geneva, Switzerland, 2004; p. 420. [Google Scholar]
- Stevens, P.F. Angiosperm Phylogeny Website. Available online: http://www.mobot.org/MOBOT/research/APweb/ (accessed on 9 December 2018).
- Bin Osman, M.; Milan, R. Mangosteen—Garcinia Mangostana; Southampton Centre for Underutilised Crops, University of Southampton: Southampton, UK, 2006; ISBN 0-85432-817-3. [Google Scholar]
- Usunomena, U. Review manuscript: A review of some African medicinal plants. Int. J. Pharma Bio Sci. 2012, 3, 1–11. [Google Scholar]
- Onasanwo, S.A.; Rotu, R.A. Antinociceptive and anti-inflammatory potentials of kolaviron: Mechanisms of action. J. Basic Clin. Physiol. Pharmacol. 2016, 27, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Nwaehujor, C.O.; Udegbunam, R.I.; Ode, J.O.; Udegbunam, S.O. Analgesic anti-inflammatory anti-pyretic activities of Garcinia hydroxybiflavanonol (GB1) from Garcinia kola. J. Korean Soc. Appl. Biol. Chem. 2015, 58, 91–96. [Google Scholar] [CrossRef]
- Tshibangu, P.T.; Kapepula, P.M.; Kapinga, M.J.K.; Lupona, H.K.; Ngombe, N.K.; Kalenda, D.T.; Jansen, O.; Wauters, J.N.; Tits, M.; Angenot, L.; et al. Fingerprinting and validation of a LC-DAD method for the analysis of biflavanones in Garcinia kola -based antimalarial improved traditional medicines. J. Pharm. Biomed. Anal. 2016, 128, 382–390. [Google Scholar] [CrossRef]
- Kalu, W.O.; Okafor, P.N.; Ijeh, I.I.; Eleazu, C. Effect of kolaviron, a biflavanoid complex from Garcinia kola on some biochemical parameters in experimentally induced benign prostatic hyperplasic rats. Biomed. Pharmacother. 2016, 83, 1436–1443. [Google Scholar] [CrossRef]
- Omotoso, G.O.; Ukwubile, I.I.; Arietarhire, L.; Sulaimon, F.; Gbadamosi, I.T. Kolaviron protects the brain in cuprizone-induced model of experimental multiple sclerosis via enhancement of intrinsic antioxidant mechanisms: Possible therapeutic applications? Pathophysiology 2018, 25, 299–306. [Google Scholar] [CrossRef]
- Nworu, C.S.; Akah, P.A.; Esimone, C.O.; Okoli, C.O.; Okoye, F.B.C. Immunomodulatory Activities of Kolaviron, a Mixture of Three Related Biflavonoids of Garcinia kola Heckel. Immunopharmacol. Immunotoxicol. 2008, 30, 317–332. [Google Scholar] [CrossRef]
- Iwu, M.M. Handbook of African Medicinal Plants; CRC Press: Boca Raton, FL, USA, 1993. [Google Scholar]
- Sacandé, M.; Pritchard, H.W. Seed research network on African trees for conservation and sustainable use. For. Genet. Resour. 2004, 31, 31–35. [Google Scholar]
- Tchoundjeu, Z.; Asaah, E.K.; Anegbeh, P.; Degrande, A.; Mbile, P.; Facheux, C.; Tsobeng, A.; Atangana, A.R.; Ngo-Mpeck, M.L.; Simons, A.J. Putting participatory domestication into practice in West And Central Africa. For. Trees Livelihoods 2006, 16, 53–69. [Google Scholar] [CrossRef]
- Franzel, S.; Kindt, R. Species priority setting procedures. In Agroforestry Tree Domestication: A Primer; Dawson, I., Harwood, C., Jamnadass, R., Beniest, J., Eds.; World Agroforestry Centre: Nairobi, Kenya, 2012; pp. 36–45. [Google Scholar]
- Cheek, M. Garcinia kola; The IUCN Red List of Threatened Species; IUCN: Gland, Switzerland, 2004; e.T34715A9884648. [Google Scholar]
- Gustafsson, M.G.H. Neotropical Clusiaceae—Neotropikey from Kew. Available online: http://www.kew.org/science/tropamerica/neotropikey/families/Clusiaceae.htm (accessed on 9 December 2018).
- Garcinia kola. Available online: https://www.prota4u.org/database/protav8.asp?h=M4&t=Garcinia,kola&p=Garcinia+kola#Synonyms (accessed on 9 December 2018).
- Guedje, N.M.; Nkongmeneck, B.-A.; Lejoly, J. Composition floristique et structure des formations à Garcinia lucida dans la région de Bipindi-Akom II (Sud-Cameroun). Acta Bot. Gallica 2002, 149, 157–178. [Google Scholar] [CrossRef]
- Guedje, N.M.; Fankap, R. Traditional utilisation of Garcinia lucida and Garcinia kola (Clusiaceae) in Cameroon. Syst. Geogr. Plants 2001, 71, 747–758. [Google Scholar] [CrossRef]
- Agyili, J.; Sacande, M.; Koffi, E.; Peprah, T. Improving the collection and germination of West African Garcinia kola Heckel seeds. New For. 2007, 34, 269–279. [Google Scholar] [CrossRef]
- Daramola, B.; Adegoke, G.O. Bitter Kola (Garcinia kola) seeds and health management potential. In Nuts and Seeds in Health and Disease Prevention; Elsevier: Amsterdam, The Netherlands, 2011; pp. 213–220. [Google Scholar]
- Magadula, J.; Mbwambo, Z. Garcinia Plant Species of African Origin, Ethnobotanical, Pharmacological and Phytochemical Studies; Open Science Publishers: New York, NY, USA, 2014. [Google Scholar]
- Mulholland, D.A.; Mwangi, E.M.; Dlova, N.C.; Plant, N.; Crouch, N.R.; Coombes, P.H. Non-toxic melanin production inhibitors from Garcinia livingstonei (Clusiaceae). J. Ethnopharmacol. 2013, 149, 570–575. [Google Scholar] [CrossRef] [PubMed]
- Joseph, K.S.; Bolla, S.; Joshi, K.; Bhat, M.; Naik, K.; Patil, S.; Bendre, S.; Gangappa, B.; Haibatti, V.; Payamalle, S.; et al. Determination of Chemical Composition and Nutritive Value with Fatty Acid Compositions of African Mangosteen (Garcinia Livingstonei). Erwerbs-Obstbau 2017, 59, 195–202. [Google Scholar] [CrossRef]
- Momo, I.J.; Kuete, V.; Dufat, H.; Michel, S.; Wandji, J. Antimicrobial activity of the methanolic extract and compounds from the stembark of Garcinia lucida vesque (Clusiaceae). Int. J. Pharm. Pharm. Sci. 2011, 3, 215–217. [Google Scholar]
- Sylvie, D.D.; Anatole, P.C.; Cabral, B.P.; Veronique, P.B. Comparison of in vitro antioxidant properties of extracts from three plants used for medical purpose in Cameroon: Acalypha racemosa, Garcinia lucida and Hymenocardia lyrata. Asian Pac. J. Trop. Biomed. 2014, 4, S625–S632. [Google Scholar] [CrossRef]
- Blay, D. Dental hygiene and livelihoods: A case of chewing sticks in Ghana. For. Prod. Livelihoods Conserv. Case Stud. Non-Timber For. Prod. Syst. 2004, 2, 25–36. [Google Scholar]
- Matig, O.E.; Ndoye, O.; Kengue, J.; Awono, A. Les Fruitiers Forestiers Comestibles du Cameroun; Bioversity International: Rome, Italy, 2006. [Google Scholar]
- Waffo, A.F.K.; Mulholland, D.; Wansi, J.D.; Mbaze, L.M.; Powo, R.; Mpondo, T.N.; Fomum, Z.T.; König, W.; Nkengfack, A.E. Afzeliixanthones A and B, Two New Prenylated Xanthones from Garcinia afzelii E NGL.(Guttiferae). Chem. Pharm. Bull. 2006, 54, 448–451. [Google Scholar] [CrossRef]
- Constant Anatole, P.; Guru, S.K.; Bathelemy, N.; Jeanne, N.; Bhushan, S.; Murayama, T.; Saxena, A.K. Ethyl acetate fraction of Garcina epunctata induces apoptosis in human promyelocytic cells (HL-60) through the ROS generation and G0/G1 cell cycle arrest: A bioassay-guided approach. Environ. Toxicol. Pharmacol. 2013, 36, 865–874. [Google Scholar] [CrossRef] [PubMed]
- Onayade, O.A.; Looman, A.M.G.; Scheffer, J.J.; Gbile, Z.O. Lavender lactone and other volatile constituents of the oleoresin from seeds of Garcinia kola Heckel. Flavour Fragr. J. 1998, 13, 409–412. [Google Scholar] [CrossRef]
- Vivien, J.; Faure, J.-J. Arbres des Forêts Denses d’Afrique Central; Ministére des relations extérieures, cooperation et développement, Agence de cooperation culturelle et technique: Paris, France, 1985.
- Manourova, A. Diversity and Nutritional Characterization of Garcinia kola Heckel in Southwest Cameroon. Master’s Thesis, Czech University of Life Sciences Prague, Prague, Czech Republic, 2017. [Google Scholar]
- Kottek, M.; Grieser, J.; Beck, C.; Rudolf, B.; Rubel, F. World Map of the Köppen-Geiger climate classification updated. Meteorol. Z. 2006, 15, 259–263. [Google Scholar] [CrossRef]
- Babalola, F.D.; Agbeja, B.O. Marketing and distribution of Garcinia kola (Bitter kola) in southwest Nigeria: Opportunity for development of a biological product. Egypt. J. Biol. 2010, 12, 12–17. [Google Scholar]
- Climate Data for Cities Worldwide—Climate-Data.org. Available online: https://en.climate-data.org/ (accessed on 9 December 2018).
- Bechem, E.E.T.; Chuyong, G.B.; Fon, B.T. A Survey of Mycorrhizal Colonization in the 50-ha Korup Forest Dynamic Plot in Cameroon. Am. J. Plant Sci. 2014, 5, 1403–1415. [Google Scholar] [CrossRef]
- Anegbeh, P.O.; Iruka, C.; Nikirika, C. Enhancing germination of bitter cola (Garcinia kola) Heckel: Prospects for agroforesty farmers in the Niger Delta. Sci. Afr. 2006, 5, 38–44. [Google Scholar]
- Eyog-Matig, O.; Aoudji, A.K.N.; Linsoussi, C. Garcinia kola Heckel seeds dormancy-breaking. Appl. Ecol. Environ. Res. 2007, 5, 63–71. [Google Scholar] [CrossRef]
- Adebisi, A.A. Bitter cola: The African wonder nut. In Riches of the Forest: For Health, Life and Spirit in Africa; Binnqüist, C.L., Ed.; CIFOR: Bogor, Indonesia, 2004; pp. 18–20. [Google Scholar]
- Dosunmu, M.I.; Johnson, E.C. Chemical evaluation of the nutritive value and changes in ascorbic acid content during storage of the fruit of “bitter kola” (Garcinia kola). Food Chem. 1995, 54, 67–71. [Google Scholar] [CrossRef]
- Dah-Nouvlessounon, D.; Adoukonou-Sagbadja, H.; Nafan, D.; Adjanohoun, A.; Noumavo, P.A.; Sina, H.; Daouda, B.O.; Baba-Moussa, L. Morpho-agronomic variability of three kola trees accessions [Cola nitida (Vent.) Schott et Endl., Cola acuminata (P. Beauv.) Schott et Endl., and Garcinia kola Heckel] from Southern Benin. Genet. Resour. Crop Evol. 2016, 63, 561–579. [Google Scholar] [CrossRef]
- International Plant Genetic Resources Institute (IPGRI). Descriptors for Mangosteen (Garcinia mangostana); IPGRI: Rome, Italy, 2003; ISBN 92-9043-587-9. [Google Scholar]
- Dadjo, C.; Toyi, M.; Nyende, A.B.; Salako, K.V.; Assogbadjo, A.E. Impact of land-use on tree and fruit morphometric variation of the bitter kola (Garcinia kola Heckel) in Benin: Insight for domestication and production. J. Hortic. For. 2018, 10, 127–134. [Google Scholar] [CrossRef]
- Kanmegne, G.; Mbouobda, H.D.; Temfack, B.; Koffi, E.K.; Omokolo, D.N. Impact of biochemical and morphological variations on germination traits in Garcinia kola Heckel seeds collected from Cameroon. Res. J. Seed Sci. 2010, 3, 82–92. [Google Scholar] [CrossRef]
- Okwu, D.E. Phytochemicals, vitamins and mineral contents of two Nigerian medicinal plants. Int. J. Mol. Med. Adv. Sci. 2005, 1, 375–381. [Google Scholar]
- Adesuyi, A.O.; Elumm, I.K.; Adaramola, F.B.; Nwokocha, A.G.M. Nutritional and phytochemical screening of Garcinia kola. Adv. J. Food Sci. Technol. 2012, 4, 9–14. [Google Scholar]
- Towns, A.M. Fertility and Fontanels: Women’s Knowledge of Medicinal Plants for Reproductive Health and Childcare in Western Africa; Naturalis Biodiversity Center, Sector Botany, Faculty of Science, Leiden University: Leiden, The Netherlands, 2014. [Google Scholar]
- Jusu, A.; Sanchez, A.C. Medicinal Plant Trade in Sierra Leone: Threats and Opportunities for Conservation. Econ. Bot. 2014, 68, 16–29. [Google Scholar] [CrossRef]
- Fondoun, J.M.; Manga, T.T. Farmers indigenous practices for conserving Garcinia kola and Gnetum africanum in southern Cameroon. Agrofor. Syst. 2000, 48, 289–302. [Google Scholar] [CrossRef]
- Oluwatosin, A.; Tolulope, A.; Ayokulehin, K.; Patricia, O.; Aderemi, K.; Catherine, F.; Olusegun, A. Antimalarial potential of kolaviron, a biflavonoid from Garcinia kola seeds, against Plasmodium berghei infection in Swiss albino mice. Asian Pac. J. Trop. Med. 2014, 7, 97–104. [Google Scholar] [CrossRef]
- Adegboye, M.F.; Akinpelu, D.A.; Okoh, A.I. The bioactive and phytochemical properties of Garcinia kola (Heckel) seed extract on some pathogens. Afr. J. Biotechnol. 2008, 7, 3934–3938. [Google Scholar]
- Ijomone, O.M.; Nwoha, P.U.; Olaibi, O.K.; Obi, A.U.; Alese, M.O. Neuroprotective Effects of Kolaviron, a Biflavonoid Complex of Garcinia kola, on Rats Hippocampus against Methamphetamine-Induced Neurotoxicity. Maced. J. Med. Sci. 2012, 5, 10–16. [Google Scholar] [CrossRef]
- Eyebe, A.; Simeon, A.E.; Angu, K.A.; Endamana, D. Integrating Biodiversity Conservation into National Development Policy: A Case study of Cameroon; IIED Poverty Conservation Learning Group: London, UK, 2012. [Google Scholar]
- Leakey, R.R.B. Potential for novel food products from agroforestry trees: A review. Food Chem. 1999, 66, 1–14. [Google Scholar] [CrossRef]
- Nzegbule, E.; Mbakwe, R. Effect of pre-sowing and incubation treatment on germination of Garcinia kola (Heckel) seeds. Fruits 2001, 56, 437–442. [Google Scholar] [CrossRef]
- Leakey, R.R. Living with the Trees of Life: Towards the Transformation of Tropical Agriculture; CABI: Wallingford, UK, 2012. [Google Scholar]
- Adebisi, A.A. A Case study of Garcinia kola nut production-to-consumption system in J4 area of Omo forest reserve, South-west Nigeria. For. Prod. Livelihoods Conserv. Case Stud. Non-Timber For. Prod. Syst. 2004, 2, 115–132. [Google Scholar] [CrossRef]
- Arogba, S.S. Comparative Analyses of the Moisture Isotherms, Proximate Compositions, Physical and Functional Properties of Dried Cola nitida and Garcinia kola kernels. J. Food Compos. Anal. 2000, 13, 139–148. [Google Scholar] [CrossRef]
- Eleyinmi, A.F.; Bressler, D.C.; Amoo, I.A.; Sporns, P.; Oshodi, A.A. Chemical composition of bitter cola (Garcinia kola) seed and hulls. Pol. J. Food Nutr. Sci. 2006, 15, 395. [Google Scholar]
- Onyekwelu, J.C.; Oyewale, O.; Stimm, B.; Mosandl, R. Antioxidant, nutritional and anti-nutritional composition of Garcinia kola and Chrysophyllum albidum from rainforest ecosystem of Ondo State, Nigeria. J. For. Res. 2015, 26, 417–424. [Google Scholar] [CrossRef]
- Stadlmayr, B.; Charrondière, U.R.; Eisenwagen, S.; Jamnadass, R.; Kehlenbeck, K. Nutrient composition of selected indigenous fruits from sub-Saharan Africa: Nutrient composition of selected indigenous fruits from sub-Saharan Africa. J. Sci. Food Agric. 2013, 93, 2627–2636. [Google Scholar] [CrossRef]
- Shewmaker, G.E.; Undersander, D.; Lawrence, L.M.; Lacefield, G.D. Alfalfa: The High-Quality Hay for Horses; Bulletin, National Alfalfa Alliance: Kennewick, WA, USA, 2005; p. 12. [Google Scholar]
- Odebunmi, E.O.; Oluwaniyi, O.O.; Awolola, G.V.; Adediji, O.D. Proximate and nutritional composition of kola nut (Cola nitida), bitter cola (Garcinia cola) and alligator pepper (Afromomum melegueta). Afr. J. Biotechnol. 2009, 8. [Google Scholar] [CrossRef]
- Esiegwu, A.C.; Okoli, I.C.; Emenalom, O.O.; Esonu, B.O.; Udedibie, A.B.I. The emerging nutricitical benefits of the african wonder nut (Garcinia kola Heckel): A review. Glob. J. Anim. Sci. Res. 2014, 2, 170–183. [Google Scholar]
- Konziase, B. Protective activity of biflavanones from Garcinia kola against Plasmodium infection. J. Ethnopharmacol. 2015, 172, 214–218. [Google Scholar] [CrossRef]
- Kagbo, H.D.; Ejebe, D.E. Phytochemistry and preliminary toxicity studies of the methanol extract of the stem bark of Garcinia kola (Heckel). Internet J. Toxicol. 2010, 7, 1–18. [Google Scholar]
- Tchimene, K.M.; Anaga, A.O.; Ugwoke, C.E.C.; Ezugwu, C.O.; Okunji, C.O.; Iwu, M.M. Bio-flavonoids and Garcinoic Acid from Garcinia kola seeds with Promising Anti-Inflammatory Potentials. Pharmacogn. J. 2015, 8, 56–58. [Google Scholar] [CrossRef]
- Ibironke, G.F.; Fasanmade, A.A. Analgesic and central nervous system depressant activities of kolaviron (A Garcinia kola biflavonoid complex). Afr. J. Biomed. Res. 2015, 18, 217–223. [Google Scholar]
- Abarikwu, S.O. Kolaviron, a natural flavonoid from the seeds of Garcinia kola, reduces LPS-induced inflammation in macrophages by combined inhibition of IL-6 secretion, and inflammatory transcription factors, ERK1/2, NF-κB, p38, Akt, p-c-JUN and JNK. Biochim. Biophys. Acta BBA Gen. Subj. 2014, 1840, 2373–2381. [Google Scholar] [CrossRef] [PubMed]
- Farombi, E.O.; Abolaji, A.O.; Farombi, T.H.; Oropo, A.S.; Owoje, O.A.; Awunah, M.T. Garcinia kola seed biflavonoid fraction (Kolaviron), increases longevity and attenuates rotenone-induced toxicity in Drosophila melanogaster. Pestic. Biochem. Physiol. 2018, 145, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Akinmoladun, A.C.; Akinrinola, B.L.; Olaleye, M.T.; Farombi, E.O. Kolaviron, a Garcinia kola Biflavonoid Complex, Protects Against Ischemia/Reperfusion Injury: Pertinent Mechanistic Insights from Biochemical and Physical Evaluations in Rat Brain. Neurochem. Res. 2015, 40, 777–787. [Google Scholar] [CrossRef] [PubMed]
- Odukanmi, O.A.; Salami, A.T.; Ashaolu, O.P.; Adegoke, A.G.; Olaleye, S.B. Kolaviron attenuates ischemia/reperfusion injury in the stomach of rats. Appl. Physiol. Nutr. Metab. 2018, 43, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Iwu, M.M.; Igboko, O.A.; Onwuchekwa, U.A.; Okunji, C.O. Evaluation of the antihepatotoxic activity of the biflavonoids of Garcinia kola seed. J. Ethnopharmacol. 1987, 21, 127–138. [Google Scholar] [CrossRef]
- Braide, V.B. Antihepatotoxic biochemical effects of kolaviron, a biflavonoid of Garcinia kola seeds. Phytother. Res. 1991, 5, 35–37. [Google Scholar] [CrossRef]
- Oyenihi, O.R.; Brooks, N.L.; Oguntibeju, O.O. Effects of kolaviron on hepatic oxidative stress in streptozotocin induced diabetes. BMC Complement. Altern. Med. 2015, 15. [Google Scholar] [CrossRef]
- Alabi, Q.K.; Akomolafe, R.O.; Olukiran, O.S.; Adeyemi, W.J.; Nafiu, A.O.; Adefisayo, M.A.; Omole, J.G.; Kajewole, D.I.; Odujoko, O.O. The Garcinia kola biflavonoid kolaviron attenuates experimental hepatotoxicity induced by diclofenac. Pathophysiology 2017, 24, 281–290. [Google Scholar] [CrossRef]
- Farombi, E.O.; Shrotriya, S.; Surh, Y.-J. Kolaviron inhibits dimethyl nitrosamine-induced liver injury by suppressing COX-2 and iNOS expression via NF-κB and AP-1. Life Sci. 2009, 84, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Indabawa, I.; Arzai, A. Antibacterial Activity of Garcinia kola and Cola nitida Seed Extracts. Bayero J. Pure Appl. Sci. 2011, 4. [Google Scholar] [CrossRef]
- Aderibigbe, S.A. Antimicrobial Activities of Garcinia kola Seed Oil against Some Clinical Microbial Isolates. Int. Res. J. Pharm. 2012, 2, 68–72. [Google Scholar]
- Jouda, J.-B.; Tamokou, J.-D.; Mbazoa, C.D.; Douala-Meli, C.; Sarkar, P.; Bag, P.K.; Wandji, J. Antibacterial and cytotoxic cytochalasins from the endophytic fungus Phomopsis sp. harbored in Garcinia kola (Heckel) nut. BMC Complement. Altern. Med. 2016, 16. [Google Scholar] [CrossRef] [PubMed]
- Sani, A.; Zakariyya, U.A.; Mahe, A.; Singh, D. In vitro study of antitrypanosomal activity of ethanolic leaf extract of Garcinia kola against Trypanosoma brucei brucei. Asian J. Pharm. Clin. Res. 2018, 11, 417–419. [Google Scholar] [CrossRef]
- Niemenak, N.; Onomo, P.E.; Fotso; Lieberei, R.; Ndoumou, D.O. Purine alkaloids and phenolic compounds in three Cola species and Garcinia kola grown in Cameroon. S. Afr. J. Bot. 2008, 74, 629–638. [Google Scholar] [CrossRef]
- Popoola, T.D.; Awodele, O.; Omisanya, A.; Obi, N.; Umezinwa, C.; Fatokun, A.A. Three indigenous plants used in anti-cancer remedies, Garcinia kola Heckel (stem bark), Uvaria chamae P. Beauv.(root) and Olax subscorpioidea Oliv.(root) show analgesic and anti-inflammatory activities in animal models. J. Ethnopharmacol. 2016, 194, 440–449. [Google Scholar] [CrossRef]
- Gyimah, A. Effect of pretreatment methods on germination of Garcinia kola Heckel seeds. Ghana J. For. 2000, 9, 39–44. [Google Scholar]
- Kouakou, K.L.; Dao, J.P.; Kouassi, K.L.; Beugré, M.M.; Koné, M.; Baudoin, J.-P.; Bi, Z.; Irié, A. Propagation of Garcinia kola (Heckel) by stem and root cutting. Silva Fenn. 2016, 50, 1588. [Google Scholar] [CrossRef]
- Kanmegne, G.; Omokolo, N.D. Germination of Garcinia kola (Heckel) seeds in response to different hormone treatments. Fruits 2008, 63, 155–161. [Google Scholar] [CrossRef]
- Termote, C.; Everaert, G.; Bwama Meyi, M.; Dhed’a Djailo, B.; Van Damme, P. Wild Edible Plant Markets in Kisangani, Democratic Republic of Congo. Hum. Ecol. 2012, 40, 269–285. [Google Scholar] [CrossRef]
- Adedokun, M.; Ojo, T.; Oyelakin, F. Socio-economic Contributions and Marketing of Garcinia kola (bitter kola) in Ijebu- Ode Ogun State, Nigeria. Int. J. Afr. Asian Stud. 2018, 42, 45–52. [Google Scholar]
- Pérez, M.R.; Ndoye, O.; Eyebe, A.; Puntodewo, A. Spatial characterisation of non-timber forest products markets in the humid forest zone of Cameroon. Int. For. Rev. 2000, 2, 71–83. [Google Scholar]
- Awono, A.; Atyi, R.E.; Foundjem-Tita, D.; Levang, P. Vegetal non-timber forest products in Cameroon, contribution to the national economy. Int. For. Rev. 2016, 18, 66–77. [Google Scholar] [CrossRef]
- Pribyl, O.; Verner, V.; Degrande, A.; Foundjem-Tita, D.; Manourova, A.; Van Damme, P. Market survey of Garcinia kola (bitter kola) in Yaoundé city, Cameroon. In Proceedings of the Future Agriculture: Socio-Ecological Transitions and Bio-Cultural Shifts, Bonn, Germany, 20–22 September 2017; p. 447. [Google Scholar]
- Tchoundjeu, Z.; Asaah, E.K.; Bayala, J.; Kalinganire, A.; Mngomba, S. Vegetative propagation techniques. In Agroforestry Tree Domestication: A Primer; Dawson, I., Harwood, C., Jamnadass, R., Beniest, J., Eds.; World Agroforestry Centre: Nairobi, Kenya, 2012. [Google Scholar]
- Tchoundjeu, Z.; Degrande, A.; Leakey, R.R.; Nimino, G.; Kemajou, E.; Asaah, E.; Facheux, C.; Mbile, P.; Mbosso, C.; Sado, T. Impacts of participatory tree domestication on farmer livelihoods in West and Central Africa. For. Trees Livelihoods 2010, 19, 217–234. [Google Scholar] [CrossRef]
- Leakey, R.R.B.; Simons, A.J. The domestication and commercialization of indigenous trees in agroforestry for the alleviation of poverty. Agrofor. Syst. 1998, 38, 165–176. [Google Scholar] [CrossRef]
- Akinnifesi, F.K.; Ajayi, O.C.; Sileshi, G.; Kadzere, I.; Akinnifesi, A.I. Domesticating and Commercializing Indigenous Fruit and Nut Tree Crops for Food Security and Income Generation in Sub-Saharan Africa. Presented at the New Crops International Symposium, Southampton, UK, 3–4 September 2007. [Google Scholar]
- Ofori, D.A.; Gyau, A.; Dawson, I.K.; Asaah, E.; Tchoundjeu, Z.; Jamnadass, R. Developing more productive African agroforestry systems and improving food and nutritional security through tree domestication. Curr. Opin. Environ. Sustain. 2014, 6, 123–127. [Google Scholar] [CrossRef]
- Clement, C.; De Cristo-Araújo, M.; Coppens D’Eeckenbrugge, G.; Alves Pereira, A.; Picanço-Rodrigues, D. Origin and Domestication of Native Amazonian Crops. Diversity 2010, 2, 72–106. [Google Scholar] [CrossRef]
- Leakey, R.; van Damme, P. The role of tree domestication in green market product value chain development. For. Trees Livelihoods 2014, 23, 116–126. [Google Scholar] [CrossRef]
- Scianna, J.D.; Winslow, S.R.; Majerus, M.E.; Gruber, L.M.; Reid, S.A. Asexual plant propagation: Special techniques and considerations for successful high altitude revegetation. In Proceedings of the USDA Natural Resources Conservation Workshop 13, Colorado State University, Colorado Water Resources Research Institute, Ft. Collins, CO, USA, 4–6 March 1998; pp. 105–117. [Google Scholar]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).