Tuning the Elastic Properties of Polymer Networks Based on a Selected Biphenyl Epoxy Precursor by Altering the Hardener—Thermal and Dielectric Approach
Abstract
1. Introduction
1.1. Designing Epoxy Resins Through Structure and Function
1.2. Combining DSC and BDS for Characterization of Polymer Networks
2. Materials and Methods
2.1. Synthesis of Propane-2,2-diylo-4,4′-diphenyl Bis(10,11-epoxyundecanoate)—DKUU
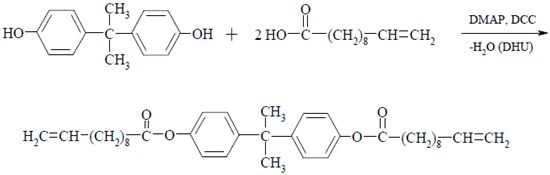
- 1H-NMR (CDCl3, δ(ppm), J): 7.21 (4H, d, J = 8.7 Hz, aromatic), 6.96 (4H, d, J = 8.7 Hz, aromatic), 5.80 (2H, m, –CH=CH2), 4.96 (4H, m, –CH=CH2), 2.53 (4H, m, CH2-COO), 2.04 (4H, m, –CH2–CH=CH2), 1.73 (4H, m, CH2–CH2–COO), 1.42 (20H, m, (CH2)5), 1.36 (6H, t, C–(CH3)2).
- FT-IR (KBr, ν(cm−1)): 3080, 1603–1465, 1017, 848 (aromatic), 2964–2851 (CH2), 1750 (C=O), 1279–1081 (C–O), 1641 (CH=CH2).
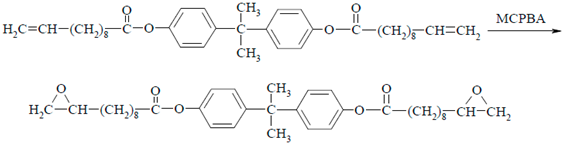
- 1H-NMR (CDCl3, δ(ppm), J): 7.20 (4H, d, J = 8.7 Hz, aromatic), 6.96 (4H, d, J = 8.8 Hz, aromatic), 2.90 (2H, m, CH of epoxy), 2.74 (2H, m, CH2 of epoxy), 2.60 (4H, m, CH2–COO), 2.48 (2H, m, CH2 of epoxy), 1.75 (4H, m, –CH2–CH2–COO), 1.55 (4H, m, –CH2-epoxy group), 1.41 (20H, m, (CH2)5), 1.38 (6H, t, C–(CH3)2).
- FT-IR (KBr), ν (cm−1): 3061, 1601–1465, 1017, 851 (aromatic), 2960–2850 (CH2), 1742, (C=O), 1288–1082 (C–O), 925 (epoxide).
2.2. Sample Preparation for Curing
2.3. Experimental Methods
3. Results and Discussion
3.1. Characterization of the Epoxy Precursor—DKUU
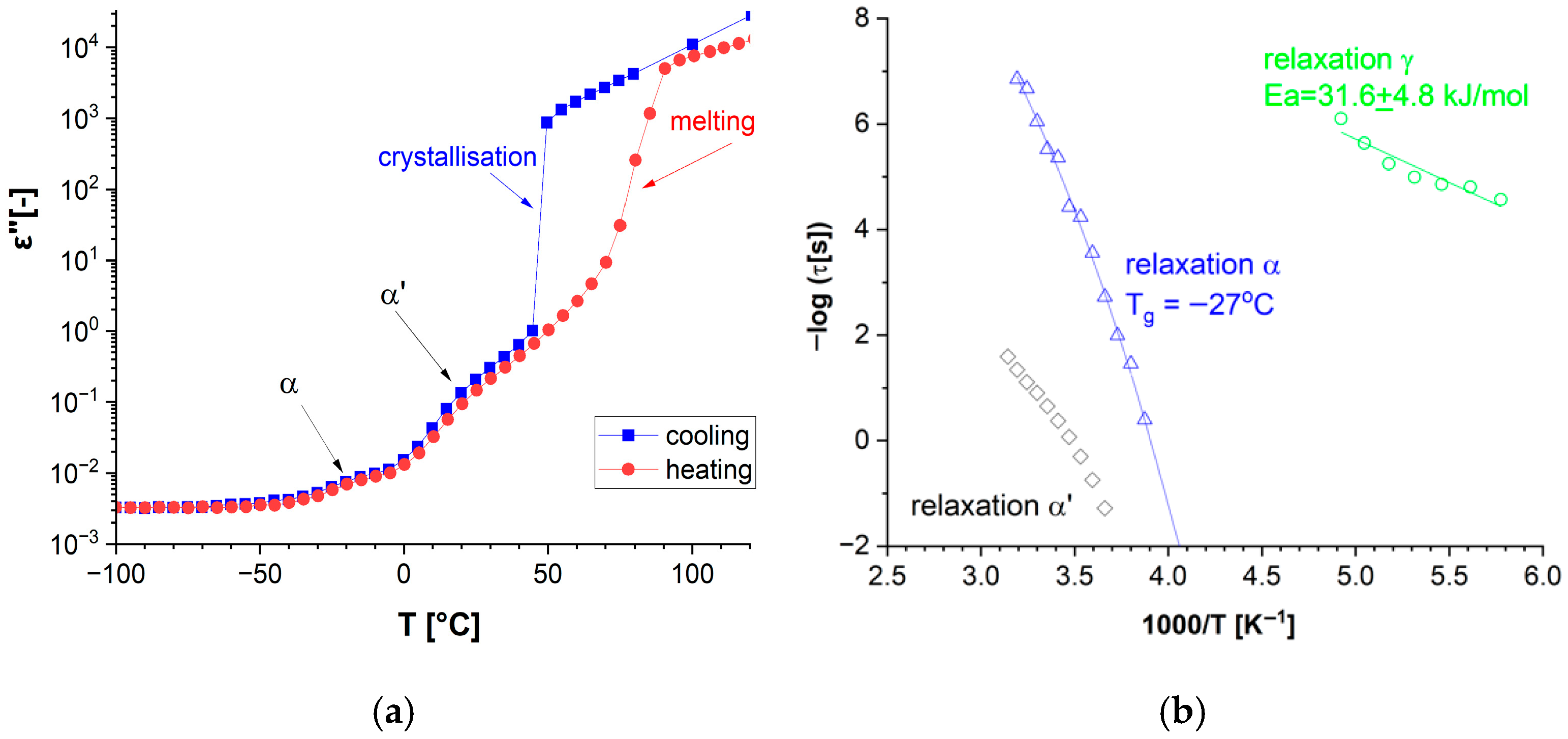
3.1.1. Thermal Analysis
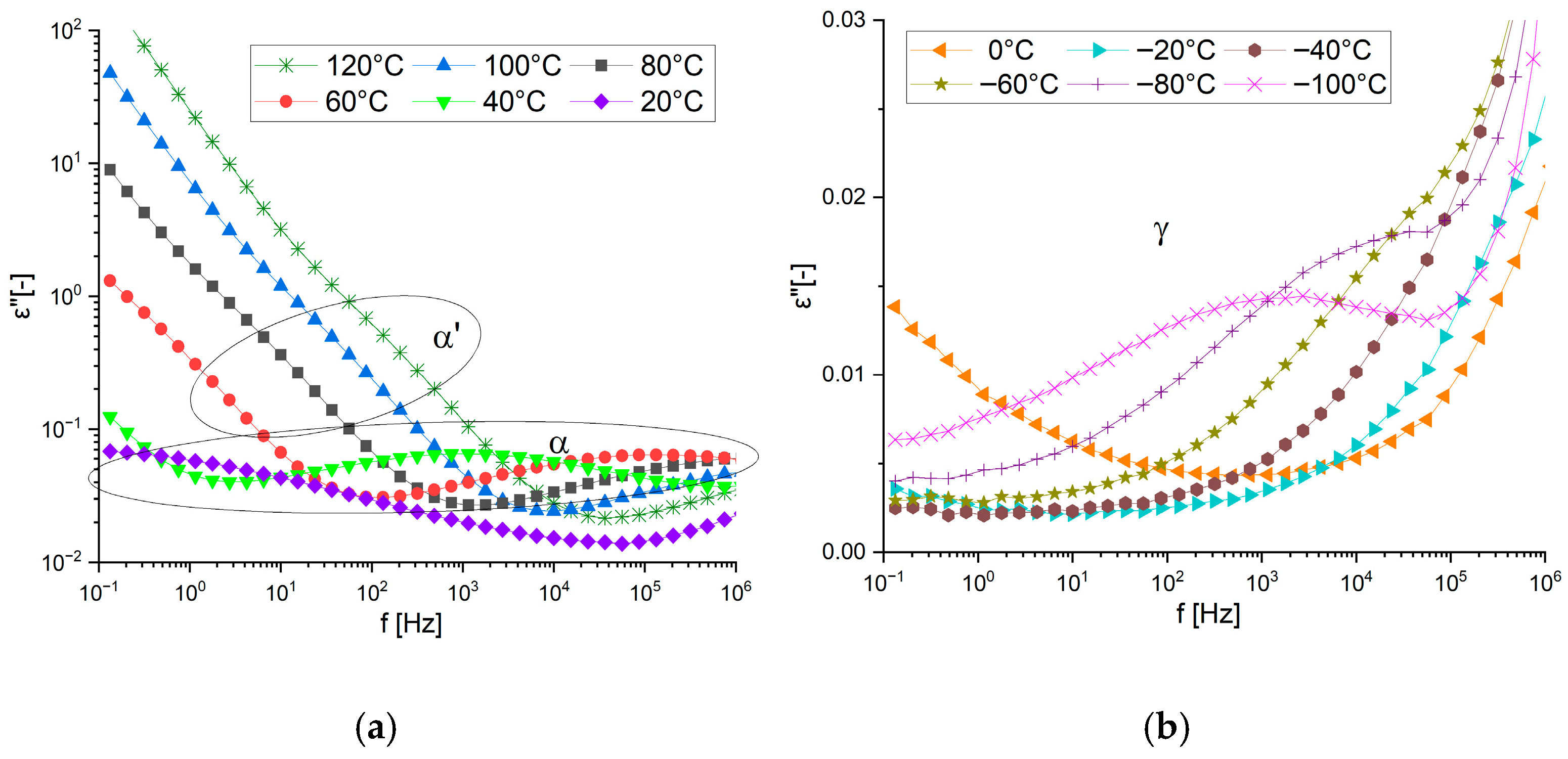
3.1.2. Dielectric Response
3.2. Observation of the Curing Process
3.3. Dielectric Properties of the Cured Products
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| BDS | Broadband Dielectric Spectroscopy |
| DC | Direct Current |
| DDM | 4,4′-diaminodiphenylmethane |
| DSC | Differential Scanning Calorimetry |
| HN | Havriliak–Negami (formula) |
| SA | Suberic acid |
| VFT | Vogel–Fulcher–Tammann (formula) |
References
- Chow, W.S.; Mohd Ishak, Z.A. Smart polymer nanocomposites: A review. Express Polym. Lett. 2020, 14, 416–435. [Google Scholar] [CrossRef]
- Kim, H.C.; Mun, S.; Ko, H.-U.; Zhai, L.; Kafy, A.; Kim, J. Renewable smart materials. Smart Mater. Struct. 2016, 25, 073001. [Google Scholar] [CrossRef]
- Saraswat, A.; Kumar, S. Cutting-edge applications of polyaniline composites towards futuristic energy supply devices. Eur. Polym. J. 2023, 200, 112501. [Google Scholar] [CrossRef]
- Jin, F.-L.; Li, X.; Park, S.-J. Synthesis and application of epoxy resins: A review. J. Ind. Eng. Chem. 2015, 29, 1–11. [Google Scholar] [CrossRef]
- Li, Y.; Ambrogi, V.; Cerruti, P.; Goswami, M.; Yang, Z.; Kessler, M.R.; Rios, O. Functional liquid crystalline epoxy networks and composites: From materials design to applications. Int. Mater. Rev. 2021, 67, 201–229. [Google Scholar] [CrossRef]
- Mohan, P. A critical review: The modification, properties, and applications of epoxy resins. Polym. Plast. Technol. Eng. 2013, 52, 107–125. [Google Scholar] [CrossRef]
- Kausar, A. High performance epoxy/polyester-based nanocomposite coatings for multipurpose applications: A review. J. Plast. Film Sheeting 2020, 36, 391–408. [Google Scholar] [CrossRef]
- El-Ghaffara, M.A.A.; Abdelwahab, N.A.; Fekry, A.M.; Sanad, M.A.; Sabaa, M.W.; Soliman, S.M.A. Polyester-epoxy resin/conducting polymer/barium sulfate hybrid composite as a smart eco-friendly anti-corrosive powder coating. Prog. Org. Coat. 2020, 144, 105664. [Google Scholar] [CrossRef]
- Yang, H.; Yuan, G.; Jiao, E.; Wang, K.; Diao, W.J.; Li, Z.; Wu, K.; Shi, J. Low dielectric constant and high thermal stability of liquid crystal epoxy polymers based on functionalized poly(phenylene oxide). Eur. Polym. J. 2023, 198, 112378. [Google Scholar] [CrossRef]
- Yang, Y.; Xu, Y.; Ji, Y.; Wei, Y. Functional epoxy vitrimers and composites. Prog. Mater. Sci. 2021, 120, 100710. [Google Scholar] [CrossRef]
- Yang, X.; Zhong, X.; Zhanga, J.; Gu, J. Intrinsic high thermal conductive liquid crystal epoxy film simultaneously combining with excellent intrinsic self-healing performance. J. Mater. Sci. Technol. 2021, 68, 209–215. [Google Scholar] [CrossRef]
- Jones, A.R.; Cintora, A.; White, S.R.; Sottos, N.R. Autonomic Healing of Carbon Fiber/Epoxy Interfaces. ACS Appl. Mater. Interfaces 2014, 6, 6033–6039. [Google Scholar] [CrossRef]
- Chen, X.; Dam, M.A.; Ono, K.; Mal, A.; Shen, H.; Nutt, S.R.; Sheran, K.; Wudl, F. A Thermally Re-mendable Cross-Linked Polymeric Material. Science 2002, 295, 1698–1702. [Google Scholar] [CrossRef]
- Fischer, H. Self-repairing material systems―A dream or a reality? Nat. Sci. 2010, 2, 873–901. [Google Scholar] [CrossRef]
- Puig, J.; Erba, I.E.D.; Schroeder, W.F.; Hoppe, C.E.; Williams, R.J.J. Epoxy-Based Organogels for Thermally Reversible Light Scattering Films and Form-Stable Phase Change Materials. ACS Appl. Mater. Interfaces 2017, 9, 11126–11133. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Jang, J. The effect of mesogenic length on the curing behavior and properties of liquid crystalline epoxy resins. Polymer 2006, 47, 3036–3042. [Google Scholar] [CrossRef]
- Lee, J.Y.; Jang, J. Anisotropically ordered liquid crystalline epoxy network on carbon fiber surface. Polym. Bull. 2007, 59, 261–268. [Google Scholar] [CrossRef]
- Liu, J.; Wang, C.; Campbell, G.A.; Earls, J.D.; Priester, R.D., Jr. Effects of liquid crystalline structure formation on the curing kinetics of an epoxy resin. J. Polym. Sci. A Polym. Chem. 1997, 35, 1105–1124. [Google Scholar] [CrossRef]
- Castell, P.; Serra, A.; Galià, M.; Giamberini, M.; Carfagna, C. Anisotropic thermosets from liquid-crystalline azomethynic epoxy resins and primary aromatic diamines. J. Polym. Sci. A Polym. Chem. 2003, 41, 1–12. [Google Scholar] [CrossRef]
- Du, Y.; Zhao, G.; Shi, G.; Wang, Y.; Li, W.; Ren, S. Effect of crosslink structure on mechanical properties, thermal stability and flame retardancy of natural flavonoid based epoxy resins. Eur. Polym. J. 2022, 162, 110898. [Google Scholar] [CrossRef]
- Varganici, C.D.; Rosu, L.; Rosu, D.; Rosca, I.; Ignat, M.E.; Ignat, L. Surface Degradation of DGEBA Epoxy Resins Cured with Structurally Different Amine Hardeners: Effects of UV Radiation. Polymers 2024, 16, 67. [Google Scholar] [CrossRef]
- Fu, K.; Xie, Q.; Lü, F.; Duan, Q.; Wang, X.; Zhu, Q.; Huang, Z. Molecular Dynamics Simulation and Experimental Studies on the Thermomechanical Properties of Epoxy Resin with Different Anhydride Curing Agents. Polymers 2019, 11, 975. [Google Scholar] [CrossRef]
- Michelotti, F.W.; Knuth, C.J.; Bavley, A. Some Anhydride Curing Agents for Epoxy Resins. J. Chem. Eng. Data 1959, 4, 79–82. [Google Scholar] [CrossRef]
- Mossety-Leszczak, B.; Włodarska, M. Liquid-Crystalline Epoxy Thermosets as Matrices for Ordered Nanocomposites—A Summary of Experimental Studies. Polym. Compos. 2017, 38, 277–286. [Google Scholar] [CrossRef]
- Okrasa, L.; Włodarska, M.; Kisiel, M.; Mossety-Leszczak, B. Modification of the Dielectric and Thermal Properties of Organic Frameworks Based on Nonterminal Epoxy Liquid Crystal with Silicon Dioxide and Titanium Dioxide. Polymers 2024, 16, 1320. [Google Scholar] [CrossRef]
- Włodarska, M.; Mossety-Leszczak, B.; Kisiel, M.; Zając, W.; Okrasa, L. Changes in molecular relaxations and network properties of a triaromatic liquid crystal epoxy resin with nonterminal functional groups. J. Polym. Sci. 2023, 61, 3244–3255. [Google Scholar] [CrossRef]
- Lascano, D.; Quiles-Carrillo, L.; Balart, R.; Boronat, T.; Montanes, N. Kinetic Analysis of the Curing of a Partially Biobased Epoxy Resin Using Dynamic Differential Scanning Calorimetry. Polymers 2019, 11, 391. [Google Scholar] [CrossRef]
- Lascano, D.; Lerma-Canto, A.; Fombuena, V.; Balart, R.; Montanes, N.; Quiles-Carrillo, L. Kinetic Analysis of the Curing Process of Biobased Epoxy Resin from Epoxidized Linseed Oil by Dynamic Differential Scanning Calorimetry. Polymers 2021, 13, 1279. [Google Scholar] [CrossRef]
- Ma, H.; Zhang, X.; Ju, F.; Tsai, S.B. A Study on Curing Kinetics of Nano-Phase Modified Epoxy Resin. Sci. Rep. 2018, 8, 3045. [Google Scholar] [CrossRef] [PubMed]
- Kremer, F.; Schönhals, A. Broadband Dielectric Spectroscopy; Kremer, F., Schönhals, A., Eds.; Springer: Berlin/Heidelberg, Germany, 2003. [Google Scholar]
- Rault, J. Origin of the Vogel–Fulcher–Tammann law in glassforming materials: The α–β bifurcation. J. Non-Cryst. Solids 2012, 271, 177–217. [Google Scholar] [CrossRef]
- Rault, J. Relaxation of glasses: The Kohlrausch exponent. J. Non-Cryst. Solids 2011, 357, 339–345. [Google Scholar] [CrossRef]
- Prevosto, D.; Capacciolli, S.; Sharifi, S.; Kessairi, K.; Lucchesi, M.; Rolla, P.A. Secondary dynamics in glass formers: Relation with the structural dynamics and the glass transition. J. Non-Cryst. Solids 2007, 353, 4278–4282. [Google Scholar] [CrossRef]
- Havriliak, S.; Negami, S. A complex plane representation of dielectric and mechanical relaxation processes in some polymers. Polymer 1967, 8, 161–210. [Google Scholar] [CrossRef]
- Johari, G.P.; Goldstein, M. Viscous liquids and the glass transition. II. Secondary relaxations in glasses of rigid molecules. J. Chem. Phys. 1970, 53, 2372–2388. [Google Scholar] [CrossRef]
- Mohomed, K.; Gerasimov, T.G.; Moussy, F.; Harmon, J.P. A broad spectrum analysis of the dielectric properties of poly(2-hydroxyethyl methacrylate). Polymer 2005, 46, 3847–3855. [Google Scholar] [CrossRef]
- Donth, E. The Glass Transition: Relaxation Dynamics in Liquids and Disordered Materials; Springer: New York, NY, USA, 2001. [Google Scholar]
- Böhmer, R.; Ngai, K.L.; Angell, C.A.; Plazek, D.J. Nonexponential relaxations in strong and fragile glass formers. J. Chem. Phys. 1993, 99, 4201–4209. [Google Scholar] [CrossRef]
- Kovacs, A.J. La contraction isotherme du volume des polymères amorphes. J. Polym. Sci. 1958, 30, 131–147. [Google Scholar] [CrossRef]
- Kranbuehl, D.E. In Situ Frequency Dependent Dielectric Sensing of Cure. In Processing of Composites; Dave, R.S., Loos, A.C., Eds.; Hanser: Munich, Germany, 2000; pp. 137–157. [Google Scholar] [CrossRef][Green Version]
- Kranbuehl, D.; Delos, S.; Yi, E.; Mayer, J.; Jarvie, T.; Winfree, W.; Hou, T. Dynamic dielectric analysis: Nondestructive material evaluation and cure cycle monitoring. Polym. Eng. Sci. 1986, 26, 338–345. [Google Scholar] [CrossRef]
- Senturia, S.D.; Sheppard, N.F. Dielectric analysis of thermoset cure. In Epoxy Resins and Composites IV: Advances in Polymer Science; Dušek, K., Ed.; Springer: Berlin/Heidelberg, Germany, 1986; Volume 80, pp. 1–47. [Google Scholar] [CrossRef]
- Neises, B.; Steglich, W. Simple method for the esterification of carboxylic acids. Angew. Chem. Int. Ed. 1978, 17, 522–524. [Google Scholar] [CrossRef]
- Mossety-Leszczak, B.; Kisiel, M.; Lechowicz, J.B.; Buszta, N.; Ostatek, R.; Włodarska, M. Analysis of curing reaction of liquid-crystalline epoxy compositions by using temperature-modulated DSC TOPEM®. J. Therm. Anal. Calorim. 2019, 138, 2435–2444. [Google Scholar] [CrossRef]
- Shen, S.; Skordos, A.A. Role of acid hydrocarbon chain length on the cure kinetics and thermal degradation of epoxy-dicarboxylic acid vitrimers. Eur. Polym. J. 2025, 228, 113812. [Google Scholar] [CrossRef]









| Mixture | Curing Conditions | Post-Curing Treatment | |
|---|---|---|---|
| Temperature [°C] | Time [min] | Tp [°C] Heating [10 °C/min] | |
| DKUU/DDM | 150 (0, 1) | 180 (1) | - |
| DKUU/SA | 150 (0, 1) | 180 (1) | 250 (0, 1) |
| Mixture | Tg [°C] from DSC | Tg [°C] from BDS |
|---|---|---|
| DKUU | −19.3 (0.8) | −27 (23) |
| DKUU/DDM | 50.2 (0.5) | 48.9 (6.2) |
| DKUU/SA | 12.5 (0.5) | 14.2 (8.9) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Włodarska, M.; Okrasa, L.; Mossety-Leszczak, B. Tuning the Elastic Properties of Polymer Networks Based on a Selected Biphenyl Epoxy Precursor by Altering the Hardener—Thermal and Dielectric Approach. Materials 2026, 19, 1358. https://doi.org/10.3390/ma19071358
Włodarska M, Okrasa L, Mossety-Leszczak B. Tuning the Elastic Properties of Polymer Networks Based on a Selected Biphenyl Epoxy Precursor by Altering the Hardener—Thermal and Dielectric Approach. Materials. 2026; 19(7):1358. https://doi.org/10.3390/ma19071358
Chicago/Turabian StyleWłodarska, Magdalena, Lidia Okrasa, and Beata Mossety-Leszczak. 2026. "Tuning the Elastic Properties of Polymer Networks Based on a Selected Biphenyl Epoxy Precursor by Altering the Hardener—Thermal and Dielectric Approach" Materials 19, no. 7: 1358. https://doi.org/10.3390/ma19071358
APA StyleWłodarska, M., Okrasa, L., & Mossety-Leszczak, B. (2026). Tuning the Elastic Properties of Polymer Networks Based on a Selected Biphenyl Epoxy Precursor by Altering the Hardener—Thermal and Dielectric Approach. Materials, 19(7), 1358. https://doi.org/10.3390/ma19071358





