Bioactive Wollastonite-Diopside Foams from Preceramic Polymers and Reactive Oxide Fillers
Abstract
:1. Introduction
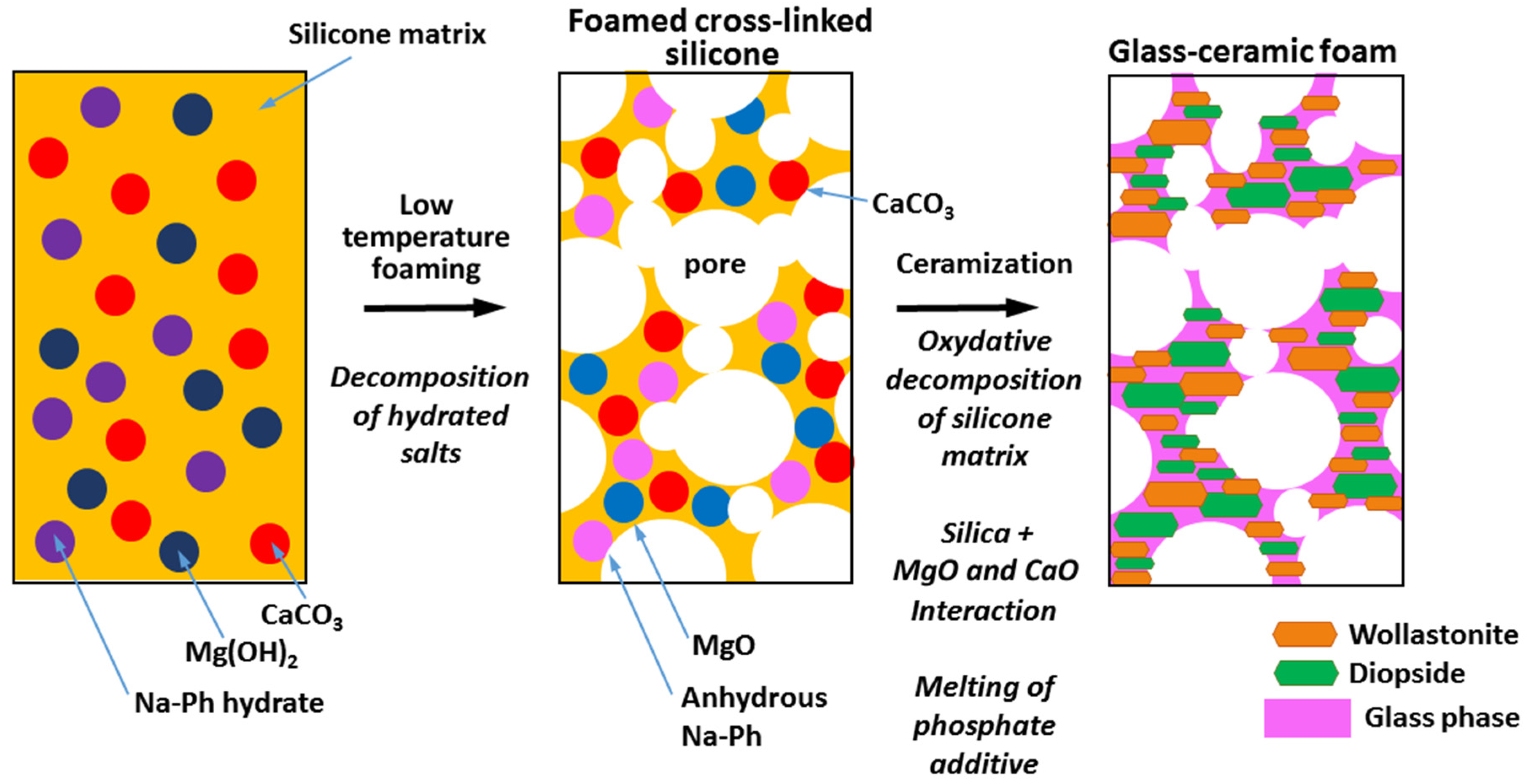
2. Experimental Procedure
2.1. Starting Materials
| Composition (% mol) | ||||
|---|---|---|---|---|
| SiO2 | CaO | MgO | Na2O | Li2O |
| 55.3 | 22.0 | 12.0 | 9.0 | 1.7 |
2.2. Preparation of Foams
2.3. Preparation of Pellets
2.4. Cell Culture and Seeding
2.5. Analysis of Cell Viability
2.6. Statistical Analysis
2.7. Characterization
3. Results and Discussion
3.1. Foaming and Phase Development
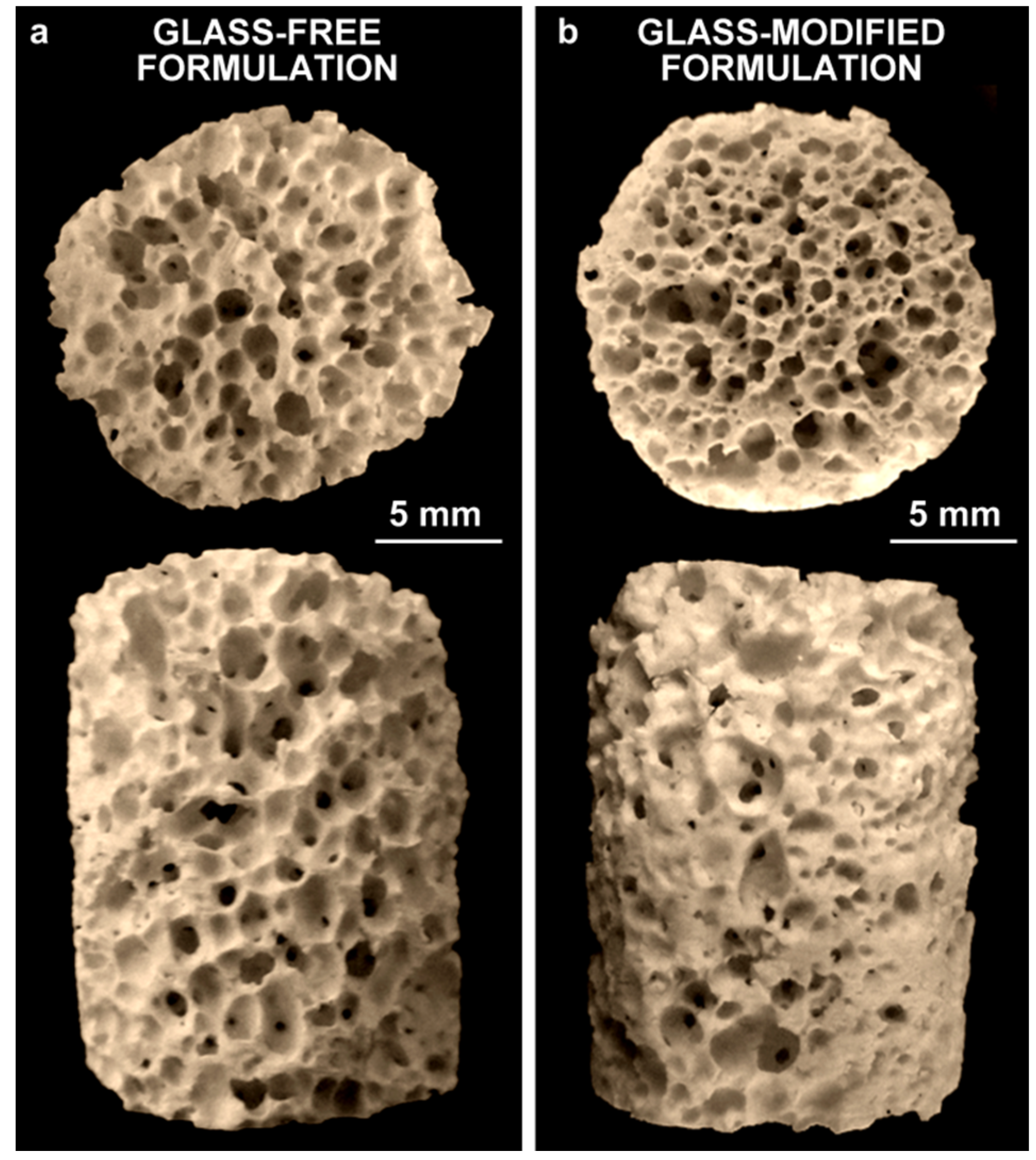
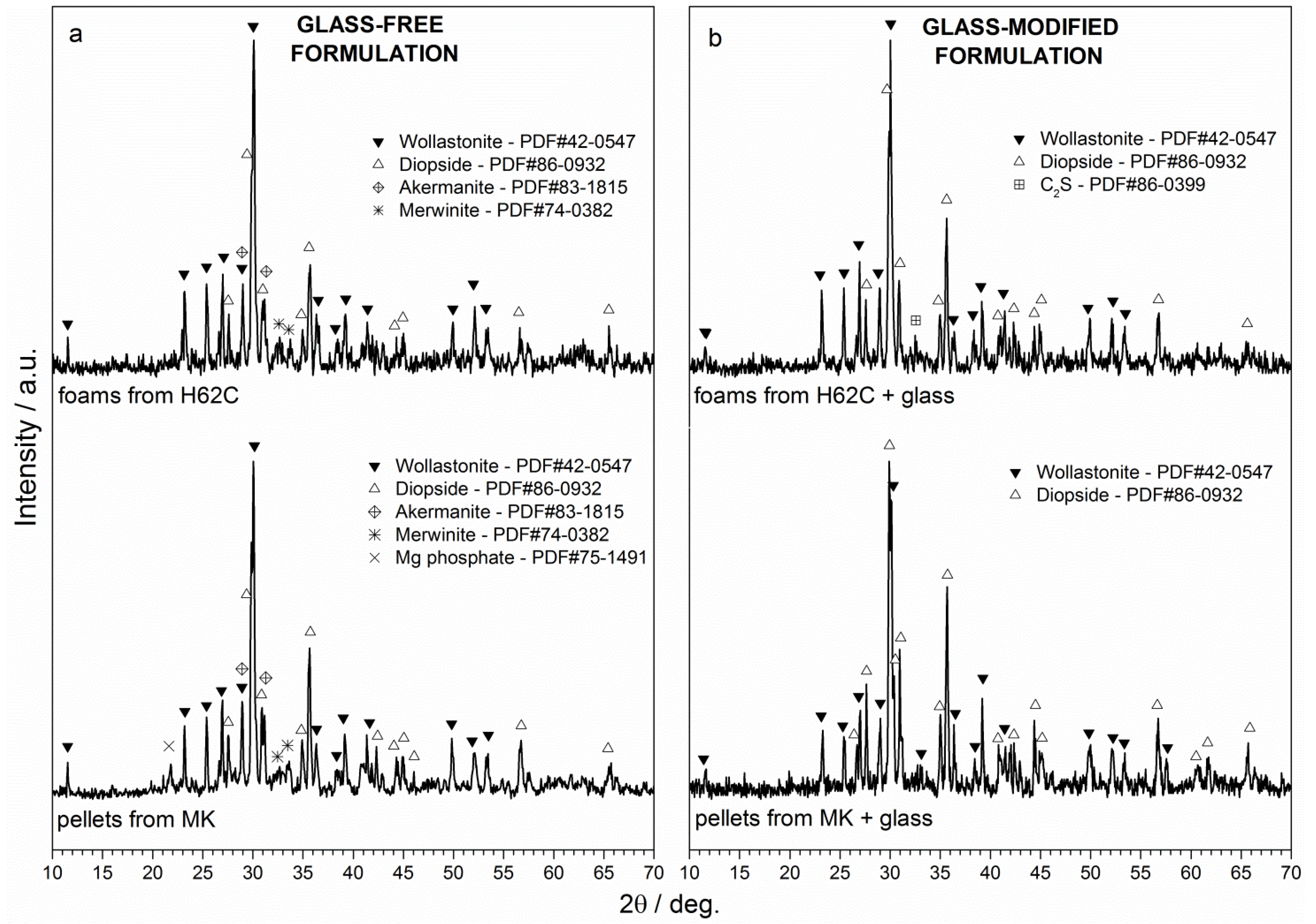
| Foam Formulation | Bulk Density (g/cm3) | Open Porosity (%) | Crushing Strength (MPa) |
|---|---|---|---|
| H62C + fillers (borax) * | 0.73 ± 0.02 | 77.0 | 1.8 ± 0.3 |
| H62C + fillers (Na-phosphate) | 0.70 ± 0.02 | 76.5 | 1.4 ± 0.1 |
| H62C + fillers + 10 wt% glass (Na-phosphate) | 0.63 ± 0.10 | 79.4 | 3.1 ± 0.7 |

3.2. Impacts of Preceramic Polymer and Glass on Phase Development
| Formulations | Wollastonite (wt%) | Diopside (wt%) | |
|---|---|---|---|
| Theoretical | CaO·SiO2 + CaO·MgO·2SiO2 | 35 | 65 |
| 1 | H62C + fillers | 56 | 44 |
| 2 | H62C + fillers + 10 wt% glass | 40 | 60 |
| 3 | MK + fillers | 49 | 51 |
| 4 | MK + fillers + 10 wt% glass | 42 | 58 |
| Formulations | Crystalline Phase (wt%) | Amorphous Phase (wt%) | |
|---|---|---|---|
| Pure Ca/Mg-rich glass | 66 | 34 | |
| 1 | H62C + fillers | 88 | 12 |
| 2 | H62C + fillers + 10 wt% glass | 98 | 2 |
| 3 | MK + fillers | 92 | 8 |
| 4 | MK + fillers + 10 wt% glass | 96 | 4 |
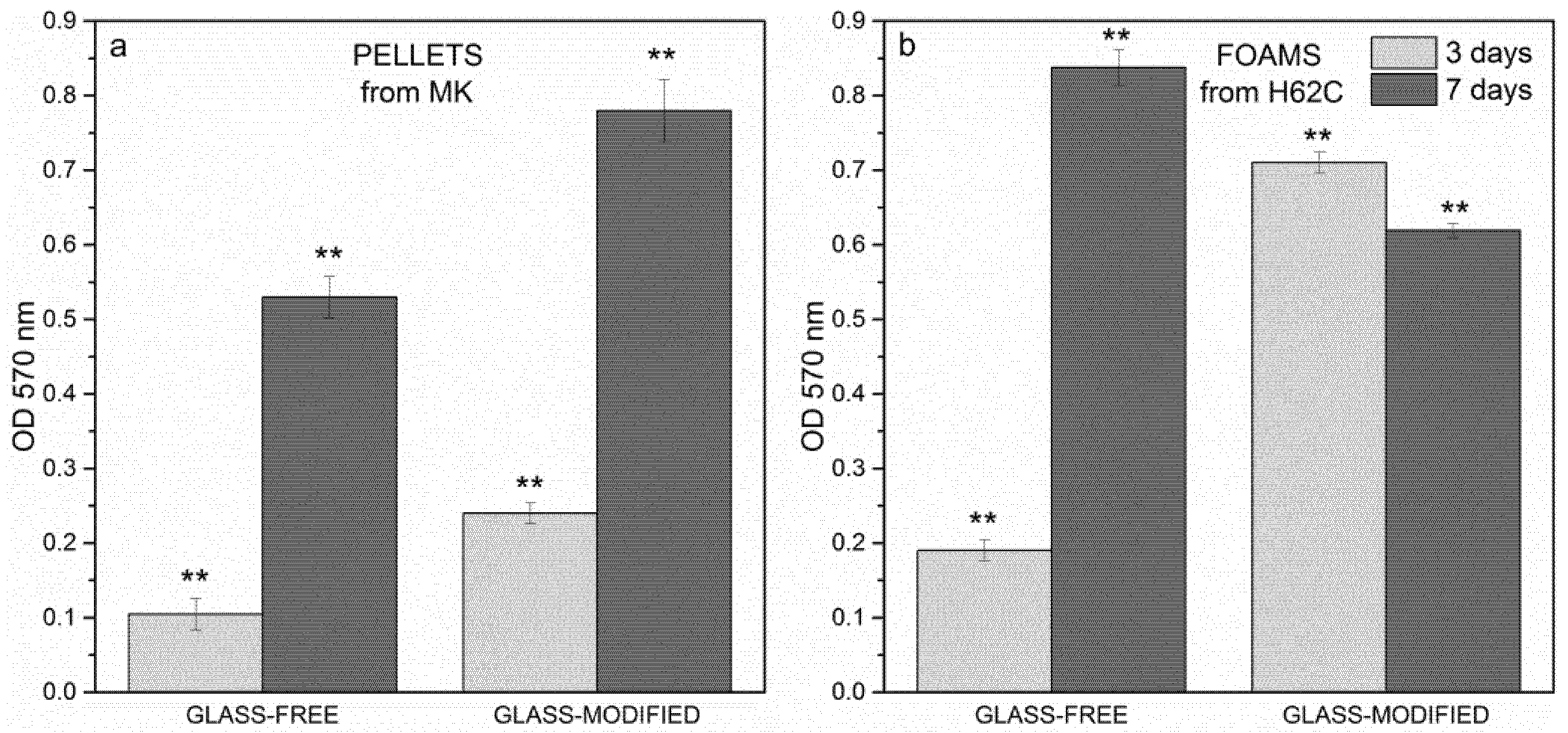
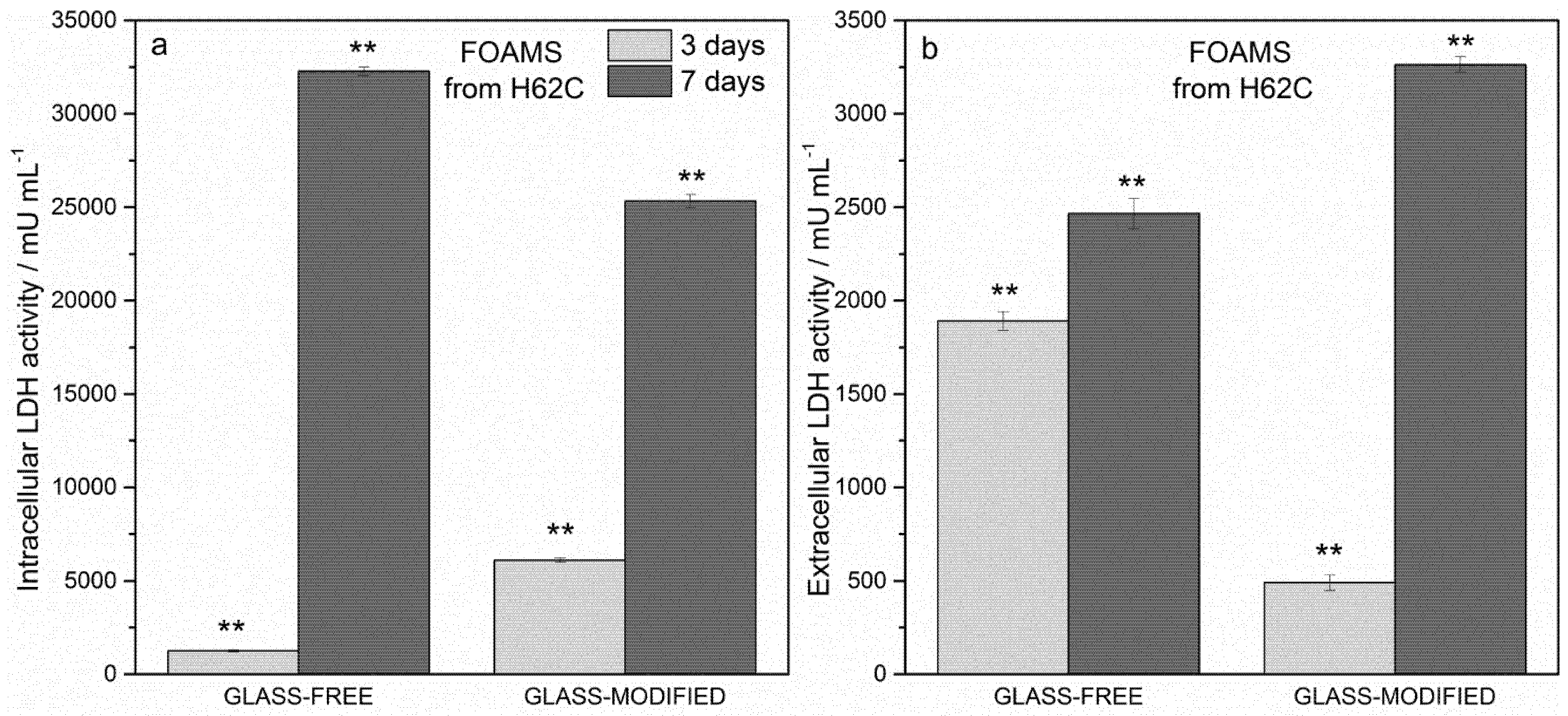
3.3. In Vitro Biological Characterization

4. Conclusions
- -
- Wollastonite-diopside ceramics can be fabricated by firing mixtures based on preceramic polymers, in the form of silicone resins (acting as silica sources), mixed with powdered metal oxide precursors;
- -
- The choice of silicone polymers with different natures and chemistry (liquid H62C, solid MK) does not affect the ceramic product in terms of main phase assemblage;
- -
- A liquid silicone can be easily foamed by water release, in turn due to the decomposition of hydrated sodium phosphate; the ceramic conversion implies the transformation of the silicone foam into a glass-ceramic foam, incorporating silicate crystals embedded in the glass phase provided by the same phosphate additive;
- -
- The liquid phase developed upon firing can be increased by the introduction of a glass filler; the positive impact on the structural integrity of samples is not accompanied by any change in the phase assemblage, operating with a glass crystallizing itself in wollastonite and diopside;
- -
- Both dense and foamed wollastonite-diopside ceramic samples showed positive results in terms of cell viability, according to the MTT assay and LDH activity tests; the incorporation of glass in the formulations proved not to be detrimental to cell survival and proliferation;
- -
- While the incorporation of glass in the formulation was not crucial for viability at Day 7, it was definitively effective at improving the biocompatibility of the samples throughout the cell culture period up to Day 3.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Colombo, P.; Mera, G.; Riedel, R.; Sorarù, G.D. Polymer-derived ceramics: 40 years of research and innovation in advanced ceramics. J. Am. Ceram. Soc. 2010, 93, 1805–1837. [Google Scholar]
- Colombo, P.; Bernardo, E.; Parcianello, G. Multifunctional advanced ceramics from preceramic polymers and nano-sized active fillers. J. Eur. Ceram. Soc. 2013, 33, 453–469. [Google Scholar] [CrossRef]
- Bernardo, E.; Fiocco, L.; Parcianello, G.; Storti, E.; Colombo, P. Advanced ceramics from preceramic polymers modified at the nano-scale: A review. Materials 2014, 7, 1927–1956. [Google Scholar] [CrossRef]
- De Aza, P.N.; Guitian, F.; de Aza. Bioactivity of wollastonite ceramics: In vitro evaluation. Scr. Metall. Mater. 1994, 31, 1001–1005. [Google Scholar]
- Lin, K.; Zhai, W.; Ni, S.; Chang, J.; Zeng, Y.; Qian, W. Study of mechanical property and in vitro biocompatibility of CaSiO3 ceramics. Ceram. Inter. 2005, 31, 323–326. [Google Scholar] [CrossRef]
- Wu, C.; Chang, J. Degradation, bioactivity and cytocompatibility of diopside, akermanite and bredigite ceramics. J. Biomed. Mater. Res.-B Appl. Biomater. 2007, 83, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Ventura, J.M.G.; Tulyaganov, D.U.; Agathopoulos, S.; Ferreira, J.M.F. Sintering and crystallization of akermanite-based glass–ceramics. Mater. Lett. 2006, 60, 1488–1491. [Google Scholar] [CrossRef]
- Nonami, T.; Tsutsumi, S. Study of diopside ceramics for biomaterials. J. Mater. Sci. Mater. Med. 1999, 10, 475–479. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Ramaswamy, Y.; Zreiqat, H. Porous diopside (CaMgSi2O6) scaffold: A promising bioactive material for bone tissue engineering. Acta Biomater. 2010, 6, 2237–2245. [Google Scholar] [CrossRef]
- Jones, J.R.; Hench, L.L. Regeneration of trabecular bone using porous ceramics. Curr. Opin. Solid State Mater. Sci. 2003, 7, 301–307. [Google Scholar] [CrossRef]
- Jones, J.R.; Lee, P.D.; Hench, L.L. Hierarchical porous materials for tissue engineering. Philos. Trans. R. Soc. A 2006, 364, 263–281. [Google Scholar] [CrossRef]
- Bernardo, E.; Carlotti, J.-F.; Dias, P.M.; Fiocco, L.; Colombo, P.; Treccani, L.; Hess, U.; Rezwan, K. Novel akermanite-based bioceramics from preceramic polymers and oxide fillers. Ceram. Int. 2014, 40, 1029–1035. [Google Scholar] [CrossRef]
- Bernardo, E.; Colombo, P.; Dainese, E.; Lucchetta, G.; Bariani, P.F. Novel 3D wollastonite-based scaffolds from preceramic polymers containing micro- and nano-sized reactive particles. Adv. Eng. Mater. 2012, 14, 269–274. [Google Scholar] [CrossRef]
- Bernardo, E.; Parcianello, G.; Colombo, P.; Matthews, S. Wollastonite foams from an extruded preceramic polymer mixed with CaCO3 microparticles assisted by supercritical carbon dioxide. Adv. Eng. Mater. 2013, 5, 60–65. [Google Scholar] [CrossRef]
- Fiocco, L.; Elsayed, H.; Bernardo, E.; Daguano, J.K.M.F.; Soares, V.O. Silicone resins mixed with active oxide fillers and Ca-Mg Silicate glass as alternative/integrative precursors for wollastonite-diopside glass-ceramic foams. J. Non-Cryst. Sol. 2015, 416, 44–49. [Google Scholar] [CrossRef]
- Bhattacharya, G.; Zhang, S.; Jayaseelan, D.D.; Lee, W.E. Mineralizing effect of Li2B4O7 and Na2B4O7 on magnesium aluminate spinel formation. J. Am. Ceram. Soc. 2007, 90, 97–106. [Google Scholar] [CrossRef]
- Rahaman, M.N.; Day, D.E.; Bal, B.S.; Fu, Q.; Jung, S.B.; Bonewald, L.F.; Tomsia, A.P. Bioactive glass in tissue engineering. Acta Biomater. 2001, 7, 2355–2373. [Google Scholar]
- Hoppe, A.; Güldal, N.S.; Boccaccini, A.R. A review of the biological response to ionic dissolution products from bioactive glasses and glass-ceramics. Biomaterials 2011, 32, 2757–2774. [Google Scholar] [CrossRef] [PubMed]
- Fu, Q.; Rahaman, M.N.; Bal, B.S.; Bonewald, L.F.; Kuroki, K.; Brown, R.F. Silicate, borosilicate and borate bioactive glass scaffolds with controllable degradation rates for bone tissue engineering applications. II. In vitro and in vivo biological evaluation. J. Biomed. Mater. Res. A 2010, 95, 172–179. [Google Scholar] [CrossRef]
- Gorustovich, A.A.; Lopez, J.M.P.; Guglielmotti, M.B.; Cabrini, R.L. Biological performance of boron-modified bioactive glass particles implanted in rat tibia bone marrow. Biomed. Mater. 2006, 1, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.B.; Day, D.E.; Brown, R.F.; Bonewald, L.F. Potential toxicity of bioactive borate glasses in-vitro and in-vivo. In Advances in Bioceramics and Porous Ceramics V; Narayan, R., Colombo, R.P., Halbig, M., Mathur, S., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2012. [Google Scholar]
- Baino, F.; Brovarone, C.V. Three-dimensional glass-derived scaffolds for bone tissue engineering: Current trends and forecasts for the future. J. Biomed. Mater. Res. A 2010, 97, 514–535. [Google Scholar]
- Kaur, G.; Pandey, O.P.; Singh, K.; Homa, D.; Scott, B.; Pickrell, G. A review of bioactive glasses: Their structure, properties, fabrication, and apatite formation. J. Biomed. Mater. Res. A 2013, 102, 254–274. [Google Scholar] [CrossRef] [PubMed]
- Rahaman, M.N.; Liang, W.; Day, D.E.; Marion, N.W.; Reilly, G.C.; Mao, J.J. Preparation and bioactive characteristics of porous borate glass substrates. Ceram. Eng. Sci. Proc. 2005, 26, 3–10. [Google Scholar]
- Kaur, G.; Pickrell, G.; Kimsawatde, G.; Homa, D.; Allbee, H.A.; Sriranganathan, N. Synthesis, cytotoxicity, and hydroxypatite formation in 27-Tris-SBF for sol-gel based CaO-P2O5-SiO2-B2O3-ZnO bioactive glasses. Sci. Rep. 2014, 4, 1–14. [Google Scholar] [CrossRef]
- Khorami, M.; Hesaraki, S.; Behnamghader, A.; Nazarian, H.; Shahrabi, S. In vitro bioactivity and biocompatibility of lithium substituted 45S5 bioglass. Mat. Sci. Eng. C 2011, 31, 1584–1592. [Google Scholar] [CrossRef]
- Miguez-Pacheco, V.; Büttner, T.; Maçon, A.L.B.; Jones, J.R.; Fey, T.; de Ligny, D.; Greil, P.; Chevalier, J.; Malchere, A.; Boccaccini, A.R. Development and characterization of lithium-releasing silicate bioactive glasses and their scaffolds for bone repair. J. Non-Cryst. Sol. 2015, in press. [Google Scholar] [CrossRef]
- Denizot, F.; Lang, R. Rapid colorimetric assay for cell growth and survival. Modifications to the tetrazolium dye procedure giving improved sensitivity and reliability. J. Immunol. Methods 1986, 89, 271–277. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fiocco, L.; Elsayed, H.; Ferroni, L.; Gardin, C.; Zavan, B.; Bernardo, E. Bioactive Wollastonite-Diopside Foams from Preceramic Polymers and Reactive Oxide Fillers. Materials 2015, 8, 2480-2494. https://doi.org/10.3390/ma8052480
Fiocco L, Elsayed H, Ferroni L, Gardin C, Zavan B, Bernardo E. Bioactive Wollastonite-Diopside Foams from Preceramic Polymers and Reactive Oxide Fillers. Materials. 2015; 8(5):2480-2494. https://doi.org/10.3390/ma8052480
Chicago/Turabian StyleFiocco, Laura, Hamada Elsayed, Letizia Ferroni, Chiara Gardin, Barbara Zavan, and Enrico Bernardo. 2015. "Bioactive Wollastonite-Diopside Foams from Preceramic Polymers and Reactive Oxide Fillers" Materials 8, no. 5: 2480-2494. https://doi.org/10.3390/ma8052480
APA StyleFiocco, L., Elsayed, H., Ferroni, L., Gardin, C., Zavan, B., & Bernardo, E. (2015). Bioactive Wollastonite-Diopside Foams from Preceramic Polymers and Reactive Oxide Fillers. Materials, 8(5), 2480-2494. https://doi.org/10.3390/ma8052480









