Hyaluronic-Acid Nanocapsules with Plant Extracts: Characterization and Antimicrobial Activity Against Skin Microbiota
Highlights
- HA nanocapsules with plant extracts were successfully synthesized.
- Nanocomposites showed stable physicochemical and structural properties.
- Extract type determined distinct antimicrobial activity profiles.
- Nanocapsules are suitable carriers for topical bioactive delivery.
- Formulations can be tailored for cosmetic or dermatological uses.
- Extract-specific activity enables targeted skincare applications.
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Nanocomposites
2.2.1. Plant Materials and Extraction
2.2.2. Preparation of Hyaluronic Acid Gel
2.2.3. Preparation of Nanoemulsions
2.2.4. Preparation of the Control (C) Sample (H0)
2.2.5. Preparation of Nanocomposite Formulations
2.2.6. Sample Preparation for Analysis
2.3. Scanning Electron Microscopy
2.4. FTIR Spectroscopy
2.5. UV-Vis Spectroscopy
2.6. Color Measurement
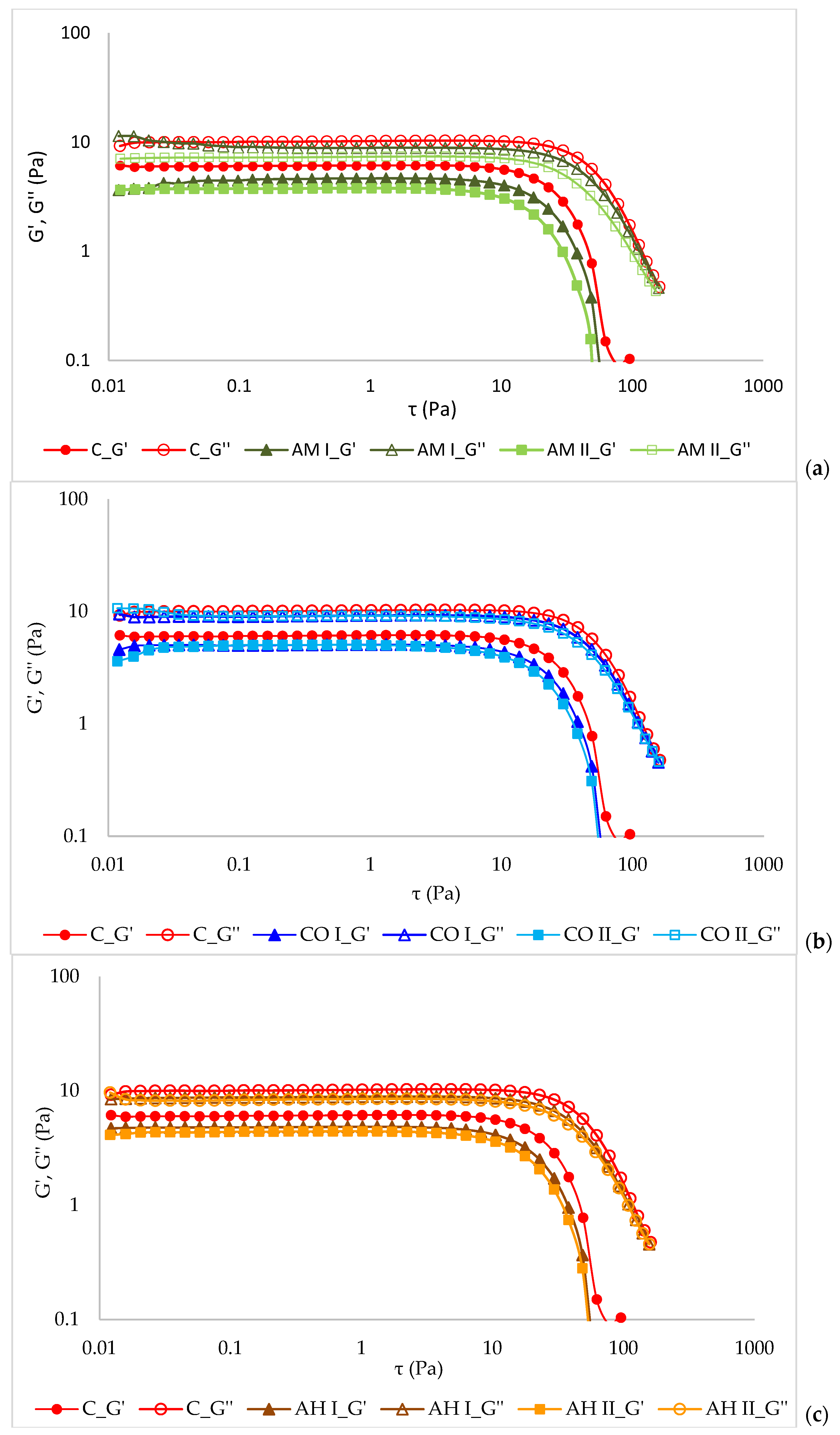
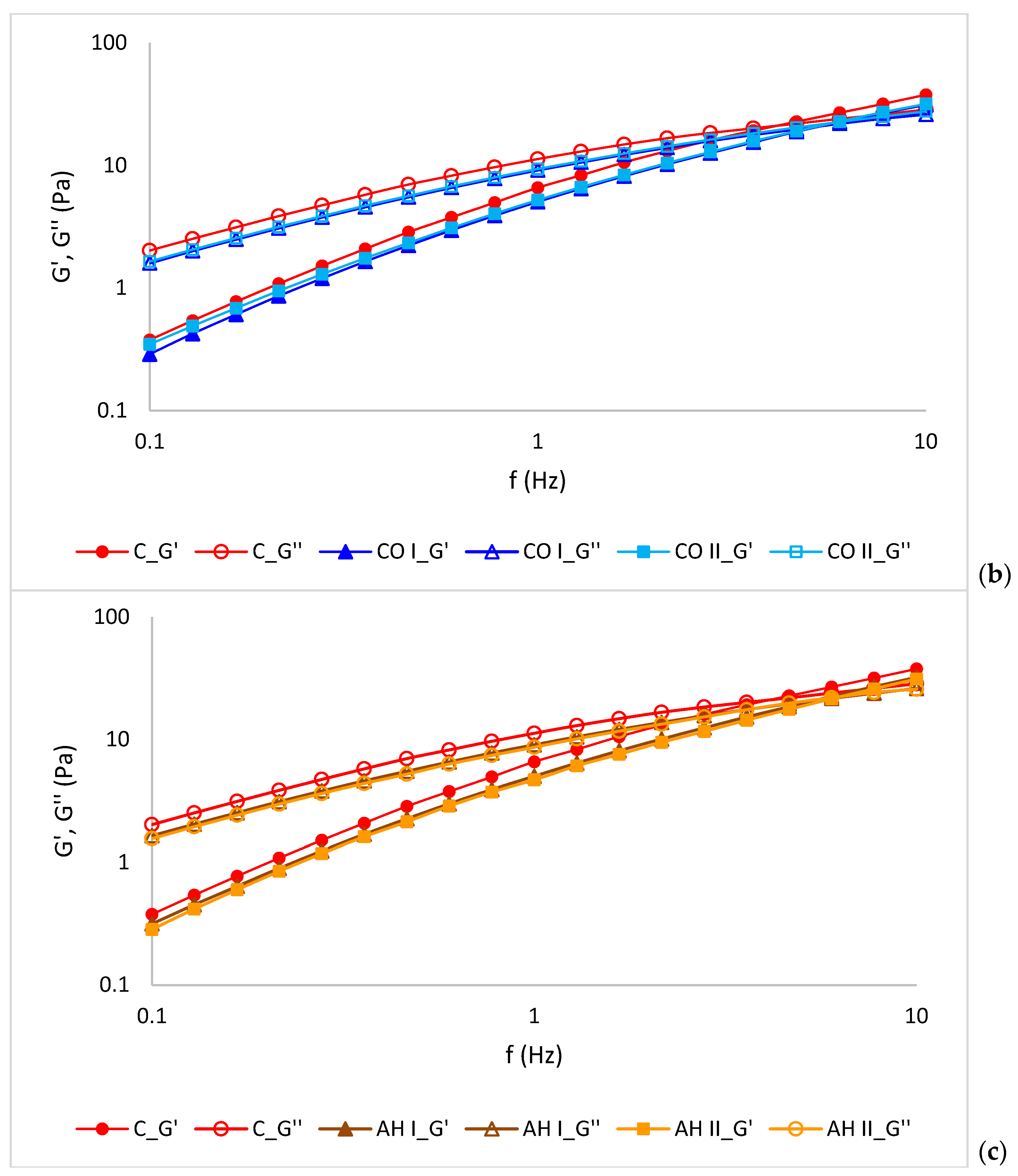
2.7. Rheological Measurement
2.8. Texture Analyses
2.9. Antimicrobial Activity Assays
2.10. Statistical Analyses
3. Results and Discussion
3.1. Scanning Electron Microscopy-Based Determination of the Obtained Nanocapsules
3.2. FTIR Spectroscopy
3.3. UV-VIS
- Aesculus hippocastanum (AH): The spectrum is dominated by a sharp, intense peak at ~264 nm. This absorption corresponds to the Band II (benzoyl system) transitions, likely attributable to coumarin glycosides (e.g., esculin, fraxin) and phenolic acids, which are the primary bioactive constituents of horse chestnut [30].
- Calendula officinalis (CO): While exhibiting the UV peaks characteristic of phenolics, the CO spectrum is distinguished by a broader absorption profile that extends into the visible region (>400 nm). This elevated baseline and “tailing” effect are indicative of the presence of carotenoids (e.g., lutein, beta-carotene), which are well-known chromophores in calendula flowers [28,31].
3.4. Color Parameters of Emulsions
3.5. Rheological Properties of Emulsions
3.6. Texture Properties of Emulsions
3.7. Preliminary Screening of Plant-Based Emulsions and Reference Antiseptics
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Adamczyk, K.; Garncarczyk, A.A.; Antończak, P.P. The microbiome of the skin. Dermatol. Rev. Dermatol. 2018, 105, 285–297. [Google Scholar] [CrossRef]
- Belkaid, Y.; Segre, J.A. Dialogue between skin microbiota and immunity. Science 2014, 346, 954–959. [Google Scholar] [CrossRef] [PubMed]
- Wanke, I.; Skabytska, Y.; Kraft, B.; Peschel, A.; Biedermann, T.; Schittek, B. Staphylococcus aureus skin colonization is promoted by barrier disruption and leads to local inflammation. Exp. Dermatol. 2013, 22, 153–155. [Google Scholar] [CrossRef]
- Kononowicz, W.S.; Malara, B. Post-treatment physiological effects, adverse effects and complications after the selected aesthetic treatments. Rev. Lit. 2021, 10, 211–216. [Google Scholar] [CrossRef]
- Cedillo-Cortezano, M.; Martinez-Cuevas, L.R.; López, J.A.M.; Barrera López, I.L.; Escutia-Perez, S.; Petricevich, V.L. Use of Medicinal Plants in the Process of Wound Healing: A Literature Review. Pharmaceuticals 2024, 17, 303. [Google Scholar] [CrossRef]
- Pourshahidi, S.; Sheykhbahaei, N. Effectiveness of herbal based medications in the treatment of oral lichen planus: A review article. J. Herb. Med. 2021, 29, 100458. [Google Scholar] [CrossRef]
- King, M. The Management of Bruising following Nonsurgical Cosmetic Treatment. J. Clin. Aesthet. Dermatol. 2017, 10, E1–E4. [Google Scholar] [PubMed]
- Sherban, A.; Wang, J.V.; Geronemus, R.G. Growing role for arnica in cosmetic dermatology: Lose the bruise. J. Cosmet. Dermatol. 2021, 20, 2062–2068. [Google Scholar] [CrossRef]
- Walendziak, W.; Villegas, N.R.; Douglas, T.E.L.; Kozlowska, J. Phytochemical studies of plant extracts enclosed in chitosan microparticles and the effect of phytoformulations on skin condition. Eur. Polym. J. 2025, 233, 113968. [Google Scholar] [CrossRef]
- Butkeviciute, A.; Ramanauskiene, K.; Kurapkiene, V.; Janulis, V. Dermal Penetration Studies of Potential Phenolic Compounds Ex Vivo and Their Antioxidant Activity In Vitro. Plants 2022, 11, 1901. [Google Scholar] [CrossRef]
- Alhalwani, A.; Husain, A.; Saemaldahar, A.; Makhdoum, F.; Alhakami, M.; Ashi, R.; Wali, R.; Alsharif, S.; Khan, M.A.; Jastaniah, N.; et al. The impact of alcohol hand sanitizer use on skin health between healthcare worker: Cross-sectional study. Ski. Res. Technol. 2024, 30, e13527. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, S.; Patel, A.; Kesselman, M.M.; Demory, M.L. Hand Sanitizer: Stopping the Spread of Infection at a Cost. Cureus 2024, 16, e61846. [Google Scholar] [CrossRef]
- Pal, V.K.; Lee, S.; Naidu, M.; Lee, C.; Kannan, K. Occurrence of and dermal exposure to benzene, toluene and styrene found in hand sanitizers from the United States. Environ. Int. 2022, 167, 107449. [Google Scholar] [CrossRef]
- Tominc, G.C.; Dalmagro, M.; Pereira, E.D.; Adamczuk, M.S.; Bonato, F.G.; Almeida, R.M.; Schneider, R.; Negri, M.F.; Gonçalves, D.D.; Hoscheid, J. Formulation and Characterization of Nanoemulsion Incorporating Chamomilla recutita L. Extract Stabilized with Hyaluronic Acid. Pharmaceutics 2024, 16, 701. [Google Scholar] [CrossRef]
- Hashempur, M.H.; Ghorat, F.; Karami, F.; Jahanbin, A.; Nouraei, H.; Abbasi, M.; Jafari, M.; Zare, A.; Barzegar, S.; Zareshahrabadi, Z. Topical Delivery Systems for Plant-Derived Antimicrobial Agents: A Review of Current Advances. Int. J. Biomater. 2025, 2025, 4251091. [Google Scholar] [CrossRef]
- Chiaula, V.; Jeppe Madsen, J.; Madsen, F.B.; Mazurek, P.; Nielsen, A.C.; Skov, A.L. Antimicrobial silicone skin adhesives facilitated by controlled octenidine release from glycerol compartments. Int. J. Smart Nano Mater. 2023, 14, 369–389. [Google Scholar] [CrossRef]
- Balazs, E.A.; Denlinger, J.L. Clinical Uses of Hyaluronan. In Ciba Foundation Symposium 143—The Biology of Hyaluronan; John Wiley and Sons: Hoboken, NJ, USA, 2007; pp. 265–285. [Google Scholar]
- Krystyjan, M.; Majka, P.; Sobolewska-Zielińska, J.; Turek, K.; Michalski, O.; Khachatryan, K.; Khachatryan, G. Enrichment of Apple–Plum Fruit Mousse with Vitamin D3 and Sea Buckthorn Oil Using Pectin-Based Encapsulation: A Study of Physicochemical and Sensory Properties. Int. J. Mol. Sci. 2025, 26, 11480. [Google Scholar] [CrossRef]
- Krystyjan, M.; Khachatryan, G.; Khachatryan, K.; Lenart-Boroń, A.; Socha, R.; Potoniec, Z. The Fortification of Fruit Mousses with Encapsulated Bioactive Compounds in a Potato Starch Matrix as an Example of Novel Functional Foods. Int. J. Mol. Sci. 2026, 27, 1106. [Google Scholar] [CrossRef] [PubMed]
- Krystyjan, M.; Khachatryan, G.; Ciesielski, W.; Buksa, K.; Sikora, M. Preparation and characteristics of mechanical and functional properties of starch/Plantago psyllium seeds mucilage films. Starch—Stärke 2017, 69, 1700014. [Google Scholar] [CrossRef]
- Krystyjan, M.; Ciesielski, W.; Khachatryan, G.; Sikora, M.; Tomasik, P. Structure, rheological, textural and thermal properties of potato starch—Inulin gels. LWT Food Sci. Technol. 2015, 60, 131–136. [Google Scholar] [CrossRef]
- Khachatryan, G.; Khachatryan, L.; Krystyjan, M.; Lenart-Boroń, A.; Krzan, M.; Kulik, K.; Białecka, A.; Grabacka, M.; Nowak, N.; Khachatryan, K. Preparation of Nano/Microcapsules of Ozonated Olive Oil in Hyaluronan Matrix and Analysis of Physicochemical and Microbiological (Biological) Properties of the Obtained Biocomposite. Int. J. Mol. Sci. 2022, 23, 14005. [Google Scholar] [CrossRef]
- Khachatryan, G.; Khachatryan, K.; Krystyjan, M.; Krzemińska-Fiedorowicz, L.; Lenart-Boroń, A.; Białecka, A.; Krupka, M.; Krzan, M.; Blaszyńska, K.; Hanula, M.; et al. Synthesis and Investigation of Physicochemical and Biological Properties of Films Containing Encapsulated Propolis in Hyaluronic Matrix. Polymers 2023, 15, 1271. [Google Scholar] [CrossRef]
- Lenart-Boroń, A.; Stankiewicz, K.; Bulanda, K.; Czernecka, N.; Heliasz, M.; Hunter, W.; Ratajewicz, A.; Khachatryan, K.; Khachatryan, G. In Vitro Antibacterial Activity of Ozonated Olive Oil against Bacteria of Various Antimicrobial Resistance Profiles Isolated from Wounds of Companion Animals. Int. J. Mol. Sci. 2024, 25, 3557. [Google Scholar] [CrossRef]
- Dias, M.I.; Albiston, C.; Añibarro-Ortega, M.; Ferreira, I.C.F.R.; Pinela, J.; Barros, L. Sonoextraction of phenolic compounds and saponins from Aesculus hippocastanum seed kernels: Modeling and optimization. Ind. Crops Prod. 2022, 185, 115142. [Google Scholar] [CrossRef]
- Mahmoud, M.H. Encapsulation of Pomegranate Seed Oil Using W/O/W Nano-Emulsion Technique Followed by Spray Drying and Its Application in Jelly Form. J. Microbiol. Biotechnol. Food Sci. 2020, 10, 449–453. [Google Scholar] [CrossRef]
- Cortez-Trejo, M.C.; Wall-Medrano, A.; Gaytán-Martínez, M.; Mendoza, S. Microencapsulation of pomegranate seed oil using a succinylated taro starch: Characterization and bioaccessibility study. Food Biosci. 2021, 41, 100929. [Google Scholar] [CrossRef]
- Adiba, A.; Razouk, R.; Haddioui, A.; Ouaabou, R.; Hamdani, A.; Kouighat, M.; Hssaini, L. FTIR spectroscopy-based lipochemical fingerprints involved in pomegranate response to water stress. Heliyon 2023, 9, e16687. [Google Scholar] [CrossRef] [PubMed]
- Ivanova, D.; Deneva, V.; Zheleva-Dimitrova, D.; Balabanova-Bozushka, V.; Nedeltcheva, D.; Gevrenova, R.; Antonov, L. Quantitative Characterization of Arnicae flos by RP-HPLC-UV and NIR Spectroscopy. Foods 2019, 8, 9. [Google Scholar] [CrossRef]
- Mekonnen, K.D. Fourier transform infrared spectroscopy as a tool for identifying the unique characteristic bands of lipid in oilseed components: Confirmed via Ethiopian indigenous desert date fruit. Heliyon 2023, 9, e14699. [Google Scholar] [CrossRef] [PubMed]
- Jerman, I.; Ogrizek, L.; Pihir, J.; Senica, M. Interactions of Highly Diluted Arnica montana Extract with Water Across Glass Interfaces. Int. J. Mol. Sci. 2025, 26, 1115. [Google Scholar] [CrossRef] [PubMed]
- Petrova, M.; Geneva, M.; Trendafilova, A.; Miladinova-Georgieva, K.; Dimitrova, L.; Sichanova, M.; Nikolova, M.; Ivanova, V.; Dimitrova, M.; Sozoniuk, M. Antioxidant Capacity and Accumulation of Caffeoylquinic Acids in Arnica montana L. In Vitro Shoots After Elicitation with Yeast Extract or Salicylic Acid. Plants 2025, 14, 967. [Google Scholar] [CrossRef]
- Sausserde, R.; Kampuss, K. Composition of Carotenoids in Calendula (Calendula officinalis L.) Flowers. Foodbalt 2014, 13–18. [Google Scholar]
- Haładyn, K.; Wojdyło, A.; Nowicka, P. Isolation of Bioactive Compounds (Carotenoids, Tocopherols, and Tocotrienols) from Calendula officinalis L., and Their Interaction with Proteins and Oils in Nanoemulsion Formulation. Molecules 2024, 29, 4184. [Google Scholar] [CrossRef]
- Jarzębski, M.; Smułek, W.; Siejak, P.; Kobus-Cisowska, J.; Pieczyrak, D.; Baranowska, H.M.; Jakubowicz, J.; Sopata, M.; Białopiotrowicz, T.; Kaczorek, E. Aesculus hippocastanum L. extract as a potential emulsion stabilizer. Food Hydrocoll. 2019, 97, 105237. [Google Scholar] [CrossRef]
- Michalak, M.; Błońska-Sikora, E.M.; Paradowska, K.; Zielińska, A. Pharmaceutical availability of hydrogels with extracts of Arnica montana, Aesculus hippocastanum and Ruscus aculeatus and their potential use as antioxidant polyphenol-rich material. Med. Stud. Med. 2023, 39, 223–229. [Google Scholar] [CrossRef]
- Kasprzak, M.M.; Berski, W.; Krystyjan, M.; Jamróz, E.; Florczuk, A.; Tkaczewska, J.; Zając, M.; Domagała, J.; Lett, A.M.; Ptasznik, S. Effects of fibre addition and processing on the stability, rheology and in vitro gastric digestion of whey protein-xanthan gum stabilised emulsions with high oil phase. LWT 2023, 178, 114465. [Google Scholar] [CrossRef]
- Krystyjan, M.; Dobosz-Kobędza, A.; Sikora, M.; Baranowska, H.M. Influence of Xanthan Gum Addition on the Short- and Long-Term Retrogradation of Corn Starches of Various Amylose Content. Polymers 2022, 14, 452. [Google Scholar] [CrossRef] [PubMed]
- Quintana, J.M.; Califano, A.N.; Zaritzky, N.E.; Partal, P.; Franco, J.M. Linear And Nonlinear Viscoelastic Behavior of Oil-In-Water Emulsions Stabilized with Polysaccharides. J. Texture Stud. 2002, 33, 215–236. [Google Scholar] [CrossRef]
- Milas, M.; Rinaudo, M.; Roure, I.; Al-Assaf, S.; Phillips, G.O.; Williams, P.A. Comparative rheological behavior of hyaluronan from bacterial and animal sources with cross-linked hyaluronan (hylan) in aqueous solution. Biopolymers 2001, 59, 191–204. [Google Scholar] [CrossRef] [PubMed]
- Khachatryan, G.; Khachatryan, K.; Krystyjan, M.; Krzan, M.; Khachatryan, L. Functional properties of composites containing silver nanoparticles embedded in hyaluronan and hyaluronan-lecithin matrix. Int. J. Biol. Macromol. 2020, 149, 417–423. [Google Scholar] [CrossRef]
- Venerová, T.; Pekař, M. Rheological properties of gels formed by physical interactions between hyaluronan and cationic surfactants. Carbohydr. Polym. 2017, 170, 176–181. [Google Scholar] [CrossRef]
- Grabowski, M.; Gmyrek, D.; Żurawska, M.; Trusek, A. Hyaluronic Acid: Production Strategies, Gel-Forming Properties, and Advances in Drug Delivery Systems. Gels 2025, 11, 424. [Google Scholar] [CrossRef]
- Di Meo, C.; Stellavato, A.; d’Agostino, M.; D’Agostino, A.; Schiraldi, C.; La Gatta, A. Hyaluronan size and concentration: Effect on key biophysical and biochemical features. Int. J. Biol. Macromol. 2024, 282, 137125. [Google Scholar] [CrossRef]
- Kharaziha, M.; Scheibel, T.; Salehi, S. Multifunctional naturally derived bioadhesives: From strategic molecular design toward advanced biomedical applications. Prog. Polym. Sci. 2024, 150, 101792. [Google Scholar] [CrossRef]
- Almuqbil, R.M.; Aldhubiab, B. Bioadhesive Nanoparticles in Topical Drug Delivery: Advances, Applications, and Potential for Skin Disorder Treatments. Pharmaceutics 2025, 17, 229. [Google Scholar] [CrossRef] [PubMed]
- Al-Smadi, K.; Ali, M.; Zhu, J.; Abdoh, A.; Phan, K.; Mohammed, Y. Advances in Characterization of Transdermal and Topical Products using Texture Analyzer Systems. AAPS PharmSciTech 2025, 26, 157. [Google Scholar] [CrossRef] [PubMed]
- Kriplani, P.; Guarve, K.; Baghael, U.S. Arnica montana L.—A plant of healing: Review. J. Pharm. Pharmacol. 2017, 69, 925–945. [Google Scholar] [CrossRef]
- Ak, G.; Zengin, G.; Sinan, K.I.; Mahomoodally, M.F.; Picot-Allain, M.C.; Cakır, O.; Bensari, S.; Yılmaz, M.A.; Gallo, M.; Montesano, D. A Comparative Bio-Evaluation and Chemical Profiles of Calendula officinalis L. Extracts Prepared via Different Extraction Techniques. Appl. Sci. 2020, 10, 5920. [Google Scholar] [CrossRef]
- Owczarek-Januszkiewicz, A.; Kicel, A.; Olszewska, M.A. Aesculus hippocastanum in the pharmaceutical industry and beyond—Phytochemistry, bioactivity, present application, and future perspectives. Ind. Crops Prod. 2023, 193, 116187. [Google Scholar] [CrossRef]
- Andrzejczuk, S.; Sozoniuk, M.; Sugier, D. Preliminary Assessment of Arnica montana L. Extract: Antimicrobial Activity Against Acinetobacter baumannii and Biofilm-Related Gene Expression Profiling. Genes 2025, 16, 1473. [Google Scholar] [CrossRef]
- Žitek, T.; Postružnik, V.; Knez, Ž.; Golle, A.; Dariš, B.; Knez Marevci, M. Arnica montana L. Supercritical Extraction Optimization for Antibiotic and Anticancer Activity. Front. Bioeng. Biotechnol. 2022, 10, 897185. [Google Scholar] [CrossRef] [PubMed]
- Ejiohuo, O.; Folami, S.; Maigoro, A.Y. Calendula in modern medicine: Advancements in wound healing and drug delivery applications. Eur. J. Med. Chem. Rep. 2024, 12, 100199. [Google Scholar] [CrossRef]
- Melnyk, N.; Skowrońska, W.; Popowski, D.; Piwowarski, J.; Granica, S. From Tradition to Mechanism: Anti-inflammatory and Microbiota-Modulating Effects of Calendula officinalis and Matricaria recutita Extracts on the Skin. Prospect. Pharm. Sci. 2025, early access. [Google Scholar] [CrossRef]
- Dridi, A.; Reis, F.S.; Pires, T.C.S.P.; Calhelha, R.C.; Pereira, C.; Zaghdoudi, K.; Ferreira, I.C.F.R.; Barros, L.; Barreira, J.C.M. Aesculus hippocastanum L.: A Simple Ornamental Plant or a Source of Compelling Molecules for Industry? Separations 2023, 10, 160. [Google Scholar] [CrossRef]
- Roy, A.; Geetha, R.V.; Lakshmi, T. Evaluation of the Antimycotic Activity of Aqueous and Ethanolic Extracts of Aesculus hippocastanum—An In Vitro Study. Int. J. Drug Dev. Res. 2011, 3, 335–338. [Google Scholar]
- Dahiya, P.; Purkayastha, S. Phytochemical Screening and Antimicrobial Activity of Some Medicinal Plants Against Multi-drug Resistant Bacteria from Clinical Isolates. Indian J. Pharm. Sci. 2012, 74, 443–450. [Google Scholar] [CrossRef] [PubMed]







| Raw Material | Extraction Yield (%) |
|---|---|
| Arnica montana | 49.02 |
| Calendula officinalis | 44.25 |
| Aesculus hippocastanum | 42.38 |
| Samples | L* (D65) | a* (D65) | b* (D65) | C* | h* | ∆E* |
|---|---|---|---|---|---|---|
| H0 | 97.00 ± 0.55 a | 0.06 ± 0.03 f | 0.97 ± 0.05 g | 0.97 ± 0.05 g | 1.51 ± 0.03 c | - |
| AM I | 57.30 ± 0.02 e | −0.66 ± 0.01 g | 28.35 ± 0.04 d | 28.36 ± 0.04 c | 178.45 ± 0.00 a | 48.23 ± 0.04 |
| AM II | 45.26 ± 0.06 f | 2.71 ± 0.01 b | 28.83 ± 0.06 c | 16.77 ± 0.02 f | 1.55 ± 0.00 b | 34.99 ± 0.01 |
| CO I | 67.45 ± 0.01 b | 0.80 ± 0.01 d | 29.67 ± 0.05 b | 29.68 ± 0.05 b | 1.54 ± 0.00 b | 41.20 ± 0.03 |
| CO II | 65.03 ± 0.45 d | 3.50 ± 0.02 a | 34.79 ± 0.19 a | 34.97 ± 0.19 a | 1.47 ± 0.00 e | 46.67 ± 0.30 |
| AH I | 65.78 ± 0.02 c | 0.41 ± 0.01 e | 16.67 ± 0.32 f | 16.77 ± 0.02 e | 1.55 ± 0.00 b | 18.31 ± 0.02 |
| AH II | 67.56 ± 0.03 b | 1.58 ± 0.01 c | 19.45 ± 0.07 e | 19.52 ± 0.07 d | 1.49 ± 0.00 d | 15.66 ± 0.07 |
| H0 | AM I | AM II | CO I | CO II | AH I | AH II | |||||
 | |||||||||||
| Preparation | Gram-Positive | Gram-Negative | Yeasts |
|---|---|---|---|
| Arnica (I) | 15.50 (6.9) | 3.31 (4.6) | 0.50 (1.7) |
| Arnica (II) | 21.81 (10.9) | 8.46 (5.3) | 5.25 (9.8) |
| Calendula (I) | 1.53 (3.7) | 0.69 (2.5) | 0.50 (1.7) |
| Calendula (II) | 2.38 (5.2) | 0 (0) | 5.17 (9.3) |
| Chestnut (I) | 6.50 (7.2) | 0 (0) | 6.50 (8.9) |
| Chestnut (II) | 10.50 (8.3) | 0.77 (2.8) | 11.08 (10.7) |
| Control (H0) | 0 (0) | 0 (0) | 0 (0) |
| Stye eye lipogel | 17.41 (20.2) | 0 (0) | 4.17 (9.9) |
| Surface disinfectant | 36.00 (16.3) | 15.38 (7.2) | 24.00 (19.8) |
| Skin disinfectant | 44.13 (10.0) | 23.77 (3.8) | 46.75 (15.3) |
| Hydrogen peroxide | 53.75 (7.29) | 42.77 (12.9) | 43.25 (19.6) |
| Microbial Group (n) | Parameter | AI | AII | Control (H0) |
|---|---|---|---|---|
| Gram-positives (n = 76) | mean | 11.96 | 15.30 | 0 |
| median | 12 | 14 | 0 | |
| SD | 7.22 | 10.35 | 0 | |
| Gram-negatives (n = 19) | mean | 4.84 | 8.74 | 0 |
| median | 0 | 10 | 0 | |
| SD | 5.52 | 5.81 | 0 | |
| Candida yeasts (n = 15) | mean | 2.07 | 5.93 | 0 |
| median | 0 | 0 | 0 | |
| SD | 4.54 | 9.27 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Lenart-Boroń, A.; Ratajewicz, A.; Czernecka-Borchowiec, N.; Kopacz, A.; Schejbal, Z.; Khachatryan, G.; Khachatryan, K.; Krystyjan, M.; Bulanda, K.; Stankiewicz, K. Hyaluronic-Acid Nanocapsules with Plant Extracts: Characterization and Antimicrobial Activity Against Skin Microbiota. Materials 2026, 19, 1288. https://doi.org/10.3390/ma19071288
Lenart-Boroń A, Ratajewicz A, Czernecka-Borchowiec N, Kopacz A, Schejbal Z, Khachatryan G, Khachatryan K, Krystyjan M, Bulanda K, Stankiewicz K. Hyaluronic-Acid Nanocapsules with Plant Extracts: Characterization and Antimicrobial Activity Against Skin Microbiota. Materials. 2026; 19(7):1288. https://doi.org/10.3390/ma19071288
Chicago/Turabian StyleLenart-Boroń, Anna, Anna Ratajewicz, Natalia Czernecka-Borchowiec, Anna Kopacz, Zofia Schejbal, Gohar Khachatryan, Karen Khachatryan, Magdalena Krystyjan, Klaudia Bulanda, and Klaudia Stankiewicz. 2026. "Hyaluronic-Acid Nanocapsules with Plant Extracts: Characterization and Antimicrobial Activity Against Skin Microbiota" Materials 19, no. 7: 1288. https://doi.org/10.3390/ma19071288
APA StyleLenart-Boroń, A., Ratajewicz, A., Czernecka-Borchowiec, N., Kopacz, A., Schejbal, Z., Khachatryan, G., Khachatryan, K., Krystyjan, M., Bulanda, K., & Stankiewicz, K. (2026). Hyaluronic-Acid Nanocapsules with Plant Extracts: Characterization and Antimicrobial Activity Against Skin Microbiota. Materials, 19(7), 1288. https://doi.org/10.3390/ma19071288










