Exploring the Synergistic Effects of Ultrafine Polyaniline Nanofibers and Oxygen-Modified Multi-Walled Carbon Nanotubes on Enhancing Pseudocapacitive Electrochemical Performance for Advanced Supercapacitors
Abstract
1. Introduction
2. Materials and Methods
2.1. PANI Ultrafine Nanofiber (PANI-NF) and PANI-NF/O-MWCNT Synthesis
2.1.1. Chemical Process (DCP)
2.1.2. Free-Template Hydrothermal Process (HT)
2.2. Sample Characterization
2.3. Working Electrode Preparation and Electrochemical (EC) Measurements
3. Results and Discussion
3.1. Optical and Structural Properties of PANI-NF and the PANI/O-MWCNT Nanocomposites
3.2. Electrochemical Properties of PANI-NF and the PANI/O-MWCNT Nanocomposites
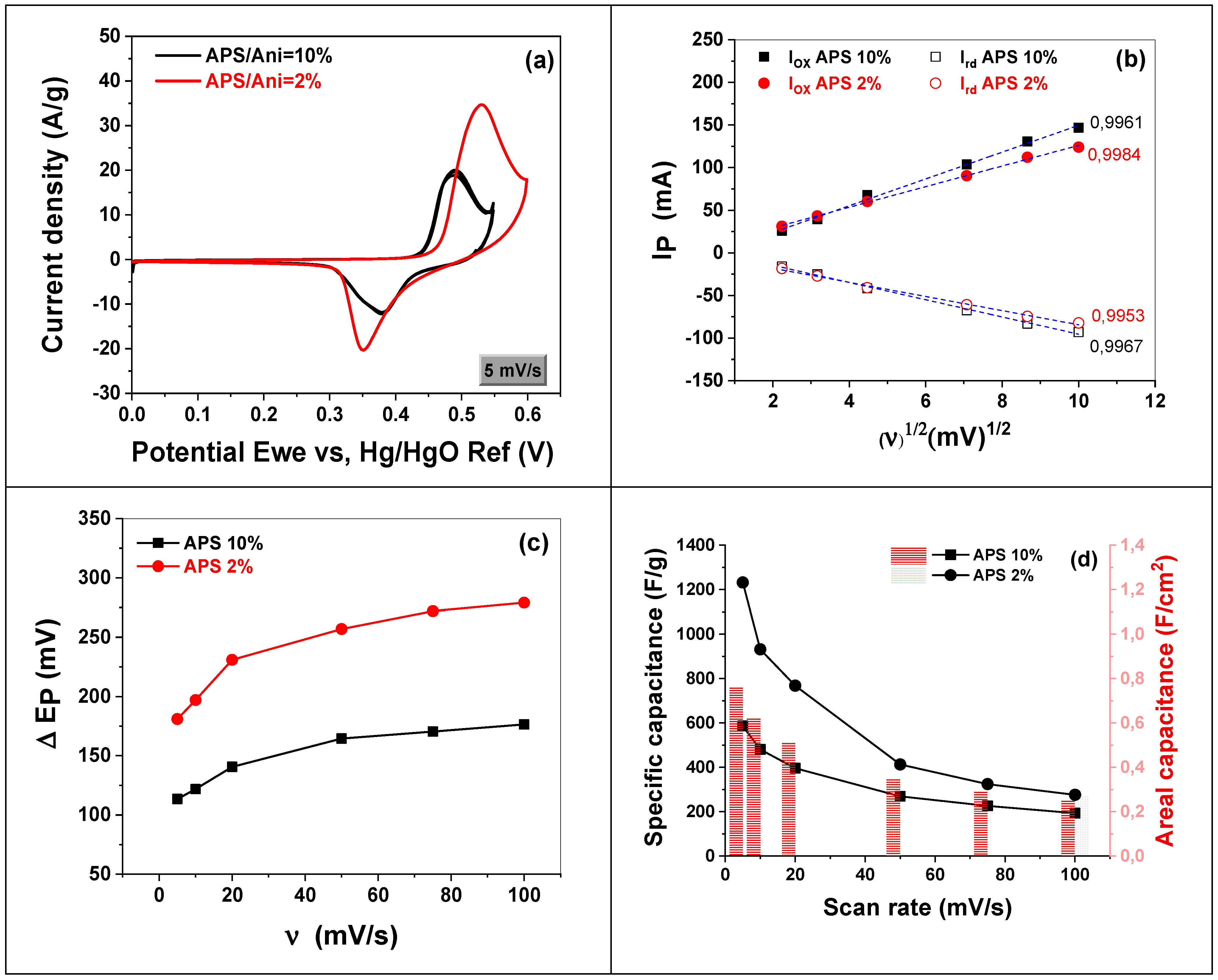
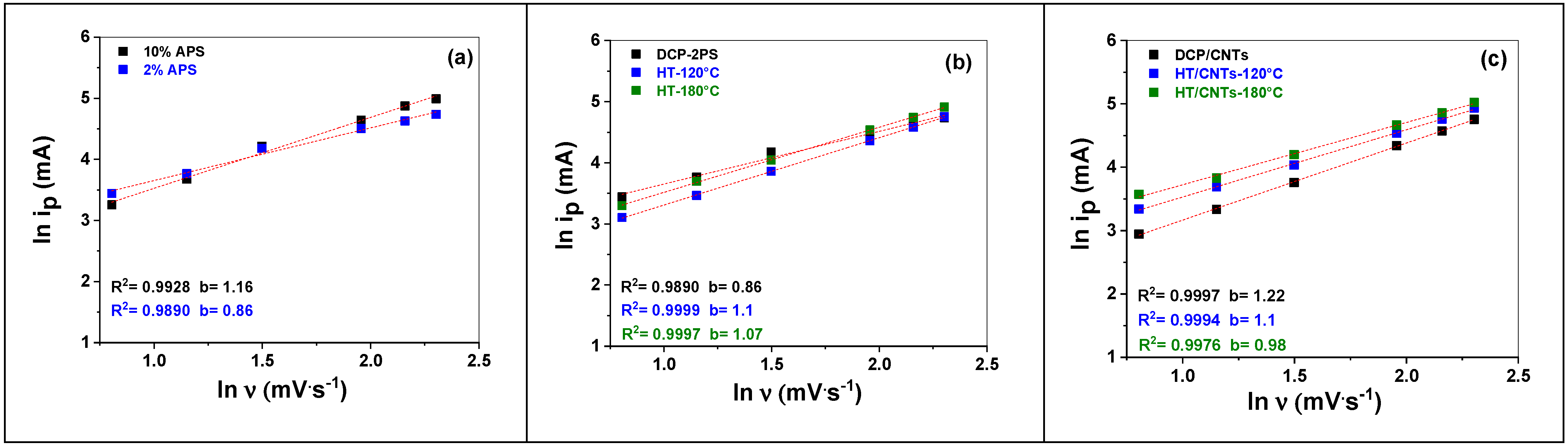
3.2.1. APS Effect
3.2.2. Growth Method Effect
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rao, N.; Singh, R.; Bashambu, L. Carbon-based nanomaterials: Synthesis and prospective applications. Mater. Today 2021, 44, 608–614. [Google Scholar] [CrossRef]
- Majumdar, D. Polyaniline as Proficient Electrode Material for Supercapacitor Applications: PANI Nanocomposites for Supercapacitor Applications. In Polymer Nanocomposites for Advanced Engineering and Military Applications; Ramdani, N., Ed.; IGI Global Scientific Publishing: London, UK, 2019; pp. 190–219. [Google Scholar] [CrossRef]
- Baig, N.; Kammakakam, I.; Falath, W. Nanomaterials: A review of synthesis methods, properties, recent progress, and challenges. Mater. Adv. 2021, 2, 1821. [Google Scholar] [CrossRef]
- Liu, J.; Wang, J.; Xu, C.; Jiang, H.; Li, C.; Zhang, L.; Lin, J.; Shen, Z.X. Advanced Energy Storage Devices: Basic Principles, Analytical Methods, and Rational Materials Design. Adv. Sci. 2018, 5, 1700322. [Google Scholar] [CrossRef] [PubMed]
- Simon, P.; Gogotsi, Y. Perspectives for electrochemical capacitors and related devices. Nat. Mater. 2020, 19, 1151–1163. [Google Scholar] [CrossRef]
- He, X.; Zhang, X. A comprehensive review of supercapacitors: Properties, electrodes, electrolytes and thermal management systems based on phase change materials. J. Energy Storage 2022, 56, 106023. [Google Scholar] [CrossRef]
- Usman, M.; Adnan, M.; Ahsan, M.T.; Javed, S.; Butt, M.S.; Akram, M.A. In Situ Synthesis of a Polyaniline/Fe-Ni Codoped Co3O4 Composite for the Electrode Material of Supercapacitors with Improved Cyclic Stability. ACS Omega 2021, 6, 1190–1196. [Google Scholar] [CrossRef]
- Sunaina, S.; Prakash, C.; Aman, J. Biomass derived carbon for supercapacitor applications: Review. J. Energy Storage 2021, 39, 102646. [Google Scholar] [CrossRef]
- Chu, X.; Yang, W.; Li, H. Recent advances in polyaniline-based micro-supercapacitors. Mater. Horiz. 2023, 10, 670–697. [Google Scholar] [CrossRef]
- Fleischmann, S.; Mitchell, J.B.; Wang, R.; Zhan, C.; Jiang, D.E.; Presser, V.; Augustyn, V. Pseudocapacitance: From Fundamental Understanding to High Power Energy Storage Materials. Chem. Rev. 2020, 120, 6738–6782. [Google Scholar] [CrossRef]
- Kumar, N.; Kim, S.B.; Lee, S.Y.; Park, S.J. Recent Advanced Supercapacitor: A Review of Storage Mechanisms, Electrode Materials, Modification, and Perspectives. J. Nanomater. 2022, 12, 3708. [Google Scholar] [CrossRef] [PubMed]
- Czagany, M.; Hompoth, S.; Keshri, A.K.; Pandit, N.; Galambos, I.; Gacsi, Z.; Baumli, P. Supercapacitors: An Efficient Way for Energy Storage Application. Materials 2024, 17, 702. [Google Scholar] [CrossRef]
- Kavishka, D.; Dulsha, K.A. A review of supercapacitors: Materials, technology, challenges, and renewable energy applications. J. Energy Storage 2024, 96, 112563. [Google Scholar] [CrossRef]
- Mariano, L.C.; Salvatierra, R.V.; Cava, C.E.; Koehler, M.; Zarbin, A.J.G.; Roman, L.S. Electrical properties of self-assembled films of polyaniline/carbon nanotubes composites. J. Phys. Chem. C 2014, 118, 24811–24818. [Google Scholar] [CrossRef]
- Li, M.; Xiang, S.; Chang, X.; Chang, C. Resorcinol-formaldehyde carbon spheres/polyaniline composite with excellent electrochemical performance for supercapacitors. J. Solid State Electrochem. 2017, 21, 485–494. [Google Scholar] [CrossRef]
- Liu, D.; Wang, H.; Du, P.; Wei, W.; Wang, Q.; Liu, P. Flexible and robust reduced graphene oxide/carbon nanoparticles/polyaniline (RGO/CNs/PANI) composite films: Excellent candidates as free-standing electrodes for high-performance supercapacitors. Electrochim. Acta 2018, 259, 161–169. [Google Scholar] [CrossRef]
- Anand, S.; Ahmad, M.W.; Saidi, A.K.A.A.; Yang, D.J.; Choudhury, A. Polyaniline nanofiber decorated carbon nanofiber hybrid mat for flexible electrochemical supercapacitor. Mater. Chem. Phys. 2020, 254, 123480. [Google Scholar] [CrossRef]
- Jiang, K.; Gerhardt, R.A. Fabrication and Supercapacitor Applications of Multiwall Carbon Nanotube Thin Films. C J. Carbon Res. 2021, 7, 70. [Google Scholar] [CrossRef]
- Lv, Y.; Wang, J.; Ji, D.; Li, J.; Zhao, S.; Zhao, Y.; Cai, Z.; He, X.; Sun, X. Carbonaceous electrode materials for supercapacitor: Preparation and surface functionalization. Front. Energy Res. 2023, 10, 957032. [Google Scholar] [CrossRef]
- He, Y.; Wang, X.; Huang, H.; Zhang, P.; Chen, B.; Guo, Z. In-situ electro polymerization of porous conducting polyaniline fibrous network for solid-state supercapacitor. Appl. Surf. Sci. 2019, 469, 446–455. [Google Scholar] [CrossRef]
- Bhandari, S. Chapter 2-Polyaniline: Structure and Properties Relationship. In Polyaniline Blends, Composites, and Nanocomposites; Della Pina, C., Falletta, E., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 23–60. [Google Scholar] [CrossRef]
- Shen, Y.; Qin, Z.; Li, T.; Zeng, F.; Chen, Y.; Liu, N. Boosting the supercapacitor performance of polyaniline nanofibers through sulfonic acid assisted oligomer assembly during seeding polymerization process. Electrochim. Acta 2020, 356, 136841. [Google Scholar] [CrossRef]
- Beygisangchin, M.; Rashid, S.A.; Shafie, S.; Sadrolhosseini, A.R.; Lim, H.N. Preparations, properties, and applications of polyaniline and polyaniline thin films—A review. Polymers 2021, 13, 2003. [Google Scholar] [CrossRef]
- Mahato, N.; Mohapatra, D.; Cho, M.H.; Ahn, K.S. Semi-Polycrystalline–Polyaniline Empowered Electrochemical Capacitor. Energies 2022, 15, 2001. [Google Scholar] [CrossRef]
- Korent, A.; Soderžnik, K.Ž.; Rožman, K.Ž. In-Situ Spectroelectrochemical Study of Conductive Polyaniline Forms for Sensor Applications. Proceedings 2020, 56, 32. [Google Scholar] [CrossRef]
- Majeed, A.H.; Mohammed, L.A.; Hammoodi, O.G.; Sehgal, S.; Alheety, M.A.; Saxena, K.K.; Dadoosh, S.A.; Mohammed, I.K.; Jasim, M.M.; Salmaan, N.U. A Review on Polyaniline: Synthesis, Properties, Nanocomposites, and Electrochemical Applications. In. J. Polym. Sci. 2022, 2022, 19 pages. [Google Scholar] [CrossRef]
- Giri, H.; Dowell, T.J.; Almtiri, M.; Scott, C.N. Polyaniline Derivatives and Their Applications. In Trends and Developments in Modern Applications of Polyaniline; Nastase, F., Ed.; Intechopen: London, UK, 2023. [Google Scholar] [CrossRef]
- Cao, X.; Zeng, H.Y.; Xu, S.; Yuan, J.; Han, J.; Xiao, G.F. Facile fabrication of the polyaniline/layered double hydroxide nanosheet composite for supercapacitors. Appl. Clay Sci. 2019, 168, 175–183. [Google Scholar] [CrossRef]
- Goswami, S.; Nandy, S.; Fortunato, E.; Martins, R. Polyaniline and its composites engineering: A class of multifunctional smart energy materials. J. Solid State Chem. 2023, 317, 123679. [Google Scholar] [CrossRef]
- Moyseowicz, A.; Gryglewicz, G. Hydrothermal-assisted synthesis of a porous polyaniline/reduced graphene oxide composite as a high-performance electrode material for supercapacitors. Compos. B Eng. 2019, 159, 4–12. [Google Scholar] [CrossRef]
- Heme, H.N.; Alif, M.S.N.; Rahat, S.M.; Shuchi, S.B. Recent progress in polyaniline composites for high capacity energy storage: A review. J. Energy Storage 2021, 42, 103018. [Google Scholar] [CrossRef]
- Fenoy, G.E.; Van Der Schueren, B.; Scotto, J.; Boulmedais, F.; Ceolín, M.R.; Bégin-Colin, S.; Bégin, D.; Marmisollé, W.A.; Azzaroni, O. Layer-by-layer assembly of iron oxide-decorated few-layer graphene/PANI: PSS composite films for high performance supercapacitors operating in neutral aqueous electrolytes. Electrochim. Acta 2018, 283, 1178–1187. [Google Scholar] [CrossRef]
- Luo, J.; Chen, Y.; Zheng, Y.; Wang, C.; Wei, W.; Liu, X. Hollow graphene-polyaniline hybrid spheres using sulfonated graphene as Pickering stabilizer for high-performance supercapacitors. Electrochim. Acta 2018, 272, 221–232. [Google Scholar] [CrossRef]
- Lee, K.S.; Park, C.W.; Phiri, I.; Ko, J.M. New design for Polyaniline@Multiwalled carbon nanotubes composites with bacteria doping for supercapacitor electrodes. Polymers 2020, 210, 123014. [Google Scholar] [CrossRef]
- Prasanna, B.P.; Avadhani, D.N.; Chaitra, K.; Nagaraju, N.; Kathyayini, N. Synthesis of polyaniline/MWCNTs by interfacial polymerization for superior hybrid supercapacitance performance. J. Polym. Res. 2018, 25, 123. [Google Scholar] [CrossRef]
- Das, D.; Borthakur, L.J.; Nath, B.C.; Saikia, B.J.; Mohan, K.J.; Dolui, S.K. Designing hierarchical NiO/PAni-MWCNT core-shell nanocomposites for high-performance super capacitor electrodes. RSC Adv. 2016, 6, 44878–44887. [Google Scholar] [CrossRef]
- Chakraborty, I.; Chakrabarty, N.; Senapati, A.; Chakraborty, A.K. CuO@NiO/Polyaniline/MWCNT Nanocomposite as High-Performance Electrode for Supercapacitor. J. Phys. Chem. C 2018, 122, 27180–27190. [Google Scholar] [CrossRef]
- Guellati, O.; Fonseca, A.; Bounour, W.; Guerioune, M.; Mekhalif, Z.; Delhalle, J.; Benaldjia, A.; Nagy, J.B. Carbon Nanotube Catalytic Deposition Synthesis. In Proceedings of the ICTON Mediterranean Winter Conference, Sousse, Tunisia, 6–8 December 2007; pp. 1–5. [Google Scholar] [CrossRef]
- Mohsen, R.M.; Morsi, S.M.M.; Selim, M.M.; Ghoneim, A.M.; El-Sherif, H.M. Electrical, thermal, morphological, and antibacterial studies of synthesized polyaniline/zinc oxide nanocomposites. Polym. Bull. 2019, 76, 1–21. [Google Scholar] [CrossRef]
- Bolagam, R.; Boddula, R.; Srinivasan, P. Synthesis of highly crystalline polyaniline with the use of (Cyclohexylamino)-1-propanesulfonic acid for supercapacitor. J. Appl. Electrochem. 2015, 45, 51–56. [Google Scholar] [CrossRef]
- Nguyen, V.H.; Shim, J.J. Green synthesis and characterization of carbon nanotubes/polyaniline nanocomposites. J. Spectrosc. 2015, 2015, 9. [Google Scholar] [CrossRef]
- Mahato, N.; Parveen, N.; Cho, M.H. Synthesis of highly crystalline polyaniline nanoparticles by simple chemical route. Mater. Lett. 2015, 161, 372–374. [Google Scholar] [CrossRef]
- Gu, Z.J.; Wang, J.T.; Li, L.L.; Chen, L.F.; Shen, Q. Formation of polyaniline nanotubes with different pore shapes using α-, β- And γ-cyclodextrins as templates. Mater. Lett. 2014, 117, 66–68. [Google Scholar] [CrossRef]
- Yu, H.; Xin, G.; Ge, X.; Bulin, C.; Li, R.; Xing, R.; Zhang, B. Porous graphene-polyaniline nanoarrays composite with enhanced interface bonding and electrochemical performance. Compos. Sci. Technol. 2018, 154, 76–84. [Google Scholar] [CrossRef]
- Liu, P.; Yan, J.; Gao, X.; Huang, Y.; Zhang, Y. Construction of layer-by-layer sandwiched graphene/polyaniline nanorods/carbon nanotubes heterostructures for high performance supercapacitors. Electrochim. Acta 2018, 272, 77–87. [Google Scholar] [CrossRef]
- Noh, J.; Jekal, S.; Yoon, C.M. Polyaniline-Coated Mesoporous Carbon Nanosheets with Fast Capacitive Energy Storage in Symmetric Supercapacitors. Adv. Sci. 2023, 10, 2301923. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.; Lei, Y.; Chen, X.; Song, H.; Liu, R. A novel silica aerogel microspheres loaded with ammonium persulfate gel breaker for mid-deep reservoirs. J. Sol-Gel Sci. Technol. 2018, 88, 105–113. [Google Scholar] [CrossRef]
- Qiu, B.; Wang, J.; Li, Z.; Wang, X.; Li, X. Influence of Acidity and Oxidant Concentration on the Nanostructures and Electrochemical Performance of Polyaniline during Fast Microwave-Assisted Chemical Polymerization. Polymers 2020, 12, 310. [Google Scholar] [CrossRef]
- Kumar, A.; Kumar, V.; Kumar, M.; Awasthi, K. Synthesis and characterization of hybrid PANI/MWCNT nanocomposites for EMI applications. Polym. Compos. 2018, 39, 3858–3868. [Google Scholar] [CrossRef]
- Potphode, D.D.; Sinha, L.; Shirage, P.M. Redox additive enhanced capacitance: Multi-walled carbon nanotubes/polyaniline nanocomposite based symmetric supercapacitors for rapid charge storage. Appl. Surf. Sci. 2019, 469, 162–172. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, L.; Qin, Z. Polyaniline nanoarrays grown on holey graphene constructed by frozen interfacial polymerization as binder−free and flexible gel electrode for high−performance supercapacitor. Carbon 2024, 225, 119100. [Google Scholar] [CrossRef]
- Yang, L.; Zhou, Y.; Xu, X.; Shen, Y.; Yan, H.; Qin, Z. Interior design of hierarchical micro/nanostructures for enhancing energy storage ability of polyanilines through frozen interfacial polymerization. Electrochim. Acta 2021, 386, 138448. [Google Scholar] [CrossRef]
- Li, T.; Qin, Z.; Liang, B.; Tian, F.; Zhao, J.; Liu, N.; Zhu, M. Morphology-dependent capacitive properties of three nanostructured polyanilines through interfacial polymerization in various acidic media. Electrochim. Acta 2015, 177, 343–351. [Google Scholar] [CrossRef]
- Bogdanović, U.; Vodnik, V.V.; Ahrenkiel, S.P.; Stoiljković, M.; Ćirić-Marjanović, G.; Nedeljković, J.M. Interfacial synthesis and characterization of gold/polyaniline nanocomposites. Synth. Met. 2014, 195, 122–131. [Google Scholar] [CrossRef]
- Shah, A.U.H.A.; Kamran, M.; Bilal, S.; Ullah, R. Cost effective chemical oxidative synthesis of soluble and electroactive polyaniline salt and its application as anticorrosive agent for steel. Materials 2019, 12, 1527. [Google Scholar] [CrossRef]
- Zhu, X.; Hou, K.; Chen, C.; Zhang, W.; Sun, H.; Zhang, G.; Gao, Z. Structural-controlled synthesis of polyaniline nanoarchitectures using hydrothermal method. High Perform. Polym. 2015, 27, 207–216. [Google Scholar] [CrossRef]
- Chakraborty, P.; Kothari, A.; Nagarajan, R. Highly ordered polyaniline as an efficient dye remover. Adsorp. Sci. Technol. 2018, 36, 429–440. [Google Scholar] [CrossRef]
- Xiong, S.; Wang, Y.; Chu, J.; Wang, X.; Zhang, R.; Gong, M.; Wu, B.; Li, Z. One-pot hydrothermal synthesis of polyaniline nanofibers/reduced graphene oxide nanocomposites and their supercapacitive properties. High Perform. Polym. 2019, 31, 1238–1247. [Google Scholar] [CrossRef]
- Du, X.; Luo, F.; Guo, Y.; Zhu, Q.; Xiao, F.; Wu, K.; Lu, M. Fabrication of graphene/single-wall carbon nanotubes/polyaniline composite gels as binder-free electrode materials. J. Appl. Polym. Sci. 2019, 136, 46948. [Google Scholar] [CrossRef]
- Yu, J.; Xie, F.; Wu, Z.; Huang, T.; Wu, J.; Yan, D.; Huang, C.; Li, L. Flexible metallic fabric supercapacitor based on graphene/polyaniline composites. Electrochim. Acta 2018, 259, 968–974. [Google Scholar] [CrossRef]
- Zhang, D.; Wu, Z.; Li, P.; Zong, X.; Dong, G.; Zhang, Y. Facile fabrication of polyaniline/multi-walled carbon nanotubes/molybdenum disulfide ternary nanocomposite and its high-performance ammonia-sensing at room temperature. Sens. Actuators B Chem. 2018, 258, 895–905. [Google Scholar] [CrossRef]
- Andreas, R.; Lesbani, A.; Yusuf, F.A. The characteristics (compositions, morphological, and structure) of nanocomposites polyaniline (PANI)/ZnO. IOP Conf. Ser. Mater. Sci. Eng. 2019, 509, 012126. [Google Scholar] [CrossRef]
- Wang, X.; Wu, D.; Song, X.; Du, W.; Zhao, X.; Zhang, D. Review on carbon/polyaniline hybrids: Design and synthesis for supercapacitor. Molecules 2019, 24, 2263. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, G.M.D. Raman dispersion in polyaniline nanofibers. Vib. Spectrosc. 2017, 90, 89–95. [Google Scholar] [CrossRef]
- Farag, A.A.; Kabel, K.I.; Elnaggar, E.M.; Al-Gamal, A.G. Influence of polyaniline/multiwalled carbon nanotube composites on alkyd coatings against the corrosion of carbon steel alloy. Corros. Rev. 2017, 35, 85–94. [Google Scholar] [CrossRef]
- Pourjavadi, A.; Doroudian, M.; Ahadpour, A.; Pourbadiei, B. Preparation of flexible and free-standing graphene-based current collector via a new and facile self-assembly approach: Leading to a high performance porous graphene/polyaniline supercapacitor. Energy 2018, 152, 178–189. [Google Scholar] [CrossRef]
- Sharma, K.; Pareek, K.; Rohan, R.; Kumar, P. Flexible supercapacitor based on three-dimensional cellulose/graphite/polyaniline composite. Int. J. Energy Res. 2019, 43, 604–611. [Google Scholar] [CrossRef]
- Peng, C.; Liu, F.; Chen, C. Synthesis of graphene/nano-sulphur/polyaniline ternary nanocomposite for high-performance supercapacitor electrodes. Polym. Polym. Compos. 2019, 27, 3–10. [Google Scholar] [CrossRef]
- Ćirić-Marjanović, G.; Trchová, M.; Stejskal, J. The chemical oxidative polymerization of aniline in water: Raman spectroscopy. J. Raman Spectrosc. 2008, 39, 1375–1387. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, Y.; Wang, Y.; Liu, J. Facial preparation of covalent modified reduced graphene oxide/polyaniline composite and its stable-enhanced electrochemical performance. Heliyon 2023, 9, e13002. [Google Scholar] [CrossRef]
- Liu, J.; Bi, H.; Morais, P.C.; Zhang, X.; Zhang, F.; Hu, L. Room-temperature magnetism in carbon dots and enhanced ferromagnetism in carbon dots-polyaniline nanocomposite. Sci. Rep. 2017, 7, 2165. [Google Scholar] [CrossRef] [PubMed]
- Lapkowski, M.; Berrada, K.; Quillard, S.; Louarn, G.; Lefrant, S.; Pron, A. Electrochemical oxidation of polyaniline in nonaqueous electrolytes–in-situ Raman spectroscopic studies. Macromolecules 1995, 28, 1233–1238. [Google Scholar] [CrossRef]
- Jin, L.; Jiang, Y.; Zhang, M.; Li, H.; Xiao, L.; Li, M.; Ao, Y. Oriented Polyaniline Nanowire Arrays Grown on Dendrimer (PAMAM) Functionalized Multiwalled Carbon Nanotubes as Supercapacitor Electrode Materials. Sci. Rep. 2018, 8, 6268. [Google Scholar] [CrossRef]
- Liu, X.; Zheng, Y.; Wang, X. Controllable Preparation of Polyaniline–Graphene Nanocomposites using Functionalized Graphene for Supercapacitor Electrodes. J. Chem. Eur. 2015, 21, 10408–10415. [Google Scholar] [CrossRef]
- Rauhala, T.; Davodi, F.; Sainio, J.; Sorsa, O.; Kallio, T. On the stability of polyaniline/carbon nanotube composites as binder-free positive electrodes for electrochemical energy storage. Electrochim. Acta 2020, 336, 135735. [Google Scholar] [CrossRef]
- Nawaz, S.; Siddiq, M.; Kausar, A.; Hussain, S.T.; Abbas, F. Facile Synthesis and Properties of Multilayered Polyaniline/Polypyrrole/Epoxy/Polystyrene/Functionalized Carbon Nanotube Composites. Polym. Plast. Technol. Eng. 2014, 53, 661–670. [Google Scholar] [CrossRef]
- Zhao, J.; Qin, Z.; Li, T.; Li, Z.; Zhou, Z.; Zhu, M. Influence of acetone on nanostructure and electrochemical properties of interfacial synthesized polyaniline nanofibers. Prog. Nat. Sci. Mater. 2015, 25, 316–322. [Google Scholar] [CrossRef]
- Teng, Y.; Li, S.; Xue, C.; Zhang, H.; Zhu, L.; Tang, Y. Synthesis of Polyaniline/Graphene Oxide/Azobenzene Composite and Its Adjustable Photoelectric Properties. Adv. Polym. Technol. 2020, 2020, 9. [Google Scholar] [CrossRef]
- Yeszhan, Y.; Duisenbekov, S.; Kurmangaliyeva, D.; Kazhigitova, D.; Askar, P.; Tileuberdi, Y.; Konarov, A.; Adilov, S.; Nuraje, N. Enhanced electrochemical performance of a polyaniline-based supercapacitor by a bicontinuous microemulsion nanoreactor approach. RSC Adv. 2025, 15, 1205–1211. [Google Scholar] [CrossRef]
- Zeng, F.; Qin, Z.; Liang, B.; Li, T.; Liu, N.; Zhu, M. Polyaniline nanostructures tuning with oxidants in interfacial polymerization system. Prog. Nat. Sci. Mater. Int. 2015, 25, 512–519. [Google Scholar] [CrossRef]
- Wang, Y.; Gao, X.; Fu, Y.; Wu, X.; Wang, Q.; Zhang, W.; Luo, C. Enhanced microwave absorption performances of polyaniline/graphene aerogel by covalent bonding. Compos. B Eng. 2019, 169, 221–228. [Google Scholar] [CrossRef]
- Mahat, M.M.; Mawad, D.; Nelson, G.W.; Fearn, S.; Palgrave, R.G.; Payne, D.J.; Stevens, M.M. Elucidating the deprotonation of polyaniline films by X-ray photoelectron spectroscopy. J. Mater. Chem. C 2015, 3, 7180–7186. [Google Scholar] [CrossRef]
- Smolin, Y.Y.; Soroush, M.; Lau, K.K.S. Oxidative chemical vapor deposition of polyaniline thin films. Beilstein J. Nanotechnol. 2017, 8, 1266–1276. [Google Scholar] [CrossRef] [PubMed]
- Kang, E.T.; Neoh, K.G.; Tan, K.L. Polyaniline with high intrinsic oxidation state. Surf. Interface Anal. 1993, 20, 833–840. [Google Scholar] [CrossRef]
- Tantawy, H.R.; Kengne, B.A.F.; McIlroy, D.N.; Nguyen, T.; Heo, D.; Qiang, Y.; Aston, D.E. X-ray photoelectron spectroscopy analysis for the chemical impact of solvent addition rate on electromagnetic shielding effectiveness of HCl-doped polyaniline nanopowders. J. Appl. Phys. 2015, 118, 17550. [Google Scholar] [CrossRef]
- Lv, H.; Wei, Z.; Han, C.; Yang, X.; Tang, Z.; Zhang, Y.; Zhi, C.; Li, H. Cross-linked polyaniline for production of long lifespan aqueous iron||organic batteries with electrochromic properties. Nat. Commun. 2023, 14, 3117. [Google Scholar] [CrossRef] [PubMed]
- Qin, X.; Wan, J.; Zhang, Q.; Zhang, Y.; Yu, H.; Shi, S. Polyaniline-modified graphitic carbon nitride as electrode materials for high-performance supercapacitors. Carbon Lett. 2023, 33, 781–790. [Google Scholar] [CrossRef]
- Jeon, J.W.; Kwon, S.R.; Lutkenhaus, J.L. Polyaniline nanofiber/electrochemically reduced graphene oxide layer-by-layer electrodes for electrochemical energy storage. J. Mater. Chem. A 2015, 3, 3757–3767. [Google Scholar] [CrossRef]
- Zou, Y.; Zhang, Z.; Zhong, W.; Yang, W. Hydrothermal direct synthesis of polyaniline, graphene/polyaniline and N-doped graphene/polyaniline hydrogels for high-performance flexible supercapacitors. J. Mater. Chem. A 2018, 6, 9245–9256. [Google Scholar] [CrossRef]
- Waware, U.S.; Arukula, R.; Hamouda, A.M.S.; Kasak, P. Electrochemical and X-ray photoelectron spectroscopic investigations of conductive polymers. Ionics 2020, 26, 831–838. [Google Scholar] [CrossRef]
- Qiu, Y.; Jia, X.; Zhang, M.; Li, H. A new strategy for fabricating well-distributed Polyaniline/Graphene composite fibers toward flexible high-performance supercapacitors. Nanomaterials 2022, 12, 3297. [Google Scholar] [CrossRef]
- Gedela, V.R.; Srikanth, V.V.S.S. Polyaniline nanostructures expedient as working electrode materials in supercapacitors. Appl. Phys. A 2014, 115, 189–197. [Google Scholar] [CrossRef]
- Kumar, S.N.; Gaillard, F.; Bouyssoux, G.; Sartre, A. high-resolution XPS studies of electrochemically synthesized conducting polyaniline films. Synth. Met. 1990, 36, 111–127. [Google Scholar] [CrossRef]
- Yue, J.; Epstein, A.J. XPS Study of self-doped conducting Polyaniline and parent systems. Macromolecules 1991, 24, 4441–4445. [Google Scholar] [CrossRef]
- Marmisollé, W.A.; Gregurec, D.; Moya, S.; Azzaroni, O. Polyanilines with pendant amino groups as electrochemically active copolymers at neutral pH. Chem. Electro. Chem. 2015, 2, 2011–2019. [Google Scholar] [CrossRef]
- Tan, H.X.; Xu, X.C. Conductive properties and mechanism of various polymers doped with carbon nanotube/polyaniline hybrid nanoparticles. Compos. Sci. Technol. 2016, 128, 155–160. [Google Scholar] [CrossRef]
- Wu, J.C.; Chen, S.S.; Yu, T.C.; Wu, K.C.W.; Hou, C.H. Effective electrochemically controlled removal of fluoride ions using electrodeposited polyaniline-carbon nanotube composite electrodes. Sep. Purif. Technol. 2021, 254, 117561. [Google Scholar] [CrossRef]
- Qin, T.; Deng, L.; Zhang, P.; Tang, M.; Li, C.; Xie, H.; Huang, S.; Gao, X. Enhancement of electrochromic properties of polyaniline induced by copper ions. Nanoscale Res. Lett. 2022, 17, 51. [Google Scholar] [CrossRef]
- Zhang, J.; Xing, A.; Jia, B.; Liu, X. Synthesis of conductive polyaniline nanofibers in one step by protonic acid and iodine doping. High Perform. Polym. 2019, 31, 893–900. [Google Scholar] [CrossRef]
- Ansari, M.O.; Alshahrie, A.; Ansari, S.A. Facile route to porous polyaniline@nanodiamond-graphene based nanohybrid structures for DC electrical conductivity retention and supercapacitor applications. J. Polym. Res. 2019, 26, 76. [Google Scholar] [CrossRef]
- Libich, J.; Máca, J.; Vondrák, J.; Čech, O.; Sedlaříková, M. Supercapacitors: Properties and applications. J. Energy Storage 2018, 17, 224–227. [Google Scholar] [CrossRef]
- Laviron, E. General expression of the linear potential sweep voltammogram in the case of diffusionless electrochemical systems. J. Electroanal. Chem. 1979, 101, 19–28. [Google Scholar] [CrossRef]
- Benchikh, I.; Ezzat, A.O.; Sabantina, L.; Benmimoun, Y.; Benyoucef, A. Investigation of hybrid electrodes of polyaniline and reduced Graphene oxide with bio-waste-derived activated carbon for supercapacitor applications. Polymers 2024, 16, 421. [Google Scholar] [CrossRef]
- Umar, A.; Ahmed, F.; Ullah, N.; Ansari, S.A.; Hussain, S.; Ibrahim, A.A.; Qasem, H.; Kumar, S.A.; Alhamami, M.A.; Almehbad, N.; et al. Exploring the potential of reduced graphene oxide/polyaniline (rGO@PANI) nanocomposites for high-performance supercapacitor application. Electrochim. Acta 2024, 479, 143743. [Google Scholar] [CrossRef]
- Marwat, M.A.; Ishfaq, S.; Adam, K.M.; Tahir, B.; Khan, M.H.S.M.F.; Karim, M.R.A.; Din, Z.U.; Abdullah, S.; Ghazanfar, E. Enhancing supercapacitor performance of Ni-Co-Mn metal-organic frameworks by compositing it with polyaniline and reduced graphene oxide. RSC Adv. 2024, 14, 2102–2115. [Google Scholar] [CrossRef]
- Li, T.; Zhou, Y.; Liang, B.; Jin, D.; Liu, N.; Qin, Z.; Zhu, M. One–pot synthesis and electrochemical properties of polyaniline nanofibers through simply tuning acid–base environment of reaction medium. Electrochim. Acta 2017, 249, 33–42. [Google Scholar] [CrossRef]
- Jasna, M.; Pillai, M.M.; Abhilash, A.; Midhun, P.S.; Jayalekshmi, S.; Jayaraj, M.K. Polyaniline wrapped carbon nanotube/exfoliated MoS2 nanosheet composite as a promising electrode for high power supercapacitors. Carbon Trends 2022, 7, 100154. [Google Scholar] [CrossRef]
- Guo, F.; Mi, H.; Zhou, J.; Zhao, Z.; Qiu, J. Hybrid pseudocapacitor materials from polyaniline@multi-walled carbon nanotube with ultrafine nanofiber-assembled network shell. Carbon 2015, 95, 323–329. [Google Scholar] [CrossRef]
- Pawar, D.C.; Bagde, A.G.; Thorat, J.P.; Lokhande, C.D. Synthesis of reduced graphene oxide (rGO)/polyaniline (PANI) composite electrode for energy storage: Aqueous asymmetric supercapacitor. Eur. Polym. J. 2024, 218, 113366. [Google Scholar] [CrossRef]
- Cho, W.H.; Cheng, I.C.; Chen, J.Z. Performance Comparison of Reduced Graphene Oxide (rGO)-polyaniline (PANI) Supercapacitors with LiCl, Li2SO4, and H2SO4 Electrolytes. J. Electrochem. Soc. 2023, 170, 010532. [Google Scholar] [CrossRef]
- Banda, H.; Dou, J.H.; Chen, T.; Zhang, Y.; Dincă, M. Dual-Ion intercalation and high volumetric capacitance in a two-dimensional non-porous coordination polymer. Angew. Chem. Int. Ed. 2021, 60, 27119–27125. [Google Scholar] [CrossRef]
- Thomas, L.; Pete, S.; Chaitra, K.; Venkatesh, K.; Gopalkrishna, B.; Nagaraju, K. Facile synthesis of PANI-MWCNT-Ni(OH)2 ternary composites and study of their performance as electrode material for supercapacitors. Diam. Relat. Mater. 2020, 106, 107853. [Google Scholar] [CrossRef]
- Colak, S.G.; Gungor, A.; Colak, M.O.A.; Simsek, U.B.; Genc, R.; Erdem, E. Exploring PANI/CBTS nanofiber composites as supercapacitor electrodes: Structure-performance correlation. Adv. Sustain. Syst. 2025, 9, e00427. [Google Scholar] [CrossRef]
- Acharya, A.A.D.; Thakur, Y.S. Tuning electrochemical performance of polyaniline-based supercapacitors by inclusion of protonic acid and electrolyte concentration. J. Indian. Chem. Soc. 2025, 102, 101836. [Google Scholar] [CrossRef]
- Fan, H.; Zhao, N.; Wang, H.; Xu, J.; Pan, F. 3D conductive network-based free-standing PANI-RGO-MWCNTs hybrid film for high-performance flexible supercapacitor. J. Mater. Chem. A 2014, 2, 12340–12347. [Google Scholar] [CrossRef]
- Shen, K.; Ran, F.; Zhang, X.; Liu, C.; Wang, N.; Niu, X.; Liu, Y.; Zhang, D.; Kong, L.; Kang, L.; et al. Supercapacitor electrodes based on nano-polyaniline deposited on hollow carbon spheres derived from cross-linked co-polymers. Synth. Met. 2015, 209, 369–376. [Google Scholar] [CrossRef]
- Chauhan, N.P.S.; Mozafari, M.; Chundawat, N.S.; Meghwal, K.; Ameta, R.; Ameta, S.C. High-performance supercapacitors based on polyaniline-graphene nanocomposites: Some approaches, challenges and opportunities. J. Ind. Eng. Chem. 2016, 36, 13–29. [Google Scholar] [CrossRef]
- Singu, B.S.; Srinivasan, P.; Yoon, K.R. Emulsion polymerization method for polyaniline-multiwalled carbon nanotube nanocomposites as supercapacitor materials. J. Solid State Electrochem. 2016, 20, 3447–3457. [Google Scholar] [CrossRef]
- Chen, N.; Ren, Y.; Kong, P.; Tan, L.; Feng, H.; Luo, Y. In situ one-pot preparation of reduced graphene oxide/polyaniline composite for high-performance electrochemical capacitors. Appl. Surf. Sci. 2017, 392, 71–79. [Google Scholar] [CrossRef]
- Du, P.; Lin, L.; Wang, H.; Liu, D.; Wei, W.; Li, J.; Liu, P. Fabrication of porous polyaniline modified MWCNTs core-shell structure for high performance supercapacitors with high rate capability. Mater. Des. 2017, 127, 76–83. [Google Scholar] [CrossRef]
- Zheng, X.; Yu, H.; Xing, R.; Ge, X.; Sun, H.; Li, R.; Zhang, Q. Multi-growth site graphene/polyaniline composites with highly enhanced specific capacitance and rate capability for supercapacitor application. Electrochim. Acta 2018, 260, 504–513. [Google Scholar] [CrossRef]
- Devi, M.; Kumar, A. Enhanced electrochemical performance of in situ reduced graphene oxide-polyaniline nanotubes hybrid nanocomposites using redox-additive aqueous electrolyte. J. Phys. D Appl. Phys. 2018, 51, 085501. [Google Scholar] [CrossRef]
- Li, J.; Qiu, S.; Liu, B.; Chen, H.; Xiao, D.; Li, H. Strong interaction between polyaniline and carbon fibers for flexible supercapacitor electrode materials. J. Power Sources 2021, 483, 228219. [Google Scholar] [CrossRef]
- Zhang, S.; Pan, N. Supercapacitors Performance Evaluation. Adv. Energy Mater. 2015, 5, 1401401. [Google Scholar] [CrossRef]
- Raza, W.; Ali, F.; Raza, N.; Luo, Y.; Kim, K.H.; Yang, J.; Kumar, S.; Mehmood, A.; Kwon, E.E. Recent advancements in supercapacitor technology. Nano Energy 2018, 52, 441–473. [Google Scholar] [CrossRef]
- Wang, S.; Zhang, J.; Gharbi, O.; Vivier, V.; Gao, M.; Orazem, M.E. Electrochemical impedance spectroscopy. Nat. Rev. Methods Primers 2021, 1, 41. [Google Scholar] [CrossRef]
- Gul, H.; Shah, A.H.A.; Bilal, S. Achieving ultrahigh cycling stability and extended potential window for supercapacitors through asymmetric combination of conductive polymer nanocomposite and activated carbon. Polymers 2019, 11, 1678. [Google Scholar] [CrossRef] [PubMed]
- Tayel, M.B.; Harb, M.E.; Soliman, M.M.; Ebrahim, S.A. Fabrication of supercapacitor using double layers of graphene and polyaniline electrodes for energy storage. J. Electr. Eng. 2016, 16, 18. [Google Scholar]
- Shao, Y.; Li, J.; Li, Y.; Wang, H.; Zhang, Q.; Kaner, R.B. Flexible quasi-solid-state planar micro-supercapacitors based on cellular graphene films. Mater. Horiz. 2017, 4, 1145–1150. [Google Scholar] [CrossRef]
- Purkait, T.; Singh, G.; Kumar, D.; Singh, M.; Dey, R.S. High-performance flexible supercapacitors based on electrochemically tailored three-dimensional reduced graphene oxide networks. Sci. Rep. 2018, 8, 640. [Google Scholar] [CrossRef]
- Kobayashi, M.; Tashiro, K.; Tadokoro, H. Molecular vibrations of three crystal forms of poly(vinylidene fluoride). Macromolecules 1975, 8, 158e171. [Google Scholar] [CrossRef]
- Bachmann, M.A.; Koenig, J.L. Vibrational analysis of phase III of poly (vinylidene fluoride). J. Chem. Phys. 1981, 74, 5896e5910. [Google Scholar] [CrossRef]
- Lu, F.; Zhang, C.; Lu, B.; Yu, K.; Liu, J.; Kang, H.; Liu, R.; Lan, G. Cellobiose as a model compound for cellulose to study the interactions in cellulose/lithium chloride/N-methyl-2-pyrrolidone systems. Cellulose 2017, 24, 1621–1629. [Google Scholar] [CrossRef]
- McMurray, I.M.; Nettles, J.R.; Uzelmeier, A.W.; Swartz, J.A.; Newby, J.J. An analysis of the N-methyl-2-pyrrolidone: Water complex using computational and matrix isolation FTIR methods. J. Molec. Spectrosc. 2025, 408, 111985. [Google Scholar] [CrossRef]













| Samples | D (cm−1) | G (cm−1) | ID/IG |
|---|---|---|---|
| O-MWCNTs | 1332 | 1592 | 1.07 |
| PANI/O-MWCNTs (DCP) | 1326 | 1589 | 0,53 |
| PANI/O-MWCNTs (HT-120 °C) | 1345 | 1588 | 0.78 |
| PANI/O-MWCNTs (HT-180 °C) | 1350 | 1572 | 0.88 |
| Electroactive Material | Electrolyte | Specific Capacitance (F·g−1) | Reference |
|---|---|---|---|
| PANI-ultrafine NFs PANI-ultrafine NFs/O-MWCNTs | 6M KOH | 2074 (2.07 F/cm2)-CV 1410 (1.41 F/cm2)-GCD | This work |
| PANI/RGO/MWCNTs | 1M H2SO4 | 498 | [115] |
| Nano-PANI/HCS | 1M H2SO4 | 435 | [116] |
| PANI/MWCNT | 1M H2SO4 | 663 | [108] |
| GO/PANI SG/PANI | 1M HCl | 1095 478 | [117] |
| PANI/MWCNT | 1M H2SO4 | 360 | [118] |
| NiO/PANI-MWCNT | 10 mL KCl | 356.54 | [36] |
| rGO/PANI | 1M H2SO4 | 524.4 | [119] |
| CNTs/PANI | 1M H2SO4 | 406 | [120] |
| PANI | 1M H2SO4 | 857.2 | [106] |
| PANI/MWCNT | 1M H2SO4 | 1551 | [35] |
| MSG/PANI | 1M H2SO4 | 912 | [121] |
| PANI:PSS/Fe-FLG | 0.1M HCl 0.1M KCL | 768.6 659.2 | [32] |
| RGO/PANI NTs | 1M KCl 1M H2SO4 | 876.43 1081.03 | [122] |
| GSA/PANI HS | 1M H2SO4 | 546 | [33] |
| Porous G/PANI nanoarrays | 1M H2SO4 | 752 | [44] |
| GH/SWCNTs/PANI | 1M H2SO4 | 145 | [59] |
| CuO@NiO/PANI/MWCNT | 3M NaOH | 1372 | [37] |
| PANI/ND@GN | 1M KOH | 150.20 | [100] |
| PANI/rGO-HT | 1M H2SO4 | 420 | [30] |
| LDH/PANI | (0.5 mol/L) K3[Fe(CN)6] | 592.5 | [28] |
| PANI | 1M H2SO4 | 387.7 | [7] |
| PANI-MWCNTs-Ni(OH)2 | 1M KOH | 1917 | [112] |
| PANI/CNF | 1M H2SO4 | 493.75 | [17] |
| PANI-g-CF | 1M H2SO4 | 1178 | [123] |
| PANI doped with HSO4− or Cl− ions | 1M KOH | 155.75 and 167.75 | [114] |
| PANI nanofibers | H3PO4/PVA | 280.4 | [79] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Djefaflia, F.; Guellati, O.; Merzoug, A.N.; Harat, A.; El Haskouri, J.; Janowska, I.; Baibarac, M. Exploring the Synergistic Effects of Ultrafine Polyaniline Nanofibers and Oxygen-Modified Multi-Walled Carbon Nanotubes on Enhancing Pseudocapacitive Electrochemical Performance for Advanced Supercapacitors. Materials 2026, 19, 1356. https://doi.org/10.3390/ma19071356
Djefaflia F, Guellati O, Merzoug AN, Harat A, El Haskouri J, Janowska I, Baibarac M. Exploring the Synergistic Effects of Ultrafine Polyaniline Nanofibers and Oxygen-Modified Multi-Walled Carbon Nanotubes on Enhancing Pseudocapacitive Electrochemical Performance for Advanced Supercapacitors. Materials. 2026; 19(7):1356. https://doi.org/10.3390/ma19071356
Chicago/Turabian StyleDjefaflia, Fahima, Ouanassa Guellati, Assia Nait Merzoug, Aicha Harat, Jamal El Haskouri, Izabela Janowska, and Mihaela Baibarac. 2026. "Exploring the Synergistic Effects of Ultrafine Polyaniline Nanofibers and Oxygen-Modified Multi-Walled Carbon Nanotubes on Enhancing Pseudocapacitive Electrochemical Performance for Advanced Supercapacitors" Materials 19, no. 7: 1356. https://doi.org/10.3390/ma19071356
APA StyleDjefaflia, F., Guellati, O., Merzoug, A. N., Harat, A., El Haskouri, J., Janowska, I., & Baibarac, M. (2026). Exploring the Synergistic Effects of Ultrafine Polyaniline Nanofibers and Oxygen-Modified Multi-Walled Carbon Nanotubes on Enhancing Pseudocapacitive Electrochemical Performance for Advanced Supercapacitors. Materials, 19(7), 1356. https://doi.org/10.3390/ma19071356









