Enhanced Photoluminescent Properties and Crystalline Morphology of LiBaPO4:Tm3+ Phosphor through Microwave Sintering Method
Abstract
:1. Introduction
2. Results and Discussion
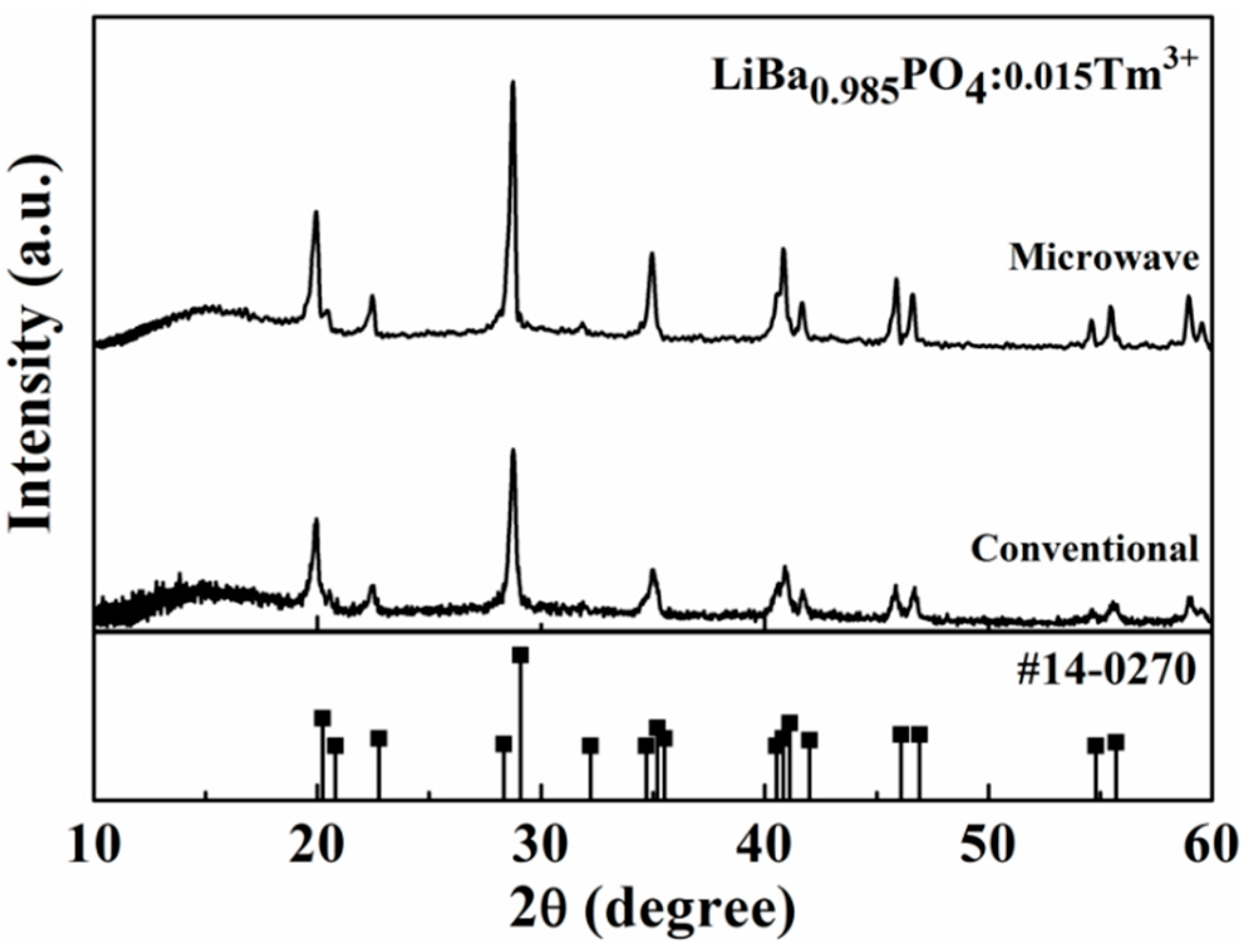
2.1. Structure
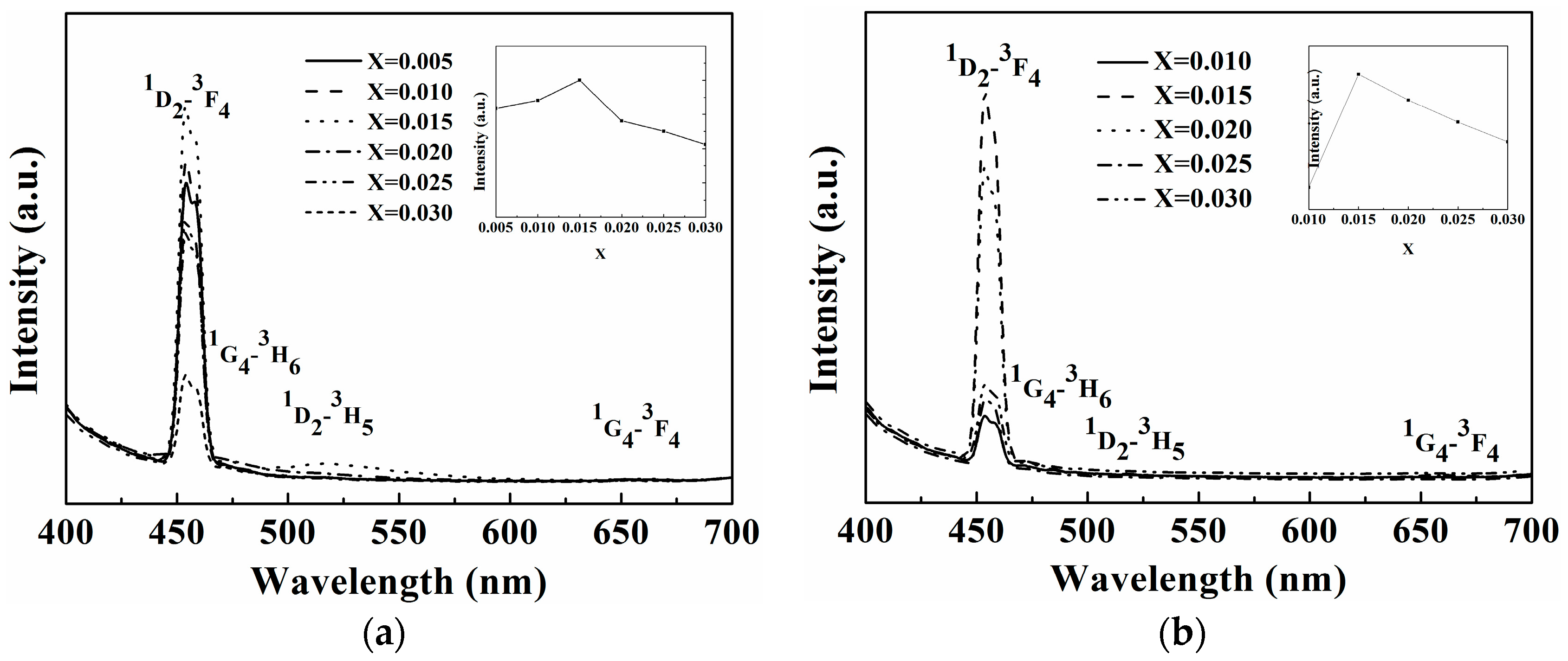
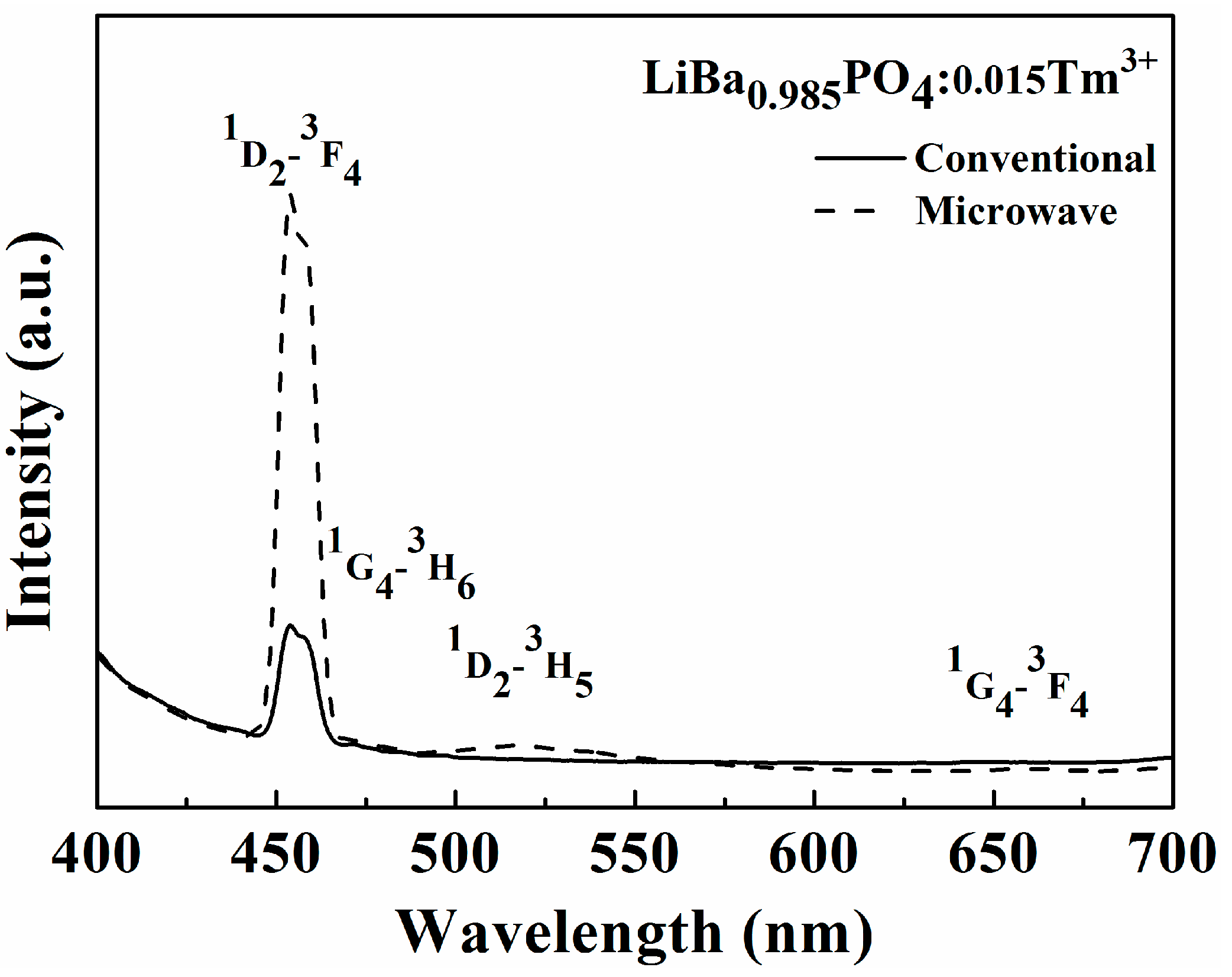
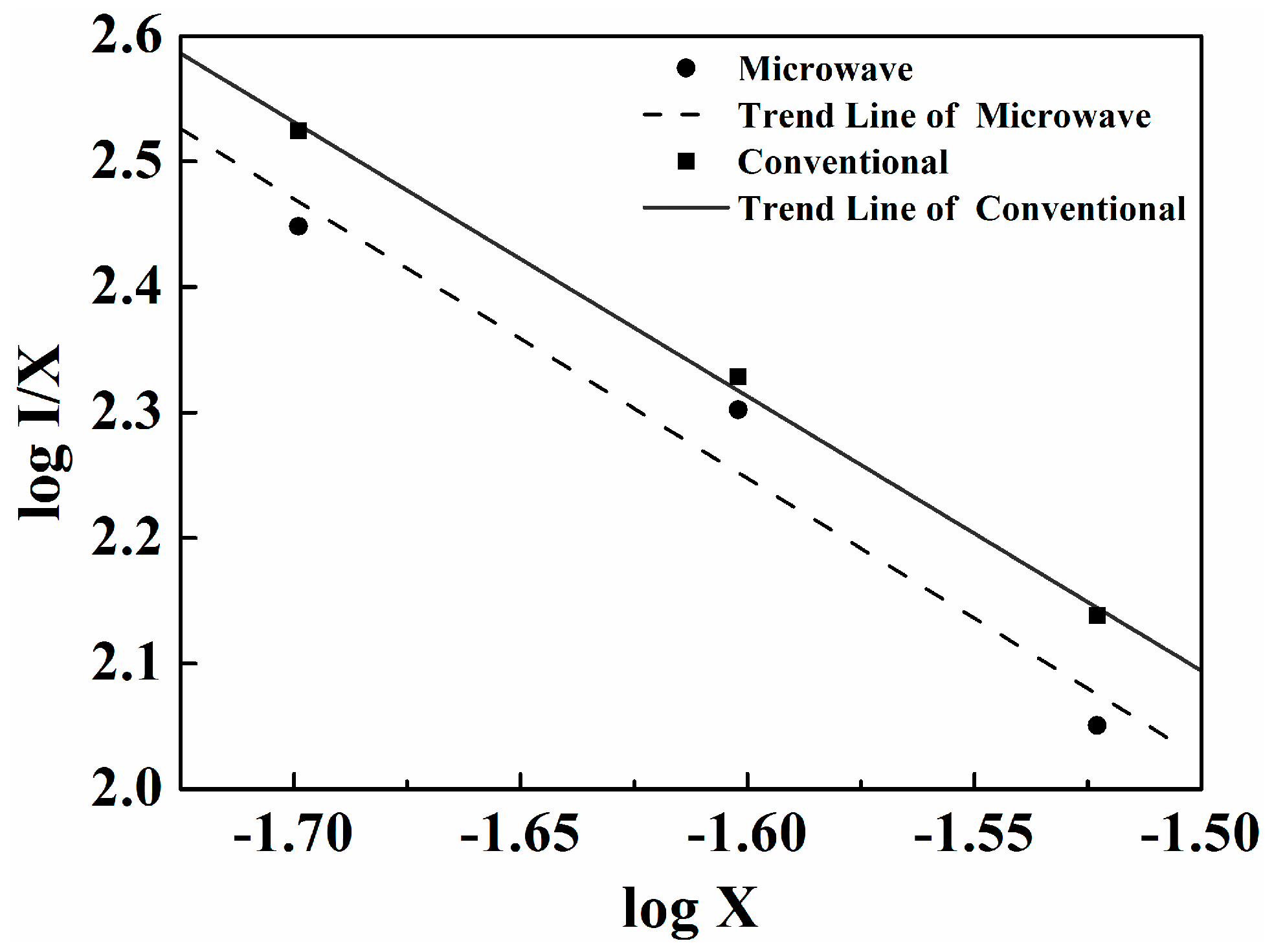
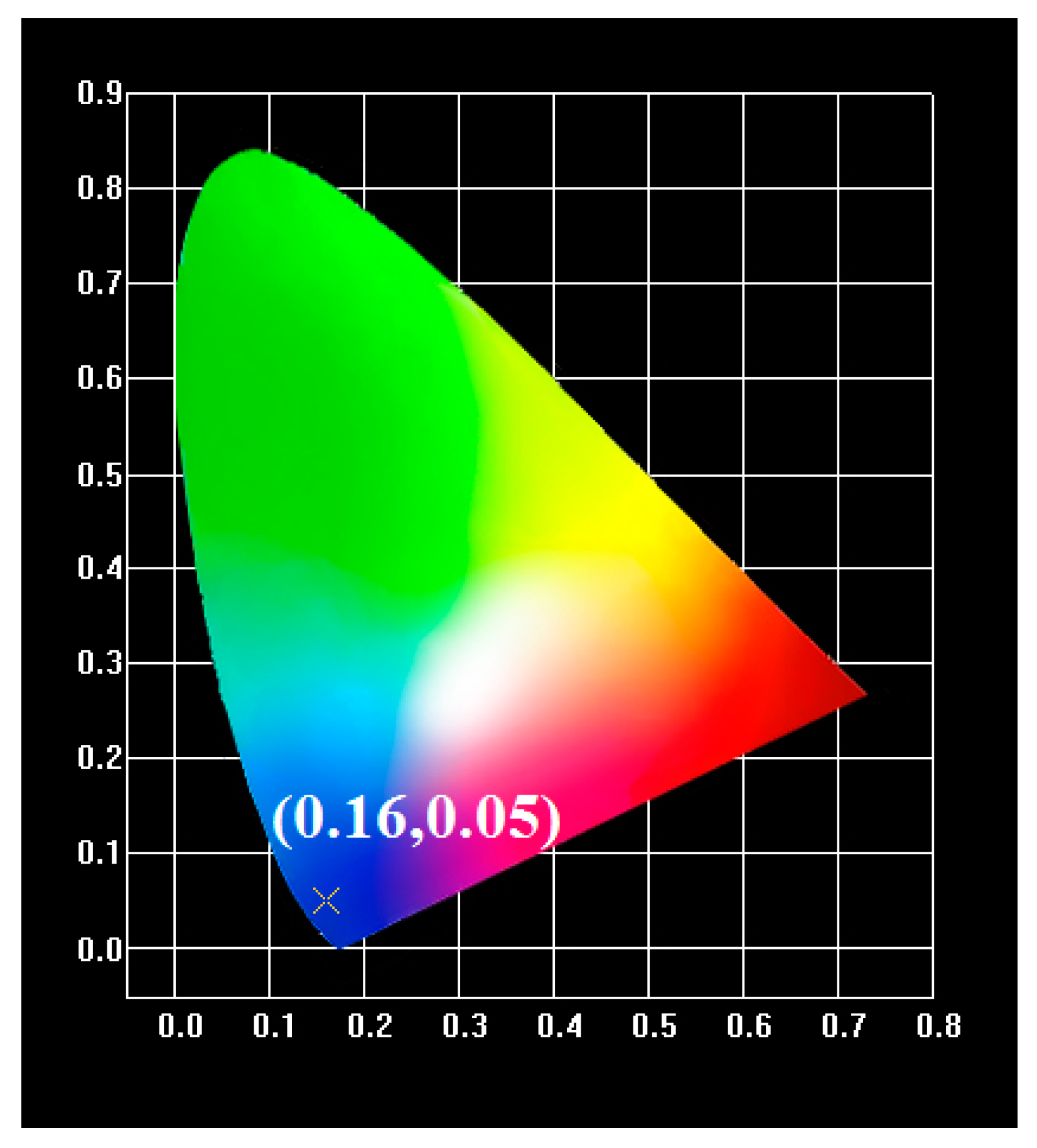
2.2. Photoluminescence Properties
3. Experimental Procedure
3.1. Sample Preparation
3.2. Characterization
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sheu, J.K.; Chang, S.J.; Kuo, C.H.; Su, Y.K.; Wu, L.W.; Lin, Y.C.; Lai, W.C.; Tsai, J.M.; Chi, G.C.; Wu, R.K. White-light emission from near UV InGaN-GaN LED chip precoated with blue/green/red phosphors. IEEE Photonics Technol. Lett. 2003, 15, 18–20. [Google Scholar] [CrossRef]
- Su, Y.K.; Peng, Y.M.; Yang, R.Y.; Chen, J.L. Effects of NaCl flux on microstructure and luminescent characteristics of KSrPO4:Eu2+ phosphors. Opt. Mater. 2012, 34, 1598–1602. [Google Scholar] [CrossRef]
- Peng, Y.M.; Su, Y.K.; Yang, R.Y. The charge transfer transition phenomenon and microstructure of Eu3+-doped NaCaPO4 phosphors sintered with NH4Cl flux via solid-state reaction. Mater. Res. Bull. 2013, 48, 1946–1951. [Google Scholar] [CrossRef]
- Yang, R.Y.; Peng, Y.M.; Su, Y.K. Novel red-emitting microwave-assisted-sintered LiSrPO4:Eu3+ phosphors for application in near-UV white light-emitting diodes. J. Electron. Mater. 2013, 42, 2910–2914. [Google Scholar] [CrossRef]
- Yang, R.Y.; Peng, Y.M.; Lai, H.L.; Chu, C.J.; Chiou, B.; Su, Y.K. Effect of the different concentrations of Eu3+ ions on the microstructure and photoluminescent properties of Zn2SiO4:xEu3+ phosphors and synthesized with TEOS solution as silicate source. Opt. Mater. 2013, 35, 1719–1723. [Google Scholar] [CrossRef]
- Peng, Y.M.; Su, Y.K.; Yang, R.Y. Improving thermal stability of KSrPO4:Tb3+ phosphors prepared by microwave assisted sintering. Opt. Mater. 2013, 35, 2102–2106. [Google Scholar] [CrossRef]
- Li, Y.Z.; Wang, Y.H.; Wang, Z.F.; Zhang, Z.Y. UV–VUV-excited photoluminescence of Tm3+ substituted β-rhenanite as a blue-emitting phosphor. J. Lumin. 2010, 130, 1225–1229. [Google Scholar] [CrossRef]
- Weng, M.H.; Yang, R.Y.; Peng, Y.M.; Chen, J.L. Yellowish green-emitting KSrPO4:Tb3+ phosphors with various doping concentrations prepared by using microwave assisted sintering. Ceram. Int. 2012, 38, 1319–1323. [Google Scholar] [CrossRef]
- Yang, R.Y.; Chen, H.Y.; Hsiung, C.M.; Chang, S.J. Crystalline morphology and photoluminescent properties of YInGe2O7:Eu3+ phosphors prepared from microwave and conventional sintering. Ceram. Int. 2011, 37, 749–752. [Google Scholar] [CrossRef]
- Chen, H.Y.; Yang, R.Y.; Chang, S.J. Improving crystalline morphology and photoluminescent properties of BaY2ZnO5:Eu3+ phosphors prepared using microwave assisted sintering. Mater. Lett. 2010, 64, 2548–2550. [Google Scholar] [CrossRef]
- Chen, H.Y.; Yang, R.Y.; Chang, S.J.; Yang, Y.K. Microstructure and photoluminescent properties of Sr2SiO4:Eu3+ phosphors with various NH4Cl flux concentrations. Mater. Res. Bull. 2012, 47, 1412–1416. [Google Scholar] [CrossRef]
- Yang, R.Y.; Lai, H.L. Microstructure, and luminescence properties of LiBaPO4:Dy3+ phosphors with various Dy3+ concentrations prepared by microwave assisted sintering. J. Lumin. 2014, 145, 49–54. [Google Scholar] [CrossRef]
- Sun, J.; Zhang, X.; Xia, Z.; Du, H. Luminescent properties of LiBaPO4:RE (RE = Eu2+, Tb3+, Sm3+) phosphors for white light-emitting diodes. J. Appl. Phys. 2012, 111, 013101. [Google Scholar] [CrossRef]
- Van Uitert, L.G. Characterization of energy transfer interactions between rare earth ions. J. Electronchem. Soc. 1967, 114, 1048–1053. [Google Scholar] [CrossRef]
- Ozawa, L.; Jaffe, P.M. The mechanism of the emission color shift with activator concentration in +3 activated phosphors. J. Electronchem. Soc. 1971, 118, 1678–1679. [Google Scholar] [CrossRef]
- Dexter, D.L. A theory of sensitized luminescence in solids. J. Chem. Phys. 1953, 21, 836–850. [Google Scholar] [CrossRef]

© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lai, H.-L.; Weng, M.-H.; Yang, R.-Y.; Chang, S.-J. Enhanced Photoluminescent Properties and Crystalline Morphology of LiBaPO4:Tm3+ Phosphor through Microwave Sintering Method. Materials 2016, 9, 356. https://doi.org/10.3390/ma9050356
Lai H-L, Weng M-H, Yang R-Y, Chang S-J. Enhanced Photoluminescent Properties and Crystalline Morphology of LiBaPO4:Tm3+ Phosphor through Microwave Sintering Method. Materials. 2016; 9(5):356. https://doi.org/10.3390/ma9050356
Chicago/Turabian StyleLai, Hsuan-Lin, Min-Hang Weng, Ru-Yuan Yang, and Shoou-Jinn Chang. 2016. "Enhanced Photoluminescent Properties and Crystalline Morphology of LiBaPO4:Tm3+ Phosphor through Microwave Sintering Method" Materials 9, no. 5: 356. https://doi.org/10.3390/ma9050356
APA StyleLai, H.-L., Weng, M.-H., Yang, R.-Y., & Chang, S.-J. (2016). Enhanced Photoluminescent Properties and Crystalline Morphology of LiBaPO4:Tm3+ Phosphor through Microwave Sintering Method. Materials, 9(5), 356. https://doi.org/10.3390/ma9050356







