Thermomechanical Treatment-Enabled Short-Circuit Diffusion Enhances Molten-Carbonate Corrosion Resistance of an Alumina-Forming Austenitic Alloy
Abstract
1. Introduction
2. Experimental and Methods
2.1. Material Preparation
2.2. Molten Salt Corrosion Tests
2.3. Corrosion Rate Calculation
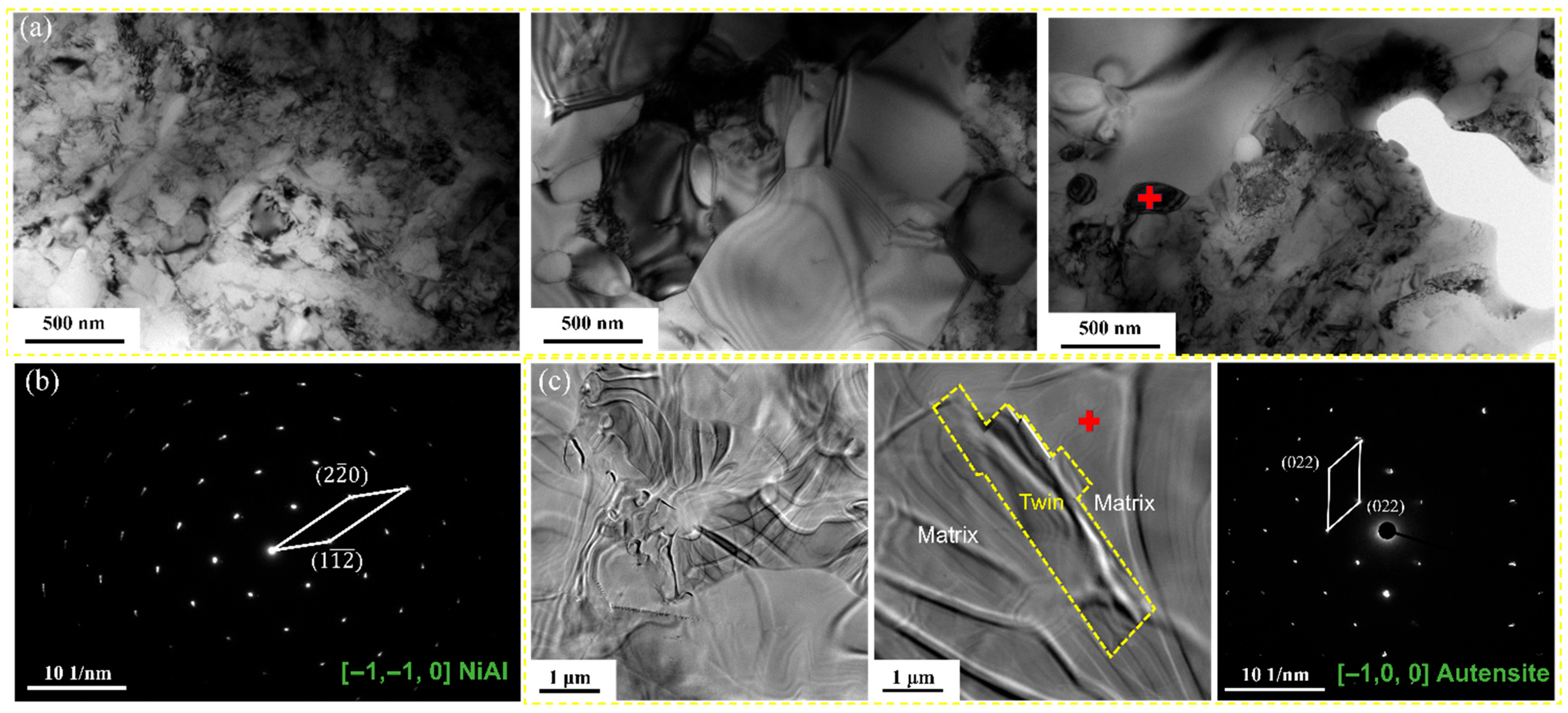
2.4. Microstructural Characterization
3. Results
3.1. Corrosion Kinetics
3.2. Phase Identification of Corrosion Products
3.3. Surface Morphology Evolution
3.4. Cross-Sectional Microstructure and Element Distribution
4. Discussion
4.1. Microstructural Evolution Induced by Thermomechanical Treatment
4.2. The Decisive Role of Defects on Al Diffusion Kinetics
4.3. Thermodynamic Analysis of Corrosion Reactions
4.4. Failure Model and Kinetic Implications
4.5. Long-Term Stability and Limitation Analysis
5. Conclusions
- Efficacy of defect engineering: A microstructure-oriented TMT strategy was successfully demonstrated. The specimen annealed at 800 °C exhibited superior corrosion resistance, with a low steady-state corrosion rate of ~62 μm/yr, significantly outperforming the coarse-grained 1000 °C counterpart. This confirms that TMT is a viable pathway to enhance the applicability of AFA alloys in aggressive molten salts without relying on expensive alloying additions.
- Mechanism of kinetic acceleration: The superior performance of the 800 °C specimen is governed by short-circuit diffusion. While classical GBE theory favors low-energy CSL boundaries for corrosion resistance, this study reveals that, for diffusion-limited AFA systems in molten carbonates, a network of dislocation pipes and subgrain boundaries increases the effective diffusivity of Al. This kinetic advantage lowers the critical solute concentration required for passivation, enabling the rapid establishment of a protective scale even with a marginal bulk Al content.
- Thermodynamic and mechanical stability of scales: The accelerated Al supply in the 800 °C specimen promotes the formation of a continuous, thermodynamically stable LiAlO2-rich inner layer. This layer acts as a highly protective barrier that effectively suppresses the basic fluxing of chromium ( formation). In contrast, the 1000 °C specimen forms a porous LiFeO2/NiO scale, which fails due to high PBR-induced stresses and internal stress accumulation, leading to breakaway oxidation.
- Long-term stability strategy: The study identifies a strategic trade-off: while enhanced diffusion accelerates scale formation, it also risks faster depletion of the Al reservoir. However, in the 800 °C specimen, dispersed NiAl precipitates act as localized Al buffers, mitigating the risk of premature Al depletion. This defect-assisted passivation strategy provides a metallurgical guideline for designing durable AFA alloys for next-generation CSP applications operating in high-temperature molten salts.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Deo Prasad, M.D.; Kuru, A.; Oldfield, P.; Ding, L.; Noller, C.; He, B. Race to Net Zero Carbon: A Climate Emergency Guide. Science 2019, 326, 434–437. [Google Scholar]
- Choi, Y.; Rayl, J.; Tammineedi, C.; Brownson, J.R. PV Analyst: Coupling ArcGIS with TRNSYS to assess distributed photovoltaic potential in urban areas. Sol. Energy 2011, 85, 2924–2939. [Google Scholar] [CrossRef]
- Javanshir, A.; Sarunac, N.; Razzaghpanah, Z. Thermodynamic analysis and optimization of single and combined power cycles for concentrated solar power applications. Energy 2018, 157, 65–75. [Google Scholar] [CrossRef]
- Padilla, R.V.; Too, Y.C.S.; Benito, R.; Stein, W. Exergetic analysis of supercritical CO2 Brayton cycles integrated with solar central receivers. Appl. Energy 2015, 148, 348–365. [Google Scholar] [CrossRef]
- Zhang, Q.; Cao, D.; Jiang, K.; Du, X.; Xu, E. Heat transport characteristics of a peak shaving solar power tower station. Renew. Energy 2020, 156, 493–508. [Google Scholar] [CrossRef]
- Mehos, M.; Turchi, C.; Vidal, J.; Wagner, M.; Ma, Z.; Ho, C.; Kruizenga, A. Concentrating Solar Power Gen3 Demonstration Roadmap; Technical Report NREL/TP-5500-67464; National Renewable Energy Lab.: Golden, CO, USA, 2017. [Google Scholar]
- Encinas-Sánchez, V.; de Miguel, M.T.; García-Martín, G.; Lasanta, M.I.; Pérez, F.J. Corrosion resistance of Cr/Ni alloy to a molten carbonate salt at various temperatures for the next generation high-temperature CSP plants. Sol. Energy 2018, 171, 286–292. [Google Scholar] [CrossRef]
- Gao, L.; Han, F.; Chen, S.; Ye, X.X.; Leng, B. High-temperature corrosion behavior of Inconel 617 with Ni-claddings in molten FLiNaK salt. Mater. Corros. 2022, 73, 486–496. [Google Scholar] [CrossRef]
- Fernández, A.G.; Cabeza, L.F. Corrosion monitoring and mitigation techniques on advanced thermal energy storage materials for CSP plants. Sol. Energy Mater. Sol. Cells 2019, 192, 179–187. [Google Scholar] [CrossRef]
- Fernández, G.; Pineda, F.; Walczak, M.; Cabeza, L.F. Corrosion evaluation of alumina-forming alloys in carbonate molten salt for CSP plants. Renew. Energy 2019, 140, 227–233. [Google Scholar] [CrossRef]
- Li, C.X.; Bell, T. Corrosion properties of plasma nitrided AISI 410 martensitic stainless steel in 3.5% NaCl and 1% HCl aqueous solutions. Corros. Sci. 2006, 48, 2036–2049. [Google Scholar] [CrossRef]
- Xu, R.; Zhang, C.; Wang, G.; Wu, Y.; Lu, Y. Molten salt stress corrosion of metal alloys—An updated review. Sol. Energy Mater. Sol. Cells 2025, 282, 113379. [Google Scholar] [CrossRef]
- Pan, Y.; Sun, B.; Chen, H.; Liu, Z.; Dai, W.; Yang, X.; Li, X. Stress corrosion cracking behavior and mechanism of 2205 duplex stainless steel under applied polarization potentials. Corros. Sci. 2024, 231, 111978. [Google Scholar] [CrossRef]
- De Miguel, M.T.; Lasanta, M.I.; García-Martín, G.; Díaz, R.; Pérez, F.J. Temperature effect and alloying elements impact on the corrosion behaviour of the alloys exposed to molten carbonate environments for CSP application. Corros. Sci. 2022, 201, 110274. [Google Scholar] [CrossRef]
- Hamdy, E.; Strach, M.; Olovsjö, J.N.; Geers, C. Differentiation in corrosion performance of alumina forming alloys in alkali carbonate melts. Corros. Sci. 2021, 192, 109857. [Google Scholar] [CrossRef]
- Rapp, R.A. Chemistry and electrochemistry of the hot corrosion of metals. Corrosion 1986, 42, 568–577. [Google Scholar] [CrossRef]
- Spiegel, M. Salt melt induced corrosion of metallic materials in waste incineration plants. Mater. Corros. 1999, 50, 373–393. [Google Scholar] [CrossRef]
- Gomez-Vidal, J.C.; Tirawat, R. Corrosion of alloys in a chloride molten salt (NaCl-LiCl) for solar thermal technologies. Sol. Energy Mater. Sol. Cells 2016, 157, 234–244. [Google Scholar]
- Maric, M.; Muránsky, O.; Karatchevtseva, I.; Ungár, T.; Hester, J.; Studer, A.; Hill, M.R. The effect of cold-rolling on the microstructure and corrosion behaviour of 316L alloy in FLiNaK molten salt. Corros. Sci. 2018, 142, 133–144. [Google Scholar] [CrossRef]
- Sarvghad, M.; Chenu, T.; Will, G. Comparative interaction of cold-worked versus annealed inconel 601 with molten carbonate salt at 450 °C. Corros. Sci. 2017, 116, 88–97. [Google Scholar] [CrossRef]
- Sarvghad, M.; Muránsky, O.; Steinberg, T.A.; Hester, J.; Hill, M.R.; Will, G. On the effect of cold-rolling on the corrosion of SS316L alloy in a molten carbonate salt. Sol. Energy Mater. Sol. Cells 2019, 202, 110136. [Google Scholar]
- ASTM G1-03; Standard Practice for Preparing, Cleaning, and Evaluation Corrosion Test Specimens. ASTM International: West Conshohocken, PA, USA, 2003.
- Birks, N.; Meier, G.H.; Pettit, F.S. Introduction to the High Temperature Oxidation of Metals, 2nd ed.; Cambridge University Press: Cambridge, UK, 2006. [Google Scholar]
- Vossen, J.P.T.; Janssen, A.H.H.; De Wit, J.H.W. Corrosion behavior of nickel-iron alloys in molten carbonate. J. Electrochem. Soc. 1996, 143, 58. [Google Scholar] [CrossRef]
- Cahn, J.W. Nucleation on dislocations. Acta Metall. 1957, 5, 169–172. [Google Scholar] [CrossRef]
- Qiao, Y.; Wang, J.; Zhang, Z.; Quan, X.; Liu, J.; Fang, X.; Han, P. Improved Oxidation Resistance of a New Aluminum-Containing Austenitic Stainless Steel at 800 °C in Air. Oxid. Met. 2017, 88, 301–314. [Google Scholar] [CrossRef]
- Teng, Z.K.; Miller, M.K.; Ghosh, G.; Liu, C.T.; Huang, S.; Russell, K.F.; Liaw, P.K. Characterization of nanoscale NiAl-type precipitates in a ferritic steel by electron microscopy and atom probe tomography. Scr. Mater. 2010, 63, 61–64. [Google Scholar] [CrossRef]
- Guo, M.; Cao, G.; Pan, H.; Guo, J.; Chen, C.; Zhang, B.; Hu, J. Recent progress in synthesis of MAX phases and oxidation & corrosion mechanism: A review. Mater. Res. Lett. 2024, 12, 765–796. [Google Scholar] [CrossRef]
- Pint, B.A.; Yamamoto, Y.; Brady, M.P. Effect of Water Vapor on Oxidation Performance of Alumina-Forming Austenitic Stainless Steels. Oxid. Met. 2010, 73, 237–246. [Google Scholar]
- Grabke, H.J. Oxidation of NiAl and FeAl. ISIJ Int. 1989, 29, 529–538. [Google Scholar] [CrossRef]
- Watanabe, T. An approach to grain boundary design for strong and ductile polycrystals. Res. Mech. 1984, 11, 47–84. [Google Scholar]
- Dai, Q.; Ye, X.X.; Ai, H.; Chen, S.; Jiang, L.; Liang, J.; Yu, K.; Leng, B.; Li, Z.; Zhou, X. Corrosion of Incoloy 800H alloys with nickel cladding in FLiNaK salts at 850 °C. Corros. Sci. 2018, 133, 349–357. [Google Scholar] [CrossRef]
- Kaur, I.; Gust, W. Fundamentals of Grain and Interphase Boundary Diffusion; Ziegler Press: Stuttgart, Germany, 1989. [Google Scholar]
- Hart, E.W. On the role of dislocations in bulk diffusion. Acta Metall. 1957, 5, 597. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Brady, M.P.; Lu, Z.P.; Maziasz, P.J.; Liu, C.T.; Pint, B.A.; Payzant, E.A. Creep-resistant, Al2O3-forming austenitic stainless steels. Science 2007, 316, 433–436. [Google Scholar]
- Pérez, F.J.; Duday, D.; Hierro, M.P.; Gómez, C.; Agüero, A.; Garcı, M.C.; Muela, R.; Pascual, A.S.; Martinez, L. Hot corrosion study of coated separator plates of molten carbonate fuel cells by slurry aluminides. Surf. Coat. Technol. 2002, 161, 293–301. [Google Scholar]
- Audigié, P.; Encinas-Sánchez, V.; Juez-Lorenzo, M.; Rodríguez, S.; Gutiérrez, M.; Pérez, F.J.; Agüero, A. High temperature molten salt corrosion behavior of aluminide and nickel-aluminide coatings for heat storage in concentrated solar power plants. Surf. Coat. Technol. 2018, 349, 1148–1157. [Google Scholar]
- Pilling, N.B.; Bedworth, R.E. The Oxidation of Metals at High Temperatures. J. Inst. Met. 1923, 29, 529–582. [Google Scholar]
- Wagner, C. Theoretical analysis of the diffusion processes determining the oxidation rate of alloys. J. Electrochem. Soc. 1952, 99, 369. [Google Scholar] [CrossRef]
- Peng, X.; Yan, J.; Zhou, Y.; Wang, F. Effect of grain refinement on the resistance of 304 stainless steel to breakaway oxidation in wet air. Acta Mater. 2005, 53, 5079–5088. [Google Scholar] [CrossRef]
- Pint, B.A.; More, K.L.; Tortorelli, P.F.; Porter, W.D.; Wright, I.G. Optimizing the imperfect oxidation performance of iron aluminides. Mater. Sci. Forum 2001, 369–372, 411–418. [Google Scholar] [CrossRef]
- Haynes, J.A.; Pint, B.A.; Porter, W.D.; Wright, I.G. Comparison of thermal expansion and oxidation behavior of various high-temperature coating materials and superalloys. Mater. High Temp. 2004, 21, 87–94. [Google Scholar] [CrossRef]






| Elements | C | Al | Cr | Ni | Ti | Fe |
|---|---|---|---|---|---|---|
| Content | 0.028 | 3.33 | 12.49 | 31.41 | 0.1 | Bal. |
| Location | O | Al | Fe | Ni | Cr |
|---|---|---|---|---|---|
| Point 1 | 58.3 | 1.9 | 36.7 | 2.3 | 0.5 |
| Point 2 | 48.8 | 3.2 | 24.5 | 15.4 | 7.9 |
| Point 3 | 49.1 | 1.4 | 49.1 | 0.4 | 0.1 |
| Point 4 | 57.2 | 0.7 | 39.6 | 0.9 | 0.2 |
| Point 5 | 47.5 | 1.4 | 46.2 | 4.7 | 0.0 |
| Point 6 | 56.9 | 2.9 | 33.9 | 3.9 | 2.2 |
| Point 7 | 52.7 | 1.7 | 41.6 | 3.8 | 0.0 |
| Location | O | Al | Fe | Ni | Cr |
|---|---|---|---|---|---|
| Point 1 | 50.3 | 1.6 | 42.1 | 2.5 | 2.8 |
| Point 2 | 42.7 | 1.2 | 49.7 | 1.8 | 4.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Jiang, H.; Yu, H.; Zheng, Y.; Zhan, F.; La, P. Thermomechanical Treatment-Enabled Short-Circuit Diffusion Enhances Molten-Carbonate Corrosion Resistance of an Alumina-Forming Austenitic Alloy. Materials 2026, 19, 1206. https://doi.org/10.3390/ma19061206
Jiang H, Yu H, Zheng Y, Zhan F, La P. Thermomechanical Treatment-Enabled Short-Circuit Diffusion Enhances Molten-Carbonate Corrosion Resistance of an Alumina-Forming Austenitic Alloy. Materials. 2026; 19(6):1206. https://doi.org/10.3390/ma19061206
Chicago/Turabian StyleJiang, Haocheng, Haicun Yu, Yuehong Zheng, Faqi Zhan, and Peiqing La. 2026. "Thermomechanical Treatment-Enabled Short-Circuit Diffusion Enhances Molten-Carbonate Corrosion Resistance of an Alumina-Forming Austenitic Alloy" Materials 19, no. 6: 1206. https://doi.org/10.3390/ma19061206
APA StyleJiang, H., Yu, H., Zheng, Y., Zhan, F., & La, P. (2026). Thermomechanical Treatment-Enabled Short-Circuit Diffusion Enhances Molten-Carbonate Corrosion Resistance of an Alumina-Forming Austenitic Alloy. Materials, 19(6), 1206. https://doi.org/10.3390/ma19061206






