Calcium Orthophosphate Cements and Concretes
Abstract
:1. Introduction
| Ca/P ionic ratio | Compound and its abbreviation | Chemical formula | Solubility at 25 °C, – log(Ks) | Solubility at 25 °C, g/L | Stability in aqueous solutions at 25 °C (pH range) |
|---|---|---|---|---|---|
| 0.5 | Monocalcium phosphate monohydrate (MCPM) | Ca(H2PO4)2·H2O | 1.14 | ~ 18 | 0.0 – 2.0 |
| 0.5 | Monocalcium phosphate anhydrous (MCPA) | Ca(H2PO4)2 | 1.14 | ~ 17 | [c] |
| 1.0 | Dicalcium phosphate dihydrate (DCPD), mineral brushite | CaHPO4·2H2O | 6.59 | ~ 0.088 | 2.0 – 6.0 |
| 1.0 | Dicalcium phosphate anhydrous (DCPA), mineral monetite | CaHPO4 | 6.90 | ~ 0.048 | [c] |
| 1.33 | Octacalcium phosphate (OCP) | Ca8(HPO4)2(PO4)4·5H2O | 96.6 | ~ 0.0081 | 5.5 – 7.0 |
| 1.5 | α-Tricalcium phosphate (α-TCP) | α-Ca3(PO4)2 | 25.5 | ~ 0.0025 | [a] |
| 1.5 | β-Tricalcium phosphate (β-TCP) | β-Ca3(PO4)2 | 28.9 | ~ 0.0005 | [a] |
| 1.2 – 2.2 | Amorphous calcium phosphate (ACP) | CaxHy(PO4)z·nH2O, n = 3 – 4.5; 15 – 20% H2O | [b] | [b] | ~ 5 – 12 [d] |
| 1.5 – 1.67 | Calcium-deficient hydroxyapatite (CDHA)[e] | Ca10-x(HPO4)x(PO4)6-x(OH)2-x[f] (0 < x < 1) | ~ 85.1 | ~ 0.0094 | 6.5 – 9.5 |
| 1.67 | Hydroxyapatite (HA) | Ca10(PO4)6(OH)2 | 116.8 | ~ 0.0003 | 9.5 – 12 |
| 1.67 | Fluorapatite (FA) | Ca10(PO4)6F2 | 120.0 | ~ 0.0002 | 7 – 12 |
| 2.0 | Tetracalcium phosphate (TTCP), mineral hilgenstockite | Ca4(PO4)2O | 38 - 44 | ~ 0.0007 | [a] |
2. Calcium Orthophosphate Cements
2.1. General Information and Brief History
2.2. Composition and Crystallization
2.3. Chemistry
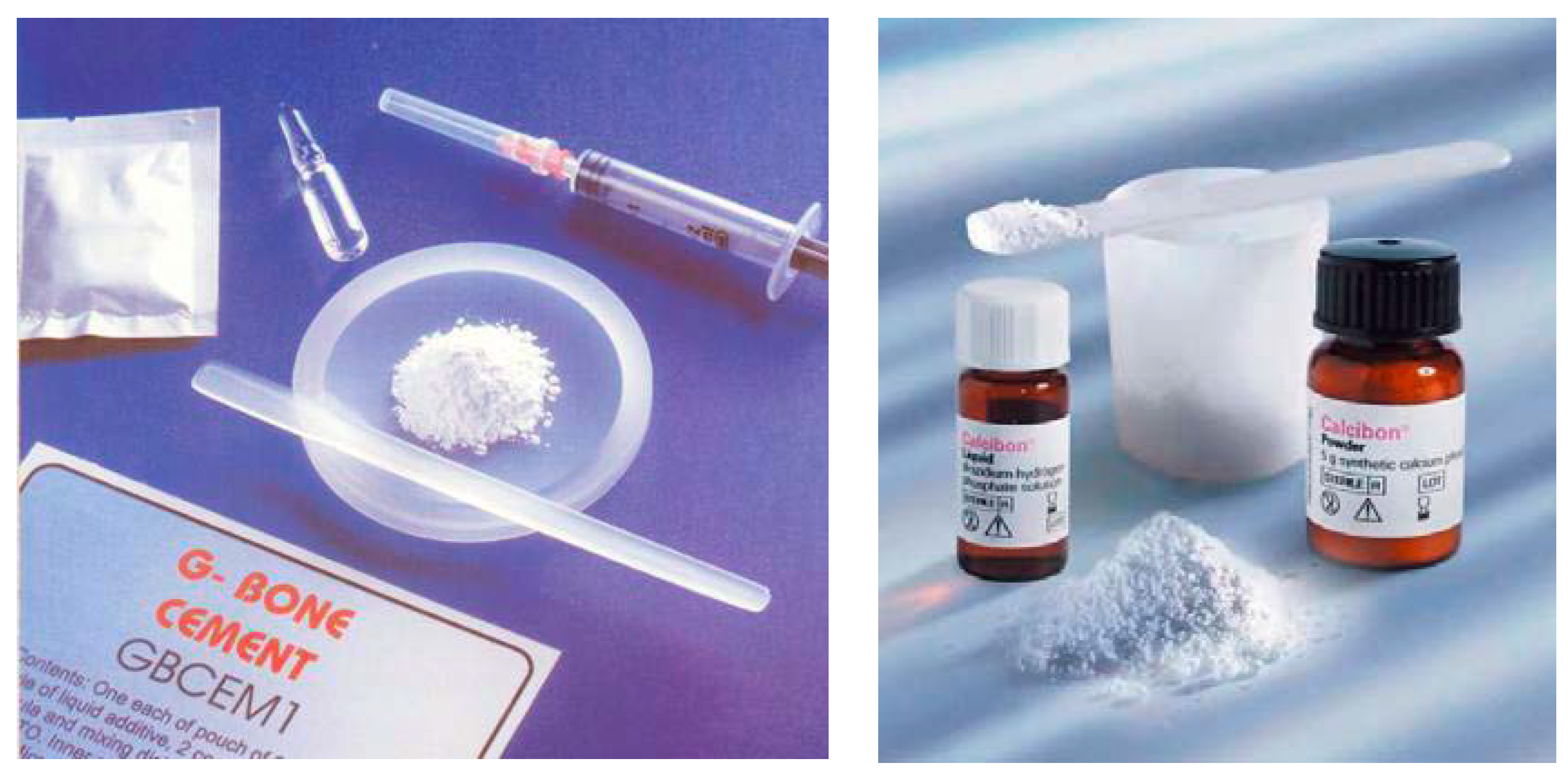
2.4. Market
| Product* | Manufacturer | Applications* |
|---|---|---|
| BoneSourceTM** | Striker Howmedica Osteonics (Rutherford, NJ) | Craniofacial |
| α-Bone Substitute Material (α-BSM®)*** | Etex Corporation (Cambridge, MA) | Filling of bone defects and voids, dental, craniofacial |
| Skeletal Repair Systems (SRS®) | Norian Corporation (Cupertino, CA) | Skeletal distal radius fractures, craniofacial |
3. Two Major Types of Calcium Orthophosphate Cements
3.1. Apatite Cements
3.2. Brushite Cements
4. Various Properties of Calcium Orthophosphate Cements
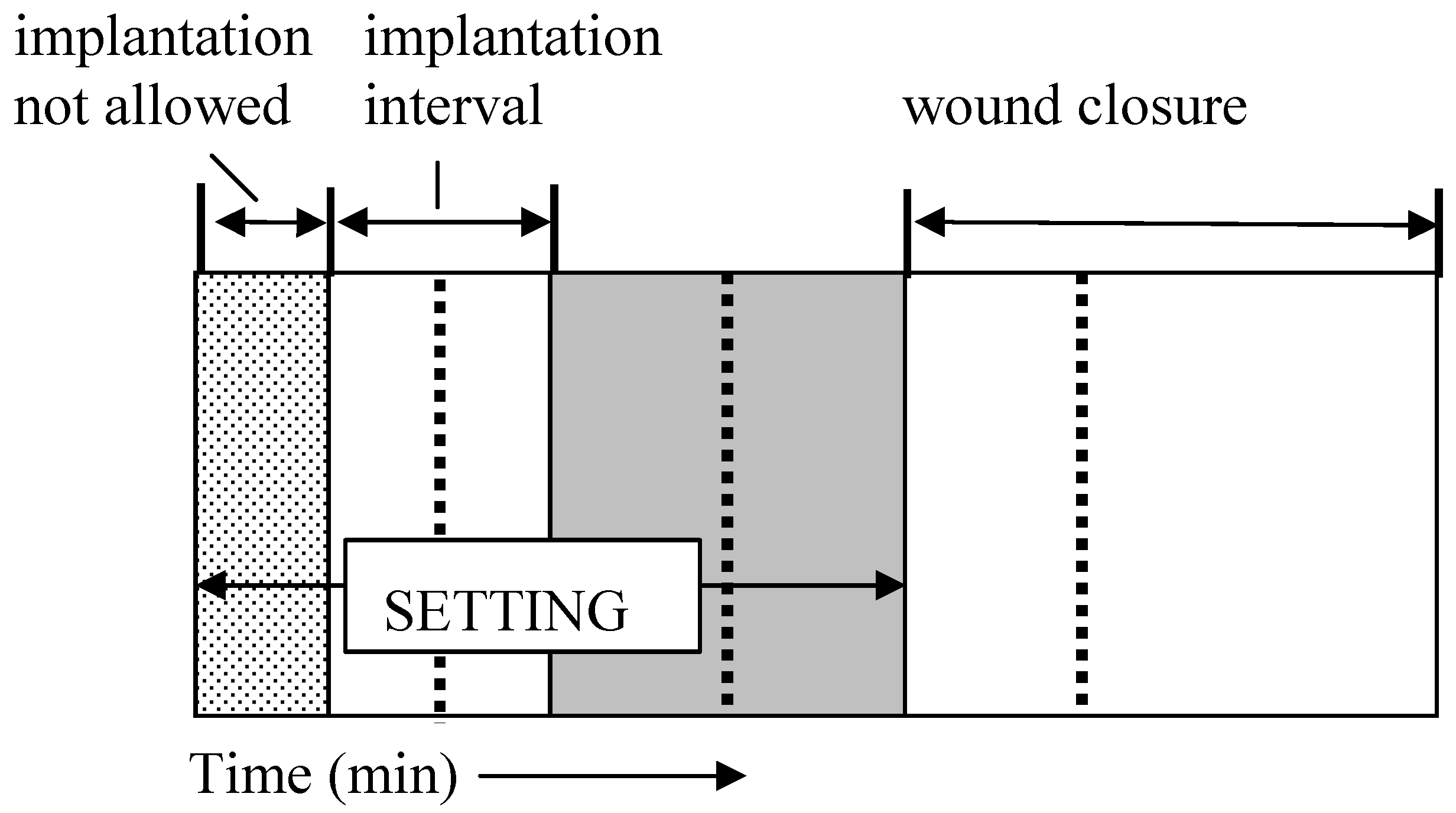
4.1. Setting and Hardening
4.2. Handling
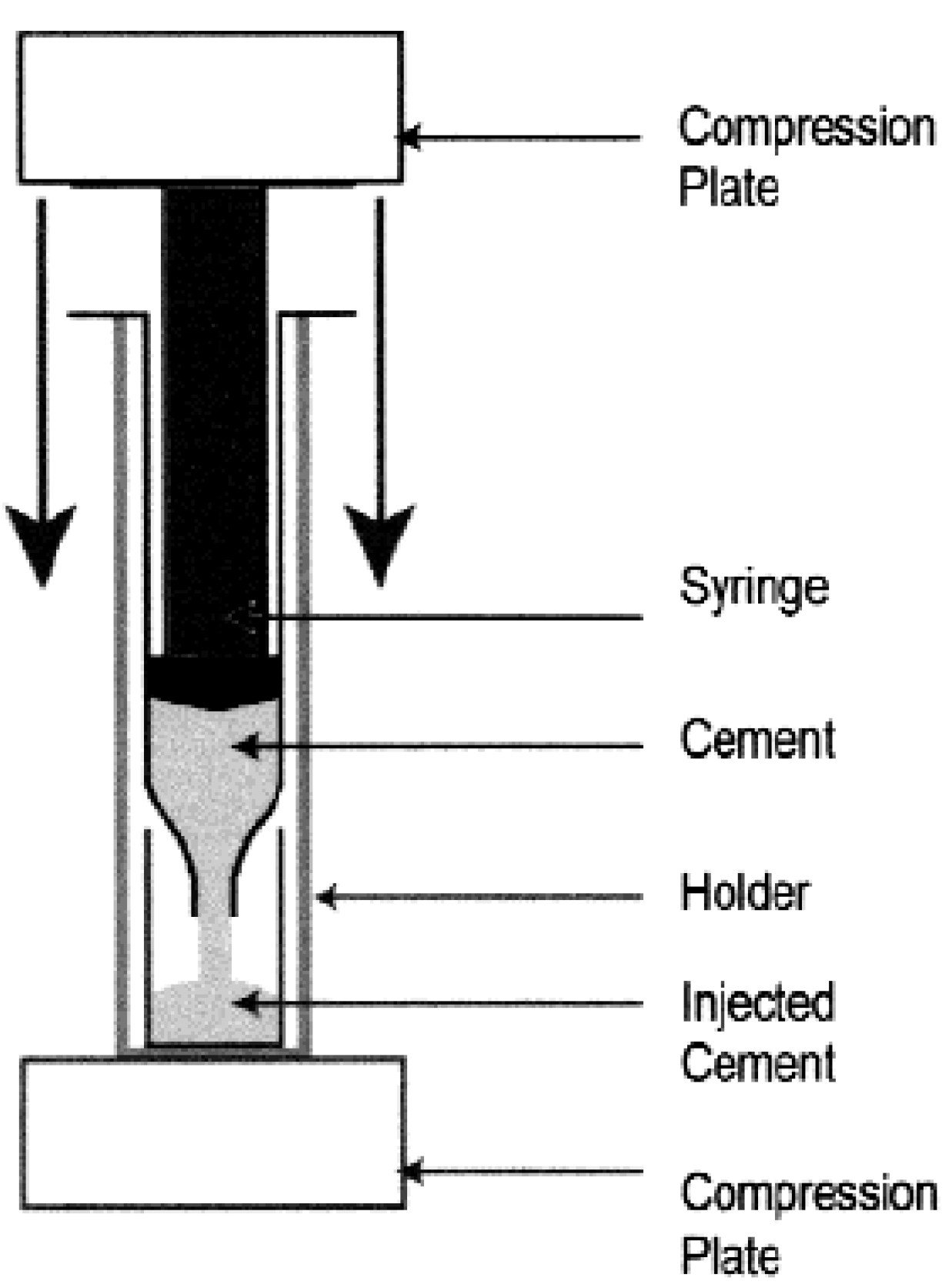
4.3. Physical and Rheological Properties
4.4. Using Additives
| Strategy | Approach | Sub-approaches |
|---|---|---|
| 1. Dissolution rate | 1.1. Change contact area between reagent and mixing liquid | 1.1.1. Change milling duration |
| 1.1.2. Use nano- or micro-sized powders | ||
| 1.2. Change solubility in the mixing liquid | 1.2.1. Use more/less soluble phase | |
| 1.2.2. Change of reaction pH | ||
| 1.3. Change saturation of the mixing liquid | ||
| 1.4. Use dissolution inhibitors in the mixing liquid | ||
| 1.5. Modify reagent surface | 1.5.1. Chemical change (pre-reaction) | |
| 1.5.2. Physical change (dissolution pits) | ||
| 2. Nucleation rate | 2.1. Use crystallization nuclei | |
| 2.2. Change the saturation of the reaction product in the mixing liquid | 2.2.1. Change of saturation | |
| 2.2.2. Change of end-product solubility | ||
| 2.3. Use nucleation inhibitors | ||
| 3. Growth rate | 3.1. Change the saturation of the reaction product in the mixing liquid | 3.1.1. Change of saturation |
| 3.1.2. Change of end-product solubility | ||
| 3.2. Use crystal growth inhibitors |
5. Bioresorption and Replacement of the Cements by Bones

6. The Mechanical Properties
7. Reinforced Calcium Orthophosphate Cement Composites and Concretes
8. Clinical and Medical Applications
8.1. Dental Applications
8.2. Craniofacial and Maxillofacial Applications
8.3. Orthopedic Applications
8.4. Vertebroplasty and Kyphoplasty
8.5. Drug Delivery
8.6. Brief Conclusions on the Medical Applications
9. Future Developments
| Advantages | Disadvantages |
|---|---|
|
|
10. Conclusions
References and Notes
- Albee, F.; Morrison, H. Studies in bone growth. Ann. Surg. 1920, 71, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Haldeman, K.; Moore, J. Influence of a local excess of calcium and phosphorus on the healing of fractures. Arch. Surg. 1934, 29, 385–396. [Google Scholar] [CrossRef]
- Ray, R.; Degge, J.; Gloyd, P.; Mooney, G. Bone regeneration. J. Bone Joint Surg. Am. 1952, 34A, 638–647. [Google Scholar]
- Getter, L.; Bhaskar, S.; Cutright, D.; Perez, B.; Brady, J.; Driskell, T.; O’Hara, M. Three biodegradable calcium orthophosphate slurry implants in bone. J. Oral Surg. 1972, 30, 263–268. [Google Scholar] [PubMed]
- Roy, D.; Linnehan, S. Hydroxyapatite formed from coral skeletal carbonate by hydrothermal exchange. Nature 1974, 247, 220–222. [Google Scholar] [CrossRef] [PubMed]
- Koster, K.; Karbe, E.; Kramer, H.; Heide, H.; Konig, R. Experimenteller Knochenersatz durch resorbierbare Calciumphosphat-Keramik. Langenbecks Arch. Chir. 1976, 341, 77–86. [Google Scholar] [CrossRef] [PubMed]
- Peelen, J.; Rejda, B.; Vermeiden, J.; de Groot, K. Sintered tri-calcium orthophosphate as bioceramic. Sci. Ceram. 1977, 9, 226–236. [Google Scholar]
- Jarcho, M.; Kay, J.; Gumaer, K.; Doremus, R.; Drobeck, H. Tissue, cellular and subcellular events at a bone-ceramic hydroxyapatite interface. J. Bioeng. 1977, 1, 79–92. [Google Scholar] [PubMed]
- Jarcho, M. Calcium phosphate ceramics as hard tissue prosthetics. Clin. Orthop. 1981, 157, 259–278. [Google Scholar] [PubMed]
- LeGeros, R.Z. Calcium phosphates in oral biology and medicine; Karger: Basel, Switzerland, 1991; p. 210. [Google Scholar]
- Dorozhkin, S.V. Calcium orthophosphates. J. Mater. Sci. 2007, 42, 1061–1095. [Google Scholar] [CrossRef]
- LeGeros, R.Z.; Chohayeb, A.; Shulman, A. Apatitic calcium phosphates: possible dental restorative materials. J. Dent. Res. 1982, 61, 343. [Google Scholar]
- Brown, W.E.; Chow, L.C. A new calcium phosphate setting cement. J. Dent. Res. 1983, 62, 672. [Google Scholar]
- Brown, W.E.; Chow, L.C. A new calcium phosphate water setting cemen. In Cements Research Progress; Brown, P.W., Ed.; American Ceramic Society: Westerville, OH, USA, 1986; pp. 352–379. [Google Scholar]
- Brown, W.E.; Chow, L.C. Dental restorative cement pastes. U.S. Patent No. 4,518,430, 21 May 1985. [Google Scholar]
- Gruninger, S.E.; Siew, C.; Chow, L.C.; O’Young, A.; Tsao, N.K.; Brown, W.E. Evaluation of the biocompatibility of a new calcium phosphate setting cement. J. Dent. Res. 1984, 63, 200. [Google Scholar]
- Tas, A.C. Porous, biphasic CaCO3-calcium phosphate biomedical cement scaffolds from calcite (CaCO3) powder. Int. J. Appl. Ceram. Technol. 2007, 4, 152–163. [Google Scholar] [CrossRef]
- Driskell, T.D.; Heller, A.L.; Koenigs, J.F. Dental treatments. U.S. Patent No. 3,913,229, 21 October 1975. [Google Scholar]
- Kingery, W.D. Cold setting properties. J. Am. Ceram. Soc. 1950, 33, 242–247. [Google Scholar] [CrossRef]
- Driessens, F.C.M.; Planell, J.A.; Gil, F.J. Encyclopedic Handbook of Biomaterials and Bioengineering, Part B, Applications; Wise, D.L., Trantolo, D.J., Altobelli, D.E., Yaszemski, M.J., Gresser, J.D., Schwarz, E.R., Eds.; Marcel Dekker: New York, USA, 1995; Volume 2, pp. 855–877. [Google Scholar]
- Schmitz, J.P.; Hollinger, J.O.; Milan, S.B. Reconstruction of bone using calcium phosphate bone cements: a critical review. J. Oral Maxillofac. Surg. 1999, 57, 1122–1126. [Google Scholar] [CrossRef] [PubMed]
- Gbureck, U.; Barralet, J.E.; Spatz, K.; Grover, L.M.; Thull, R. Ionic modification of calcium phosphate cement viscosity. Part I: Hypodermic injection and strength improvement of apatite cement. Biomaterials 2004, 25, 2187–2195. [Google Scholar] [CrossRef] [PubMed]
- Tagaya, M.; Goto, H.; Iinuma, M.; Wakamatsu, N.; Tamura, Y.; Doi, Y. Development of self-setting Te-Cp/alpha-TCP cement for pulpotomy. Dent. Mater. J. 2005, 24, 555–561. [Google Scholar] [CrossRef]
- Driessens, F.C.M.; Boltong, M.G.; Khairoun, I.; de Maeyer, E.A.P.; Ginebra, M.P.; Wenz, R.; Planell, J.A.; Verbeeck, R.M.H. Applied aspects of calcium phosphate bone cemen. In Biomaterials Engineering and Devices: Human Applications; Wise, D.L., Trantolo, D.J., Lewandrowski, K.U., Gresser, J.D., Cattaneo, M.V., Eds.; Humana Press: Totowa, NJ, USA, 2000; Vol. 2, pp. 253–260. [Google Scholar]
- Driessens, F.C.M.; Planell, J.A.; Boltong, M.G.; Khairoun, I.; Ginebra, M.P. Osteotransductive bone cements. Proc. Inst. Mech. Eng. H: J. Eng. Med. 1998, 212, 427–435. [Google Scholar] [CrossRef]
- Frankenburg, E.P.; Goldstein, S.A.; Bauer, T.W.; Harris, S.A.; Poser, R.D. Biomechanical and histological evaluation of a calcium phosphate cement. J. Bone Joint Surg. Am. 1998, 80A, 1112–1124. [Google Scholar] [PubMed]
- Frayssinet, P.; Gineste, L.; Conte, P.; Fages, J.; Rouquet, N. Short-term implantation effects of a DCPD-based calcium phosphate cement. Biomaterials 1998, 19, 971–977. [Google Scholar] [CrossRef] [PubMed]
- Rey, C.; Tofighi, A.; Mounic, S.; Combes, C.; Lee, D. Biomimetism and calcium phosphate cements. In Actualités en Biomatériaux; Mainard, D., Louis, J.P., Eds.; Romillat: Paris, France, 2002; Volume 6, pp. 27–37. [Google Scholar]
- Combes, C.; Bareille, R.; Rey, C. Calcium carbonate-calcium phosphate mixed cement compositions for bone reconstruction. J. Biomed. Mater. Res. A 2006, 79A, 318–328. [Google Scholar] [CrossRef] [PubMed]
- Bohner, M.; Gbureck, U.; Barralet, J.E. Technological issues for the development of more efficient calcium phosphate bone cements: a critical assessment. Biomaterials 2005, 26, 6423–6429. [Google Scholar] [CrossRef] [PubMed]
- Ikenaga, M.; Hardouin, P.; Lemaître, J.; Andrianjatovo, H.; Flautre, B. Biomechanical characterization of a biodegradable calcium phosphate hydraulic cement: a comparison with porous biphasic calcium phosphate ceramics. J. Biomed. Mater. Res. 1998, 40, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Ginebra, M.P.; Traykova, T.; Planell, J.A. Calcium phosphate cements: competitive drug carriers for the musculoskeletal system? Biomaterials 2006, 27, 2171–2177. [Google Scholar] [CrossRef] [PubMed]
- Ginebra, M.P.; Traykova, T.; Planell, J.A. Calcium phosphate cements as bone drug delivery systems: a review. J. Control. Rel. 2006, 113, 102–110. [Google Scholar] [CrossRef]
- It is interesting to note that calcium sulfate (gypsum or plaster of Paris) has been implanted into fracture gaps and voids of bones for more than 100 years [35,36,37].
- Dreesmann, H. Knochenplombierung bei Hohlenforigen Defekten des Knochens. Beitr. Klin. Chir. 1892, 9, 804–810. [Google Scholar]
- Peltier, L.F. The use of plaster of Paris to fill defects in bone. Clin. Orthop. 1961, 21, 1–29. [Google Scholar] [PubMed]
- Kelly, C.M.; Wilkins, R.M.; Gitelis, S.; Hartjen, C.; Watson, J.T.; Kim, P.T. The use of a surgical grade calcium sulfate as a bone graft substitute: results of a multicenter trial. Clin. Orthop. 2001, 382, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Bohner, M. New hydraulic cements based on α-tricalcium phosphate – calcium sulfate dihydrate mixtures. Biomaterials 2004, 25, 741–749. [Google Scholar] [CrossRef] [PubMed]
- Fernández, E.; Vlad, M.D.; Gel, M.M.; Lopez, J.; Torres, R.; Cauich, J.V.; Bohner, M. Modulation of porosity in apatitic cements by the use of α-tricalcium phosphate – calcium sulphate dihydrate mixtures. Biomaterials 2005, 26, 3395–3404. [Google Scholar]
- Nilsson, M.; Fernández, E.; Sarda, S.; Lidgren, L.; Planell, J.A. Characterization of a novel calcium phosphate/sulphate bone cement. J. Biomed. Mater. Res. 2002, 61, 600–607. [Google Scholar] [CrossRef] [PubMed]
- Grover, L.M.; Gbureck, U.; Wright, A.J.; Tremaynec, M.; Barralet, J.E. Biologically mediated resorption of brushite cement in vitro. Biomaterials 2006, 27, 2178–2185. [Google Scholar] [CrossRef] [PubMed]
- Grover, L.M.; Gbureck, U.; Wright, A.J.; Barralet, J.E. Cement formulations in the calcium phosphate H2O – H3PO4 – H4P2O7 system. J. Am. Ceram. Soc. 2005, 88, 3096–3103. [Google Scholar] [CrossRef]
- Grover, L.M.; Gbureck, U.; Young, A.M.; Wright, A.J.; Barralet, J.E. Temperature dependent setting kinetics and mechanical properties of β-TCP – pyrophosphoric acid bone cement. J. Mater. Chem. 2005, 46, 4955–4962. [Google Scholar] [CrossRef]
- Lilley, K.J.; Gbureck, U.; Wright, A.J.; Knowles, J.C.; Farrar, D.F.; Barralet, J.E. Brushite cements from polyphosphoric acid, calcium phosphate systems. J. Am. Ceram. Soc. 2007, 90, 1892–1898. [Google Scholar] [CrossRef]
- Fernández, E.; Planell, J.A.; Best, S.M. Precipitation of carbonated apatite in the cement system α-Ca3(PO4)2 – Ca(H2PO4)2 – CaCO3. J. Biomed. Mater. Res. 1999, 47, 466–471. [Google Scholar] [CrossRef] [PubMed]
- Calafiori, A.R.; di Marco, G.; Martino, G.; Marotta, M. Preparation and characterization of calcium phosphate biomaterials. J. Mater. Sci. Mater. Med. 2007, 18, 2331–2338. [Google Scholar] [CrossRef] [PubMed]
- Kon, M.; Hirakata, L.M.; Miyamoto, Y.; Kasahara, H.; Asaoka, K. Strengthening of calcium phosphate cement by compounding calcium carbonate whiskers. Dent. Mater. J. 2005, 24, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Serraj, S.; Michailesco, P.; Margerit, J.; Bernard, B.; Boudeville, P. Study of a hydraulic calcium phosphate cement for dental applications. J. Mater. Sci. Mater. Med. 2002, 13, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Nurit, L.; Margerit, J.; Terol, A.; Boudeville, P. pH-metric study of the setting reaction of monocalcium phosphate monohydrate/calcium oxide-based cements. J. Mater. Sci. Mater. Med. 2002, 13, 1007–1014. [Google Scholar] [CrossRef] [PubMed]
- Boudeville, P.; Serraj, S.; Leloup, J.M.; Margerit, J.; Pauvert, B.; Terol, A. Physical properties and self-setting mechanism of calcium phosphate cements from calcium bis-dihydrogenophosphate monohydrate and calcium oxide. J. Mater. Sci. Mater. Med. 1999, 10, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Michaïlesco, P.; Kouassi, M.; Briak, H.E.; Armynot, A.; Boudeville, P. Antimicrobial activity and tightness of a DCPD – CaO-based hydraulic calcium phosphate cement for root canal filling. J. Biomed. Mater. Res. B Appl. Biomater. 2005, 74, 760–767. [Google Scholar] [CrossRef] [PubMed]
- Briak, H.E.; Durand, D.; Nurit, J.; Munier, S.; Pauvert, B.; Boudeville, P. Study of a hydraulic dicalcium phosphate dihydrate/calcium oxide-based cement for dental applications. J. Biomed. Mater. Res. :Appl. Biomater. 2002, 63, 447–453. [Google Scholar] [CrossRef]
- Briak, H.E.; Durand, D.; Boudeville, P. Study of a hydraulic DCPA/CaO-based cement for dental applications. J. Mater. Sci. Mater. Med. 2008, 19, 737–744. [Google Scholar] [CrossRef] [PubMed]
- Takagi, S.; Chow, L.C.; Ishikawa, K. Formation of hydroxyapatite in new calcium phosphate cements. Biomaterials 1998, 19, 1593–1599. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Troczynski, T.; Liu, D.M. Influence of apatite seeds on the synthesis of calcium phosphate cement. Biomaterials 2002, 23, 2751–2760. [Google Scholar] [CrossRef] [PubMed]
- Hsu, H.C.; Chiu, C.Y.; Tuan, W.H.; Lee, H.Y. Structural stability of calcium phosphate cement during aging in water. Mater. Sci. Eng. C 2008, 28, 429–433. [Google Scholar] [CrossRef]
- Roemhildt, M.L.; McGee, T.D.; Wagner, S.D. Novel calcium phosphate composite bone cement, strength and bonding properties. J. Mater. Sci. Mater. Med. 2003, 14, 137–141. [Google Scholar] [CrossRef]
- Roemhildt, M.L.; Wagner, S.D.; McGee, T.D. Characterization of a novel calcium phosphate composite bone cement: flow, setting, and aging properties. J. Mater. Sci. Mater. Med. 2006, 17, 1127–1132. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ye, J.; Wang, Y.; Chen, L. Self-setting properties of a β-dicalcium silicate reinforced calcium phosphate cement. J. Biomed. Mater. Res. B Appl. Biomater. 2007, 82B, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Huan, Z.; Chang, J. Novel tricalcium silicate/monocalcium phosphate monohydrate composite bone cement. J. Biomed. Mater. Res. B Appl. Biomater. 2007, 82B, 352–359. [Google Scholar] [CrossRef] [PubMed]
- Huan, Z.; Chang, J. Calcium-phosphate-silicate composite bone cement, self-setting properties and in vitro bioactivity. J. Mater. Sci. Mater. Med. 2009, 20, 833–841. [Google Scholar] [CrossRef] [PubMed]
- Huan, Z.; Chang, J. Novel bioactive composite bone cements based on the β-tricalcium phosphate – monocalcium phosphate monohydrate composite cement system. Acta Biomater 2009, in press. [Google Scholar]
- Guo, D.; Xu, K.; Zhao, X.; Han, Y. Development of a strontium-containing hydroxyapatite bone cement. Biomaterials 2005, 26, 4073–4083. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ye, J. Variation of crystal structure of hydroxyapatite in calcium phosphate cement by the substitution of strontium ions. J. Mater. Sci. Mater. Med. 2008, 19, 1183–1186. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.; Su, J.C.; Wei, J.; Guo, H.; Liu, C.S. Injectable bioactive calcium-magnesium phosphate cement for bone regeneration. Biomed. Mater. 2008, 3, 044105:1–044105:7. [Google Scholar]
- Wu, F.; Wei, J.; Guo, H.; Chen, F.P.; Hong, H.; Liu, C.S. Self-setting bioactive calcium-magnesium phosphate cement with high strength and degradability for bone regeneration. Acta Biomater. 2008, 4, 1873–1884. [Google Scholar] [CrossRef] [PubMed]
- Pina, S.; Olhero, S.M.; Gheduzzi, S.; Miles, A.W.; Ferreira, J.M.F. Influence of setting liquid composition and liquid-to-powder ratio on properties of a Mg-substituted calcium phosphate cement. Acta Biomater 2009, in press. [Google Scholar]
- Acarturk, O.; Lehmicke, M.; Aberman, H.; Toms, D.; Hollinger, J.O.; Fulmer, M.T. Bone healing response to an injectable calcium phosphate cement with enhanced radiopacity. J. Biomed. Mater. Res. B Appl. Biomater. 2008, 86, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Gbureck, U.; Knappe, O.; Grover, L.M.; Barralet, J.E. Antimicrobial potency of alkali ion substituted calcium phosphate cements. Biomaterials 2005, 26, 6880–6886. [Google Scholar] [CrossRef] [PubMed]
- Driessens, F.C.M.; Boltong, M.G.; de Mayer, E.A.P.; Wenz, R.; Nies, B.; Planell, J.A. The Ca/P range of nanoapatitic calcium phosphate cements. Biomaterials 2002, 23, 4011–4017. [Google Scholar] [CrossRef] [PubMed]
- Gbureck, U.; Thull, R.; Barralet, J.E. Alkali ion substituted calcium phosphate cement formation from mechanically activated reactants. J. Mater. Sci. Mater. Med. 2005, 16, 423–427. [Google Scholar] [CrossRef] [PubMed]
- Doi, Y.; Shimizu, Y.; Moriwaki, Y.; Aga, M.; Iwanaga, H.; Shibutani, T.; Yamamoto, K.; Iwayama, Y. Development of a new calcium phosphate cement that contains sodium calcium phosphate. Biomaterials 2001, 22, 847–854. [Google Scholar] [CrossRef] [PubMed]
- Lilley, J.; Gbureck, U.; Knowles, J.C.; Farrar, D.F.; Barralet, J.E. Cement from magnesium substituted hydroxyapatite. J. Mater. Sci. Mater. Med. 2005, 16, 455–460. [Google Scholar] [CrossRef] [PubMed]
- Ni, G.X.; Lu, W.W.; Tang, B.; Ngan, A.H.W.; Chiu, K.Y.; Cheung, K.M.C.; Li, Z.Y.; Luk, K.D.K. Effect of weight-bearing on bone-bonding behavior of strontium-containing hydroxyapatite bone cement. J. Biomed. Mater. Res. A 2007, 83, 570–576. [Google Scholar] [CrossRef] [PubMed]
- Alkhraisat, M.H.; Mariño, F.T.; Rodríguez, C.R.; Jerez, L.B.; Cabarcos, E.L. Combined effect of strontium and pyrophosphate on the properties of brushite cements. Acta Biomater. 2008, 4, 664–670. [Google Scholar] [CrossRef] [PubMed]
- Yao, Z.P.; Liu, W.G.; Ni, G.X. Biology characteristics and clinical application of strontium substituted hydroxyapatite bone cement. J. Clin. Rehabil. Tissue Eng. Res. 2008, 12, 7151–7154. [Google Scholar]
- Li, S.; Liu, B.; Cheng, J.; Hu, J. Composite cement of magnesium-bearing phosphoaluminate-hydroxyapatite reinforced by treated raw silk fiber. Cem. Concr. Compos. 2008, 30, 347–352. [Google Scholar] [CrossRef]
- Fadeeva, I.V.; Barinov, S.M.; Komlev, V.S.; Fedotov, D.A.; Durisin, J.; Medvecky, L. Apatite formation in the reaction-setting mixture of Ca(OH)2 – KH2PO4 system. J. Biomed. Mater. Res. A 2004, 70, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Tas, A.C. Use of vaterite and calcite in forming calcium phosphate cement scaffolds. Ceram. Eng. Sci. Proc. 2008, 28, 135–150. [Google Scholar]
- Fernández, E.; Vlad, M.D.; Hamcerencu, M.; Darie, A.; Torres, R.; Lopez, J. Effect of iron on the setting properties of α-TCP bone cements. J. Mater. Sci. 2005, 40, 3677–3682. [Google Scholar] [CrossRef]
- Vlad, M.D.; del Valle, L.J.; Poeata, I.; Barracó, M.; López, J.; Torres, R.; Fernández, E. Injectable iron-modified apatitic bone cement intended for kyphoplasty, cytocompatibility study. J. Mater. Sci. Mater. Med. 2008, 19, 3575–3583. [Google Scholar] [CrossRef] [PubMed]
- http://en.wikipedia.org/wiki/Cement accessed in January 2009.
- There are some differences between TTCP + DCPD and TTCP + DCPA cements. Due to a higher solubility of DCPD (Table 1), a TTCP + DCPD cement sets faster than a TTCP + DCPA cement. Besides, injectability of a TTCP + DCPD cement is better [84,85].
- Burguera, E.F.; Xu, H.H.K.; Weir, M.D. Injectable and rapid-setting calcium phosphate bone cement with dicalcium phosphate dihydrate. J. Biomed. Mater. Res. B Appl. Biomater. 2006, 77, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Burguera, E.F.; Guitian, F.; Chow, L.C. A water setting tetracalcium phosphate – dicalcium phosphate dihydrate cement. J. Biomed. Mater. Res. A 2004, 71, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Driessens, F.C.M.; Boltong, M.G.; Bermudez, O.; Planell, J.A. Formulation and setting times of some calcium orthophosphate cements, a pilot study. J. Mater. Sci. Mater. Med. 1993, 4, 503–508. [Google Scholar] [CrossRef]
- Chow, L.C.; Markovic, M.; Takagi, S. Calcium phosphate cements. In Cements research progress; Struble, L.J., Ed.; American Ceramic Society: Westerville, OH, USA, 1998; Chapter 7; pp. 215–238. [Google Scholar]
- Driessens, F.C.M.; Boltong, M.G.; Bermudez, O.; Planell, J.A.; Ginebra, M.P.; Fernández, E. Effective formulations for the preparation of calcium phosphate bone cements. J. Mater. Sci. Mater. Med. 1994, 5, 164–170. [Google Scholar] [CrossRef]
- Kurashina, K.; Hirano, M.; Kotani, A.; Klein, C.P.A.T.; de Groot, K. In vivo study of calcium phosphate cements, implantation of an α-tricalcium phosphate/dicalcium phosphate dibasic/tetracalcium phosphate monoxide cement paste. Biomaterials 1997, 18, 539–543. [Google Scholar] [CrossRef] [PubMed]
- Friedman, C.D.; Costantino, P.D.; Takagi, S.; Chow, L.C. BoneSourceTM hydroxyapatite cement, a novel biomaterial for craniofacial skeletal tissue engineering and reconstruction. J. Biomed. Mater. Res. 1998, 43, 428–432. [Google Scholar] [CrossRef] [PubMed]
- Khairoun, I.; Boltong, M.G.; Driessens, F.C.M.; Planell, J.A. Effect of calcium carbonate on the compliance of apatitic calcium phosphate bone cement. Biomaterials 1997, 18, 1535–1539. [Google Scholar] [CrossRef] [PubMed]
- Fernández, E.; Gil, F.J.; Best, S.M.; Ginebra, M.P.; Driessens, F.C.M.; Planell, J.A. Improvement of the mechanical properties of new calcium phosphate bone cements in the CaHPO4 – α-Ca3(PO4)2 system, compressive strength and microstructural development. J. Biomed. Mater. Res. 1998, 41, 560–567. [Google Scholar] [CrossRef] [PubMed]
- Fukase, Y.; Eanes, E.D.; Takagi, S.; Chow, L.C.; Brown, W.E. Setting reactions and compressive strengths of calcium phosphate cements. J. Dent. Res. 1990, 69, 1852–1856. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Monroe, E.A. Calcium phosphate dental cements. Mat. Res. Soc. Symp. Proc. 1991, 179, 25–39. [Google Scholar] [CrossRef]
- Ishikawa, K.; Miyamoto, Y.; Kon, M.; Nagayama, M.; Asaoka, K. Non-decay type fast-setting calcium orthophosphate cement, composite with sodium alginate. Biomaterials 1995, 16, 527–532. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.H.K.; Quinn, J.B.; Takagi, S.; Chow, L.C. Processing and properties of strong and non-rigid calcium phosphate cement. J. Dent. Res. 2002, 81, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.K.; Lim, B.S.; Kim, C.W. Mechanical properties of calcium phosphate based dental filling and regeneration materials. J. Oral Rehabil. 2003, 30, 418–425. [Google Scholar] [CrossRef] [PubMed]
- Ginebra, M.P.; Fernández, E.; de Mayer, E.A.P.; Verbeeck, R.M.H.; Boltong, M.G.; Ginebra, J.; Driessens, F.C.M.; Planell, J.A. Setting reaction and hardening of an apatitic calcium phosphate cement. J. Dent. Res. 1997, 76, 905–912. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Shen, W.; Gu, Y.; Hu, L. Mechanism of the hardening process for a hydroxyapatite cement. J. Biomed. Mater. Res. 1997, 35, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Driessens, F.C.M.; de Mayer, E.A.P.; Fernández, E.; Boltong, M.G.; Berger, G.; Verbeeck, R.M.H.; Ginebra, M.P.; Planell, J.A. Amorphous calcium phosphate cements and their transformation into calcium deficient hydroxyapatite. Bioceramics 1996, 9, 231–234. [Google Scholar]
- Driessens, F.C.M.; Planell, J.A.; Gil, F. Calcium phosphate bone cements. In Encyclopedic Handbook of Biomaterials and Bioengineering; Wise, D.L., Trantolo, D.J., Altobelli, D.E., Yaszemski, M.J., Cresser, J.D., Schwartz, E.R., Eds.; Marcel Dekker: New York, USA, 1995; Part B, Volume 2, pp. 855–877. [Google Scholar]
- Lemaître, J. Injectable calcium phosphate hydraulic cements: New developments and potential applications. Inn. Tech. Biol. Med. 1995, 16, 109–120. [Google Scholar]
- In the vast majority cases, a precipitated poorly crystalline HA and CDHA are undistinguishable and might be considered as synonyms [11].
- To honor Prof. George Jarvis Brush (1831 – 1912), an American mineralogist, Yale University, New Haven, Connecticut, USA.
- Xia, Z.; Grover, L.M.; Huang, Y.; Adamopoulos, I.E.; Gbureck, U.; Triffitt, J.T.; Shelton, R.M.; Barralet, J.E. In vitro biodegradation of three brushite calcium phosphate cements by a macrophage cell-line. Biomaterials 2006, 27, 4557–4565. [Google Scholar] [CrossRef] [PubMed]
- Bermudez, O.; Boltong, M.G.; Driessens, F.C.M.; Planell, J.A. Development of an octocalcium phosphate cement. J. Mater. Sci. Mater. Med. 1994, 5, 144–146. [Google Scholar] [CrossRef]
- In early 1990-s, depending on the type of calcium orthophosphate formed after the setting, five groups of the cement formulations were thought to exist: DCPD, CDHA, HA, ACP and OCP [88,108], while currently only two cement groups remain.
- Khairoun, I.; Boltong, M.G.; Driessens, F.C.M.; Planell, J.A. Limited compliance of some apatitic calcium phosphate bone cements with clinical requirements. J. Mater. Sci. Mater. Med. 1998, 9, 667–671. [Google Scholar] [CrossRef] [PubMed]
- Lacout, J.; Mejdoubi, E.; Hamad, M. Crystallization mechanisms of calcium orthophosphate cement for biological uses. J. Mater. Sci. Mater. Med. 1996, 7, 371–374. [Google Scholar] [CrossRef]
- Song, Y.; Feng, Z.; Wang, T. In situ study on the curing process of calcium phosphate bone cement. J. Mater. Sci. Mater. Med. 2007, 18, 1185–1193. [Google Scholar] [CrossRef] [PubMed]
- Weiss, D.D.; Sachs, M.A.; Woodard, C.R. Calcium phosphate bone cements: A comprehensive review. J. Long Term Eff. Med. Implants 2003, 13, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Fernández, E.; Gil, F.J.; Ginebra, M.P.; Driessens, F.C.M.; Planell, J.A.; Best, S.M. Calcium phosphate bone cements for clinical applications. Part I: Solution chemistry. J. Mater. Sci. Mater. Med. 1999, 10, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Brown, W.E.; Chow, L.C. A new calcium phosphate water-setting cemen. In Cements Research Progress; Brown, P.W., Ed.; American Ceramic Society: Westerville, OH, USA, 1986; pp. 351–379. [Google Scholar]
- Hatim, Z.; Freche, M.; Keribech, A.; Lacout, J.L. The setting mechanism of a phosphocalcium biological cement. Ann. Chim. Sci. Mat. 1998, 23, 65–68. [Google Scholar] [CrossRef]
- Ishikawa, K.; Asaoka, K. Estimation of ideal mechanical strength and critical porosity of calcium phosphate cement. J. Biomed. Mater. Res. 1995, 29, 1537–1543. [Google Scholar] [CrossRef] [PubMed]
- Chow, L.C. Development of self-setting calcium phosphate cements. J. Ceram. Soc. Jpn. 1991, 99, 954–964. [Google Scholar] [CrossRef]
- Chow, L.C. Calcium phosphate cements: Chemistry, properties and applications. Mat. Res. Soc. Symp. Proc. 2000, 599, 27–37. [Google Scholar] [CrossRef]
- Chow, L.C. Calcium phosphate cements. In Octacalcium Phosphate; Chow, L.C., Eanes, E.D., Eds.; Karger: Basel, Switzerland, 2001; Volume 18, pp. 148–163. [Google Scholar]
- Brown, P.W.; Fulmer, M.T. Kinetics of hydroxyapatite formation at low temperature. J. Am. Ceram. Soc. 1991, 74, 934–940. [Google Scholar] [CrossRef]
- TenHuisen, K.S.; Brown, P.W. The formation of hydroxyapatite-ionomer cements at 38 °C. J. Dent. Res. 1994, 3, 598–606. [Google Scholar]
- Ishikawa, K.; Takagi, S.; Chow, L.C.; Suzuki, K. Reaction of calcium phosphate cements with different amounts of tetracalcium phosphate and dicalcium phosphate anhydrous. J. Biomed. Mater. Res. 1999, 46, 504–510. [Google Scholar] [CrossRef] [PubMed]
- Matsuya, S.; Takagi, S.; Chow, L.C. Effect of mixing ratio and pH on the reaction between Ca4(PO4)2O and CaHPO4. J. Mater. Sci. Mater. Med. 2000, 11, 305–311. [Google Scholar] [CrossRef] [PubMed]
- Burguera, E.F.; Guitian, F.; Chow, L.C. Effect of the calcium to phosphate ratio of tetracalcium phosphate on the properties of calcium phosphate bone cement. J. Biomed. Mater. Res. A 2008, 85A, 674–683. [Google Scholar] [CrossRef] [PubMed]
- Lemaître, J.; Mirtchi, A.A.; Mortier, A. Calcium phosphate cements for medical use: state of the art and perspectives of development. Silic. Ind. 1987, 9-10, 141–146. [Google Scholar]
- Bajpai, P.; Fuchs, C.; McCullum, D. Development of tricalcium orthophosphate ceramic cemen. In Quantitative characterization and performance of porous implants for hard tissue applications; Lemons, J., Ed.; Am. Soc. Test. Mater.: Philadelphia, USA, 1987; ASTM STP 953; pp. 377–388. [Google Scholar]
- Mirtchi, A.A.; Lemaître, J.; Terao, N. Calcium phosphate cements: study of the β-tricalcium phosphate – monocalcium phosphate system. Biomaterials 1989, 10, 475–480. [Google Scholar] [CrossRef] [PubMed]
- Bohner, M.; van Landuyt, P.; Merkle, H.P.; Lemaître, J. Composition effects on the pH of a hydraulic calcium orthophosphate cement. J. Mater. Sci. Mater. Med. 1997, 8, 675–681. [Google Scholar] [CrossRef] [PubMed]
- Bohner, M.; Lemaître, J.; Ring, T.A. Effects of sulfate, pyrophosphate and citrate ions on the physiochemical properties of cements made of β-tricalcium phosphate – phosphoric acid – water mixtures. J. Am. Ceram. Soc. 1996, 79, 1427–1434. [Google Scholar] [CrossRef]
- Grover, L.M.; Hofmann, M.P.; Gbureck, U.; Kumarasami, B.; Barralet, J.E. Frozen delivery of brushite calcium phosphate cements. Acta Biomater. 2008, 4, 1916–1923. [Google Scholar] [CrossRef] [PubMed]
- Fernández, E.; Gil, F.J.; Best, S.M.; Ginebra, M.P.; Driessens, F.C.M.; Planell, J.A. The cement setting reaction in the CaHPO4 – α-Ca3(PO4)2 system: an X-ray diffraction study. J. Biomed. Mater. Res. 1998, 42, 403–406. [Google Scholar] [CrossRef] [PubMed]
- Fernández, E.; Gil, F.J.; Ginebra, M.P.; Driessens, F.C.M.; Planell, J.A.; Best, S.M. Production and characterisation of new calcium phosphate bone cements in the CaHPO4 – α-Ca3(PO4)2 system: pH, workability and setting times. J. Mater. Sci. Mater. Med. 1999, 10, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Barralet, J.E.; Lilley, K.J.; Grover, L.M.; Farrar, D.F.; Ansell, C.; Gbureck, U. Cements from nanocrystalline hydroxyapatite. J. Mater. Sci. Mater. Med. 2004, 15, 407–411. [Google Scholar] [CrossRef] [PubMed]
- Lilley, K.J.; Gbureck, U.; Wright, A.J.; Farrar, D.F.; Barralet, J.E. Cement from nanocrystalline hydroxyapatite, effect of calcium phosphate ratio. J. Mater. Sci. Mater. Med. 2005, 16, 1185–1190. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ye, J.; Wang, Y.; Wu, X.; Bai, B. Control of crystallinity of hydrated products in a calcium phosphate bone cement. J. Biomed. Mater. Res. A 2007, 81A, 781–790. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ye, J.; Wang, H. Effects of additives on the rheological properties and injectability of a calcium phosphate bone substitute material. J. Biomed. Mater. Res. B Appl. Biomater. 2006, 78, 259–264. [Google Scholar] [CrossRef] [PubMed]
- Tofighi, A.; Schaffer, K.; Palazzolo, R. Calcium phosphate cement (CPC): A critical development path. Key Eng. Mater. 2008, 361-363, 303–306. [Google Scholar] [CrossRef]
- de Maeyer, E.A.P.; Verbeeck, R.M.H.; Vercruysse, C.W.J. Conversion of octacalcium phosphate in calcium phosphate cements. J. Biomed. Mater. Res. 2000, 52, 95–106. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ye, J.; Wang, Y. Hydration mechanism of a novel PCCP + DCPA cement system. J. Mater. Sci. Mater. Med. 2008, 19, 813–816. [Google Scholar] [CrossRef] [PubMed]
- Zoulgami, M.; Lucas, A.; Briard, P.; Gaudé, J. A self-setting single-component calcium phosphate cement. Biomaterials 2001, 22, 1933–1937. [Google Scholar] [CrossRef] [PubMed]
- Knaack, D.; Goad, M.E.; Aiolova, M.; Rey, C.; Tofighi, A.; Chakravarthy, P.; Lee, D.D. Resorbable calcium phosphate bone substitute. J. Biomed. Mater. Res. 1998, 43, 399–409. [Google Scholar] [CrossRef] [PubMed]
- Tofighi, A.; Mounic, S.; Chakravarthy, P.; Rey, C.; Lee, D. Setting reactions involved in injectable cements based on amorphous calcium phosphate. Key Eng. Mater. 2001, 192-195, 769–772. [Google Scholar] [CrossRef]
- Monma, H.; Kanazawa, T. The hydration of α-tricalcium phosphate. Yogyo Kyokai Shi 1976, 84, 209–213. [Google Scholar] [CrossRef]
- Fernández, E.; Ginebra, M.P.; Boltong, M.G.; Driessens, F.C.M.; Ginebra, J.; de Maeyer, E.A.P.; Verbeeck, R.M.H.; Planell, J.A. Kinetic study of the setting reaction of a calcium phosphate bone cement. J. Biomed. Mater. Res. 1996, 32, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Gbureck, U.; Barralet, J.E.; Radu, L.; Klinger, H.G.; Thull, R. Amorphous α-tricalcium phosphate, preparation and aqueous setting reaction. J. Am. Ceram. Soc. 2004, 87, 1126–1132. [Google Scholar] [CrossRef]
- Bohner, M.; Malsy, A.K.; Camire, C.L.; Gbureck, U. Combining particle size distribution and isothermal calorimetry data to determine the reaction kinetics of α-tricalcium phosphate – water mixtures. Acta Biomater. 2006, 2, 343–348. [Google Scholar] [CrossRef] [PubMed]
- Brunner, T.J.; Grass, R.N.; Bohner, M.; Stark, W.J. Effect of particle size, crystal phase and crystallinity on the reactivity of tricalcium phosphate cements for bone reconstruction. J. Mater. Chem. 2007, 38, 4072–4078. [Google Scholar] [CrossRef]
- Alves, H.L.R.; dos Santos, L.A.; Bergmann, C.P. Injectability evaluation of tricalcium phosphate bone cement. J. Mater. Sci. Mater. Med. 2008, 19, 2241–2246. [Google Scholar] [CrossRef] [PubMed]
- Jack, V.; Buchanan, F.J.; Dunne, N.J. Particle attrition of α-tricalcium phosphate, effect on mechanical, handling, and injectability properties of calcium phosphate cements. Proc. Inst. Mech. Eng. H: J. Eng. Med. 2008, 222, 19–28. [Google Scholar] [CrossRef]
- Gbureck, U.; Grolms, O.; Barralet, J.E.; Grover, L.M.; Thull, R. Mechanical activation and cement formation of β-tricalcium phosphate. Biomaterials 2003, 24, 4123–4131. [Google Scholar] [CrossRef] [PubMed]
- Gbureck, U.; Barralet, J.E.; Hofmann, M.P.; Thull, R. Nanocrystalline tetracalcium phosphate cement. J. Dent. Res. 2004, 83, 425–428. [Google Scholar] [CrossRef] [PubMed]
- Gbureck, U.; Barralet, J.E.; Hofmann, M.P.; Thull, R. Mechanical activation of tetracalcium phosphate. J. Am. Ceram. Soc. 2004, 87, 311–313. [Google Scholar] [CrossRef]
- Tsai, C.H.; Ju, C.P.; Lin, J.H.C. Morphology and mechanical behavior of TTCP-derived calcium phosphate cement subcutaneously implanted in rats. J. Mater. Sci. Mater. Med. 2008, 19, 2407–2415. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.H.; Lin, R.M.; Ju, C.P.; Lin, J.H.C. Bioresorption behavior of tetracalcium phosphate-derived calcium phosphate cement implanted in femur of rabbits. Biomaterials 2008, 29, 984–993. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.H.; Lin, J.H.C.; Ju, C.P. γ-Radiation-induced changes in structure and properties of tetracalcium phosphate and its derived calcium phosphate cement. J. Biomed. Mater. Res. B Appl. Biomater. 2007, 80B, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Chow, L.C.; Markovic, M.; Frukhtbeyn, S.A.; Takagi, S. Hydrolysis of tetracalcium phosphate under a near-constant composition condition – effects of pH and particle size. Biomaterials 2005, 26, 393–401. [Google Scholar] [CrossRef] [PubMed]
- TenHuisen, K.S.; Brown, P.W. Formation of calcium-deficient hydroxyapatite from α-tricalcium phosphate. Biomaterials 1998, 19, 2209–2217. [Google Scholar] [CrossRef] [PubMed]
- Ginebra, M.P.; Fernández, E.; Driessens, F.C.M.; Planell, J.A. Modeling of the hydrolysis of α-TCP. J. Am. Ceram. Soc. 1999, 82, 2808–2812. [Google Scholar] [CrossRef]
- Durucan, C.; Brown, P.W. α-tricalcium phosphate hydrolysis to hydroxyapatite at and near physiological temperature. J. Mater. Sci. Mater. Med. 2000, 11, 365–371. [Google Scholar] [CrossRef] [PubMed]
- Durucan, C.; Brown, P.W. Kinetic model for α-tricalcium phosphate hydrolysis. J. Am. Ceram. Soc. 2002, 85, 2013–2018. [Google Scholar] [CrossRef]
- Fulmer, M.T.; Brown, P.W. Hydrolysis of dicalcium phosphate dihydrate to hydroxyapatite. J. Mater. Sci. Mater. Med. 1998, 9, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Ginebra, M.P.; Driessens, F.C.M.; Planell, J.A. Effect of the particle size on the micro and nanostructural features of a calcium phosphate cement, a kinetic analysis. Biomaterials 2004, 25, 3453–3462. [Google Scholar] [CrossRef] [PubMed]
- Koshino, T.; Kubota, W.; Morii, T. Bone formation as a reaction to hydraulic hydroxyapatite thermal decomposition product used as bone cement in rabbits. Biomaterials 1995, 16, 125–128. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Huang, Y.; Chen, J. The physicochemical properties of the solidification of calcium phosphate cement. J. Biomed. Mater. Res. B Appl. Biomater. 2004, 69B, 73–78. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Gai, W.; Pan, S.; Liu, Z. The exothermal behavior in the hydration process of calcium phosphate cement. Biomaterials 2003, 24, 2995–3003. [Google Scholar] [CrossRef] [PubMed]
- Charrière, E.; Terrazzoni, S.; Pittet, C.; Mordasini, P.; Dutoit, M.; Lemaître, J.; Zysset, P. Mechanical characterization of brushite and hydroxyapatite cements. Biomaterials 2001, 22, 2937–2945. [Google Scholar] [CrossRef] [PubMed]
- Morgan, H.; Dauskardt, R.H. Notch strength insensitivity of self-setting hydroxyapatite bone cements. J. Mater. Sci. Mater. Med. 2003, 14, 647–653. [Google Scholar] [CrossRef] [PubMed]
- Bohner, M. Calcium orthophosphates in medicine, from ceramics to calcium phosphate cements. Injury 2000, 31, SD37–SD47. [Google Scholar] [CrossRef]
- Bohner, M. Physical and chemical aspects of calcium phosphates used in spinal surgery. Eur. Spine J. 2001, 10, S114–S121. [Google Scholar] [CrossRef] [PubMed]
- von Gonten, A.S.; Kelly, J.R.; Antonucci, J.M. Load-bearing behavior of a simulated craniofacial structure fabricated from a hydroxyapatite cement and bioresorbable fiber-mesh. J. Mater. Sci. Mater. Med. 2000, 11, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Gisep, A.; Kugler, S.; Wahl, D.; Rahn, B. The mechanical characterization of a bone defect model filled with ceramic cements. J. Mater. Sci. Mater. Med. 2004, 15, 1065–1071. [Google Scholar] [CrossRef] [PubMed]
- Takagi, S.; Chow, L.C.; Markovic, M.; Friedman, C.D.; Costantino, P.D. Morphological and phase characterizations of retrieved calcium phosphate cement implants. J. Biomed. Mater. Res.: Appl. Biomater. 2001, 58, 36–41. [Google Scholar] [CrossRef]
- Ambard, A.J.; Mueninghoff, L. Calcium phosphate cement, review of mechanical and biological properties. J. Prosthodont. 2006, 15, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Lewis, G. Injectable bone cements for use in vertebroplasty and kyphoplasty, state-of-the-art review. J. Biomed. Mater. Res. B Appl. Biomater. 2006, 76B, 456–468. [Google Scholar] [CrossRef] [PubMed]
- Kenny, S.M.; Buggy, M. Bone cements and fillers: A review. J. Mater. Sci. Mater. Med. 2003, 14, 923–938. [Google Scholar] [CrossRef] [PubMed]
- Constantz, B.R.; Ison, I.C.; Fulmer, M.T.; Poser, R.D.; Smith, S.T.; van Wagoner, M.; Ross, J.; Goldstein, S.A.; Jupiter, J.B.; Rosenthal, D.I. Skeletal repair by in situ formation of the mineral phase of bone. Science 1995, 267, 1796–1799. [Google Scholar] [CrossRef]
- Bohner, M. Reactivity of calcium phosphate cements. J. Mater. Chem. 2007, 38, 3980–3986. [Google Scholar] [CrossRef]
- Bohner, M.; Brunner, T.J.; Stark, W.J. Controlling the reactivity of calcium phosphate cements. J. Mater. Chem. 2008, 18, 5669–5675. [Google Scholar] [CrossRef]
- Yuan, H.; Li, Y.; de Bruijn, J.D.; de Groot, K.; Zhang, X. Tissue responses of calcium phosphate cement, a study in dogs. Biomaterials 2000, 21, 1283–1290. [Google Scholar] [CrossRef] [PubMed]
- Takechi, M.; Miyamoto, Y.; Ishikawa, K.; Toh, T.; Yuasa, T.; Nagayama, M.; Suzuki, K. Initial histological evaluation of anti-washout type fast-setting calcium phosphate cement following subcutaneous implantation. Biomaterials 1998, 19, 2057–2063. [Google Scholar] [CrossRef] [PubMed]
- Fulmer, M.T.; Brown, P.W. Effects of Na2HPO4 and NaH2PO4 on hydroxyapatite formation. J. Biomed. Mater. Res. 1993, 27, 1095–1102. [Google Scholar] [CrossRef] [PubMed]
- Otsuka, M.; Matsuda, Y.; Suwa, Y.; Fox, J.L.; Higuchi, W.I. Effect of particle size of metastable calcium phosphates on mechanical strength of a novel self-setting bioactive calcium phosphate cement. J. Biomed. Mater. Res. 1995, 29, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Shao, H.; Chen, F.; Zheng, H. Effects of granularity of raw materials on the hydration and hardening process of calcium phosphate cement. Biomaterials 2003, 24, 4103–4113. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.C.; Lin, J.H.C.; Ju, C.P. Transmission electron microscopic study on setting mechanism of tetracalcium phosphate/dicalcium phosphate anhydrous-based calcium phosphate cement. J. Biomed. Mater. Res. 2003, 64, 664–671. [Google Scholar] [CrossRef]
- Fernández, E.; Gil, F.J.; Ginebra, M.P.; Driessens, F.C.M.; Planell, J.A.; Best, S.M. Calcium phosphate bone cements for clinical applications. Part II: precipitate formation during setting reactions. J. Mater. Sci. Mater. Med. 1999, 10, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Brown, W.E. Crystal growth of bone mineral. Clin. Orthop. Rel. Res. 1966, 44, 205–220. [Google Scholar] [CrossRef]
- Tung, M.S.; Brown, W.E. An intermediate state in hydrolysis of amorphous calcium phosphate. Calcif. Tissue Int. 1983, 35, 783–790. [Google Scholar] [CrossRef] [PubMed]
- Brown, W.E.; Eidelman, N.; Tomazic, B.B. Octacalcium phosphate as a precursor in biomineral formation. Adv. Dent. Res. 1987, 1, 306–313. [Google Scholar] [PubMed]
- Constantz, B.R.; Barr, B.M.; Ison, I.C.; Fulmer, M.T.; Baker, J.; McKinney, L.A.; Goodman, S.B.; Gunasekaren, S.; Delaney, D.C.; Ross, J.; Poser, R.D. Histological, chemical and crystallographic analysis of four calcium orthophosphate cements in different rabbit osseous sites. J. Biomed. .Mater. Res.:Appl. Biomater. 1998, 43, 451–461. [Google Scholar]
- Elliott, J.C. Structure and chemistry of the apatites and other calcium orthophosphates; Elsevier: Amsterdam, Holland, 1994; p. 404. [Google Scholar]
- Bohner, M.; Merkle, H.P.; van Landuyt, P.; Trophardy, G.; Lemaître, J. Effect of several additives and their admixtures on the physico-chemical properties of a calcium phosphate cement. J. Mater. Sci. Mater. Med. 2000, 11, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Vereecke, G.; Lemaître, J. Calculation of the solubility diagrams in the system Ca(OH)2 – H3PO4 – KOH – HNO3 – CO2 – H2O. J. Cryst. Growth 1990, 104, 820–832. [Google Scholar] [CrossRef]
- Klein, C.P.; de Groot, K.; Driessen, A.A.; van der Lubbe, H.B. Interaction of biodegradable β-whitlockite ceramics with bone tissue, an in vivo study. Biomaterials 1985, 6, 189–192. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Shen, W.; Chen, J. Solution property of calcium phosphate cement hardening body. Mater. Chem. Phys. 1999, 58, 78–83. [Google Scholar] [CrossRef]
- Apelt, D.; Theiss, F.; El-Warrak, AO.; Zlinszky, K.; Bettschart-Wolfisberger, R.; Bohner, M.; Matter, S.; Auer, J.A.; von Rechenberg, B. In vivo behavior of three different injectable hydraulic calcium phosphate cements. Biomaterials 2004, 25, 1439–1451. [Google Scholar] [CrossRef] [PubMed]
- Barralet, J.E.; Grover, L.M.; Gbureck, U. Ionic modification of calcium phosphate cement viscosity. Part II: hypodermic injection and strength improvement of brushite cement. Biomaterials 2004, 25, 2197–2203. [Google Scholar] [CrossRef] [PubMed]
- Sarda, S.; Fernández, E.; Nilsson, M.; Balcells, M.; Planell, J.A. Kinetic study of citric acid influence on calcium phosphate bone cements as water-reducing agent. J. Biomed. Mater. Res. 2002, 61, 653–659. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.; Ye, J.; Wang, Y. Improved injectability and in vitro degradation of a calcium phosphate cement containing poly(lactide-co-glycolide) microspheres. Acta Biomater. 2008, 4, 1837–1845. [Google Scholar] [CrossRef] [PubMed]
- Grover, L.M.; Knowles, J.C.; Fleming, G.J.P.; Barralet, J.E. In vitro ageing of brushite calcium phosphate cement. Biomaterials 2003, 24, 4133–4141. [Google Scholar] [CrossRef] [PubMed]
- Mariño, F.T.; Mastio, J.; Rueda, C.; Blanco, L.; Cabarcos, E.L. Increase of the final setting time of brushite cements by using chondroitin 4-sulfate and silica gel. J. Mater. Sci. Mater. Med. 2007, 18, 1195–1201. [Google Scholar] [CrossRef] [PubMed]
- Mariño, F.T.; Torres, J.; Hamdan, M.; Rodríguez, C.R.; Cabarcos, E.L. Advantages of using glycolic acid as a retardant in a brushite forming cement. J. Biomed. Mater. Res. B Appl. Biomater. 2007, 83B, 571–579. [Google Scholar] [CrossRef] [PubMed]
- Flautre, B.; Delecourt, C.; Blary, M.; van Landuyt, P.; Lemaître, J.; Hardouin, P. Volume effect on biological properties of a calcium phosphate hydraulic cement, experimental study on sheep. Bone 1999, 25, S35–S39. [Google Scholar] [CrossRef]
- Bohner, M. pH variations of a solution after injecting brushite cements. Key Eng. Mater. 2001, 192-195, 813–816. [Google Scholar] [CrossRef]
- Xie, J.; Riley, C.; Chittur, K. Effect of albumin on brushite transformation to hydroxyapatite. J. Biomed. Mater. Res. 2001, 57, 357–365. [Google Scholar] [CrossRef] [PubMed]
- Frayssinet, P.; Roudier, M.; Lerch, A.; Ceolin, J.L.; Depres, E.; Rouquet, N. Tissue reaction against a self-setting calcium phosphate cement set in bone or outside the organism. J. Mater. Sci. Mater. Med. 2000, 11, 811–815. [Google Scholar] [CrossRef] [PubMed]
- Ohura, K.; Bohner, M.; Hardouin, P.; Lemaître, J.; Pasquier, G.; Flautre, B. Resorption of, and bone formation from, new β-tricalcium orthophosphate – monocalcium orthophosphate cements: an in vivo study. J. Biomed. Mater. Res. 1996, 30, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Flautre, B.; Maynou, C.; Lemaître, J.; van Landuyt, P.; Hardouin, P. Bone colonization of β-TCP granules incorporated in brushite cements. J. Biomed. Mater. Res.: Appl. Biomater. 2002, 63, 413–417. [Google Scholar] [CrossRef]
- Standard test method for time of setting of hydraulic cement paste by Gillmore needles. ASTM C266-89. In Annual Book of ASTM Standards; American Society for Testing and Materials: Philadelphia, USA, 1993; Volume 04.01, pp. 189–191.
- Standard test method for time of setting of hydraulic cement paste by Vicat needle. ASTM C191-92. In Annual Book of ASTM Standards; American Society for Testing and Materials: Philadelphia, USA, 1993; Volume 04.01, pp. 158–160.
- Nilsson, M.; Carlson, J.; Fernández, E.; Planell, J.A. Monitoring the setting of calcium-based bone cements using pulse-echo ultrasound. J. Mater. Sci. Mater. Med. 2002, 13, 1135–1141. [Google Scholar] [CrossRef] [PubMed]
- Carlson, J.; Nilsson, M.; Fernández, E.; Planell, J.A. An ultrasonic pulse-echo technique for monitoring the setting of CaSO4-based bone cement. Biomaterials 2003, 24, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, M.P.; Nazhat, S.N.; Gbureck, U.; Barralet, J.E. Real-time monitoring of the setting reaction of brushite-forming cement using isothermal differential scanning calorimetry. J. Biomed. Mater. Res. B Appl. Biomater. 2006, 79B, 360–364. [Google Scholar] [CrossRef] [PubMed]
- Martin, R.I.; Brown, P.W. The effects of magnesium on hydroxyapatite formation in vitro from CaHPO4 and Ca4(PO4)2O at 37.4 °C. Calcif. Tissue Int. 1997, 60, 538–546. [Google Scholar] [CrossRef] [PubMed]
- Brunner, TJ.; Bohner, M.; Dora, C.; Gerber, C.; Stark, W.J. Comparison of amorphous TCP nanoparticles to micron-sized α-TCP as starting materials for calcium phosphate cements. J. Biomed. Mater. Res. B Appl. Biomater. 2007, 83B, 400–407. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.Y.; Wang, Y.W.; Dong, L.M.; Yu, Z.W. Thermokinetic analysis of the hydration process of calcium phosphate cement. J. Therm. Anal. Calorim. 2006, 85, 785–789. [Google Scholar] [CrossRef]
- Bohner, M.; Gbureck, U. Thermal reactions of brushite cements. J. Biomed. Mater. Res. B (Appl. Biomater.) 2008, 84B, 375–385. [Google Scholar] [CrossRef]
- Hofmann, M.P.; Young, A.M.; Nazhat, S.N.; Gbureck, U.; Barralet, J.E. Setting kinetics observation of a brushite cement by FTIR and DSC. Key Eng. Mater. 2006, 309-311, 837–840. [Google Scholar] [CrossRef]
- Liu, C.; Huang, Y.; Zheng, H. Study of the hydration process of calcium phosphate cement by AC impedance spectroscopy. J. Am. Ceram. Soc. 1999, 82, 1052–1057. [Google Scholar] [CrossRef]
- Hofmann, M.P.; Young, A.M.; Gbureck, U.; Nazhat, S.N.; Barralet, J.E. FTIR-monitoring of a fast setting brushite bone cement, effect of intermediate phases. J. Mater. Chem. 2006, 16, 3199–3206. [Google Scholar] [CrossRef]
- Hsu, H.C.; Tuan, W.H.; Lee, H.Y. In-situ observation on the transformation of calcium phosphate cement into hydroxyapatite. Mater. Sci. Eng. C 2009, in press. [Google Scholar]
- Rau, J.V.; Generosi, A.; Smirnov, V.V.; Ferro, D.; Rossi, A.V.; Barinov, S.M. Energy dispersive X-ray diffraction study of phase development during hardening of calcium phosphate bone cements with addition of chitosan. Acta Biomater. 2008, 4, 1089–1094. [Google Scholar] [CrossRef] [PubMed]
- Ginebra, M.P.; Fernández, E.; Driessens, F.C.M.; Boltong, M.G.; Muntasell, J.; Font, J.; Planell, J.A. The effects of temperature on the behaviour of an apatitic calcium phosphate cement. J. Mater. Sci. Mater. Med. 1995, 6, 857–860. [Google Scholar] [CrossRef]
- Baroud, G.; Bohner, M.; Heini, P.; Steffen, T. Injection biomechanics of bone cements used in vertebroplasty. Biomed. Mater. Eng. 2004, 14, 487–504. [Google Scholar] [PubMed]
- Leung, K.S.; Siu, W.S.; Li, S.F.; Qin, L.; Cheung, W.H.; Tam, K.F.; Po, P.; Lui, Y. An in vitro optimized injectable calcium phosphate cement for augmenting screw fixation in osteopenic goats. J. Biomed. Mater. Res. B Appl. Biomater. 2006, 78B, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Eames, W.B.; Monroe, S.D.; Roan, J.D.; Oneal, S.J. Proportioning and mixing of cements – comparison of working times. Oper. Dent. 1977, 2, 97–104. [Google Scholar] [PubMed]
- Baroud, G.; Matsushita, C.; Samara, M.; Beckman, L.; Steffen, T. Influence of oscillatory mixing on the injectability of three acrylic and two calcium phosphate bone cements for vertebroplasty. J. Biomed. Mater. Res. B Appl. Biomater. 2004, 68B, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Nomoto, T.; Haraguchi, K.; Yamaguchi, S.; Sugano, N.; Nakayama, H.; Sekino, T.; Niihara, K. Hydrolyses of calcium phosphates-allografts composite in physiological solutions. J. Mater. Sci. Mater. Med. 2006, 17, 379–385. [Google Scholar] [CrossRef] [PubMed]
- Oda, M.; Takeuchi, A.; Lin, X.; Matsuya, S.; Ishikawa, K. Effects of liquid phase on basic properties of α-tricalcium phosphate-based apatite cement. Dent. Mater. J. 2008, 27, 672–677. [Google Scholar] [CrossRef] [PubMed]
- Sarda, S.; Fernández, E.; Llorens, J.; Martinez, S.; Nilsson, M.; Planell, J.A. Rheological properties of an apatitic bone cement during initial setting. J. Mater. Sci. Mater. Med. 2001, 12, 905–909. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Shao, H.; Chen, F.; Zheng, H. Rheological properties of concentrated aqueous injectable calcium phosphate cement slurry. Biomaterials 2006, 27, 5003–5013. [Google Scholar] [CrossRef] [PubMed]
- Bohner, M.; Baroud, G. Injectability of calcium phosphate pastes. Biomaterials 2005, 26, 1553–1563. [Google Scholar] [CrossRef] [PubMed]
- Khairoun, I.; Boltong, M.G.; Driessens, F.C.M.; Planell, J.A. Some factors controlling the injectability of calcium phosphate bone cements. J. Mater. Sci. Mater. Med. 1998, 9, 425–428. [Google Scholar] [CrossRef] [PubMed]
- Burguera, E.F.; Xu, H.H.K.; Sun, L. Injectable calcium phosphate cement, effects of powder-to-liquid ratio and needle size. J. Biomed. Mater. Res. B Appl. Biomater. 2008, 84B, 493–502. [Google Scholar] [CrossRef] [PubMed]
- Habib, M.; Baroud, G.; Gitzhofer, F.; Bohner, M. Mechanisms underlying the limited injectability of hydraulic calcium phosphate paste. Acta Biomater. 2008, 4, 1465–1471. [Google Scholar] [CrossRef] [PubMed]
- Baroud, G.; Cayer, E.; Bohner, M. Rheological characterization of concentrated aqueous beta-tricalcium phosphate suspensions: the effect of liquid-to-powder ratio, milling time and additives. Acta Biomater 2005, 1, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Bohner, M.; Doebelin, N.; Baroud, G. Theoretical and experimental approach to test the cohesion of calcium phosphate pastes. Eur. Cell Mater. 2006, 12, 26–35. [Google Scholar] [PubMed]
- Bermudez, O.; Boltong, M.G.; Driessens, F.C.M.; Planell, J.A. Compressive strength and diametral tensile strength of some calcium-orthophosphate cements, a pilot study. J. Mater. Sci. Mater. Med. 1993, 4, 389–393. [Google Scholar] [CrossRef]
- del Valle, S.; Miňo, N.; Muňoz, F.; González, A.; Planell, J.A.; Ginebra, M.P. In vivo evaluation of an injectable macroporous calcium phosphate cement. J. Mater. Sci. Mater. Med. 2007, 18, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Khairoun, I.; Driessens, F.C.M.; Boltong, M.G.; Planell, J.A.; Wenz, R. Addition of cohesion promoters to calcium orthophosphate cements. Biomaterials 1999, 20, 393–398. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, Y.; Ishikawa, K.; Takechi, M.; Toh, T.; Yuasa, T.; Nagayama, M.; Suzuki, K. Histological and compositional evaluations of three types of calcium phosphate cements when implanted in subcutaneous tissue immediately after mixing. J. Biomed. Mater. Res.: Appl. Biomater. 1999, 48, 36–42. [Google Scholar] [CrossRef]
- Bigi, A.; Bracci, B.; Panzavolta, S. Effect of added gelatin on the properties of calcium phosphate cement. Biomaterials 2004, 25, 2893–2899. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, K.; Matsuya, S.; Nakagawa, M.; Udoh, K.; Suzuki, K. Basic properties of apatite cement containing spherical tetracalcium phosphate made with plasma melting method. J. Mater. Sci. Mater. Med. 2004, 15, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Ye, J.; Wang, Y. Effect of additives on the morphology of the hydrated product and physical properties of a calcium phosphate cement. J. Mater. Sci. Technol. 2008, 24, 285–288. [Google Scholar]
- Barralet, J.E.; Hofmann, M.; Grover, L.M.; Gbureck, U. High strength apatitic cement by modification with α-hydroxy acid salts. Adv. Mater. 2003, 15, 2091–2095. [Google Scholar] [CrossRef]
- Barralet, J.E.; Duncan, C.O.; Dover, M.S.; Bassett, D.C.; Nishikawa, H.; Monaghan, A.; Gbureck, U. Cortical bone screw fixation in ionically modified apatite cements. J. Biomed. Mater. Res. B Appl. Biomater. 2005, 73B, 238–243. [Google Scholar] [CrossRef] [PubMed]
- Ginebra, M.P.; Boltong, M.G.; Fernández, E.; Planell, J.A.; Driessens, F.C.M. Effect of various additives and temperature on some properties of an apatitic calcium phosphate cement. J. Mater. Sci. Mater. Med. 1995, 6, 612–616. [Google Scholar] [CrossRef]
- Wang, X.; Ye, J.; Wang, Y. Influence of a novel radiopacifier on the properties of an injectable calcium phosphate cement. Acta Biomater. 2007, 3, 757–763. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Tanaka, M.; Sakurai, M.; Maeda, M. Development of calcium phosphate cement. J. Eur. Ceram. Soc. 2006, 26, 549–552. [Google Scholar] [CrossRef]
- Markovic, M.; Takagi, S.; Chow, L.C. Formation of macropores in calcium phosphate cements through the use of mannitol crystals. Key Eng. Mater. 2001, 192-195, 773–776. [Google Scholar] [CrossRef]
- Almirall, A.; Larrecq, G.; Delgado, J.A.; Martínez, S.; Planell, J.A.; Ginebra, M.P. Fabrication of low temperature macroporous hydroxyapatite scaffolds by foaming and hydrolysis of an α-TCP paste. Biomaterials 2004, 25, 3671–3680. [Google Scholar] [CrossRef] [PubMed]
- Barralet, J.E.; Grover, L.; Gaunt, T.; Wright, A.J.; Gibson, I.R. Preparation of macroporous calcium phosphate cement tissue engineering scaffold. Biomaterials 2002, 23, 3063–3072. [Google Scholar] [CrossRef] [PubMed]
- Takagi, S.; Chow, L.C. Formation of macropores in calcium phosphate cement implants. J. Mater. Sci. Mater. Med. 2001, 12, 135–139. [Google Scholar] [CrossRef] [PubMed]
- Simon, C.G., Jr.; Khatri, C.A.; Wight, S.A.; Wang, F.W. Preliminary report on the biocompatibility of a moldable, resorbable, composite bone graft consisting of calcium phosphate cement and poly(lactide-co-glycolide) microspheres. J. Orthop. Res. 2002, 20, 473–482. [Google Scholar] [CrossRef] [PubMed]
- Ruhe, P.Q.; Hedberg, E.L.; Padron, N.T.; Spauwen, P.H.M.; Jansen, J.A.; Mikos, A.G. Biocompatibility and degradation of poly(DL-lactic-co-glycolic acid)/calcium phosphate cement composites. J. Biomed. Mater. Res. A 2005, 74, 533–544. [Google Scholar] [CrossRef] [PubMed]
- Habraken, W.J.E.M.; Wolke, J.G.C.; Mikos, A.G.; Jansen, J.A. Injectable PLGA microsphere/calcium phosphate cements, physical properties and degradation characteristics. J. Biomater. Sci. Polym. Ed. 2006, 17, 1057–1074. [Google Scholar] [CrossRef] [PubMed]
- Habraken, W.J.E.M.; Wolke, J.G.C.; Mikos, A.G.; Jansen, J.A. PLGA microsphere/calcium phosphate cement composites for tissue engineering, in vitro release and degradation characteristics. J. Biomater. Sci. Polym. Ed. 2008, 19, 1171–1188. [Google Scholar] [CrossRef] [PubMed]
- Link, D.P.; van den Dolder, J.; van den Beucken, J.J.J.P.; Cuijpers, V.M.; Wolke, J.G.C.; Mikos, A.G.; Jansen, J.A. Evaluation of the biocompatibility of calcium phosphate cement/PLGA microparticle composites. J. Biomed. Mater. Res. A 2008, 87, 760–769. [Google Scholar] [CrossRef] [PubMed]
- Link, D.P.; van den Dolder, J.; Jurgens, W.J.F.M.; Wolke, J.G.C.; Jansen, J.A. Mechanical evaluation of implanted calcium phosphate cement incorporated with PLGA microparticles. Biomaterials 2006, 27, 4941–4947. [Google Scholar] [CrossRef] [PubMed]
- Tas, A.C. Preparation of porous apatite granules from calcium phosphate cement. J. Mater. Sci. Mater. Med. 2008, 19, 2231–2239. [Google Scholar] [CrossRef] [PubMed]
- Tas, A.C. Preparation of self-setting cement-based micro- and macroporous granules of carbonated apatitic calcium phosphate. Ceram. Eng. Sci. Proc. 2006, 27, 49–60. [Google Scholar]
- Li, M.; Liu, X.; Liu, X.; Ge, B.; Chen, K. Creation of macroporous calcium phosphate cements as bone substitutes by using genipin – crosslinked gelatin microspheres. J. Mater. Sci. Mater. Med. 2009, 20, 925–934. [Google Scholar] [CrossRef] [PubMed]
- Habraken, W.J.E.M.; de Jonge, L.T.; Wolke, J.G.C.; Yubao, L.; Mikos, A.G.; Jansen, J.A. Introduction of gelatin microspheres into an injectable calcium phosphate cement. J. Biomed. Mater. Res. A 2008, 87, 643–655. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.P.; Ye, J.D.; Li, X.; Dong, H. Production of in-situ macropores in an injectable calcium phosphate cement by introduction of cetyltrimethyl ammonium bromide. J. Mater. Sci. Mater. Med. 2008, 19, 3221–3225. [Google Scholar]
- Habraken, W.J.E.M.; Zhang, Z.; Wolke, J.G.C.; Grijpma, D.W.; Mikos, A.G.; Feijen, J.; Jansen, J.A. Introduction of enzymatically degradable poly(trimethylene carbonate) microspheres into an injectable calcium phosphate cement. Biomaterials 2008, 29, 2464–2476. [Google Scholar] [CrossRef] [PubMed]
- del Real, R.P.; Wolke, J.G.C.; Vallet-Regi, M.; Jansen, J.A. A new method to produce macropores in calcium phosphate cements. Biomaterials 2002, 23, 3673–3680. [Google Scholar] [CrossRef] [PubMed]
- del Real, R.P.; Ooms, E.; Wolke, J.G.C.; Vallet-Regi, M.; Jansen, J.A. In vivo bone response to porous calcium phosphate cement. J. Biomed. Mater. Res. A 2003, 65, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Hesaraki, S.; Moztarzadeh, F.; Sharifi, D. Formation of interconnected macropores in apatitic calcium phosphate bone cement with the use of an effervescent additive. J. Biomed. Mater. Res. A 2007, 83, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Hesaraki, S.; Zamanian, A.; Moztarzadeh, F. The influence of the acidic component of the gas-foaming porogen used in preparing an injectable porous calcium phosphate cement on its properties, acetic acid versus citric acid. J. Biomed. Mater. Res. B Appl. Biomater. 2008, 86, 208–216. [Google Scholar] [CrossRef] [PubMed]
- Hesaraki, S.; Moztarzadeh, F.; Solati-Hashjin, M. Phase evaluation of an effervescent-added apatitic calcium phosphate bone cement. J. Biomed. Mater. Res. B Appl. Biomater. 2006, 79B, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Sarda, S.; Nilsson, M.; Balcells, M.; Fernández, E. Influence of surfactant molecules as air-entraining agent for bone cement macroporosity. J. Biomed. Mater. Res. A 2003, 65, 215–221. [Google Scholar] [CrossRef] [PubMed]
- Friberg, J.; Fernández, E.; Sarda, S.; Nilsson, M.; Ginebra, M.P.; Martinez, S.; Planell, J.A. An experimental approach to the study of the rheology behavior of synthetic bone calcium phosphate cements. Key Eng. Mater. 2001, 192-195, 777–780. [Google Scholar] [CrossRef]
- Andrianjatovo, H.; Lemaître, J. Effects of polysaccharides on the cement properties in the monocalcium phosphate/β-tricalcium phosphate system. Innov. Tech. Biol. Med. 1995, 16, 140–147. [Google Scholar]
- Cherng, A.; Takagi, S.; Chow, L.C. Effects of hydroxypropylmethylcellulose and other gelling agents on the handling properties of calcium phosphate cement. J. Biomed. Mater. Res. 1997, 35, 273–277. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, A.; Matsuno, H.; Yamamoto, S.; Kawasaki, T.; Kohgo, T.; Uo, M.; Watari, F.; Nakasu, M. Tissue response to a newly developed calcium phosphate cement containing succinic acid and carboxymethyl-chitin. J. Biomed. Mater. Res. A 2003, 64, 491–501. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.H.K.; Weir, M.D.; Burguera, E.F.; Fraser, A.M. Injectable and macroporous calcium phosphate cement scaffold. Biomaterials 2006, 27, 4279–4287. [Google Scholar] [CrossRef] [PubMed]
- Bigi, A.; Torricelli, P.; Fini, M.; Bracci, B.; Panzavolta, S.; Sturba, L.; Giardino, R. A biomimetic gelatin-calcium phosphate bone cement. Int. J. Artif. Organs 2004, 27, 664–673. [Google Scholar] [PubMed]
- Bigi, A.; Panzavolta, S.; Sturba, L.; Torricelli, P.; Fini, M.; Giardino, R. Normal and osteopenic bone-derived osteoblast response to a biomimetic gelatin – calcium phosphate bone cement. J. Biomed. Mater. Res. A 2006, 78, 739–745. [Google Scholar] [CrossRef] [PubMed]
- Fujishiro, Y.; Takahashi, K.; Sato, T. Preparation and compressive strength of α-tricalcium phosphate/gelatin gel composite cement. J. Biomed. Mater. Res. 2001, 54, 525–530. [Google Scholar] [CrossRef] [PubMed]
- Bigi, A.; Panzavolta, S.; Rubini, K. Setting mechanism of a biomimetic bone cement. Chem. Mater. 2004, 16, 3740–3745. [Google Scholar] [CrossRef]
- Panzavolta, S.; Torricelli, P.; Sturba, L.; Bracci, B.; Giardino, R.; Bigi, A. Setting properties and in vitro bioactivity of strontium-enriched gelatin-calcium phosphate bone cements. J. Biomed. Mater. Res. A 2008, 84, 965–972. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.X.; Shi, X.T.; Wang, Y.P.; Shi, Z.L. Performance of calcium phosphate bone cement using chitosan and gelatin as well as citric acid as hardening liquid. J. Clin. Rehabil. Tissue Eng. Res. 2008, 12, 6381–6384. [Google Scholar]
- Shie, M.Y.; Chen, D.C.H.; Wang, C.Y.; Chiang, T.Y.; Ding, S.J. Immersion behavior of gelatin-containing calcium phosphate cement. Acta Biomater. 2008, 4, 646–655. [Google Scholar] [CrossRef] [PubMed]
- Majekodunmi, A.O.; Deb, S.; Nicholson, J.W. Effect of molecular weight and concentration of poly(acrylic acid) on the formation of a polymeric calcium phosphate cement. J. Mater. Sci. Mater. Med. 2003, 14, 747–752. [Google Scholar] [CrossRef] [PubMed]
- Majekodunmi, A.O.; Deb, S. Poly(acrylic acid) modified calcium phosphate cements, the effect of the composition of the cement powder and of the molecular weight and concentration of the polymeric acid. J. Mater. Sci. Mater. Med. 2007, 18, 1883–1888. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.C.; Ju, C.P.; Wang, J.C.; Hung, C.C.; Lin, J.H.C. Brittle and ductile adjustable cement derived from calcium phosphate cement/polyacrylic acid composites. Dent. Mater. 2008, 24, 1616–1622. [Google Scholar] [CrossRef] [PubMed]
- Bohner, M.; Theiss, F.; Apelt, D.; Hirsiger, W.; Houriet, R.; Rizzoli, G.; Gnos, E.; Frei, C.; Auer, J.A.; von Rechenberg, B. Compositional changes of a dicalcium phosphate dihydrate cement after implantation in sheep. Biomaterials 2003, 24, 3463–3474. [Google Scholar] [CrossRef] [PubMed]
- Leroux, L.; Hatim, Z.; Freche, M.; Lacout, J.L. Effects of various adjuvants (lactic acid.; glycerol and chitosan) on the injectability of a calcium phosphate cement. Bone 1999, 25, S31–S34. [Google Scholar] [CrossRef]
- Barralet, J.E.; Tremayne, M.J.; Lilley, K.J.; Gbureck, U. Chemical modification of calcium phosphate cements with α-hydroxy acids and their salts. Chem. Mater. 2005, 17, 1313–1319. [Google Scholar] [CrossRef]
- Driessens, F.C.M.; Boltong, M.G.; de Maeyer, E.A.P.; Verbeeck, R.M.H.; Wenz, R. Effect of temperature and immersion on the setting of some calcium phosphate cements. J. Mater. Sci. Mater. Med. 2000, 11, 453–457. [Google Scholar] [CrossRef] [PubMed]
- Gbureck, U.; Dembski, S.; Thull, R.; Barralet, J.E. Factors influencing calcium phosphate cement shelf life. Biomaterials 2005, 26, 3691–3697. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, K.; Takagi, S.; Chow, L.C.; Ishikawa, Y. Properties and mechanisms of fast-setting calcium phosphate cements. J. Mater. Sci. Mater. Med. 1995, 6, 528–533. [Google Scholar] [CrossRef]
- Miyamoto, Y.; Ishikawa, K.; Fukao, K.; Sawada, M.; Nagayama, M.; Kon, M.; Asaoka, K. In vivo setting behavior of fast-setting calcium phosphate cement. Biomaterials 1995, 16, 855–860. [Google Scholar] [CrossRef] [PubMed]
- Takechi, M.; Miyamoto, Y.; Momota, Y.; Yuasa, T.; Tatehara, S.; Nagayama, M.; Ishikawa, K. Effects of various sterilization methods on the setting and mechanical properties of apatite cement. J. Biomed. Mater. Res. B Appl. Biomater. 2004, 69B, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Yuasa, T.; Miyamoto, Y.; Ishikawa, K.; Takechi, M.; Nagayama, M.; Suzuki, K. In vitro resorption of three apatite cements with osteoclasts. J. Biomed. Mater. Res. 2001, 54, 344–350. [Google Scholar] [CrossRef] [PubMed]
- Khairoun, I.; Magne, D.; Gauthier, O.; Bouler, J.M.; Aguado, E.; Daculsi, G.; Weiss, P. In vitro characterization and in vivo properties of a carbonated apatite bone cement. J. Biomed. Mater. Res. 2002, 60, 633–642. [Google Scholar]
- Sanzana, E.S.; Navarro, M.; Macule, F.; Suso, S.; Planell, J.A.; Ginebra, M.P. Of the in vivo behavior of calcium phosphate cements and glasses as bone substitutes. Acta Biomater. 2008, 4, 1924–1933. [Google Scholar] [CrossRef] [PubMed]
- Bodde, E.W.H.; Cammaert, C.T.R.; Wolke, J.G.C.; Spauwen, P.H.M.; Jansen, J.A. Investigation as to the osteoinductivity of macroporous calcium phosphate cement in goats. J. Biomed. Mater. Res. B Appl. Biomater. 2007, 83, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, Y.; Ishikawa, K.; Takeshi, M.; Toh, T.; Yoshida, Y.; Nagayama, M.; Kon, M.; Asaoka, K. Tissue response to fast-setting calcium phosphate cement in bone. J. Biomed. Mater. Res. 1997, 37, 457–464. [Google Scholar] [CrossRef] [PubMed]
- Young, S.; Holde, M.; Gunasekaran, S.; Poser, R.; Constantz, B.R. The correlation of radiographic, MRI and histological evaluations over two years of a carbonated apatite cement in a rabbit model. In Proceedings of the 44th Annual Meeting, Orthopedic Research Society, New Orleans, USA, March 16-19, 1998; p. 846.
- Kroese-Deutman, H.C.; Wolke, J.G.C.; Spauwen, P.H.M.; Jansen, J.A. Closing capacity of cranial bone defects using porous calcium phosphate cement implants in a rabbit animal model. J. Biomed. Mater. Res. A 2006, 79, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Bourgeois, B.; Laboux, O.; Obadia, L.; Gauthier, O.; Betti, E.; Aguado, E.; Daculsi, G.; Bouler, J.M. Calcium-deficient apatite: a first in vivo study concerning bone ingrowth. J. Biomed. Mater. Res. A 2003, 65, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Descamps, M.; Dejou, J.; Koubi, G.; Hardouin, P.; Lemaitre, J.; Proust, J.P. The biodegradation mechanism of calcium phosphate biomaterials in bone. J. Biomed. Mater. Res.: Appl. Biomater. 2002, 63, 408–412. [Google Scholar] [CrossRef]
- Wenisch, S.; Stahl, J.P.; Horas, U.; Heiss, C.; Kilian, O.; Trinkaus, K.; Hild, A.; Schnettler, R. In vivo mechanisms of hydroxyapatite ceramic degradation by osteoclasts, fine structural microscopy. J. Biomed. Mater. Res. A 2003, 67, 713–718. [Google Scholar] [CrossRef] [PubMed]
- Ooms, E.M.; Wolke, J.G.C.; van der Waerden, J.P.; Jansen, J.A. Trabecular bone response to injectable calcium phosphate (Ca-P) cement. J. Biomed. Mater. Res. 2002, 61, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Theiss, F.; Apelt, D.; Brand, B.; Kutter, A.; Zlinszky, K.; Bohner, M.; Matter, S.; Frei, C.; Auer, J.A.; von Rechenberg, B. Biocompatibility and resorption of a brushite calcium phosphate cement. Biomaterials 2005, 26, 4383–4394. [Google Scholar] [CrossRef] [PubMed]
- Heymann, D.; Pradal, G.; Benahmad, M. Cellular mechanisms of calcium phosphate degradation. Histol. Histopathol. 1999, 14, 871–877. [Google Scholar] [PubMed]
- Penel, G.; Leroy, N.; van Landuyt, P.; Flautre, B.; Hardouin, P.; Lemaître, J.; Leroy, G. Raman microspectrometry studies of brushite cement, in vivo evolution in a sheep model. Bone 1999, 25, S81–S84. [Google Scholar] [CrossRef]
- Dorozhkin, S.V. Inorganic chemistry of the dissolution phenomenon, the dissolution mechanism of calcium apatites at the atomic (ionic) level. Comment Inorg. Chem. 1999, 20, 285–299. [Google Scholar] [CrossRef]
- Dorozhkin, S.V. A review on the dissolution models of calcium apatites. Prog. Cryst. Growth Charact. 2002, 44, 45–61. [Google Scholar] [CrossRef]
- Knabe, C.; Driessens, F.C.M.; Planell, J.A.; Gildenhaar, R.; Berger, G.; Reif, D.; Fitzner, R.; Radlanski, RJ.; Gross, U. Evaluation of calcium phosphates and experimental calcium phosphate bone cements using osteogenic cultures. J. Biomed. Mater. Res. 2000, 52, 498–508. [Google Scholar] [CrossRef] [PubMed]
- Teitelbaum, S.L. Bone resorption by osteoclasts. Science 2000, 289, 1504–1508. [Google Scholar] [CrossRef] [PubMed]
- Mostov, K.; Werb, Z. Journey across the osteoclast. Science 1997, 276, 219–220. [Google Scholar] [CrossRef] [PubMed]
- Rodan, G.A.; Martin, T.J. Therapeutic approaches to bone diseases. Science 2000, 289, 1508–1514. [Google Scholar] [CrossRef] [PubMed]
- Hench, L.L. Bioceramics, from concept to clinic. J. Am. Ceram. Soc. 1991, 74, 1487–1510. [Google Scholar] [CrossRef]
- Hench, L.L. Bioceramics. J. Am. Ceram. Soc. 1998, 81, 1705–1728. [Google Scholar] [CrossRef]
- Sun, L.; Berndt, C.C.; Gross, K.A.; Kucuk, A. Material fundamentals and clinical performance of plasma-sprayed hydroxyapatite coatings, a review. J. Biomed. Mater. Res. B Appl. Biomater. 2001, 58, 570–592. [Google Scholar] [CrossRef]
- Renault, F.; Chabriere, E.; Andrieu, J.P.; Dublet, B.; Masson, P.; Rochu, D. Tandem purification of two HDL-associated partner proteins in human plasma.; paraoxonase (PON1) and phosphate binding protein (HPBP) using hydroxyapatite chromatography. J. Chromatogr. B 2006, 836, 15–21. [Google Scholar] [CrossRef]
- Yoshitake, T.; Kobayashi, S.; Ogawa, T.; Okuyama, T. Hydroxyapatite chromatography of guanidine denatured proteins: 1. guanidine containing phosphate buffer system. Chromatography 2006, 27, 19–26. [Google Scholar]
- Ooms, E.M.; Egglezos, E.A.; Wolke, J.G.C.; Jansen, J.A. Soft-tissue response to injectable calcium phosphate cements. Biomaterials 2003, 24, 749–757. [Google Scholar] [CrossRef] [PubMed]
- Ooms, E.M.; Wolke, J.G.C.; van de Heuvel, MT.; Jeschke, B.; Jansen, J.A. Histological evaluation of the bone response to calcium phosphate cement implanted in cortical bone. Biomaterials 2003, 24, 989–1000. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, N.; Ong, K.; Villarraga, M.; Schwardt, J.; Wenz, R.; Togawa, D.; Fujishiro, T.; Turner, A.S.; Seim, H.B., III; Bauer, T.W. Histological and mechanical evaluation of self-setting calcium phosphate cements in a sheep vertebral bone void model. J. Biomed. Mater. Res. A 2007, 81, 838–846. [Google Scholar] [CrossRef] [PubMed]
- Wen, C.Y.; Qin, L.; Lee, K.M.; Chan, K.M. The use of brushite calcium phosphate cement for enhancement of bone-tendon integration in an anterior cruciate ligament reconstruction rabbit model. J. Biomed. Mater. Res. B Appl. Biomater. 2009, in press. [Google Scholar]
- Fernández, E.; Ginebra, M.P.; Bermudez, O.; Boltong, M.G.; Driessens, F.C.M.; Planell, J.A. Dimensional and thermal behaviour of calcium phosphate cements during setting compared to PMMA bone cements. J. Mater. Sci. Lett. 1995, 14, 4–5. [Google Scholar] [CrossRef]
- Yamamoto, H.; Niwa, S.; Hori, M.; Hattori, T.; Sawai, K.; Aoki, S.; Hirano, M.; Takeuchi, H. Mechanical strength of calcium phosphate cement in vivo and in vitro. Biomaterials 1998, 19, 1587–1591. [Google Scholar] [CrossRef] [PubMed]
- Andrianjatovo, H.; Jose, F.; Lemaître, J. Effect of β-TCP granulometry on setting time and strength of calcium orthophosphate hydraulic cements. J. Mater. Sci. Mater. Med. 1996, 7, 34–39. [Google Scholar] [CrossRef]
- Ishikawa, K.; Takagi, S.; Chow, L.C.; Ishikawa, Y.; Eanes, E.D.; Asaoka, K. Behavior of a calcium orthophosphate cement in simulated blood plasma in vitro. Dent. Mater. 1994, 10, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Driessens, F.C.M. Chemistry and applied aspects of calcium orthophosphate bone cements. In Concepts and clinical applications of ionic cements, Proceeding of 15th European Conference on Biomaterials, Arcachon, Bordeaux, France, Sept 8, 1999.
- Miyazaki, K.; Horibe, T.; Antonucci, J.M.; Takagi, S.; Chow, L.C. Polymeric calcium phosphate cements, analysis of reaction products and properties. Dent. Mater. 1993, 9, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Miyazaki, K.; Horibe, T.; Antonucci, J.M.; Takagi, S.; Chow, L.C. Polymeric calcium phosphate cements, setting reaction modifiers. Dent. Mater. 1993, 9, 46–50. [Google Scholar] [CrossRef] [PubMed]
- dos Santos, L.A.; de Oliveira, L.C.; Rigo, E.C.S.; Carrodeguas, R.G.; Boschi, A.O.; de Arruda, A.C.F. Influence of polymeric additives on the mechanical properties of α-tricalcium phosphate cement. Bone 1999, 25, S99–S102. [Google Scholar] [CrossRef]
- Mickiewicz, R.A.; Mayes, A.M.; Knaack, D. Polymer – calcium phosphate cement composites for bone substitutes. J. Biomed. Mater. Res. 2002, 61, 581–592. [Google Scholar] [CrossRef] [PubMed]
- Fernández, E.; Sarda, S.; Hamcerencu, M.; Vlad, M.D.; Gel, M.; Valls, S.; Torres, R.; López, J. High-strength apatitic cement by modification with superplasticizers. Biomaterials 2005, 26, 2289–2296. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, T.; Yamamoto, M.; Ioku, K.; Goto, S. Relationship between compressive strength and pore structure of hardened cement pastes. Adv. Cement Res. 1997, 9, 25–30. [Google Scholar] [CrossRef]
- Costantino, P.D.; Friedman, C.D.; Jones, K.; Chow, L.C.; Sisson, G.A. Experimental hydroxyapatite cement cranioplasty. Plast. Reconstr. Surg. 1992, 90, 174–185. [Google Scholar] [CrossRef] [PubMed]
- Chow, L.C.; Hirayama, S.; Takagi, S.; Parry, E. Diametral tensile strength and compressive strength of a calcium phosphate cement, effect of applied pressure. J. Biomed. Mater. Res. Appl. Biomater. 2000, 53, 511–517. [Google Scholar] [CrossRef]
- Barralet, J.E.; Gaunt, T.; Wright, A.J.; Gibson, I.R.; Knowles, J.C. Effect of porosity reduction by compaction on compressive strength and microstructure of calcium phosphate cement. J. Biomed. Mater. Res.: Appl. Biomater. 2002, 63, 1–9. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, H.H.K.; Takagi, S.; Chow, L.C. In situ hardening hydroxyapatite-based scaffold for bone repair. J. Mater. Sci. Mater. Med. 2006, 17, 437–445. [Google Scholar] [CrossRef] [PubMed]
- Pittet, C.; Lemaître, J. Mechanical characterization of brushite cements: a Mohr circles approach. J. Biomed. Mater. Res.: Appl. Biomater. 2000, 53, 769–780. [Google Scholar] [CrossRef]
- Morgan, E.F.; Yetkinler, D.N.; Constantz, B.R.; Dauskardt, R.H. Mechanical properties of carbonated apatite bone mineral substitute: strength, fracture and fatigue behaviour. J. Mater. Sci. Mater. Med. 1997, 8, 559–570. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.H.K.; Carey, L.E.; Simon, C.G., Jr. Premixed macroporous calcium phosphate cement scaffold. J. Mater. Sci. Mater. Med. 2007, 18, 1345–1353. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.H.K.; Simon, C.G., Jr. Self-hardening calcium phosphate cement-mesh composite, Reinforcement.; macropores.; and cell response. J. Biomed. Mater. Res. A 2004, 69, 267–278. [Google Scholar] [CrossRef] [PubMed]
- Losee, J.E.; Karmacharya, J.; Gannon, F.H.; Slemp, A.E.; Ong, G.; Hunenko, O.; Gorden, A.D.; Bartlett, S.P.; Kirschner, R.E. Reconstruction of the immature craniofacial skeleton with a carbonated calcium phosphate bone cement, interaction with bioresorbable mesh. J. Craniofacial Surg. 2003, 14, 117–124. [Google Scholar] [CrossRef]
- Khairoun, I.; LeGeros, R.Z.; Daculsi, G.; Bouler, J.M.; Gauthier, O.; Guicheux, J. Macroporous, resorbable and injectable calcium phosphate-based cements (MCPC) for bone repair: Augmentation, regeneration and osteoporosis treatment. Patent No. WO/2005/077049, 25 August 2005. [Google Scholar]
- Speirs, A.D.; Oxland, T.R.; Masri, B.A.; Poursartip, A.; Duncan, C.P. Calcium phosphate cement composites in revision hip arthroplasty. Biomaterials 2005, 26, 7310–7318. [Google Scholar] [CrossRef] [PubMed]
- dos Santos, L.A.; Carrodeguas, R.G.; Boschi, A.O.; de Arruda, A.C.F. Fiber-enriched double-setting calcium phosphate bone cement. J. Biomed. Mater. Res. A 2003, 65, 244–250. [Google Scholar] [CrossRef] [PubMed]
- Gbureck, U.; Spatz, K.; Thull, R. Improvement of mechanical properties of self-setting calcium phosphate bone cements mixed with different metal oxides. Mat.-Wiss. u. Werkstofftech. 2003, 34, 1036–1040. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, H.H.K. Effects of synergistic reinforcement and absorbable fiber strength on hydroxyapatite bone cement. J. Biomed. Mater. Res. A 2005, 75, 832–840. [Google Scholar] [CrossRef] [PubMed]
- Buchanan, F.; Gallagher, L.; Jack, V.; Dunne, N. Short-fibre reinforcement of calcium phosphate bone cement. Proc. Inst. Mech. Eng. H: J. Eng. Med. 2007, 221, 203–212. [Google Scholar] [CrossRef]
- Wang, X.; Ye, J.; Wang, Y.; Chen, L. Reinforcement of calcium phosphate cement by bio-mineralized carbon nanotube. J. Am. Ceram. Soc. 2007, 90, 962–964. [Google Scholar] [CrossRef]
- According to Wikipedia, the free encyclopedia: Concrete is a construction material that consists of a cement (commonly Portland cement),aggregates (generally gravel and sand) and water. It solidifies and hardens after mixing and placement due to a chemical process known as hydration. The water reacts with the cement, which bonds the other components together, eventually creating a stone-like material. http://en.wikipedia.org/wiki/Concrete (accessed in January 2009).
- Dickens-Venz, S.H.; Takagi, S.; Chow, L.C.; Bowen, R.L.; Johnston, A.D.; Dickens, B. Physical and chemical properties of resin-reinforced calcium phosphate cements. Dent. Mater. 1994, 10, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.H.K.; Eichmiller, F.C.; Barndt, P.R. Effects of fiber length and volume fraction on the reinforcement of calcium phosphate cement. J. Mater. Sci. Mater. Med. 2001, 12, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.H.K.; Quinn, J.B.; Takagi, S.; Chow, L.C. Synergistic reinforcement of in situ hardening calcium phosphate composite scaffold for bone tissue engineering. Biomaterials 2004, 25, 1029–1037. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, A.; Yamamoto, S.; Kawasaki, T.; Kohgo, T.; Nakasu, M. Development of calcium phosphate cement using chitosan and citric acid for bone substitute materials. Biomaterials 2002, 23, 1091–1101. [Google Scholar] [CrossRef] [PubMed]
- Takagi, S.; Chow, L.C.; Hirayama, S.; Eichmiller, F.C. Properties of elastomeric calcium phosphate cement-chitosan composites. Dent. Mater. 2003, 19, 797–804. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Xu, H.H.K.; Takagi, S.; Chow, L.C. Fast setting calcium phosphate cement – chitosan composite, mechanical properties and dissolution rates. J. Biomater. Appl. 2007, 21, 299–316. [Google Scholar] [CrossRef] [PubMed]
- Pan, Z.H.; Jiang, P.P.; Fan, Q.Y.; Ma, B.; Cai, H.P. Mechanical and biocompatible influences of chitosan fiber and gelatin on calcium phosphate cement. J. Biomed. Mater. Res. B Appl. Biomater. 2007, 82, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Li, H.; Cheng, W.; Yang, Y.; Zhu, M.; Zhou, C. Novel injectable calcium phosphate/chitosan composites for bone substitute materials. Acta Biomater. 2006, 2, 557–565. [Google Scholar] [CrossRef] [PubMed]
- Pan, Z.H.; Cai, H.P.; Jiang, P.P.; Fan, Q.Y. Properties of a calcium phosphate cement synergistically reinforced by chitosan fiber and gelatin. J. Polymer Res. 2006, 13, 323–327. [Google Scholar] [CrossRef]
- Weir, M.D.; Xu, H.H.K. High-strength.; in situ-setting calcium phosphate composite with protein release. J. Biomed. Mater. Res. A 2008, 85, 388–396. [Google Scholar] [CrossRef] [PubMed]
- Lian, Q.; Li, D.C.; He, J.K.; Wang, Z. Mechanical properties and in-vivo performance of calcium phosphate cement – chitosan fibre composite. Proc. Inst. Mech. Eng. H: J. Eng. Med. 2008, 222, 347–353. [Google Scholar] [CrossRef]
- Wang, X.; Chen, L.; Xiang, H.; Ye, J. Influence of anti-washout agents on the rheological properties and injectability of a calcium phosphate cement. J. Biomed. Mater. Res. B Appl. Biomater. 2007, 81, 410–418. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Zhang, S.; Chen, T.; Liu, C.; Lin, S.; Tian, X. Calcium phosphate cement reinforced by polypeptide copolymers. J. Biomed. Mater. Res. B Appl. Biomater. 2006, 76, 432–439. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, Y.; Ishikawa, K.; Takechi, M.; Toh, T.; Yuasa, T.; Nagayama, M.; Suzuki, K. Basic properties of calcium phosphate cement containing atelocollagen in its liquid or powder phases. Biomaterials 1998, 19, 707–715. [Google Scholar] [CrossRef] [PubMed]
- Otsuka, M.; Kuninaga, T.; Otsuka, K.; Higuchi, W.I. Effect of nanostructure on biodegradation behaviors of self-setting apatite/collagen composite cements containing vitamin K2 in rats. J. Biomed. Mater. Res. B Appl. Biomater. 2006, 79, 176–184. [Google Scholar] [CrossRef] [PubMed]
- Moreau, J.L.; Weir, M.D.; Xu, H.H.K. Self-setting collagen – calcium phosphate bone cement: mechanical and cellular properties. J. Biomed. Mater. Res. A 2009, in press. [Google Scholar]
- Xu, H.H.K.; Eichmiller, F.C.; Giuseppetti, A.A. Reinforcement of a self-setting calcium phosphate cement with different fibers. J. Biomed. Mater. Res. 2000, 52, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.H.K.; Quinn, J.B.; Takagi, S.; Chow, L.C.; Eichmiller, F.C. Strong and macroporous calcium phosphate cement.; effects of porosity and fiber reinforcement on mechanical properties. J. Biomed. Mater. Res. 2001, 57, 457–466. [Google Scholar] [CrossRef] [PubMed]
- dos Santos, L.A.; Carrodeguas, R.G.; Boschi, A.O.; de Arruda, A.C.F. Dual-setting calcium phosphate cement modified with ammonium polyacrylate. Artif. Organs 2003, 27, 412–418. [Google Scholar] [CrossRef] [PubMed]
- dos Santos, L.A.; de Oliveira, L.C.; da Silva Rigo, E.C.; Carrodéguas, R.G.; Boschi, A.O.; de Arruda, A.C.F. Fiber reinforced calcium phosphate cement. Artif. Organs 2000, 24, 212–216. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.S.; Chen, C.W.; Ducheyne, P. In vitro surface reaction layer formation and dissolution of calcium phosphate cement – bioactive glass composites. Biomed. Mater. 2008, 3, 034111:1–034111:11. [Google Scholar] [CrossRef]
- Xu, H.H.K.; Simon, C.G., Jr. Self-hardening calcium phosphate composite scaffold for bone Tissue Engineering. J. Orthop. Res. 2004, 22, 535–543. [Google Scholar] [CrossRef] [PubMed]
- Burguera, E.F.; Xu, H.H.K.; Takagi, S.; Chow, L.C. High early strength calcium phosphate bone cement, Effects of dicalcium phosphate dihydrate and absorbable fibers. J. Biomed. Mater. Res. A 2005, 75, 966–975. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.H.K.; Quinn, J.B. Calcium phosphate cement containing resorbable fibers for short-term reinforcement and macroporosity. Biomaterials 2002, 23, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Gorst, N.J.S.; Perrie, Y.; Gbureck, U.; Hutton, A.L.; Hofmann, M.P.; Grover, L.M.; Barralet, J.E. Effects of fiber reinforcement on the mechanical properties of brushite cement. Acta Biomater. 2006, 2, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Julien, M.; Khairoun, I.; LeGeros, R.Z.; Delplace, S.; Pilet, P.; Weiss, P.; Daculsi, G.; Bouler, J.M.; Guicheux, J. Physico-chemical-mechanical and in vitro biological properties of calcium phosphate cements with doped amorphous calcium phosphates. Biomaterials 2007, 28, 956–965. [Google Scholar] [CrossRef] [PubMed]
- Lemaître, J.; Munting, E.; Mirtchi, A.A. Setting, hardening and resorption of calcium phosphate hydraulic cements. Rev. Stomatol. Chir. Maxillofac. 1992, 93, 163–165. [Google Scholar] [PubMed]
- Müller, F.A.; Gbureck, U.; Kasuga, T.; Mizutani, Y.; Barralet, J.E.; Lohbauer, U. Whisker-reinforced calcium phosphate cements. J. Am. Ceram. Soc. 2007, 90, 3694–3697. [Google Scholar] [CrossRef]
- Nakagawa, A.; Matsuya, S.; Takeuchi, A.; Ishikawa, K. Comparison of the effects of added α – and β-tricalcium phosphate on the basic properties of apatite cement. Dent. Mater. J. 2007, 26, 342–347. [Google Scholar] [CrossRef] [PubMed]
- Gisep, A.; Wieling, R.; Bohner, M.; Matter, S.; Schneider, E.; Rahn, B. Resorption patterns of calcium-phosphate cements in bone. J. Biomed. Mater. Res. A 2003, 66, 532–540. [Google Scholar] [CrossRef] [PubMed]
- Ion-substituted ACPs contained other ions (carbonates, Mg, Zn, F) were used in that study [375].
- Gbureck, U.; Spatz, K.; Thull, R.; Barralet, J.E. Rheological enhancement of mechanically activated α-tricalcium phosphate cements. J. Biomed. Mater. Res. B Appl. Biomater. 2005, 73, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Castaldini, A.; Cavallini, A. Setting properties of bone cement with added synthetic hydroxyapatite. Biomaterials 1985, 6, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Sogal, A.; Hulbert, S.F. Mechanical properties of a composite bone cement, polymethylmethacrylate and hydroxyapatite. In Proceedings of the 5th International Symposium on Ceramics in Medicine, Kyoto, Japan, November, 1992; Volume 5, pp. 213–224.
- Harper, E.J.; Behiri, J.C.; Bonfield, W. Flexural and fatigue properties of a bone cement based upon polyethylmethacrylate and hydroxyapatite. J. Mater. Sci. Mater. Med. 1995, 6, 799–803. [Google Scholar] [CrossRef]
- Harper, E.J. Bioactive bone cements. Proc. Inst. Mech. Eng. H: J. Eng. Med. 1998, 212, 113–120. [Google Scholar] [CrossRef]
- Shinzato, S.; Kobayashi, M.; Mousa, W.F.; Kamimura, M.; Neo, M.; Kitamura, Y.; Kokubo, T.; Nakamura, T. Bioactive polymethylmethacrylate-based bone cement: comparison of glass beads, apatite- and wollastonite-containing glass-ceramic, and hydroxyapatite fillers on mechanical and biological properties. J. Biomed. Mater. Res. 2000, 51, 258–272. [Google Scholar] [CrossRef] [PubMed]
- Harper, E.J.; Braden, M.; Bonfield, W. Mechanical properties of hydroxyapatite reinforced poly(ethylmethacrylate) bone cement after immersion in a physiological solution, Influence of a silane coupling agent. J. Mater. Sci. Mater. Med. 2000, 11, 491–497. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.H.K.; Quinn, J.B. Whisker-reinforced bioactive composites containing calcium phosphate cement fillers, Effects of filler ratio and surface treatments on mechanical properties. J. Biomed. Mater. Res. 2001, 57, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Claes, L.; Höllen, I.; Ignatius, A. Resorbable bone cements. Orthopäde 1997, 26, 459–462. [Google Scholar] [PubMed]
- Jansen, J.A.; de Ruijter, J.E.; Schaeken, H.G.; van der Waerden, J.P.C.; Planell, J.A.; Driessens, F.C.M. Evaluation of tricalciumphosphate/hydroxyapatite cement for tooth replacement, an experimental animal study. J. Mater. Sci. Mater. Med. 1995, 6, 653–657. [Google Scholar] [CrossRef]
- Larsson, S.; Bauer, TW. Use of injectable calcium phosphate cement for fracture fixation: a review. Clin. Orthop. Relat. Res. 2002, 395, 23–32. [Google Scholar]
- Gbureck, U.; Knappe, O.; Hofmann, N.; Barralet, J.E. Antimicrobial properties of nanocrystalline tetracalcium phosphate cements. J. Biomed. Mater. Res. B Appl. Biomater. 2007, 83, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Sethuraman, S.; Nair, L.S.; El-Amin, S.; Nguyen, M.T.N.; Greish, Y.E.; Bender, J.D.; Brown, P.W.; Allcock, H.R.; Laurencin, C.T. Novel low temperature setting nanocrystalline calcium phosphate cements for bone repair: osteoblast cellular response and gene expression studies. J. Biomed. Mater. Res. A 2007, 82, 884–891. [Google Scholar] [CrossRef] [PubMed]
- Link, D.P.; van den Dolder, J.; Wolke, J.G.C.; Jansen, J.A. The cytocompatibility and early osteogenic characteristics of an injectable calcium phosphate cement. Tissue Eng. 2007, 13, 493–500. [Google Scholar] [CrossRef] [PubMed]
- Oda, H.; Nakamura, K.; Matsushita, T.; Yamamoto, S.; Ishibashi, H.; Yamazaki, T.; Morimoto, S. Clinical use of a newly developed calcium phosphate cement (XSB-671D). J. Orthop. Sci. 2006, 11, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Braun, C.; Rahn, B.; Fulmer, M.T.; Steiner, A.; Gisep, A. Intra-articular calcium Phosphate cement, its fate and impact on joint tissues in a rabbit model. J. Biomed. Mater. Res. B Appl. Biomater. 2006, 79, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Costantino, P.; Friedman, C.; Jones, K.; Chow, L.C.; Pelzer, H.; Sisson, G. Hydroxyapatite cement. I. Basic chemistry and histologic properties. Arch. Otolaryngol. Head Neck Surg. 1991, 117, 379–384. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.C.; Wang, J.T.; Hong, C.Y.; Brown, W.E.; Chow, C.Y. The periapical tissue reactions to a calcium phosphate cement in the teeth of monkeys. J. Biomed. Mater. Res. 1991, 25, 485–498. [Google Scholar] [CrossRef] [PubMed]
- Sugawara, A.; Fujikawa, K.; Kusama, K.; Nishiyama, M.; Murai, S.; Takagi, S.; Chow, L.C. Histopathologic reaction of calcium phosphate cement for alveolar ridge augmentation. J. Biomed. Mater. Res. 2002, 61, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Fujikawa, K.; Sugawara, A.; Kusama, K.; Nishiyama, M.; Murai, S.; Takagi, S.; Chow, L.C. Fluorescent labeling analysis and electron probe microanalysis for alveolar ridge augmentation using calcium phosphate cement. Dent. Mater. J. 2002, 21, 296–305. [Google Scholar] [CrossRef] [PubMed]
- Sugawara, A.; Chow, L.C.; Takagi, S.; Chohayeb, H. In vitro evaluation of the sealing ability of a calcium phosphate cement when used as a root canal sealer-filler. J. Endod. 1990, 16, 162–165. [Google Scholar] [CrossRef] [PubMed]
- Noetzel, J.; Özer, K.; Reisshauer, B.H.; Anil, A.; Rössler, R.; Neumann, K.; Kielbassa, A.M. Tissue responses to an experimental calcium phosphate cement and mineral trioxide aggregate as materials for furcation perforation repair, a histological study in dogs. Clin. Oral Invest. 2006, 10, 77–83. [Google Scholar] [CrossRef]
- Zhang, W.; Walboomers, X.F.; Jansen, J.A. The formation of tertiary dentin after pulp capping with a calcium phosphate cement, loaded with PLGA microparticles containing TGF-β1. J. Biomed. Mater. Res. A 2008, 85, 439–444. [Google Scholar] [CrossRef] [PubMed]
- Comuzzi, L.; Ooms, E.; Jansen, J.A. Injectable calcium phosphate cement as a filler for bone defects around oral implants, an experimental study in goats. Clin. Oral Implants Res. 2002, 13, 304–311. [Google Scholar] [CrossRef] [PubMed]
- Shirakata, Y.; Oda, S.; Kinoshita, A.; Kikuchi, S.; Tsuchioka, H.; Ishikawa, I. Histocompatible healing of periodontal defects after application of injectable calcium phosphate bone cement. A preliminary study in dogs. J. Periodontol. 2002, 73, 1043–1053. [Google Scholar] [CrossRef] [PubMed]
- Chaung, H.M.; Hong, C.H.; Chiang, C.P.; Lin, S.K.; Kuo, Y.S.; Lan, W.H.; Hsieh, C.C. Comparison of calcium phosphate cement mixture and pure calcium hydroxide as direct pulp-capping agents. J. Formos Med. Assoc. 1996, 95, 545–550. [Google Scholar] [PubMed]
- Aral, A.; Yalçn, S.; Karabuda, Z.C.; Anιl, A.; Jansen, J.A.; Mutlu, Z. Injectable calcium phosphate cement as a graft material for maxillary sinus augmentation: an experimental pilot study. Clin. Oral Implants Res. 2008, 19, 612–617. [Google Scholar] [CrossRef] [PubMed]
- Sliindo, M.L.; Costantino, P.D.; Friedman, C.D.; Chow, L.C. Facial skeletal augmentation using hydroxyapatite cement cranioplasty. Arch. Otolaryngol. Head Neck Surg. 1993, 119, 185–190. [Google Scholar] [CrossRef] [PubMed]
- Bifano, C.A.; Edgin, W.A.; Colleton, C.; Bifano, S.L.; Constantino, P.D. Preliminary evaluation of hydroxyapatite cement as an augmentation device in the edentulous atrophic canine mandible. Oral Surg. 1998, 85, 512–516. [Google Scholar]
- Friedman, C.D.; Constantino, P.D.; Jones, K.; Chow, L.C.; Pelzer, H.; Sisson, G. Hydroxyapatite cement. II. Obliteration and reconstruction of the cat frontal sinus. Arch. Otolaryngol. Head Neck Surg. 1991, 117, 385–389. [Google Scholar] [CrossRef] [PubMed]
- Sinikovic, B.; Kramer, F.J.; Swennen, G.; Lubbers, H.T.; Dempf, R. Reconstruction of orbital wall defects with calcium phosphate cement: clinical and histological findings in a sheep model. Int. J. Oral Maxillofac. Surg. 2007, 36, 54–61. [Google Scholar] [CrossRef]
- Smartt, J.M.; Karmacharya, J.; Gannon, F.H.; Ong, G.; Jackson, O.; Bartlett, S.P.; Poser, R.D.; Kirschner, R.E. Repair of the immature and mature craniofacial skeleton with a carbonated calcium phosphate cement: assessment of biocompatibility, osteoconductivity and remodeling capacity. Plast. Reconstr. Surg. 2005, 115, 1642–1650. [Google Scholar] [CrossRef] [PubMed]
- Reddi, S.P.; Stevens, M.R.; Kline, S.N.; Villanueva, P. Hydroxyapatite cement in craniofacial trauma surgery, indications and early experience. J. Cran. Maxillofac. Trauma 1999, 5, 7–12. [Google Scholar]
- Friedman, C.D.; Costantino, P.D.; Synderman, C.H.; Chow, L.C.; Takagi, S. Reconstruction of the frontal sinus and frontofacial skeleton with hydroxyapatite cement. Arch. Facial Plast. Surg. 2000, 2, 124–129. [Google Scholar] [CrossRef] [PubMed]
- Kuemmerle, J.M.; Oberle, A.; Oechslin, C.; Bohner, M.; Frei, C.; Boecken, I.; von Rechenberg, B. Assessment of the suitability of a new brushite calcium phosphate cement for cranioplasty – an experimental study in sheep. J. Cran. Maxillofac. Surg. 2005, 33, 37–44. [Google Scholar] [CrossRef]
- Luaces-Rey, R.; García-Rozado, A.; Crespo-Escudero, J.L.; Seijas, B.P.; Arenaz-Búa, J.; López-Cedrún, J.L. Use of carbonated calcium phosphate bone cement and resorbable plates for the treatment of frontal sinus fractures: two case reports. J. Plastic Reconstr. Aesthetic Surg. 2009, 62, 272–273. [Google Scholar] [CrossRef]
- Tamimi, F.; Torres, J.; Cabarcos, E.L.; Bassett, D.C.; Habibovic, P.; Luceron, E.; Barralet, J.E. Minimally invasive maxillofacial vertical bone augmentation using brushite based cements. Biomaterials 2009, 30, 208–216. [Google Scholar] [CrossRef] [PubMed]
- Liverneaux, P. Osteoporotic distal radius curettage-filling with an injectable calcium phosphate cement. A cadaveric study. Eur. J. Orthop. Surg. Traumatol. 2005, 15, 1–6. [Google Scholar] [CrossRef]
- Liverneaux, P.; Vernet, P.; Robert, C.; Diacono, P. Cement pinning of osteoporotic distal radius fractures with an injectable calcium phosphate bone substitute, report of 6 cases. Eur. J. Orthop. Surg. Traumatol. 2006, 16, 10–16. [Google Scholar] [CrossRef]
- Thordarson, D.; Hedman, T.; Yetkinler, D.; Eskander, E.; Lawrence, T.; Poser, R. Superior compressive strength of a calcaneal fracture construct augmented with remodelable cancellous bone cement. J. Bone Joint Surg. Am. 1999, 81A, 239–246. [Google Scholar] [PubMed]
- Stankewich, C.J.; Swiontkowski, M.F.; Tencer, A.F.; Yetkinler, D.N.; Poser, R.D. Augmentation of femoral neck fracture fixation with an injectable calcium-phosphate bone mineral cement. J. Orthopaedic Res. 1996, 14, 786–793. [Google Scholar] [CrossRef]
- Goodman, S.; Bauer, T.; Carter, D.; Casteleyn, P.P.; Goldstein, S.A.; Kyle, R.F.; Larsson, S.; Stankewich, C.J.; Swiontkowski, M.F.; Tencer, A.F.; Yetkinler, D.N.; Poser, R.D. Norian SRS® cement augmentation in hip fracture treatment. Clin. Orthop. Rel. Res. 1998, 348, 42–50. [Google Scholar] [CrossRef]
- Bai, B.; Jazrawi, L.; Kummer, F.; Spivak, J. The use of an injectable, biodegradable calcium orthophosphate bone substitute for the prophylactic augmentation of osteoporotic vertebrae and the management of vertebral compression fractures. Spine 1999, 24, 1521–1526. [Google Scholar] [CrossRef] [PubMed]
- Horstmann, W.G.; Verheyen, C.C.P.M.; Leemans, R. An injectable calcium phosphate cement as a bone-graft substitute in the treatment of displaced lateral tibial plateau fractures. Injury 2003, 34, 141–144. [Google Scholar] [CrossRef] [PubMed]
- Simpson, D.; Keating, J.F. Outcome of tibial plateau fractures managed with calcium phosphate cement. Injury 2004, 35, 913–918. [Google Scholar] [CrossRef] [PubMed]
- Welch, R.D.; Zhang, H.; Bronson, D.G. Experimental tibial plateau fractures augmented with calcium phosphate cement or autologous bone graft. J. Bone Joint Surg. Am. 2003, 85A, 222–231. [Google Scholar] [PubMed]
- Keating, J.F.; Hajducka, C.L.; Harper, J. Minimal internal fixation and calcium phosphate cement in the treatment of fractures of the tibial plateau, a pilot study. J. Bone Joint Surg. Br. 2003, 85B, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Moore, D.; Maitra, R.; Farjo, L.; Graziano, G.; Goldstein, S. Restoration of pedicle screw fixation with an in situ setting calcium orthophosphate cement. Spine 1997, 22, 1696–1705. [Google Scholar] [CrossRef] [PubMed]
- Mermelstein, L.E.; McLain, R.F.; Yerby, S.A. Reinforcement of thoracolumbar burst fractures with calcium phosphate cement. Spine 1998, 23, 664–671. [Google Scholar] [CrossRef] [PubMed]
- Mermelstein, L.E.; Chow, L.C.; Friedman, C.; Crisco, J. The reinforcement of cancellous bone screws with calcium orthophosphate cement. J. Orthop. Trauma 1996, 10, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Liverneaux, P.; Khallouk, R. Calcium phosphate cement in wrist arthrodesis: three cases. J. Orthop. Sci. 2006, 11, 289–293. [Google Scholar] [CrossRef] [PubMed]
- Ooms, E.M.; Wolke, J.G.C.; van der Waerden, J.P.C.M.; Jansen, J.A. Use of injectable calcium phosphate cement for the fixation of titanium implants: an experimental study in goats. J. Biomed. Mater. Res. B Appl. Biomater. 2003, 66, 447–456. [Google Scholar] [CrossRef] [PubMed]
- Strauss, E.J.; Pahk, B.; Kummer, F.J.; Egol, K. Calcium phosphate cement augmentation of the femoral neck defect created after dynamic hip screw removal. J. Orthop. Trauma 2007, 21, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Schildhauer, T.A.; Bennett, A.P.; Wright, T.M.; Lane, J.M.; O’Leary, P.F. Intravertebral body reconstruction with an injectable in situ-setting carbonated apatite: biomechanical evaluation of a minimally invasive technique. J. Orthop. Res. 1999, 17, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Jansen, J.A.; Ooms, E.; Verdonschot, N.; Wolke, J.G.C. Injectable calcium phosphate cement for bone repair and implant fixation. Orthop. Clin. North Am. 2005, 36, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Maestretti, G.; Cremer, C.; Otten, P.; Jakob, R.P. Prospective study of standalone balloon kyphoplasty with calcium phosphate cement augmentation in traumatic fractures. Eur. Spine J. 2007, 16, 601–610. [Google Scholar] [CrossRef] [PubMed]
- Hisatome, T.; Yasunaga, Y.; Ikuta, Y.; Fujimoto, Y. Effects on articular cartilage of subchondral replacement with polymethylmethacrylate and calcium phosphate cement. J. Biomed. Mater. Res. 2002, 59, 490–498. [Google Scholar] [CrossRef] [PubMed]
- Lim, T.H.; Brebach, G.T.; Renner, S.M.; Kim, W.J.; Kim, J.G.; Lee, R.E.; Andersson, G.B.; An, H.S. Biomechanical evaluation of an injectable calcium phosphate cement for vertebroplasty. Spine 2002, 27, 1297–1302. [Google Scholar] [CrossRef] [PubMed]
- Belkoff, S.M.; Mathis, J.M.; Jasper, L.E.; Deramond, H. An ex vivo biomechanical evaluation of a hydroxyapatite cement for use with vertebroplasty. Spine 2001, 26, 1542–1546. [Google Scholar] [CrossRef] [PubMed]
- Heini, P.F.; Berlemann, U.; Kaufmann, M.; Lippuner, K.; Fankhauser, C.; van Landuyt, P. Augmentation of mechanical properties in osteoporotic vertebral bones – a biomechanical investigation of vertebroplasty efficacy with different bone cements. Eur. Spine J. 2001, 10, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Tomita, S.; Kin, A.; Yazu, M.; Abe, M. Biomechanical evaluation of kyphoplasty and vertebroplasty with calcium phosphate cement in a simulated osteoporotic compression fracture. J. Orthop. Sci. 2003, 8, 192–197. [Google Scholar] [CrossRef] [PubMed]
- Libicher, M.; Hillmeier, J.; Liegibel, U.; Sommer, U.; Pyerin, W.; Vetter, M.; Meinzer, H.P.; Grafe, I.; Meeder, P.; Nöldge, G.; Nawroth, P.; Kasperk, C. Osseous integration of calcium phosphate in osteoporotic vertebral fractures after kyphoplasty: initial results from a clinical and experimental pilot study. Osteoporos. Int. 2006, 17, 1208–1215. [Google Scholar] [CrossRef]
- Khanna, A.J.; Lee, S.; Villarraga, M.; Gimbel, J.; Steffey, D.; Schwardt, J. Biomechanical evaluation of kyphoplasty with calcium phosphate cement in a 2-functional spinal unit vertebral compression fracture model. Spine J. 2008, 8, 770–777. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.S.; Zhang, Z.M.; Mao, H.Q.; Geng, D.C.; Wang, G.L.; Gan, M.F.; Yang, H.L. Biomechanics of calcium phosphate cement in vertebroplasty. J. Clin. Rehabil. Tissue Eng. Res. 2008, 12, 8071–8074. [Google Scholar]
- Otsuka, M.; Matsuda, Y.; Suwa, Y.; Fox, J.L.; Higuchi, W.I. A novel skeletal drug-delivery system using a self-setting calcium orthophosphate cement. 3. Physicochemical properties and drug-release rate of bovine insulin and bovine albumin. J. Pharm. Sci. 1994, 83, 255–258. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.; Wong, J.; Matsuda, Y.; Fox, J.L.; Higuchi, W.I.; Otsuka, M. Self-setting hydroxyapatite cement: a novel skeletal drug-delivery system for antibiotics. J. Pharm. Sci. 1992, 81, 529–531. [Google Scholar] [CrossRef] [PubMed]
- Bohner, M.; Lemaître, J.; van Landuyt, P.; Zambelli, P.; Merkle, H.P.; Gander, B. Gentamicin-loaded hydraulic calcium orthophosphate bone cement as antibiotic delivery system. J. Pharm. Sci. 1997, 86, 565–572. [Google Scholar] [CrossRef] [PubMed]
- Bohner, M.; Lemaître, J.; Merkle, H.P.; Gander, B. Control of gentamicin release from a calcium phosphate cement by admixed poly(acrylic acid). J. Pharm. Sci. 2000, 89, 1262–1270. [Google Scholar] [CrossRef] [PubMed]
- Ratier, A.; Freche, M.; Locout, J.L.; Rodriguez, F. Behaviour of an injectable calcium phosphate cement with added tetracycline. Int. J. Pharm. 2004, 274, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Kisanuki, O.; Yajima, H.; Umeda, T.; Takakura, Y. Experimental study of calcium phosphate cement impregnated with dideoxy-kanamycin B. J. Orthop. Sci. 2007, 12, 281–288. [Google Scholar] [CrossRef] [PubMed]
- McNally, A.; Sly, K.; Lin, S.; Bourges, X.; Daculsi, G. Release of antibiotics from macroporous injectable calcium phosphate cement. Key Eng. Mater. 2008, 361-363, 359–362. [Google Scholar] [CrossRef]
- Hofmann, M.P.; Mohammed, A.R.; Perrie, Y.; Gbureck, U.; Barralet, J.E. High-strength resorbable brushite bone cement with controlled drug-releasing capabilities. Acta Biomater. 2009, 5, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Tamimi, F.; Torres, J.; Bettini, R.; Ruggera, F.; Rueda, C.; López-Ponce, M.; Cabarcos, E.L. Doxycycline sustained release from brushite cements for the treatment of periodontal diseases. J. Biomed. Mater. Res. A 2008, 85, 707–714. [Google Scholar] [CrossRef] [PubMed]
- Young, A.M.; Ng, P.Y.J.; Gbureck, U.; Nazhat, S.N.; Barralet, J.E.; Hofmann, M.P. Characterization of chlorhexidine-releasing, fast-setting, brushite bone cements. Acta Biomater. 2008, 4, 1081–1088. [Google Scholar] [CrossRef] [PubMed]
- Hesaraki, S.; Nemati, R. Cephalexin-loaded injectable macroporous calcium phosphate bone cement. J. Biomed. Mater. Res. B Appl. Biomater. 2009, in press. [Google Scholar]
- Otsuka, M.; Matsuda, Y.; Suwa, Y.; Fox, J.L.; Higuchi, W.I. A novel skeletal drug delivery system using a self-setting calcium orthophosphate cement. 5. Drug release behavior from a heterogeneous drug-loaded cement containing an anticancer drug. J. Pharm. Sci. 1994, 83, 1565–1568. [Google Scholar] [CrossRef] [PubMed]
- Otsuka, M.; Matsuda, Y.; Suwa, Y.; Fox, J.L.; Higuchi, W.I. A novel skeletal drug delivery system using a self-setting calcium orthophosphate cement. 2. Physicochemical properties and drug release rate of the cement-containing indomethacin. J. Pharm. Sci. 1994, 83, 611–615. [Google Scholar] [CrossRef] [PubMed]
- Panzavolta, S.; Torricelli, P.; Bracci, B.; Fini, M.; Bigi, A. Alendronate and pamidronate calcium phosphate bone cements, setting properties and in vitro response of osteoblast and osteoclast cells. J. Inorg. Biochem. 2009, 103, 101–106. [Google Scholar] [CrossRef] [PubMed]
- le Nihouannen, D.; Hacking, S.A.; Gbureck, U.; Komarova, S.V.; Barralet, J.E. The use of RANKL-coated brushite cement to stimulate bone remodeling. Biomaterials 2008, 29, 3253–3259. [Google Scholar] [CrossRef] [PubMed]
- Li, D.X.; Fan, H.S.; Zhu, X.D.; Tan, Y.F.; Xiao, W.Q.; Lu, J.; Xiao, Y.M.; Chen, J.Y.; Zhang, X.D. Controllable release of salmon-calcitonin in injectable calcium phosphate cement modified by chitosan oligosaccharide and collagen polypeptide. J. Mater. Sci. Mater. Med. 2007, 18, 2225–2231. [Google Scholar] [CrossRef] [PubMed]
- Kamegai, A.; Shimamura, N.; Naitou, K.; Nagahara, K.; Kanematsu, N.; Mori, M. Bone formation under the influence of bone morphogenetic protein/self-setting apatite cement composite as delivery system. Biomed. Mater. Eng. 1994, 4, 291–307. [Google Scholar] [PubMed]
- Fei, Z.; Hu, Y.; Wu, D.; Wu, H.; Lu, R.; Bai, J.; Song, H. Preparation and property of a novel bone graft composite consisting of rhBMP-2 loaded PLGA microspheres and calcium phosphate cement. J. Mater. Sci. Mater. Med. 2008, 19, 1109–1116. [Google Scholar] [CrossRef] [PubMed]
- Ruhé, P.Q.; Kroese-Deutman, H.C.; Wolke, J.G.C.; Spauwen, P.H.M.; Jansen, J.A. Bone inductive properties of rhBMP-2 loaded porous calcium phosphate cement implants in cranial defects in rabbits. Biomaterials 2004, 25, 2123–2132. [Google Scholar] [CrossRef] [PubMed]
- Bodde, E.W.H.; Boerman, O.C.; Russel, F.G.M.; Mikos, A.G.; Spauwen, P.H.M.; Jansen, J.A. The kinetic and biological activity of different loaded rhBMP-2 calcium phosphate cement implants in rats. J. Biomed. Mater. Res. A 2008, 87, 780–791. [Google Scholar] [CrossRef] [PubMed]
- Perrier, M.; Lu, Y.; Nemke, B.; Kobayashi, H.; Peterson, A.; Markel, M. Acceleration of second and fourth metatarsal fracture healing with recombinant human bone morphogenetic protein-2/calcium phosphate cement in horses. Vet. Surg. 2008, 37, 648–655. [Google Scholar] [CrossRef] [PubMed]
- Meraw, S.J.; Reeve, C.M.; Lohse, C.M.; Sioussat, T.M. Treatment of perimplant defects with combination growth factor cement. J. Periodont. 2000, 71, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Zang, X.F.; Zhao, Z.P.; Wang, J.L.; Mi, L. Co-transplantation of exogenous nerve growth factor and calcium phosphate cement composite for repairing rabbit radial bone defects. J. Clin. Rehabil. Tissue Eng. Res. 2008, 12, 8037–8041. [Google Scholar]
- Qu, X.Y.; Jiang, D.M.; Li, M.; Zhang, D.W.; Qin, J.Q.; Liu, C.K. Deproteinized osteoarticular allografts integrated with calcium phosphate cement and recombinant human vascular endothelial cell growth factor plus recombinant human bone morphogenetic protein-2, an immunological study. J. Clin. Rehabil. Tissue Eng. Res. 2008, 12, 8067–8070. [Google Scholar]
- Stallmann, H.P.; de Roo, R.; Faber, C.; Amerongen, A.V.N.; Wuisman, P.I.J.M. In vivo release of the antimicrobial peptide hLFi-11 from calcium phosphate cement. J. Orthop. Res. 2008, 26, 531–538. [Google Scholar]
- Sasaki, T.; Ishibashi, Y.; Katano, H.; Nagumo, A.; Toh, S. In vitro elution of vancomycin from calcium phosphate cement. J. Arthroplasty 2005, 20, 1055–1059. [Google Scholar] [CrossRef] [PubMed]
- Blom, E.J.; Klein-Nulend, J.; Wolke, J.G.C.; van Waas, M.A.J.; Driessens, F.C.M.; Burger, E.H. Transforming growth factor-β1 incorporation in a calcium phosphate bone cement, Material properties and release characteristics. J. Biomed. Mater. Res. 2002, 59, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Blom, E.J.; Klein-Nulend, J.; Yin, L.; van Waas, M.A.J.; Burger, E.H. Transforming growth factor-β1 incorporated in calcium phosphate cement stimulates osteotransductivity in rat calvarial bone defects. Clin. Oral Implants Res. 2001, 12, 609–616. [Google Scholar] [CrossRef]
- Link, D.P.; van den Dolder, J.; van den Beucken, J.J.; Wolke, J.G.C.; Mikos, A.G.; Jansen, J.A. Bone response and mechanical strength of rabbit femoral defects filled with injectable CaP cements containing TGF-β1 loaded gelatin microparticles. Biomaterials 2008, 29, 675–682. [Google Scholar] [CrossRef] [PubMed]
- Habraken, W.J.E.M.; Boerman, O.C.; Wolke, J.G.C.; Mikos, A.G.; Jansen, J.A. In vitro growth factor release from injectable calcium phosphate cements containing gelatin microspheres. J. Biomed. Mater. Res. A 2009, in press. [Google Scholar]
- Ruhé, P.Q.; Boerman, O.C.; Russel, F.G.M.; Mikos, A.G.; Spauwen, P.H.M.; Jansen, J.A. In vivo release of rhBMP-2 loaded porous calcium phosphate cement pretreated with albumin. J. Mater. Sci. Mater. Med. 2006, 17, 919–927. [Google Scholar] [CrossRef] [PubMed]
- Naito, K.; Obayashi, O.; Mogami, A.; Itoi, A.; Kaneko, K. Fracture of the calcium phosphate bone cement which used to enchondroma of the hand, a case report. Eur. J. Orthop. Surg. Traumatol. 2008, 18, 405–408. [Google Scholar] [CrossRef]
- Blattert, T.R.; Delling, G.; Weckbach, A. Evaluation of an injectable calcium phosphate cement as an autograft substitute for transpedicular lumbar interbody fusion: a controlled, prospective study in the sheep model. Eur. Spine J. 2003, 12, 216–223. [Google Scholar] [PubMed]
- Cavalcanti, S.C.; Santos, S.C.; Pereira, C.L.; Mazzonetto, R.; de Moraes, M.; Moreira, R.W.F. Histological and histomorphometric analyses of calcium phosphate cement in rabbit calvaria. J. Cran. Maxillofac. Surg. 2008, 36, 354–359. [Google Scholar] [CrossRef]
- Sanchez-Sotelo, J.; Munuera, L.; Madero, R. Treatment of fractures of the distal radius with a remodellable bone cement: a prospective, randomised study using Norian SRS®. J. Bone Joint Surg. Br. 2000, 82, 856–863. [Google Scholar] [CrossRef] [PubMed]
- Lobenhoffer, P.; Gerich, T.; Witte, F.; Tscherne, H. Use of an injectable calcium phosphate bone cement in the treatment of tibial plateau fractures: a prospective study of twenty-six cases with twenty-month mean follow-up. J. Orthop. Trauma 2002, 16, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Cassidy, C.; Jupiter, J.B.; Cohen, M.; Delli-Santi, M.; Fennell, C.; Leinberry, C.; Husband, J.; Ladd, A.; Seitz, W.R.; Constantz, B.R. Norian SRS® cement compared with conventional fixation in distal radial fractures, a randomized study. J. Bone Joint Surg. Am. 2003, 85, 2127–2137. [Google Scholar] [PubMed]
- Schmidt, R.; Cakir, B.; Mattes, T.; Wegener, M.; Puhl, W.; Richter, M. Cement leakage during vertebroplasty, an underestimated problem? Eur. Spine J. 2005, 14, 466–473. [Google Scholar] [CrossRef] [PubMed]
- Vlad, M.D.; Torres, R.; López, J.; Barracó, M.; Moreno, J.A.; Fernández, E. Does mixing affect the setting of injectable bone cement? An ultrasound study. J. Mater. Sci. Mater. Med. 2007, 18, 347–352. [Google Scholar] [CrossRef] [PubMed]
- Krebs, J.; Aebli, N.; Goss, B.G.; Sugiyama, S.; Bardyn, T.; Boecken, I.; Leamy, P.J.; Ferguson, S.J. Cardiovascular changes after pulmonary embolism from injecting calcium phosphate cement. J. Biomed. Mater. Res. B Appl. Biomater. 2007, 82, 526–532. [Google Scholar] [CrossRef] [PubMed]
- Russell, T.A.; Leighton, R.K. Comparison of autogenous bone graft and endothermic calcium phosphate cement for defect augmentation in tibial plateau fractures. A multicenter, prospective, randomized study. J. Bone Joint Surg. Am. 2008, 90A, 2057–2061. [Google Scholar] [CrossRef] [PubMed]
- Dickson, K.F.; Friedman, J.; Buchholz, J.G.; Flandry, F.D. The use of BoneSourceTM hydroxyapatite cement for traumatic metaphyseal bone void filling. J. Trauma 2002, 53, 1103–1108. [Google Scholar] [CrossRef] [PubMed]
- Takagi, S.; Chow, L.C.; Hirayama, S.; Sugawara, A. Premixed calcium phosphate cement pastes. J. Biomed. Mater. Res. B Appl. Biomater. 2003, 67, 689–696. [Google Scholar] [CrossRef] [PubMed]
- Carey, L.E.; Xu, H.H.K.; Simon, C.G., Jr.; Takagi, S.; Chow, L.C. Premixed rapid-setting calcium phosphate composites for bone repair. Biomaterials 2005, 26, 5002–5014. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.H.K.; Carey, L.E.; Simon, C.G., Jr.; Takagi, S.; Chow, L.C. Premixed calcium phosphate cements, synthesis.; physical properties.; and cell cytotoxicity. Dent. Mater. 2007, 23, 433–441. [Google Scholar] [CrossRef]
- Lemaître, J.; Pittet, C.; Brendlen, D. Pasty or liquid multiple constituent compositions for injectable calcium phosphate cements. Patent App. No. WO 03/041753 A1, 22 May 2003. [Google Scholar]
- Chow, L.C. Calcium phosphate materials, reactor response. Adv. Dent. Res. 1988, 2, 181–184. [Google Scholar]
- Tajima, S.; Kishi, Y.; Oda, M.; Maruta, M.; Matsuya, S.; Ishikawa, K. Fabrication of biporous low-crystalline apatite based on mannitol dissolution from apatite cement. Dent. Mater. J. 2006, 25, 616–620. [Google Scholar] [CrossRef] [PubMed]
- Bohner, M. Calcium phosphate emulsions: possible applications. Key Eng. Mater. 2001, 192-195, 765–768. [Google Scholar] [CrossRef]
- Troczynski, T. A concrete solution. Nature Mater. 2004, 3, 13–14. [Google Scholar] [CrossRef]
- Xu, H.H.K.; Burguera, E.F.; Carey, L.E. Strong.; macroporous and in situ-setting calcium phosphate cement-layered structures. Biomaterials 2007, 28, 3786–3796. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.H.K.; Takagi, S.; Quinn, J.B.; Chow, L.C. Fast-setting calcium phosphate scaffolds with tailored macropore formation rates for bone regeneration. J. Biomed. Mater. Res. A 2004, 68, 725–734. [Google Scholar] [CrossRef] [PubMed]
- Ginebra, M.P.; Rilliard, A.; Fernández, E.; Elvira, C.; san Roman, J.; Planell, J.A. Mechanical and rheological improvement of a calcium phosphate cement by the addition of a polymeric drug. J. Biomed. Mater. Res. 2001, 57, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Steffen, T.; Stoll, T.; Arvinte, T.; Schenk, R.K. Porous tricalcium phosphate and transforming growth factor used for anterior spine surgery. Eur. Spine J. 2001, 10, S132–S140. [Google Scholar] [CrossRef] [PubMed]
- Ginebra, M.P.; Delgado, J.A.; Harr, I.; Almirall, A.; del Valle, S.; Planell, J.A. Factors affecting the structure and properties of an injectable self-setting calcium phosphate foam. J. Biomed. Mater. Res. A 2007, 80, 351–361. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Su, J.; Wei, J.; Kong, H.; Liu, C. Biocompatibility and osteogenicity of degradable Ca-deficient hydroxyapatite scaffolds from calcium phosphate cement for bone tissue engineering. Acta Biomater. 2009, 5, 268–278. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Wei, J.; Kong, H.; Liu, C.; Pan, K. Biocompatibility and osteogenesis of calcium phosphate cement scaffolds for bone tissue engineering. Adv. Mater. Res. 2008, 47-50, 1383–1386. [Google Scholar] [CrossRef]
- Weir, M.D.; Xu, H.H.K.; Simon, Jr. C.G. Strong calcium phosphate cement-chitosan-mesh construct containing cell-encapsulating hydrogel beads for bone tissue engineering. J. Biomed. Mater. Res. A 2006, 77, 487–496. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.H.; Tan, W.Q.; Lin, J. Repair of madibular bone defect by combining calcium phosphate cement with bone morphogenetic protein composite as a bone graft material. Chinese J. Biomed. Engin. 2007, 26, 153–156. [Google Scholar]
- Niikura, T.; Tsujimoto, K.; Yoshiya, S.; Tadokoro, K.; Kurosaka, M.; Shiba, R. Vancomycin-impregnated calcium phosphate cement for methicillin-resistant staphylococcus aureus femoral osteomyelitis. Orthopedics 2007, 30, 320–321. [Google Scholar] [PubMed]
- Lode, A.; Wolf-Brandstetter, C.; Reinstorf, A.; Bernhardt, A.; König, U. Pompe, W.; Gelinsky, M. Calcium phosphate bone cements, functionalized with VEGF: release kinetics and biological activity. J. Biomed. Mater. Res. A 2007, 81, 474–483. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, M.; Toda, T. In vivo estimation of periapical bone reconstruction by chondroitin sulfate in calcium phosphate cement. J. Eur. Ceram. Soc. 2004, 24, 521–531. [Google Scholar] [CrossRef]
- Wang, J.L.; Mi, L.; Hou, G.H.; Zheng, Z. Repair of radial defects using calcium phosphate cements/poly lactic-co-glycolic acid materials combined with mesenchymal stem cells in rabbits. J. Clin. Rehabil. Tissue Eng. Res. 2008, 12, 8001–8005. [Google Scholar]
- Simon, C.G., Jr.; Guthrie, W.F.; Wang, F.W. Cell seeding into calcium phosphate cement. J. Biomed. Mater. Res. A 2004, 68, 628–639. [Google Scholar] [CrossRef] [PubMed]
- dos Santos, L.A.; Carrodéguas, R.G.; Rogero, S.O.; Higa, O.Z.; Boschi, A.O.; de Arruda, A.C. Alpha-tricalcium phosphate cement: “in vitro” cytotoxicity. Biomaterials 2002, 23, 2035–2042. [Google Scholar] [CrossRef] [PubMed]
- Baroud, G.; Steffen, T. A new cannula to ease cement injection during vertebroplasty. Eur. Spine J. 2005, 14, 474–479. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.M. The future of biomedical materials. J. Mater. Sci. Mater. Med. 2006, 17, 1025–1028. [Google Scholar] [CrossRef] [PubMed]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Dorozhkin, S.V. Calcium Orthophosphate Cements and Concretes. Materials 2009, 2, 221-291. https://doi.org/10.3390/ma2010221
Dorozhkin SV. Calcium Orthophosphate Cements and Concretes. Materials. 2009; 2(1):221-291. https://doi.org/10.3390/ma2010221
Chicago/Turabian StyleDorozhkin, Sergey V. 2009. "Calcium Orthophosphate Cements and Concretes" Materials 2, no. 1: 221-291. https://doi.org/10.3390/ma2010221
APA StyleDorozhkin, S. V. (2009). Calcium Orthophosphate Cements and Concretes. Materials, 2(1), 221-291. https://doi.org/10.3390/ma2010221




