Comparative Study of Metallic Iron Production from High-Phosphorus Iron Ores: Carbon-Composite Pellet Direct Reduction–Melting vs. Granular Direct Reduction–Magnetic Separation–Melting
Abstract
1. Introduction
2. Experimental
2.1. Raw Materials
2.2. Theoretical Basis of Experiments
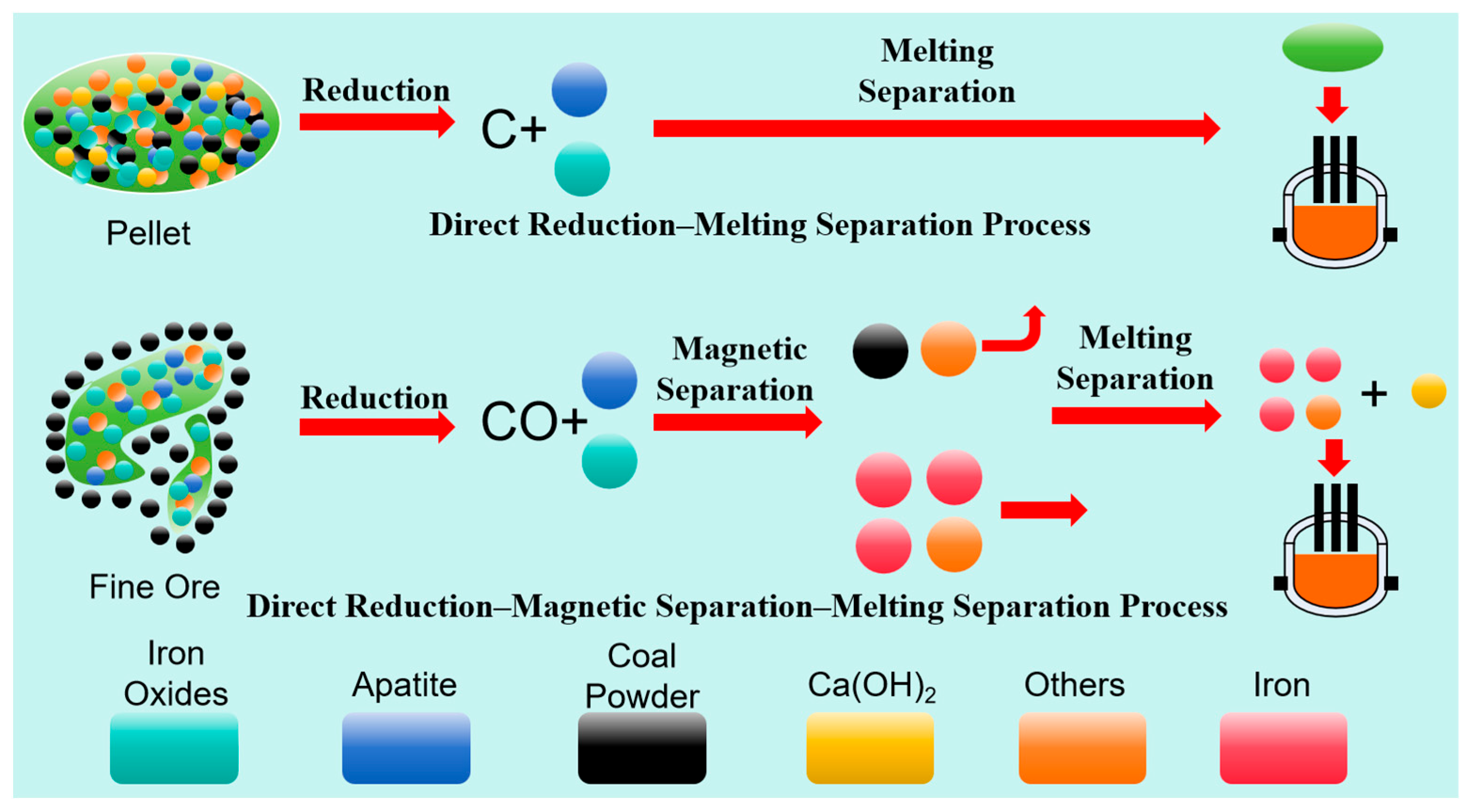
2.3. Experimental Methodology
3. Results and Discussion
3.1. Direct Reduction of the CCP-DR–MS Process
3.2. Smelting Separation of the CCP-DR–MS Process
3.3. Direct Reduction of the DR–MS–MS Process
3.4. Smelting Separation of the DR–MS–MS Process
3.5. Reasons for Differences in Dephosphorization Efficiency
4. Conclusions
- In high-phosphorus iron ore, iron primarily exists in goethite (FeO(OH)) form, while phosphorus is predominantly present as apatite. Thermal analysis reveals that goethite (FeO(OH)) in this ore undergoes decomposition at approximately 290 °C, exhibiting a mass loss rate of 10.75%. Particle size distribution measurements indicate an average ore diameter of 471 μm (approximately 35 mesh), characterizing this as a typical sandy-type high-phosphorus ore with relatively coarse granulometry.
- The direct reduction of carbon-composite pellets (26% coal addition, 180 min, 1100–1150 °C) achieved ∼95% metallization with facile slag–iron separation. However, phosphorus was synchronously reduced with iron during the process, and Ca(OH)2 addition during smelting showed negligible effects on phosphorus removal. Consequently, the resulting iron contained about 1.6% phosphorus—exceeding steelmaking specifications—rendering this route unsuitable for producing a qualified iron product.
- The granular ore direct reduction process (30% coal, 1050 °C, 120 min) attained 94.72% metallization, 82.37% iron grade, and 1.05% residual phosphorus. Crucially, phosphorus remained unreduced during CO-dominated reduction, while magnetic separation eliminated coal residues and partial gangue. Subsequent smelting with Ca(OH)2 (basicity 2.0, 1550 °C, 40 min) yielded iron with only 0.21% phosphorus (>85% removal). With further deep dephosphorization, this process produces steelmaking-grade iron, showing potential for industrial application.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Li, W.B.; Song, R.F.; Liu, H.Y. Review of China’s Progress in Benefication Technology for Refractory Iron Ores. Met. Mine 2008, 11, 1–4+132. [Google Scholar] [CrossRef]
- Jiang, T.; Jin, Y.S.; Li QYang, Y.B.; Qiu, G.Z. Dephosphorization technology of iron ores by Acidthiobacillus ferrooxidans. Chin. J. Nonferrous Met. 2007, 17, 1718. [Google Scholar] [CrossRef]
- Cheng, C.; Xue, Q.G.; Wang, G.; Zhang, Y.Y.; Wang, J.S. Phosphorus migration during direct reduction of coal composite high-phosphorus iron ore pellets. Metall. Mater. Trans. B 2016, 47, 154–163. [Google Scholar] [CrossRef]
- Bao, Q.P.; Lei, G.; Guo, Z.C. A novel direct reduction-flash smelting separation process of treating high phosphorous iron ore fines. Powder Technol. 2021, 377, 149–162. [Google Scholar] [CrossRef]
- Cao, Y.Y.; Sun, T.C.; Gao, E.X.; Xu, C.Y. Effects of Volatile Component in Coal on High-Phosphorus Oolitic Hematite in Direct Reduction Roasting Process. J. Northeast. Univ. (Nat. Sci.) 2014, 35, 1346–1349+1355. [Google Scholar] [CrossRef]
- Yu, W.; Sun, T.C.; Cui, Q.; Kou, Z. Effect of Coal Type on the Reduction and Magnetic Separation of a High-phosphorus Oolitic Hematite Ore. ISIJ Int. 2015, 55, 536–543. [Google Scholar] [CrossRef]
- Nunes, A.P.L.; Pinto, C.L.L.; Valadão, G.E.S.; de Magalhães Viana, P.R. Floatability studies of wavellite and preliminary results on phosphorus removal from a Brazilian iron ore by froth flotation. Miner. Eng. 2012, 39, 206–212. [Google Scholar] [CrossRef]
- Yu, J.T.; Guo, Z.; Tang, H. Dephosphorization Treatment of High Phosphorus Oolitic Iron Ore by Hydrometallurgical Process and Leaching Kinetics. ISIJ Int. 2013, 53, 2056–2064. [Google Scholar] [CrossRef]
- Zhang, L.; Machiela, R.; Das, P.; Zhang, M.; Eisele, T. Dephosphorization of unroasted oolitic ores through alkaline leaching at low temperature. Hydrometallurgy 2019, 184, 8. [Google Scholar] [CrossRef]
- Wu, S.C.; Su, T.C.; Li, Z.Y.; Li, X.H. Research Progress of Direct Reduction—Magnetic Separation of High Phosphorus Iron Ore. Met. Mine 2021, 2, 58–64. [Google Scholar] [CrossRef]
- Keith, Q. A review on the characterisation and processing of oolitic iron ores. Miner. Eng. 2018, 126, 89–100. [Google Scholar] [CrossRef]
- Li, K.Q.; Ni, W.; Zhu, M.; Zheng, M.J.; Li, Y. Iron extraction from oolitic iron ore by a deep reduction process. J. Iron Steel Res. Int. 2011, 18, 9–13. [Google Scholar] [CrossRef]
- Omran, M. Microwave Dephosphorisation of High Phosphorus Iron Ores of the Aswan Region, Egypt: Developing a Novel Process for High Phosphorus Iron Ore Utilization. Ph.D. Thesis, University of Oulu, Oulu, Finland, 2015. [Google Scholar] [CrossRef]
- Omran, M.; Fabritius, T.; Mattila, R. Thermally assisted liberation of high phosphorus oolitic iron ore: A comparison between microwave and conventional furnaces. Powder Technol. 2015, 269, 7–14. [Google Scholar] [CrossRef]
- Gao, J.T.; Guo, L.; Zhong, Y.W.; Ren, H.R.; Guo, Z.C. Removal of phosphorus-rich phase from high-phosphorous iron ore by melt separation at 1573 K in a super-gravity field. Int. J. Miner. Metall. Mater. 2016, 23, 743–750. [Google Scholar] [CrossRef]
- Gao, J.T.; Guo, L.; Guo, Z.C. Separation of P phase and Fe phase in high phosphorus oolitic iron ore by ultrafine grinding and gaseous reduction in a rotary furnace. Metall. Mater. Trans. B 2015, 46, 2180–2189. [Google Scholar] [CrossRef]
- Gao, P.; Li, G.F.; Han, Y.X.; Sun, Y.S. Reaction behavior of phosphorus in coal-based reduction of an oolitic hematite ore and pre-dephosphorization of reduced iron. Metals 2016, 6, 82. [Google Scholar] [CrossRef]
- Cai, X.Y.; Qian, G.M.; Zhang, B.; Chen, Q.S.; Hu, C.Q. Selective liberation of high-phosphorous oolitic hematite assisted by microwave processing and acid leaching. Minerals 2018, 8, 245. [Google Scholar] [CrossRef]
- Fisher-White, M.J.; Lovel, R.R.; Sparrow, G.J. Phosphorus removal from goethitic iron ore with a low temperature heat treatment and a caustic leach. ISIJ Int. 2012, 52, 797–803. [Google Scholar] [CrossRef]
- Yu, W.; Sun, T.; Cui, Q. Can sodium sulfate be used as an additive for the reduction roasting of high-phosphorus oolitic hematite ore? Int. J. Miner. Process. 2014, 133, 119–122. [Google Scholar] [CrossRef]
- Zhao, Y.Q.; Sun, T.C.; Zhao, H.Y.; Li, X.H.; Wang, X. Effects of CaCO3 as Additive on Coal-based Reduction of High-phosphorus Oolitic Hematite Ore. ISIJ Int. 2018, 58, 1768–1774. [Google Scholar] [CrossRef]
- Wen, Y.U.; Sun, T.; Kou, J.; Wei, Y.X.; Xu, C.Y.; Liu, Z.Z. The Function of Ca(OH)2 and Na2CO3 as Additive on the Reduction of High-Phosphorus Oolitic Hematite-coal Mixed Pellets. ISIJ Int. 2013, 53, 427–433. [Google Scholar] [CrossRef]
- Cheng, Y.X.; Sun, T.C.; Kou, J.; Li, L.Y.; Mo, X.L.; Tang, L.G. Mechanism of phosphorus removal in beneficiation of high phosphorous oolitic hematite by direct reduction roasting with dephosphorization agent. Trans. Nonferrous Met. Soc. China 2012, 22, 2806–2812. [Google Scholar] [CrossRef]
- Yang, D.W.; Sun, T.C.; Xu, C.Y. Reducing Roasting and a Synchronized Dephosphorization of a High-phosphorus Oolite Hematite. Min. Metall. Eng. 2010, 30, 29–31. [Google Scholar] [CrossRef]
- Xu, Y.; Sun, T.C.; Liu, Z.G.; Xu, C.Y. Dephosphorization effect of sodium carbonate in the process of direct reduction roasting of high phosphorous oolitic hematite. J. Northeast. Univ. (Nat. Sci.) 2014, 35, 1028–1032. [Google Scholar] [CrossRef]
- Li, G.; Zhang, S.; Rao, M.; Zhang, Y.; Jiang, T. Effects of sodium salts on reduction roasting and Fe-P separation of high-phosphorus oolitic hematite ore. Int. J. Miner. Process. 2013, 124, 26–34. [Google Scholar] [CrossRef]
- Rao, M.; Ouyang, C.; Li, G.; Zhang, S.; Zhang, Y.; Jiang, T. Behavior of phosphorus during the carbothermic reduction of phosphorus-rich oolitic hematite ore in the presence of Na2SO4. Int. J. Miner. Process. 2015, 143, 72–79. [Google Scholar] [CrossRef]
- Luo, L.Q.; Zhang, H.Q. Process mineralogy and characteristic associations of iron and phosphorus-based minerals on oolitic hematite. J. Cent. South Univ. 2017, 24, 1959–1967. [Google Scholar] [CrossRef]
- Liu, Y.Q.; Zhang, H.; Li, Z.E.; Zhang, A.M.; Zhang, X.H.; Qing, S. Impact of slag composition activity on the behavior of phosphorus in the smelting reduction process of high-phosphorus iron ores. Int. J. Hydrogen Energy 2017, 42, 24487–24494. [Google Scholar] [CrossRef]
- Young, A.M.; Walton, M.A.; Carter, T.L. Selective tolerance to discriminative stimulus effects of morphine or d-amphetamine. Behav. Pharmacol. 1992, 3, 201–210. [Google Scholar] [CrossRef] [PubMed]






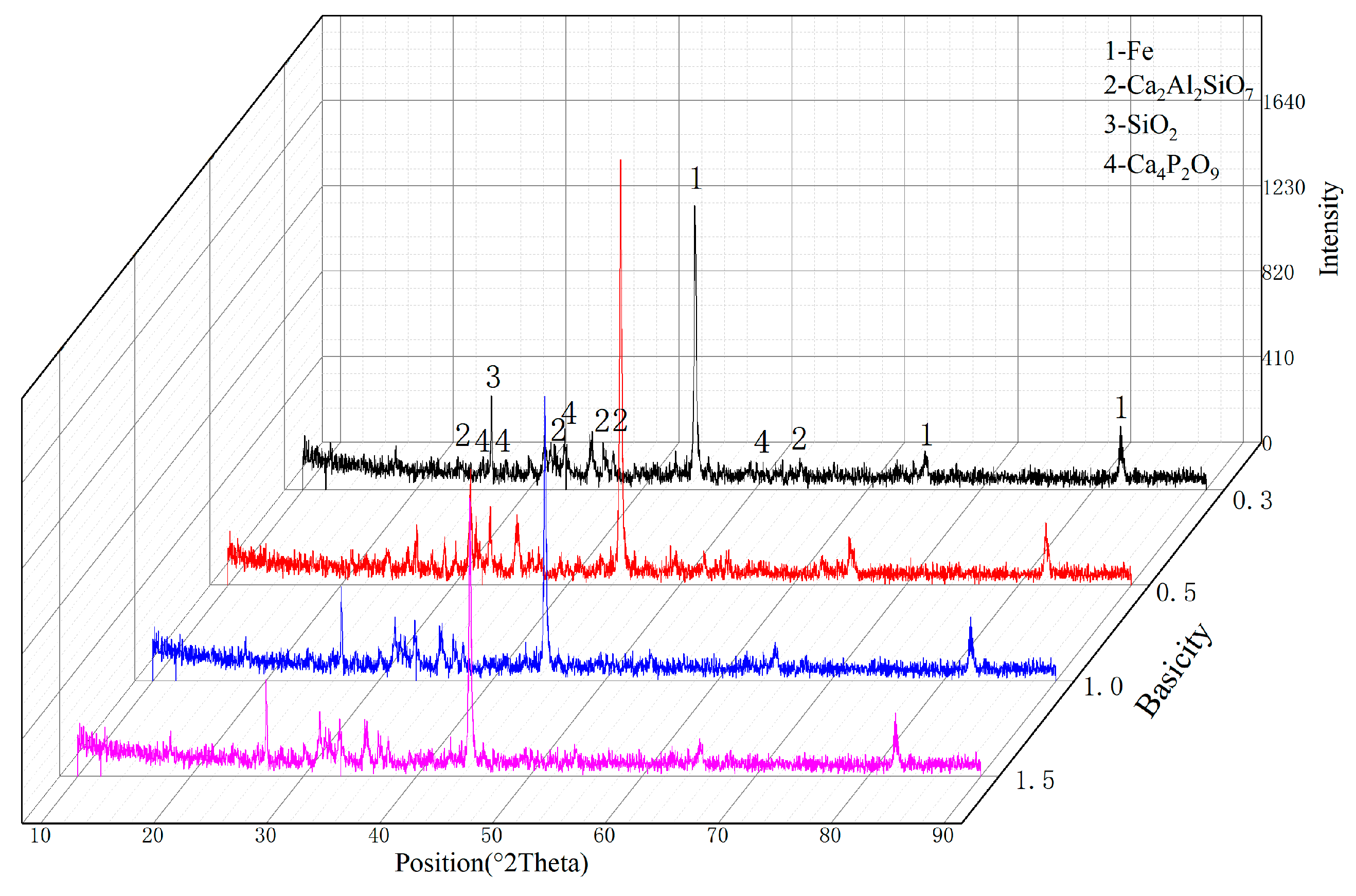








| Elements | TFe | SiO2 | CaO | MgO | Al2O3 | MnO | P2O5 | LOI |
|---|---|---|---|---|---|---|---|---|
| Content % | 52.65 | 4.28 | 1.57 | 0.14 | 1.24 | 0.67 | 1.95 | 11.25 |
| No. | Coal Blending Ratio/% | Ca(OH)2 Dosage/g | Basicity | Reduction Temperature/°C | Melting Separation Temperature/°C | Melting Separation Time/min |
|---|---|---|---|---|---|---|
| 1 | 26 | 0 | 0.3 | 1100 | 1400 | 40 |
| 2 | 26 | 1.5 | 0.5 | 1100 | 1400 | 40 |
| 3 | 26 | 5.0 | 1.0 | 1100 | 1400 | 40 |
| 4 | 26 | 9.1 | 1.5 | 1100 | 1400 | 40 |
| 5 | 26 | 0 | 0.3 | 1150 | 1450 | 40 |
| 6 | 26 | 1.5 | 0.5 | 1150 | 1450 | 40 |
| 7 | 26 | 5.0 | 1.0 | 1150 | 1450 | 40 |
| 8 | 26 | 9.1 | 1.5 | 1150 | 1450 | 40 |
| No. | Coal Blending Ratio/% | Reduction Temperature/°C | Reduction Heating Time/min | Reduction Isothermal Time/min | Melting Separation Time/min |
|---|---|---|---|---|---|
| 1 | 30 | 950 | 60 | 120 | 50 |
| 2 | 30 | 1000 | 60 | 120 | 50 |
| 3 | 30 | 1050 | 60 | 120 | 50 |
| 4 | 30 | 1100 | 60 | 120 | 50 |
| MgO | CaO | MnO | FeO | Al2O3 | Fe2O3 | SiO2 | P2O5 |
|---|---|---|---|---|---|---|---|
| 0.8 | 1.0 | 0.6 | 0.48 | 0.6 | 0.48 | 0.48 | 0.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Wang, B.; Gao, J.; Wang, F.; Yu, Y.; Qi, Y. Comparative Study of Metallic Iron Production from High-Phosphorus Iron Ores: Carbon-Composite Pellet Direct Reduction–Melting vs. Granular Direct Reduction–Magnetic Separation–Melting. Materials 2026, 19, 1499. https://doi.org/10.3390/ma19081499
Wang B, Gao J, Wang F, Yu Y, Qi Y. Comparative Study of Metallic Iron Production from High-Phosphorus Iron Ores: Carbon-Composite Pellet Direct Reduction–Melting vs. Granular Direct Reduction–Magnetic Separation–Melting. Materials. 2026; 19(8):1499. https://doi.org/10.3390/ma19081499
Chicago/Turabian StyleWang, Bin, Jianjun Gao, Feng Wang, Yue Yu, and Yuanhong Qi. 2026. "Comparative Study of Metallic Iron Production from High-Phosphorus Iron Ores: Carbon-Composite Pellet Direct Reduction–Melting vs. Granular Direct Reduction–Magnetic Separation–Melting" Materials 19, no. 8: 1499. https://doi.org/10.3390/ma19081499
APA StyleWang, B., Gao, J., Wang, F., Yu, Y., & Qi, Y. (2026). Comparative Study of Metallic Iron Production from High-Phosphorus Iron Ores: Carbon-Composite Pellet Direct Reduction–Melting vs. Granular Direct Reduction–Magnetic Separation–Melting. Materials, 19(8), 1499. https://doi.org/10.3390/ma19081499






