A Reusable SERS Substrate with Internal Standard for the Detection of N-Butylamine Gas
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of SiO2 [26]
2.3. Synthesis of SiO2@Fe2O3 [27]
2.4. Synthesis of SiO2@Fe2O3-Ag [28]
2.5. Synthesis of SiO2@Fe2O3-Ag@ZIF-8 [29]
2.6. Synthesis of Si/SiO2@Fe2O3-Ag(ET)@ZIF-8 Substrate
2.7. Characterization
2.8. SERS Measurements
3. Results and Discussion
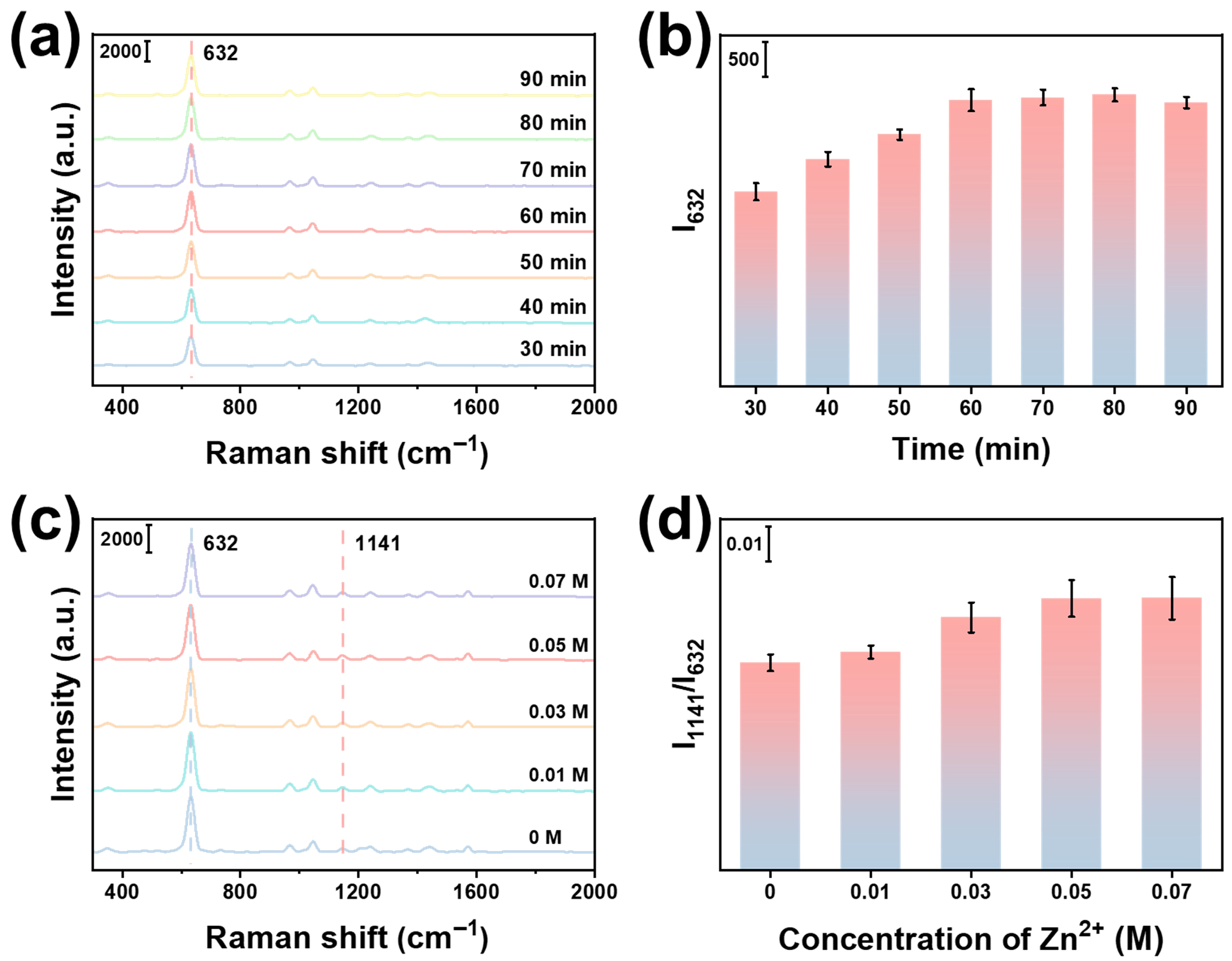
3.1. Fabrication and Characterization of SiO2@Fe2O3-Ag@ZIF-8
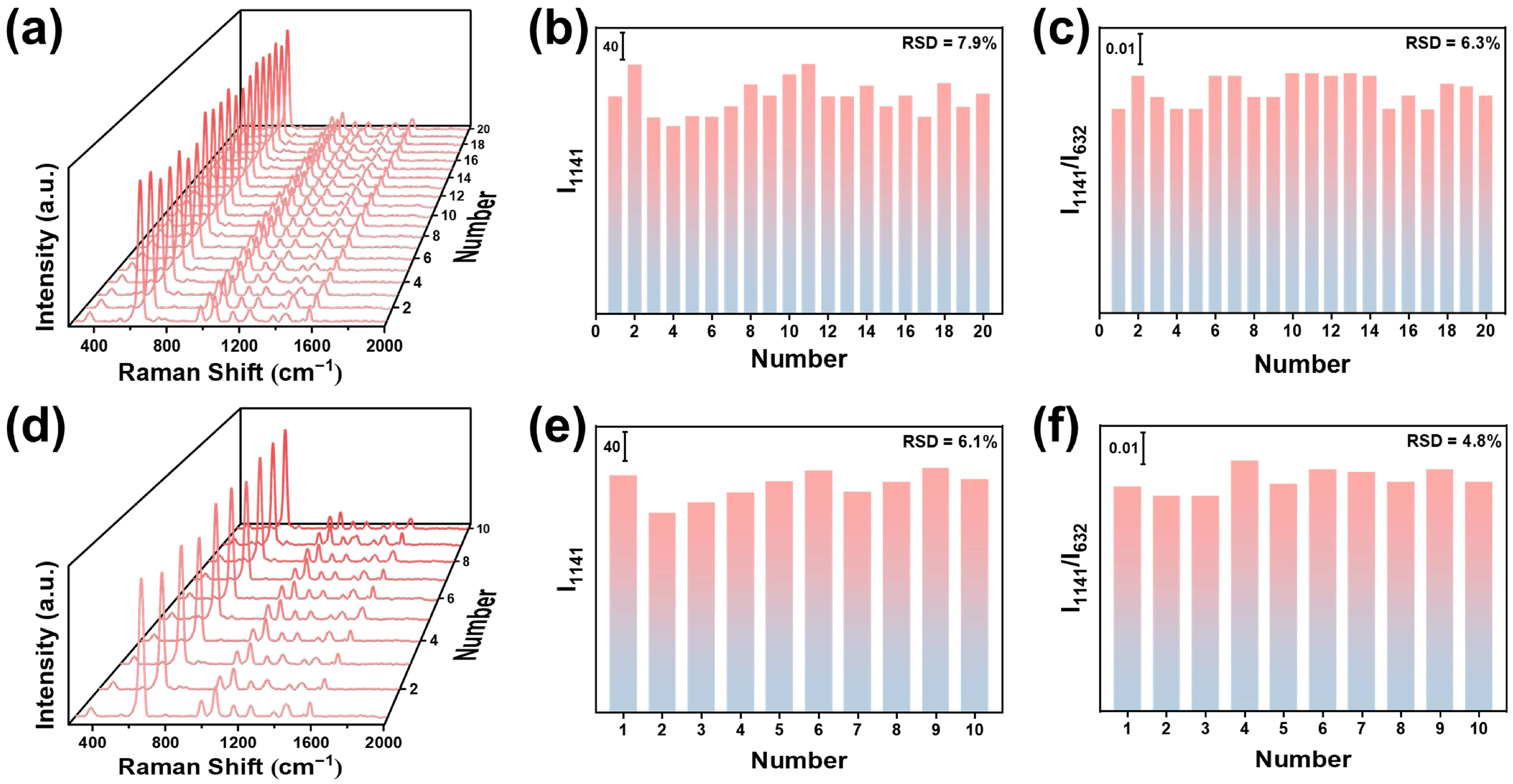
3.2. Sensing Performance of Si/SiO2@Fe2O3-Ag(ET)@ZIF-8
3.3. Photocatalytic Self-Cleaning Capability of Si/SiO2@Fe2O3-Ag(ET)@ZIF-8
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Khatib, M.; Haick, H. Sensors for volatile organic compounds. ACS Nano 2022, 16, 7080–7115. [Google Scholar] [CrossRef]
- Chen, S.; Duan, X.; Liu, C.; Liu, S.; Li, P.; Su, D.; Sun, X.; Guo, Y.; Chen, W.; Wang, Z. La-Ce-MOF nanocomposite coated quartz crystal microbalance gas sensor for the detection of amine gases and formaldehyde. J. Hazard. Mater. 2024, 467, 133672. [Google Scholar] [CrossRef] [PubMed]
- Andre, R.S.; Mercante, L.A.; Facure, M.H.M.; Sanfelice, R.C.; Fugikawa-Santos, L.; Swager, T.M.; Correa, D.S. Recent progress in amine gas sensors for food quality monitoring: Novel architectures for sensing materials and systems. ACS Sens. 2022, 7, 2104–2131. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Gao, R.; Wu, Y.; Zhang, X.; Cheng, X.; Gao, S.; Xu, Y.; Huo, L. Novel in-situ deposited V2O5 nanorods array film sensor with enhanced gas sensing performance to n-butylamine. Chem. Eng. J. 2023, 459, 141505. [Google Scholar] [CrossRef]
- Attinà, A.; Oliveri, I.P.; Di Bella, S. Detection of volatile primary aliphatic amines: Highly selective and sensitive vapoluminescent sensing of n-butylamine. Sens. Actuators B Chem. 2024, 419, 136414. [Google Scholar] [CrossRef]
- Li, C.; Jiang, X.; Hou, X. Dielectric barrier discharge molecular emission spectrometer as gas chromatographic detector for amines. Microchem. J. 2015, 119, 108–113. [Google Scholar] [CrossRef]
- Akbarinejad, A.; Ghoorchian, A.; Kamalabadi, M.; Alizadeh, N. Electrospun soluble conductive polypyrrole nanoparticles for fabrication of highly selective n-butylamine gas sensor. Sens. Actuators B Chem. 2016, 236, 99–108. [Google Scholar] [CrossRef]
- Kaneti, Y.V.; Liu, M.; Zhang, X.; Bu, Y.; Yuan, Y.; Jiang, X.; Yu, A. Synthesis of platinum-decorated iron vanadate nanorods with excellent sensing performance toward n-butylamine. Sens. Actuators B Chem. 2016, 236, 173–183. [Google Scholar] [CrossRef]
- Xu, F.; Luo, Q.; Qian, J.; Lu, Q.; Xia, J. Developing versatile and highly selective chemosensor for amines detection based on bis-thiophene methane containing cyclopalladated compounds. Sens. Actuators B Chem. 2022, 359, 131561. [Google Scholar] [CrossRef]
- Rakow, N.A.; Sen, A.; Janzen, M.C.; Ponder, J.B.; Suslick, K.S. Molecular recognition and discrimination of amines with a colorimetric array. Angew. Chem. Int. Ed. 2005, 44, 4528–4532. [Google Scholar]
- Xue, P.; Xu, Q.; Gong, P.; Qian, C.; Ren, A.; Zhang, Y.; Lu, R. Fibrous film of a two-component organogel as a sensor to detect and discriminate organic amines. Chem. Commun. 2013, 49, 5838–5840. [Google Scholar] [CrossRef] [PubMed]
- Qu, C.; Fang, H.; Yu, F.; Chen, J.; Su, M.; Liu, H. Artificial nose of scalable plasmonic array gas sensor for multi-dimensional SERS recognition of volatile organic compounds. Chem. Eng. J. 2024, 482, 148773. [Google Scholar] [CrossRef]
- Jin, X.; Zhu, Q.; Feng, L.; Li, X.; Zhu, H.; Miao, H.; Zeng, Z.; Wang, Y.; Li, Y.; Wang, L.; et al. Light-trapping SERS substrate with regular bioinspired arrays for detecting trace dyes. ACS Appl. Mater. Interfaces 2021, 13, 11535–11542. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Li, D.; Feng, S.; Liu, X.; Guo, X.; Wen, Y.; Yang, H. Highly specific and sensitive SERS detection of putrescine using Au nanobowls@Cu-MOF embedded in a hydrogel nanoreactor. Small 2025, 21, 2408030. [Google Scholar] [CrossRef]
- Benhabib, M.; Kleinman, S.L.; Peterman, M.C. Quantification of amines in refinery process water via surface-enhanced Raman spectroscopy. Energy Fuels 2023, 37, 1881–1886. [Google Scholar] [CrossRef]
- Marega, C.; Maculan, J.; Andrea Rizzi, G.; Saini, R.; Cavaliere, E.; Gavioli, L.; Cattelan, M.; Giallongo, G.; Marigo, A.; Granozzi, G. Polyvinyl alcohol electrospun nanofibers containing Ag nanoparticles used as sensors for the detection of biogenic amines. Nanotechnology 2015, 26, 075501. [Google Scholar] [CrossRef]
- Wan, Y.; Li, J.; Jiang, G.; Qi, J.; Wang, B.; Ozaki, Y.; Pi, F. Anisotropic growth in three-dimensional plasmonic networks for ultrasensitive and versatile SERS platforms. Anal. Chem. 2025, 97, 18804–18814. [Google Scholar] [CrossRef]
- Li, A.; Qiao, X.; Liu, K.; Bai, W.; Wang, T. Hollow metal organic framework improves the sensitivity and anti-interference of the detection of exhaled volatile organic compounds. Adv. Funct. Mater. 2022, 32, 2202805. [Google Scholar] [CrossRef]
- Li, M.; He, X.; Wu, C.; Wang, L.; Zhang, X.; Gong, X.; Zeng, X.; Huang, Y. Deep learning enabled SERS identification of gaseous molecules on flexible plasmonic MOF nanowire films. ACS Sens. 2024, 9, 979–987. [Google Scholar]
- Gu, Z.; Xu, Q.; Wang, X.; Lin, X.; Duan, N.; Wang, Z.; Wu, S. Food freshness prediction platform utilizing deep learning-based multimodal sensor fusion of volatile organic compounds and moisture distribution. ACS Sens. 2025, 10, 3091–3100. [Google Scholar]
- Guo, H.; Li, Y.; Pi, F. Sensitive and reproducible gold nanostar@metal-organic framework-based SERS membranes for the online monitoring of the freshness of shrimps. Analyst 2023, 148, 2081–2091. [Google Scholar] [CrossRef]
- Kim, H.; Trinh, B.T.; Kim, K.H.; Moon, J.; Kang, H.; Jo, K.; Akter, R.; Jeong, J.; Lim, E.-K.; Jung, J.; et al. Au@ZIF-8 SERS paper for food spoilage detection. Biosens. Bioelectron. 2021, 179, 113063. [Google Scholar] [CrossRef]
- Fu, J.-H.; Zhong, Z.; Xie, D.; Guo, Y.-J.; Kong, D.-X.; Zhao, Z.-X.; Zhao, Z.-X.; Li, M. SERS-active MIL-100(Fe) sensory array for ultrasensitive and multiplex detection of VOCs. Angew. Chem. Int. Ed. 2020, 59, 20489–20498. [Google Scholar] [CrossRef]
- Li, X.; Liu, H.; Gu, C.; Zhang, J.; Jiang, T. PDMS/TiO2/Ag hybrid substrate with intrinsic signal and clean surface for recyclable and quantitative SERS sensing. Sens. Actuators B Chem. 2022, 351, 130886. [Google Scholar] [CrossRef]
- Hu, M.; Li, K.; Dang, X.; Yang, C.; Li, X.; Wang, Z.; Li, K.; Cao, L.; Hu, X.; Li, Y.; et al. Phase-tunable molybdenum boride ceramics as an emerging sensitive and reliable SERS platform in harsh environments. Small 2024, 20, 2308690. [Google Scholar] [CrossRef] [PubMed]
- Plumeré, N.; Ruff, A.; Speiser, B.; Feldmann, V.; Mayer, H.A. Stöber silica particles as basis for redox modifications: Particle shape, size, polydispersity, and porosity. J. Colloid Interface Sci. 2012, 368, 208–219. [Google Scholar] [CrossRef] [PubMed]
- Naghdi, S.; Rhee, K.Y.; Jaleh, B.; Park, S.J. Altering the structure and properties of iron oxide nanoparticles and graphene oxide/iron oxide composites by urea. Appl. Surf. Sci. 2016, 364, 686–693. [Google Scholar] [CrossRef]
- Chen, L.; Zhang, W.; Wang, J.; Li, X.; Li, Y.; Hu, X.; Zhao, L.; Wu, Y.; He, Y. High piezo/photocatalytic efficiency of Ag/Bi5O7I nanocomposite using mechanical and solar energy for N2 fixation and methyl orange degradation. Green Energy Environ. 2023, 8, 283–295. [Google Scholar] [CrossRef]
- Li, X.; Zhao, H.; Huang, J.; Li, Y.; Miao, H.; Shi, G.; Wong, P.K. A high-performance TiO2 protective layer derived from non-high vacuum technology for a Si-based photocathode to enhance photoelectrochemical water splitting. J. Mater. Chem. A 2024, 12, 16605–16616. [Google Scholar] [CrossRef]
- Ding, X.; Yang, K.-L. Liquid crystal based optical sensor for detection of vaporous butylamine in air. Sens. Actuators B Chem. 2012, 173, 607–613. [Google Scholar] [CrossRef]
- Adamska, E.; Niska, K.; Wcisło, A.; Grobelna, B. Characterization and cytotoxicity comparison of silver- and silica-based nanostructures. Materials 2021, 14, 4987. [Google Scholar] [CrossRef]
- Prasad, R.; Crouse, S.H.; Rousseau, R.W.; Grover, M.A. Quantifying dense multicomponent slurries with in-line ATR-FTIR and Raman spectroscopies: A hanford case study. Ind. Eng. Chem. Res. 2023, 62, 15962–15973. [Google Scholar] [CrossRef]
- Jubb, A.M.; Allen, H.C. Vibrational spectroscopic characterization of hematite, maghemite, and magnetite thin films produced by vapor deposition. ACS Appl. Mater. Interfaces 2010, 2, 2804–2812. [Google Scholar] [CrossRef]
- Kirik, N.; Krylov, A.; Boronin, A.; Koshcheev, S.; Solovyov, L.; Rabchevskii, E.; Shishkina, N.; Anshits, A. The relationship between the structural characteristics of α-Fe2O3 catalysts and their lattice oxygen reactivity regarding hydrogen. Materials 2023, 16, 4466. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, S.; Pandey, A.; Nikam, A.N.; Nannuri, S.H.; George, S.D.; Fayaz, S.M.A.; Vincent, A.P.; Mutalik, S. ZIF-8 nano confined protein-titanocene complex core-shell MOFs for efficient therapy of neuroblastoma: Optimization, molecular dynamics and toxicity studies. Int. J. Biol. Macromol. 2021, 178, 444–463. [Google Scholar] [CrossRef] [PubMed]
- Nagpal, R.; Sugihara, M.; Magariu, N.; Tjardts, T.; Meling-Lizarde, N.; Strunskus, T.; Ameri, T.; Ameloot, R.; Adelung, R.; Lupan, O. Humidity-tolerant selective sensing of hydrogen and n-butanol using ZIF-8 coated CuO:Al film. Mater. Chem. Front. 2025, 9, 3425–3442. [Google Scholar] [CrossRef]
- Yu, X.; Zhang, J.; Wang, X.; Ma, Q.; Gao, X.; Xia, H.; Lai, X.; Fan, S.; Zhao, T.-S. Fischer-Tropsch synthesis over methyl modified Fe2O3@SiO2 catalysts with low CO2 selectivity. Appl. Catal. B 2018, 232, 420–428. [Google Scholar] [CrossRef]
- Yavuz, C.; Erten-Ela, S. Solar light-responsive α-Fe2O3/CdS/g-C3N4 ternary photocatalyst for photocatalytic hydrogen production and photodegradation of methylene blue. J. Alloys Compd. 2022, 908, 164584. [Google Scholar] [CrossRef]
- Peng, Y.; Jiang, Y.; Wang, C.; Pang, K.; Li, X.; Zhou, X.; Wang, Z. Ag@Au@4-NPH@ZIF-8 based nucleophilic addition strategy for SERS detection of gaseous acetone. Chem. Eng. J. 2025, 522, 167880. [Google Scholar] [CrossRef]
- Sadjadi, S.; Malmir, M.; Heravi, M.M. Preparation of Ag-doped g-C3N4 nano sheet decorated magnetic γ-Fe2O3@SiO2 core-shell hollow spheres through a novel hydrothermal procedure: Investigation of the catalytic activity for A3, KA2 coupling reactions and [3 + 2] cycloaddition. Appl. Organomet. Chem. 2018, 32, e4413. [Google Scholar] [CrossRef]
- Hamad, S.; Bharati Moram, S.S.; Yendeti, B.; Podagatlapalli, G.K.; Nageswara Rao, S.V.S.; Pathak, A.P.; Mohiddon, M.A.; Soma, V.R. Femtosecond laser-induced, nanoparticle-embedded periodic surface structures on crystalline silicon for reproducible and multi-utility SERS platforms. ACS Omega 2018, 3, 18420–18432. [Google Scholar] [CrossRef]
- Li, S.; Li, D.; Zhang, Q.-Y.; Tang, X. Surface enhanced Raman scattering substrate with high-density hotspots fabricated by depositing Ag film on TiO2-catalyzed Ag nanoparticles. J. Alloys Compd. 2016, 689, 439–445. [Google Scholar] [CrossRef]
- Li, Y.; Liu, K.; Li, X.; Jin, X.; Lyu, J.; Xu, Y.; Zhu, H.; Shi, G. Optical SERS sensor with dandelion flower-like Ag/ZnFe2O4 nanotubes on the Si pyramids for detecting trace dyes. Sens. Actuators B Chem. 2025, 424, 136888. [Google Scholar] [CrossRef]
- Li, Y.; Song, D.; Liu, B.; Li, X.; Huang, J.; Zhu, H.; Shi, G. A molecularly imprinted SERS sensor with both high-accuracy quantitative analysis and high-stability reuse based on spatial separation of catalytic sites. J. Mater. Chem. A 2025, 13, 32731–32740. [Google Scholar] [CrossRef]
- Wang, Y.; Feng, L.; Zhu, H.; Miao, H.; Li, Y.; Liu, X.; Shi, G. Noncontact metal-spiropyran-metal nanostructured substrates with Ag and Au@SiO2 nanoparticles deposited in nanohole arrays for surface-enhanced fluorescence and trace detection of metal ions. ACS Appl. Nano Mater. 2021, 4, 3780–3789. [Google Scholar] [CrossRef]
- Wang, D.; Zhang, N.; Yang, T.; Zhang, Y.; Jing, X.; Zhou, Y.; Long, J.; Meng, L. Amino acids and doxorubicin as building blocks for metal ion-driven self-assembly of biodegradable polyprodrugs for tumor theranostics. Acta Biomater. 2022, 147, 245–257. [Google Scholar] [CrossRef]
- Gupta, S.; Singh, R.; Bhardwaj, S.; Kuzmin, A.; Thakkur, S.; Garg, S.; Rzhevskii, A.; Vaitla, J.; Baev, A.; Siddhanta, S.; et al. Dramatic enhancement of targeted subcellular Raman imaging via synergetic nanoscale integration of resonance and surface enhanced Raman scattering mechanisms. ACS Photonics 2025, 12, 5074–5086. [Google Scholar] [CrossRef]
- Miao, L.; Zhang, B.-L.; Song, W.-T.; Chen, J.; Shi, W.-J.; Wang, R.-H.; Liu, S.; Li, Y.-J.; Zhang, J.-J. Zinc sulfate open frameworks with nonconventional room-temperature phosphorescence for selective amine vapor detection. Inorg. Chem. 2025, 64, 7214–7223. [Google Scholar] [CrossRef]
- Rekha, S.M.; Ghuge, R.S.; Vikraman, H.K.; Kiran, M.S.R.N.; Sivalingam, Y.; Bhat, S.V. ZnO/CuI heterojunction UV-photovoltaic gas sensor for self-powered IoT-integrated n-butylamine VOC detection. Adv. Mater. Technol. 2025, 10, 70049. [Google Scholar] [CrossRef]
- Cheng, L.; Yuan, Z.; Wu, R.; Li, Z.; Zheng, J.; Song, J. A leapfrog improvement in n-butanol detection achieved through single atom catalysts Co-N-C. Chem. Eng. J. 2025, 519, 164855. [Google Scholar] [CrossRef]
- Jamalabadi, H.; Mani-Varnosfaderani, A.; Alizadeh, N. Detection of alkyl amine vapors using PPy-ZnO hybrid nanocomposite sensor array and artificial neural network. Sens. Actuators A Phys. 2018, 280, 228–237. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Xu, M.; Li, X.; Xie, L.; Wang, Q.; Shi, G. A Reusable SERS Substrate with Internal Standard for the Detection of N-Butylamine Gas. Materials 2026, 19, 1207. https://doi.org/10.3390/ma19061207
Xu M, Li X, Xie L, Wang Q, Shi G. A Reusable SERS Substrate with Internal Standard for the Detection of N-Butylamine Gas. Materials. 2026; 19(6):1207. https://doi.org/10.3390/ma19061207
Chicago/Turabian StyleXu, Mingyang, Xin Li, Lin Xie, Qin Wang, and Gang Shi. 2026. "A Reusable SERS Substrate with Internal Standard for the Detection of N-Butylamine Gas" Materials 19, no. 6: 1207. https://doi.org/10.3390/ma19061207
APA StyleXu, M., Li, X., Xie, L., Wang, Q., & Shi, G. (2026). A Reusable SERS Substrate with Internal Standard for the Detection of N-Butylamine Gas. Materials, 19(6), 1207. https://doi.org/10.3390/ma19061207






