1. Introduction
Atomic Layer Deposition (ALD) is a widely used thin-film deposition technique based on self-limiting surface reactions, offering excellent thickness control, high uniformity, and outstanding conformality [
1,
2,
3]. These features make ALD particularly attractive for applications in microelectronics, optoelectronics, photonics, and advanced functional coatings. However, achieving high film quality in ALD depends not only on the choice of precursors and deposition temperature, but also critically on the dynamics of gas transport and the removal of reaction by-products during the process [
1,
4]. A key step in every ALD cycle is the purge step, whose role is to remove excess precursor molecules and reaction products from the reactor chamber and to prevent parasitic gas-phase reactions. Inadequate purging leads to precursor overlap and extended residence times, which may deteriorate film uniformity, surface morphology, and structural quality [
3,
5]. Despite its fundamental importance, the influence of purge dynamics on growth kinetics, microstructure, and crystallinity of ALD-grown films has received relatively limited attention, especially in relation to reactor volume and gas residence effects.
In typical ALD reactors, different purge strategies can be employed. In the static purge vacuum mode (SPVM), the pumping system is closed during precursor exposure, allowing the precursor to remain in the reactor volume for a longer time and increasing its partial pressure near the substrate surface. In contrast, in the dynamic purge vacuum mode (DPVM), continuous pumping and carrier gas flow are applied, ensuring faster and more efficient removal of residual species and better separation of successive ALD half-reactions. These two approaches are expected to result in different growth regimes, ranging from nearly ideal, self-limited ALD to conditions with an increased contribution of non-ideal growth that may resemble chemical vapor deposition (CVD)-like processes [
1,
3], as the precursor may not fully escape the features it has diffused into. However, a direct comparison of their impact on film growth and structure is still lacking. Oxide materials such as ZnO and ZrO
2 were deliberately selected as model systems for this study for several complementary reasons. Both materials are technologically important and widely used in ALD-based applications: ZnO is a well-known wide-bandgap semiconductor employed in optoelectronics, transparent electronics, and sensors [
6,
7,
8], whereas ZrO
2 is a key high-k dielectric and a functional oxide used in photonic and phosphor-related structures [
4,
9,
10]. Therefore, understanding how process parameters affect their growth is directly relevant for practical applications. At the same time, ZnO and ZrO
2 differ significantly in their surface chemistry, precursor reactivity, and growth mechanisms in ALD. ZnO typically exhibits relatively robust and reproducible growth behavior [
6,
7,
11], while ZrO
2 is known to be more sensitive to process conditions, such as temperature and precursor residence time, showing a transition from amorphous to crystalline phases with increasing temperature [
4,
9,
10]. This contrast makes them ideal complementary test systems for evaluating how purge dynamics influence growth per cycle, surface morphology, and crystallinity. Furthermore, both materials can be deposited over a wide temperature range, from low-temperature regimes suitable for temperature-sensitive substrates to higher temperatures that promote crystallization and improved structural quality [
4,
6,
9]. This allows a direct comparison between low- and high-temperature growth regimes and enables assessment of how purge-mode engineering and temperature optimization jointly control film structure and conformality. As demonstrated in our previous reports [
12,
13], these materials have been successfully integrated into various functional applications, in which their structural integrity is a critical factor influencing device performance. Importantly, in most previous studies the film structure in ALD has been primarily tuned by changing the precursor chemistry or the deposition temperature [
1,
4,
6,
9]. In contrast, we present that purge-mode engineering—independent of precursor chemistry or deposition temperature—is a cost-effective strategy for tailoring the microstructure, morphology, and crystallinity of oxide thin films. This approach treats the purge step not only as a technical requirement, but also as an active process parameter for deliberate microstructure control.
This work presents a systematic study of the impact of purge mode on the growth, structure, and morphology of ZnO and ZrO
2 thin films deposited by ALD. Two purge regimes—DPVM and SPVM—were compared in reactors with different effective chamber volumes (47 and 470 cm
3), enabling an assessment of gas residence and scaling effects. The oxide films were characterized by X-ray diffraction (XRD) and atomic force microscopy (AFM) to correlate growth conditions with crystallinity and surface morphology, while growth per cycle (GPC) is used to analyze the growth kinetics. By linking process parameters with microstructure and film quality, this study showed that purge-mode engineering provides a simple and cost-effective route to tailor highly conformal oxide thin films without changing precursor chemistry [
1,
4,
5]. These findings are crucial for the development of functional applications, enabling the successful integration of optimized ALD oxides into phosphor and light-emitting diode (LED) structures.
3. Results and Discussion
The influence of the purge strategy on the growth behavior is directly reflected in the growth per cycle values summarized in
Table 1. For both ZnO and ZrO
2 films, and for investigated reactor volumes, the GPC obtained in SPVM is systematically higher than that measured in DPVM. This behavior can be explained by the fundamentally different gas transport conditions in both modes. In SPVM, closing the pumping system during exposure extends gas residence time, which may promote re-adsorption and secondary reactions. This can trigger higher GPC values. Conversely, DPVM utilizes continuous pumping and carrier gas flow to effectively remove precursors and by-products.
Although this leads to a slightly lower GPC, it provides growth control and eliminates non-specific gas-phase reactions [
3,
17]. Importantly, the comparison between the two reactor chamber volumes shows that, within the experimental uncertainty, the GPC values are essentially identical for a given material and purge mode. On the contrary, the use of the smaller chamber offers clear practical advantages, such as lower precursor consumption and a higher confidence in achieving uniform deposition throughout the entire chamber volume. From a technological point of view, this makes the small-volume reactor a more efficient and economically attractive solution without compromising film quality.
The higher GPC of ZrO
2 compared to ZnO originates from differences in precursor chemistry and surface reaction mechanisms (
Figure 1). In ALD, the growth per cycle is determined not only by the surface coverage, but also by the amount of material deposited per reactive site. The zirconium precursor TDMAZ is a multifunctional molecule containing four -N(CH
3)
2 ligands, which enables efficient reaction with surface -OH groups [
1]. Although the larger molecular size of TDMAZ may introduce steric hindrance effects that limit the maximum surface coverage, each chemisorption event results in the incorporation of a relatively large amount of material due to the high atomic mass of zirconium and the formation of a ZrO
2 unit. As a result, the mass deposited per cycle can be higher for ZrO
2 than for ZnO, even if the surface site density or steric accessibility differs. The observed higher GPC of ZrO
2 is therefore attributed to the combined effect of precursor chemistry and the higher mass contribution per adsorption event, rather than to film density alone. This interpretation is consistent with the known differences in ALD growth mechanisms between metal oxides based on different metal centers [
18,
19].
Figure 4 and
Figure 5 present the XRD patterns of ZnO thin films deposited in DPVM and SPVM for both reactor volumes (470 and 47 cm
3, respectively). In all cases, the diffraction peaks, including the (10.0), (00.2), (10.1), and (11.0) planes, correspond to the hexagonal wurtzite structure of ZnO. However, clear differences in structural ordering are observed between the two regimes. Films grown in DPVM show higher peak intensities and a better signal-to-noise ratio, indicating improved crystallinity and a well-developed polycrystalline structure. In contrast, films deposited via SPVM exhibit lower peak intensities and a higher diffuse background. The dominance of selected diffraction peaks may indicate the presence of preferred orientation (texturing), rather than single-crystal growth. This behavior suggests a lower degree of structural ordering and/or the presence of fine-grained or partially disordered material [
4]. These differences can be explained by the gas transport conditions in the two regimes. In SPVM, longer precursor residence time and limited gas flow create near-static conditions that may favor growth along preferred crystallographic directions. In DPVM, continuous gas flow and shorter residence time promote more homogeneous nucleation and more uniform polycrystalline structure with multiple crystallographic orientations [
3,
17]. As a result, DPVM leads to well-defined polycrystalline films, while SPVM tends to produce more disordered and/or preferentially oriented structures, regardless of the reactor volume. The AFM images with cross-sectional profiles with cross-sectional profiles presented in
Figure 6 and
Figure 7 further support the conclusions drawn from the XRD analysis. For the reactor with a volume of 470 cm
3, both DPVM and SPVM films exhibit an RMS roughness of about 4 nm; however, the DPVM film shows a more homogeneous surface with finer and more uniformly distributed surface features, while the SPVM film contains a larger number of pronounced local surface protrusions. For the smaller reactor (47 cm
3), the RMS roughness is approximately 4 nm for DPVM and about 3 nm for SPVM. Despite the slightly lower RMS value, the SPVM film exhibits a more diffuse and less well-defined surface texture, whereas the DPVM film maintains a more uniform and better-defined microstructure [
20]. This indicates that RMS roughness alone is not sufficient to describe the surface quality and must be interpreted together with structural information from XRD and additional AFM-derived parameters (see
Table 2). Importantly, reducing the reactor volume from 470 to 47 cm
3 does not change the crystalline phase of ZnO and does not deteriorate the film quality. Instead, the positive effect of DPVM on crystallinity and microstructural uniformity is preserved in both reactor volumes.
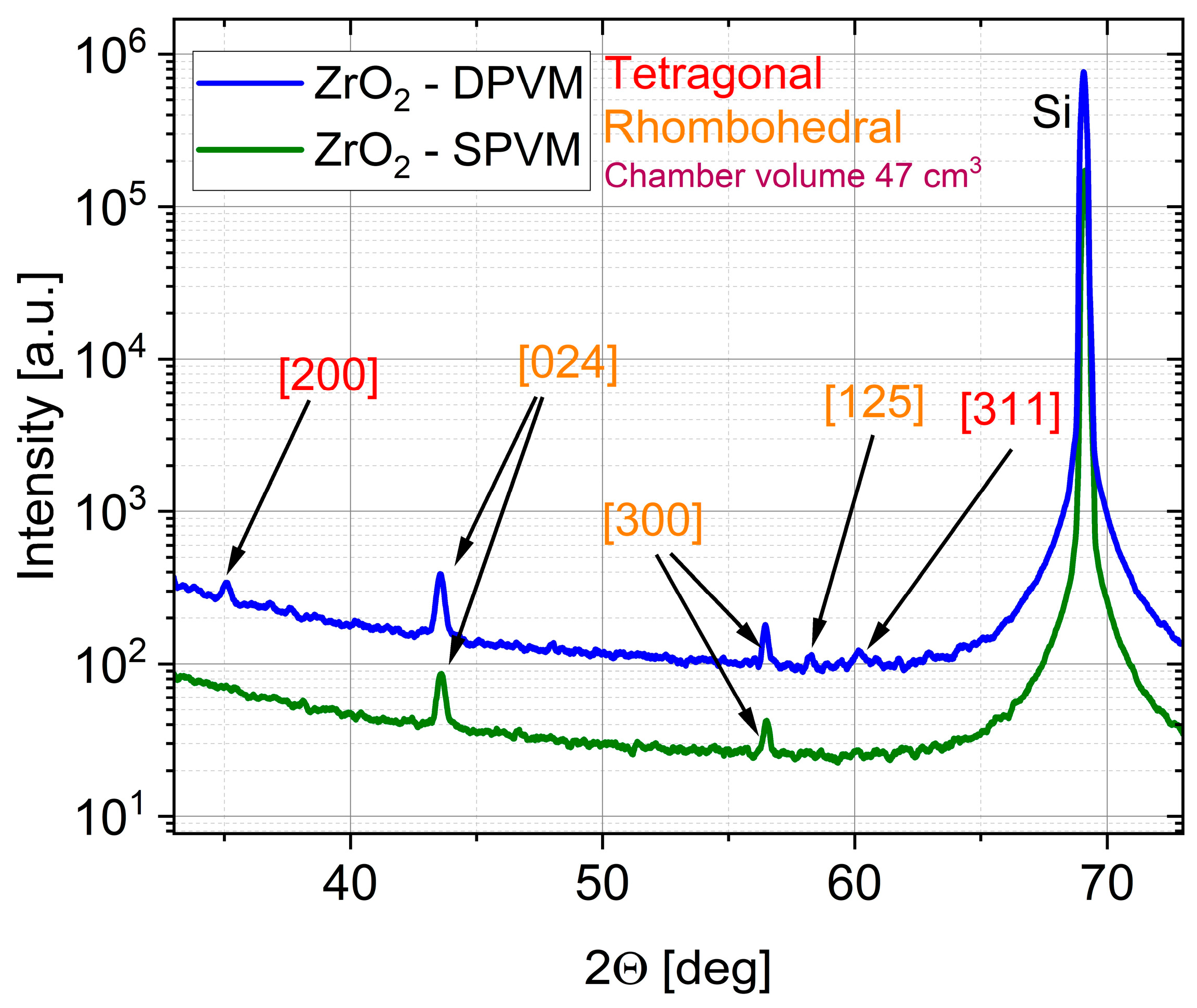
The effect of the purge mode is even more pronounced for ZrO
2, which is known to be more sensitive to growth chemistry and process conditions than ZnO. For ZrO
2 films deposited in the 47 cm
3 chamber, the XRD patterns in
Figure 8 reveal that the DPVM-deposited film exhibits distinctly higher diffraction intensities than the SPVM-deposited one. The observed reflections such as (200), (024), (300), (125), and (311) can be assigned to the tetragonal and rhombohedral phases of ZrO
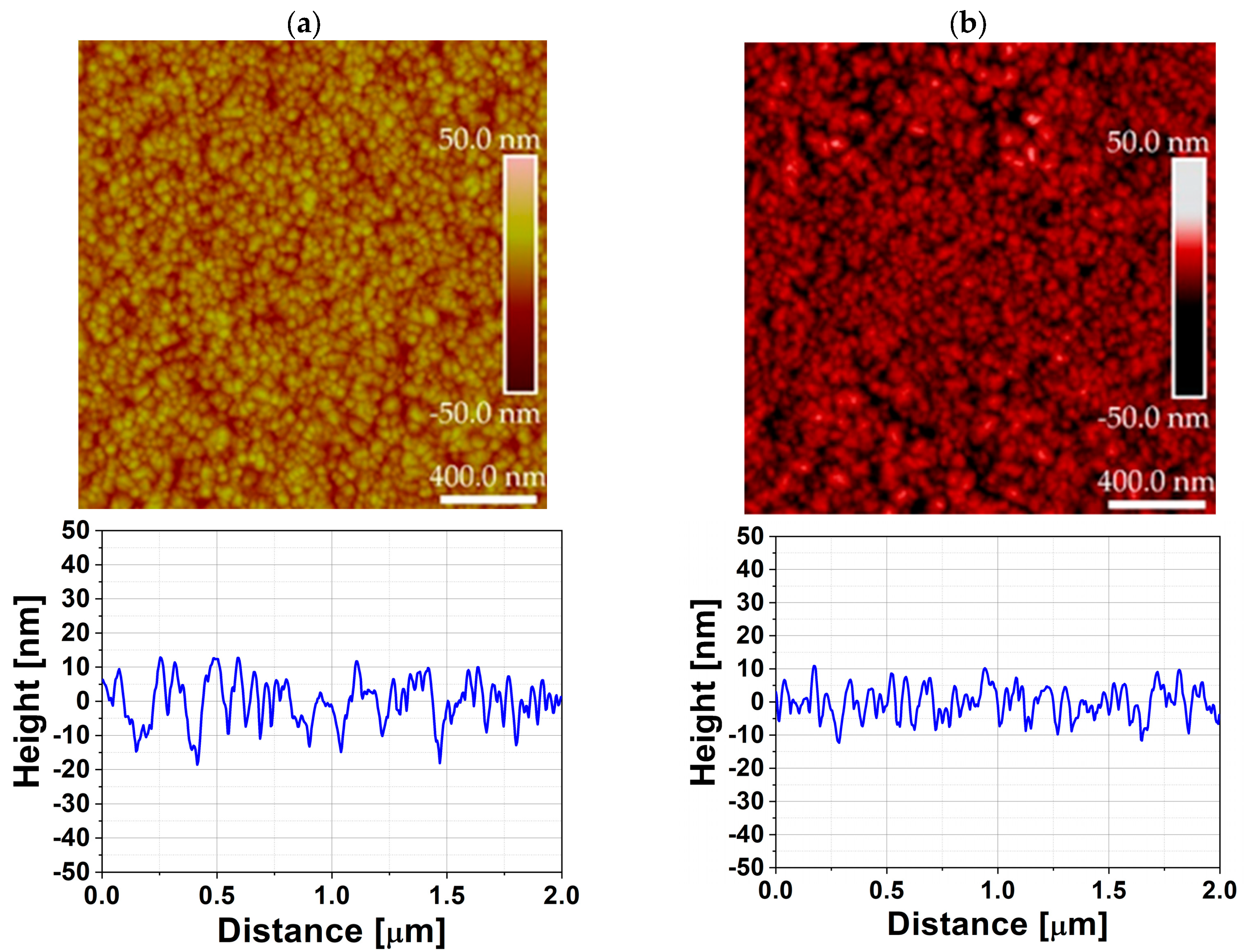
2, demonstrating that crystallization already occurs at this low temperature, particularly when DPVM is used. In contrast, the SPVM film shows much weaker diffraction peaks, indicating a lower degree of structural ordering. This is consistent with the AFM results with cross-sectional profiles in
Figure 9, which show higher RMS roughness (6 nm) values for SPVM compared to DPVM (4 nm). The DPVM film exhibits a smoother and more homogeneous surface, which reflects a more uniform nucleation and growth process [
20,
21].
AFM parameters in
Table 2 confirm that the AFM and XRD results are consistent and provide a coherent picture of the influence of the purge mode on film growth. For ZnO deposited in the 470 cm
3 chamber, DPVM leads to a much higher particle density (0.17 µm
−2) compared to SPVM (0.05 µm
−2), while maintaining a similar RMS roughness of about 4 nm and a lower R
max (36 nm vs. 42 nm). This higher nucleation density correlates well with the XRD results, where the DPVM-deposited ZnO film exhibits significantly higher diffraction intensities. In the smaller 47 cm
3 chamber, ZnO films deposited in DPVM also show a higher R
max (33 nm vs. 27 nm) and a lower particle density (0.10 µm
−2 vs. 1.10 µm
−2 in SPVM). Although the RMS roughness for the SPVM film (3 nm) is slightly lower than that obtained in DPVM (4 nm), the surface morphology appears more diffuse and less well-defined (
Figure 7). The SPVM film exhibits less distinct grain features and a more irregular surface texture, indicating reduced microstructural uniformity [
20]. In addition, the analysis of the threshold height and typical particle height/width parameters further supports the differences in the growth mechanism between DPVM and SPVM. For ZnO deposited in the 470 cm
3 chamber, the typical particle dimensions remain similar (about 20 nm in height and 200 nm in width); however, the threshold height is lower for DPVM (25 nm) than for SPVM (32 nm). This indicates a narrower height distribution and a reduced contribution of tall, protruding surface features in the DPVM case. This observation is consistent with the lower R
max and higher particle density obtained for DPVM and explains the improved crystalline ordering observed in the XRD patterns. For ZrO
2 deposited in the 47 cm
3 chamber, the differences are even more pronounced: the RMS roughness increases from about 4 nm in DPVM to 6 nm in SPVM, and R
max increases from 35 nm to 54 nm, while the particle density rises from 15 to 24 µm
−2, indicating a rougher and less homogeneous surface in SPVM. These morphological changes are directly reflected in the XRD patterns, where the DPVM film shows clearly higher diffraction intensities and better-defined peaks, evidencing improved crystallinity. Although the typical particle dimensions for ZrO
2 (about 20–25 nm in height and 100 nm in width) and the threshold height (29 nm) are comparable, the SPVM film exhibits a much larger R
max (54 nm) compared to the DPVM film (35 nm). This indicates a larger contribution of high, non-uniform surface features and a less homogeneous growth mode in SPVM. Such morphological inhomogeneity directly correlates with the weaker and broader diffraction peaks observed in XRD, evidencing a lower degree of structural ordering.
To gain deeper mechanistic insight into the reaction kinetics, the relationship between reactor chamber volume and precursor dosing was analyzed. A significant finding of this study is the high kinetic efficiency observed during reactor scaling: while the effective chamber volume was increased tenfold (from 47 cm
3 to 470 cm
3), the optimum precursor pulse times required only a twofold increase to maintain full surface saturation. Crucially, all other growth parameters—including purging times after precursor pulses and deposition temperatures—were kept identical across both chamber reactor volumes and purge modes to ensure a rigorous comparison. The experimental data from both chamber volumes suggest that the structural integrity of the films is primarily governed by the gas transport regime. To support this interpretation, the characteristic gas residence time (τ) was considered using the standard relation τ = V/Q, where V is the reactor volume and Q is the volumetric gas flow rate [
22]. According to this relation, a tenfold increase in chamber reactor volume should, in principle, lead to a proportional increase in residence time and thus require a correspondingly larger precursor pulse time to achieve saturation. However, the experimental results demonstrate that a mere twofold increase in precursor pulse time is sufficient. This indicates that surface saturation is not governed solely by the total chamber reactor volume or the bulk precursor concentration, but rather by the local availability of reactive species near the surface and the efficiency of mass transport [
19]. The SPVM, applied consistently across both reactor sizes, introduces a 1 s ‘wait time’ with a closed stop valve. This interval results in a forced stagnation of the gas phase, extending the precursor residence time far beyond the limits of natural convective transport. This dwell time allows precursor molecules to linger and diffuse throughout the volume, which may promote undesirable gas-phase collisions or precursor re-adsorption. Such a mechanistic breakdown potentially explains the systematic differences observed in the XRD and AFM data. Specifically, the sharper diffraction peaks and more uniform surface textures in DPVM-grown films likely result from a continuous convective flow that effectively clears the precursor load. In contrast, the reduced crystallinity and local surface protrusions in SPVM-grown films—observed regardless of chamber size—might be a consequence of this forced stagnation, which could disrupt the ideal self-limiting ALD regime by creating conditions that may only resemble CVD-like processes [
1,
3].
Importantly, reducing the reactor chamber volume does not alter the phase composition of ZnO, but it preserves the positive effect of DPVM on structural quality. Altogether, the combined analysis of typical particle size, threshold height, RMS roughness, and R
max for ZnO and ZrO
2 demonstrates that DPVM, even in the smaller reactor volume, promotes more uniform growth with a narrower height distribution and better-controlled microstructure, which is fully consistent with the improved crystallinity revealed by XRD. These results confirm that for both oxides, the purge mode is the dominant factor controlling the structural quality of the films. At the same time, the smaller reactor volume enables more efficient precursor utilization and enhanced control over gas-phase conditions without compromising film properties. Consequently, although SPVM yields a higher GPC than DPVM, this increase occurs at the expense of structural integrity and microstructural control. These observations confirm that DPVM facilitates a growth regime closer to ideal ALD through more effective purging and superior separation of the half-reactions. [
3,
17].
Crucially, the inferior uniformity and limited conformality inherent to the SPVM prevented the successful fabrication of functional optoelectronic devices, such as the phosphor and LED structures investigated here. We hypothesize that in SPVM-grown layers, a higher density of structural defects—including non-stoichiometric grain boundaries and localized pinholes—acts as potent non-radiative recombination centers and parasitic shunting paths [
23]. These defects lead to excessive leakage currents and thermal instability, effectively rendering SPVM films unsuitable for integration into active optoelectronic architectures [
24]. In contrast, the microstructural control afforded by DPVM suppresses these loss mechanisms, providing the film quality necessary for functional device integration.
Consequently, the following sections focus on demonstrating the feasibility of integrating these optimized DPVM-grown layers into phosphor and LED structures, as this regime ensures the structural integrity required for functional optoelectronic performance.
Although low-temperature ALD, especially in DPVM, already provides good uniformity and high conformality, higher growth temperatures remain preferable for device-quality layers [
4,
17]. At elevated temperatures, atoms on the growing surface can more easily rearrange into energetically favorable lattice positions, which promotes grain growth, reduces defects, and improves crystallinity and microstructural stability [
4]. These factors are critical for electronic and optoelectronic devices, where structural defects act as charge traps and scattering centers, degrading carrier transport, increasing leakage currents, and reducing optical efficiency. Moreover, films grown at higher temperature typically exhibit better thermal and electrical stability, which is essential for reliable long-term operation of devices such as n-i-p heterostructure LEDs and phosphor-based structures. Therefore, while low-temperature processes are indispensable for integration with temperature-sensitive substrates and for achieving highly conformal coatings, higher growth temperatures are still preferred whenever the highest structural quality and device performance are required [
4,
17].
The XRD pattern in
Figure 10 of the ZrO
2 film deposited at 240 °C in DPVM in the 47 cm
3 chamber confirms the presence of tetragonal and rhombohedral phases, indicating a high degree of crystallization already during growth. Well-defined reflections such as (006), (200), (202), (220), (300), and (311) with relatively high intensities demonstrate good structural quality and a low contribution of any amorphous phase. This indicates that the elevated growth temperature effectively promotes structural ordering of the ZrO
2 lattice. The obtained well-defined and crystalline ZrO
2 structure provides a suitable host matrix for the incorporation of rare-earth (RE) ions such as europium (Eu), terbium (Tb), and Cerium (Ce). The preserved phase purity and absence of secondary phases suggest that such a structurally ordered lattice is highly promising for rare-earth doping without significantly disturbing the crystal framework, as reported in previous studies [
25]. AFM analysis presented in
Figure 11 and
Table 3 shows that the as-grown ZrO
2:RE film exhibits an RMS roughness of about 7 nm, which decreases to approximately 5 nm after annealing at 800 °C. At the same time, the typical particle height and width remain around 20 nm and 100 nm, respectively. This is accompanied by a slight reduction in R
max (from 50 nm to 49 nm) and a decrease in threshold height (from 22 nm to 19 nm), pointing to a more uniform height distribution of surface features. The particle density remains nearly unchanged (from 99 to 91 µm
−2), suggesting that thermal treatment mainly promotes grain growth and surface smoothing rather than renucleation. Altogether, these results demonstrate that high-temperature growth and post-deposition annealing improve the structural and morphological quality of the ZrO
2 host and provide more favorable conditions for the activation of RE-related luminescent centers, which is crucial for phosphor and optoelectronic applications [
20,
25].
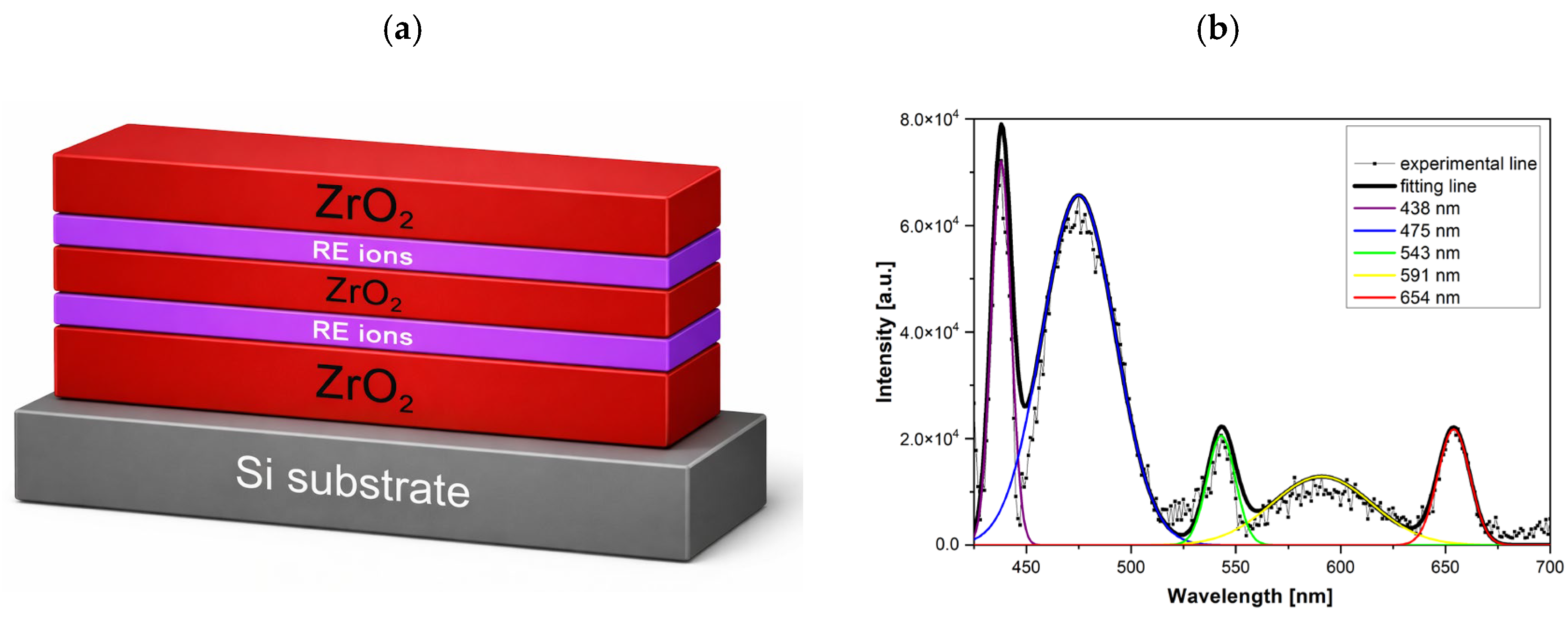
As mentioned in our previous work [
12], the schematic of the multilayer ZrO
2:RE structure shown in
Figure 12a illustrates a phosphor architecture consisting of alternating ZrO
2 layers and RE-doped active layers deposited on a silicon substrate, where the rare-earth ions are Eu, Tb, and Ce. Such multilayer systems require both precise thickness control and high structural quality, since the optical activity of RE ions is strongly influenced by the crystallinity of the host matrix and the local structural environment. The photoluminescence spectrum in
Figure 12b of the ZrO
2:RE films after annealing at 800 °C, decomposed using multi-peak Gaussian fitting, reveals emission bands centered at approximately 438, 475, 543, 591, and 654 nm. These emission bands are commonly associated with defect-related states in the ZrO
2 host as well as with optical centers related to rare-earth ions [
25,
26,
27]. Given the complexity of the defect structure of ZrO
2 and the possible overlap of emission bands, a unique assignment of each band to a specific RE ion (Eu, Tb, or Ce) is not straightforward. Nevertheless, the presence of RE dopants clearly modifies the emission spectrum and confirms the optical activity of the ZrO
2:RE system after high-temperature annealing.
As mentioned in our previous work [
13],
Figure 13 illustrates the device concept and its optical response. The schematic cross-section shows the n-ZnO/i-layer/p-GaN heterostructure diode deposited on a sapphire substrate, where both the ZnO layer and the intrinsic interlayer were grown by ALD in DPVM in a reactor with an effective chamber volume of 47 cm
3. As demonstrated in the previous sections, the DPVM process provides a growth regime closer to ideal, self-limited ALD, with improved microstructural control compared to SPVM, as evidenced by the combined AFM and XRD analyses. This is particularly important for device fabrication, where smooth, uniform, and highly conformal layers are required. The corresponding CIE chromaticity diagram for the n-ZnO/Al
2O
3/HfO
2/p-GaN heterostructure diodes confirms that our n-i-p structures enable efficient color tuning from blue emission toward visible white light. In agreement with the earlier structural analysis, ZnO layers grown at the higher temperature of 300 °C exhibit improved crystallinity and a well-defined wurtzite structure, which directly translates into more stable and reproducible optical emission [
6,
23]. For these films, the emission coordinates fall within the cold-white region of the CIE diagram, demonstrating the potential of this approach for white-light optoelectronic applications. AFM images in
Figure 14 further support the conclusions drawn from the earlier morphological analysis. The insertion of the intrinsic interlayer reduces the surface roughness from 16 nm for the p-GaN/n-ZnO structure to 7 nm for the p-GaN/i/n-ZnO structure, while the bare p-GaN substrate shows an RMS value of about 1 nm. This behavior is fully consistent with the previously discussed role of growth mode and process control: smoother and more homogeneous interfaces are obtained when growth proceeds in a well-controlled ALD regime, which is essential for minimizing interface-related defects in device structures. Finally, ZnO films grown at the relatively higher temperature of 300 °C form hexagonal columnar structures and exhibit the wurtzite phase, as evidenced by the XRD reflections (10.0), (00.2), (10.1), and (11.0), in agreement with our previous reports [
7].
This observation directly links the earlier structural discussion with device performance: higher growth temperature improves crystallinity and microstructural ordering, while DPVM ensures high conformality and uniformity. Together, these factors explain why the optimized ALD process conditions lead not only to improved film quality but also to superior and tunable electrical and optical responses of ZnO- or ZrO2-based device structures.