Chemical Modification of Cellulose Fibers for Sustainable Food Packaging: Structure–Property–Sustainability Relationships
Abstract
1. Introduction
1.1. Fiber-Based Food Packaging in the Context of Sustainability
1.2. Why Chemical Modification of Cellulose Matters
2. Cellulose Fibers for Food Packaging: Structure and Constraints
2.1. Hierarchical Structure of Cellulose Fibers
2.2. Chemical and Physical Limitations in FCM Applications
2.3. Native vs. Modified Cellulose: Design Space
3. Chemical Modification Strategies for Cellulose Fibers
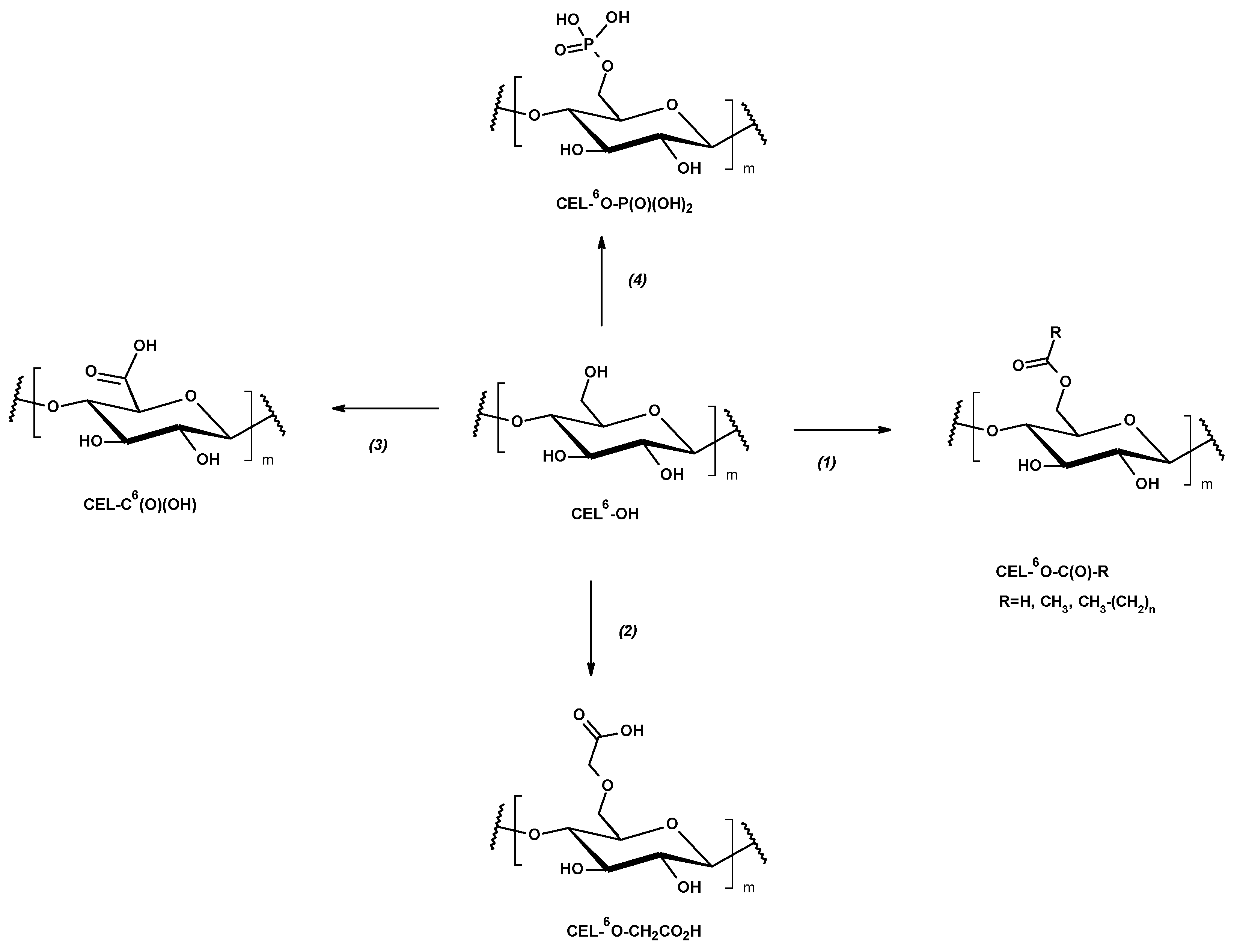
3.1. Esterification and Acylation of Cellulose Fibers
3.2. Etherification and Cationic/Anionic Functionalization
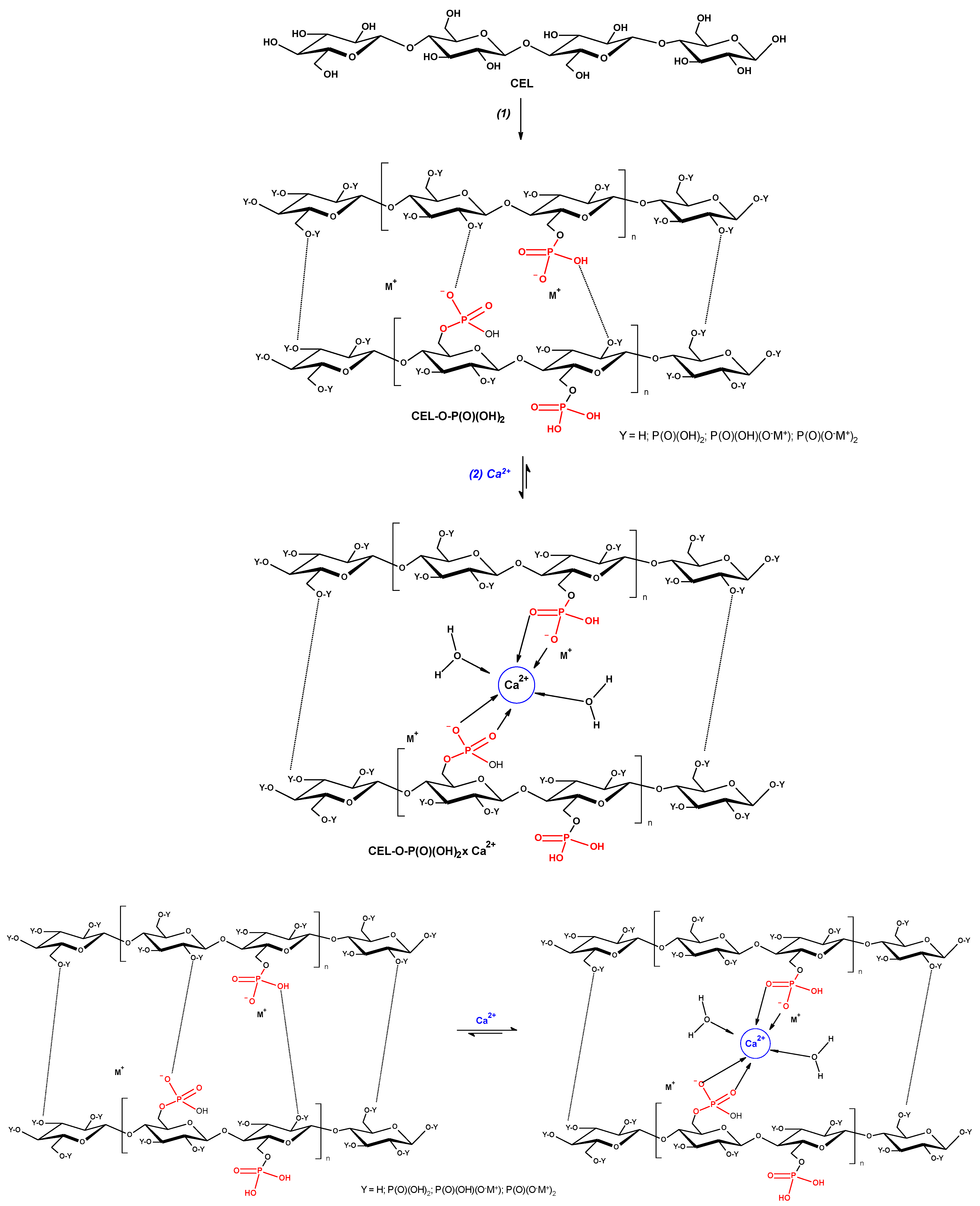
3.3. Phosphorylation and Ionic Complexation
3.4. Oxidative Functionalization
3.5. Degree of Substitution and Chemical Heterogeneity as Design Parameters
4. Structure–Property Relationships in Modified Cellulose Fibers
4.1. Effect of Chemical Structure on Fiber–Fiber Interactions
4.2. Barrier Properties (Water, Grease, Vapor)
4.3. Mechanical Performance of Modified Fiber Networks
4.4. Aging, Stability, and Process Robustness
5. Chemical Modification and Food Contact Safety
5.1. Migration and NIAS Formation
5.2. Impact of Modification on Toxicological Profiles
5.3. Safety-by-Design Approaches for Modified Cellulose
6. Sustainability Assessment of Chemically Modified Cellulose
6.1. Life Cycle Implications of Chemical Modification
6.2. Recyclability and End-of-Life Compatibility
6.3. Trade-Offs Between Performance and Circularity
- -
- Barrier functionality is achieved with minimal add-on (g/m−2) and/or selectively at the surface;
- -
- The modification does not require intensive purification steps or generate chemically burdened waste streams;
- -
7. Design Considerations for Modified Cellulose Packaging
7.1. From Chemical Modification to Quantifiable Performance Targets
7.2. Integrating Circularity and Safety as Design Constraints
7.3. Defining Practical Design Windows
- -
- Laboratory phase (g): Reaction optimization, determination of DS and MS using instrumental methods (e.g., FTIR).
- -
- Pilot phase (kg-t): Testing equipment adaptability, e.g., using composite grinding discs instead of basalt ones for improved nanostructure yield.
- -
8. Challenges and Future Directions
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AGU | Anhydro glucose unit |
| ATR-FTIR | Attenuated total reflectance Fourier-transform infrared spectroscopy |
| BfR | Bundesinstitut für Risikobewertung (German Federal Institute for Risk Assessment) |
| Ca2+ | Calcium ion |
| CEPI | Confederation of European Paper Industries |
| CMC | Carboxymethyl cellulose |
| CNFs | Cellulose nanofibrils |
| CNCs | Cellulose nanocrystals |
| Cobb60 | Water absorptiveness measured over 60 s (ISO 535) |
| DS | Degree of substitution |
| EoL | End of life |
| EU | European Union |
| FCM | Food contact material |
| FCC | Food contact chemical |
| GWP | Global warming potential |
| HEC | Hydroxyethyl cellulose |
| ISO | International Organization for Standardization |
| KIT | Grease resistance rating (TAPPI T559) |
| LCA | Life cycle assessment |
| MOAHs | Mineral oil aromatic hydrocarbons |
| MOSHs | Mineral oil saturated hydrocarbons |
| NIAS | Non-intentionally added substances |
| OML | Overall migration limit |
| OTR | Oxygen transmission rate |
| PFAS | Per- and polyfluoroalkyl substances |
| PLA | Polylactide |
| PPWR | Packaging and Packaging Waste Regulation |
| P(V) | Pentavalent phosphorus |
| RH | Relative humidity |
| SML | Specific migration limit |
| TEMPO | 2,2,6,6-tetramethylpiperidine-1-oxyl |
| TOCNFs | TEMPO-oxidized cellulose nanofibrils |
| WVTR | Water vapor transmission rate |
| XPS | X-ray photoelectron spectroscopy |
References
- European Parliament; Council of the European Union. Directive (EU) 2019/904 of 5 June 2019 on the Re-Duction of the Impact of Certain Plastic Products on the Environment. 2019. Available online: https://eur-lex.europa.eu/eli/dir/2019/904/oj/eng (accessed on 4 January 2026).
- Geueke, B.; Groh, K.; Muncke, J. Food Packaging in the Circular Economy: Overview of Chemical Safety Aspects for Commonly Used Materials. J. Clean. Prod. 2018, 193, 491–505. [Google Scholar] [CrossRef]
- Groh, K.J.; Geueke, B.; Martin, O.; Maffini, M.; Muncke, J. Overview of Intentionally Used Food Contact Chemicals and Their Hazards. Environ. Int. 2021, 150, 106225. [Google Scholar] [CrossRef] [PubMed]
- Mikkonen, K.S.; Tenkanen, M. Sustainable Food-Packaging Materials Based on Future Biorefinery Products: Xylans and Mannans. Trends Food Sci. Technol. 2012, 28, 90–102. [Google Scholar] [CrossRef]
- Siracusa, V.; Rocculi, P.; Romani, S.; Rosa, M.D. Biodegradable Polymers for Food Packaging: A Review. Trends Food Sci. Technol. 2008, 19, 634–643. [Google Scholar] [CrossRef]
- Naderi, A.; Lindström, T.; Sundström, J. Carboxymethylated Nanofibrillated Cellulose: Rheological Studies. Cellulose 2014, 21, 1561–1571. [Google Scholar] [CrossRef]
- Danish Veterinary and Food Administration. Ban on PFAS in Paper and Board Food Contact Materials (Denmark) (Factsheet; 1 July Implementation; Functional Barrier Exception). Available online: https://en.foedevarestyrelsen.dk/Media/638210239823191854/Faktaark FCM %28english%29.pdf (accessed on 4 January 2026).
- European Chemicals Agency (ECHA). PFAS Restriction Proposal Under REACH (Updated Proposal in-Formation; Submitted 13 January 2023). Available online: https://echa.europa.eu/-/echa-publishes-updated-pfas-restriction-proposal (accessed on 4 January 2026).
- Guidance on the Improved Collection and Sorting of Fibre-Based Packaging for Recycling Version 3. 2024. Available online: https://4evergreenforum.eu/wp-content/uploads/4evergreens-Guidance-on-the-Improved-Collection-and-Sorting-of-Fibre-based-Packaging-for-Recycling.pdf?ver=20260309150807 (accessed on 4 January 2026).
- Heise, K.; Koso, T.; King, A.W.T.; Nypelö, T.; Penttilä, P.; Tardy, B.L.; Beaumont, M. Spatioselective Surface Chemistry for the Production of Functional and Chemically Anisotropic Nanocellulose Colloids. J. Mater. Chem. A 2022, 10, 23413–23432. [Google Scholar] [CrossRef]
- McNamara, J.T.; Morgan, J.L.W.; Zimmer, J. A Molecular Description of Cellulose Biosynthesis. Annu. Rev. Biochem. 2015, 84, 895–921. [Google Scholar] [CrossRef] [PubMed]
- Semple, K.E.; Zhou, C.; Rojas, O.J.; Nkeuwa, W.N.; Dai, C. Moulded Pulp Fibers for Disposable Food Packaging: A State-of-the-Art Review. Food Packag. Shelf Life 2022, 33, 100908. [Google Scholar] [CrossRef]
- Hubbe, M.A.; Sjöstrand, B.; Lestelius, M.; Håkansson, H.; Swerin, A.; Henriksson, G. Swelling of Cellulosic Fibers in Aqueous Systems: A Review of Chemical and Mechanistic Factors. BioResources 2024, 19, 6859–6945. [Google Scholar] [CrossRef]
- Peltola, H.; Pääkkönen, E.; Jetsu, P.; Heinemann, S. Wood Based PLA and PP Composites: Effect of Fibre Type and Matrix Polymer on Fibre Morphology, Dispersion and Composite Properties. Compos. Part A Appl. Sci. Manuf. 2014, 61, 13–22. [Google Scholar] [CrossRef]
- Fahlén, J. The Cell Wall Ultrastructure of Wood Fibres: Effects of the Chemical Pulp Fibre Line; KTH Royal Institute of Technology: Stockholm, Sweden, 2005.
- Pitcher, M.L.; Koshani, R.; Sheikhi, A. Chemical Structure-Property Relationships in Nanocelluloses. J. Polym. Sci. 2024, 62, 9–31. [Google Scholar] [CrossRef]
- Wu, C.; Li, J.; Zhang, Y.Q.; Li, X.; Wang, S.Y.; Li, D.Q. Cellulose Dissolution, Modification, and the Derived Hydrogel: A Review. ChemSusChem 2023, 16, e202300518. [Google Scholar] [CrossRef]
- Sellman, F.A. Characterization and Utilization of Interactions in Wet and Dry Cellulose Nanofibrillar Networks; KTH Royal Institute of Technology: Stockholm, Sweden, 2024.
- Jones, J.H.; Briggs, L.J.; Carson, F.T. U. S. Department of Comme! On Physical Properties of Paper Effect of Humidity on Physical Properties of Paper. Available online: https://nvlpubs.nist.gov/nistpubs/Legacy/circ/nbscircular445.pdf (accessed on 11 January 2026).
- Berthold, L.; Niini, A.; Tanninen, P.; Leminen, V.; Majschak, J.-P. Role of Blank Moisturisation in Press Forming of Paperboard. BioResources 2024, 19, 2272–2285. [Google Scholar] [CrossRef]
- Kunam, P.K.; Ramakanth, D.; Akhila, K.; Gaikwad, K.K. Bio-Based Materials for Barrier Coatings on Paper Packaging. Biomass Convers. Biorefin. 2022, 14, 12637–12652. [Google Scholar] [CrossRef]
- Pignères, E.; Vercasson, A.; Gaucel, S.; Coffigniez, F.; Gontard, N.; Angellier-Coussy, H.; Guillard, V. How to Get Reliable Oxygen Transmission Rate Values for Coated Cardboards? Prog. Org. Coat. 2024, 186, 108048. [Google Scholar] [CrossRef]
- ISO 187:2022(En); Paper, Board and Pulps-Standard Atmosphere for Conditioning and Testing and Procedure for Monitoring the Atmosphere and Conditioning of Samples. ISO: Geneva, Switzerland, 2022. Available online: https://www.iso.org/obp/ui/#iso:std:iso:187:ed-3:v1:en (accessed on 4 January 2026).
- Water Vapor Transmission Rate of Paper and Paperboard at 23°C and 50% RH, Test Method T 448 Om-21. Available online: https://imisrise.tappi.org/TAPPI/Products/01/T/0104T448.aspx (accessed on 4 January 2026).
- OECD. PFASs and Alternatives in Food Packaging (Paper and Paperboard): Report on the Commercial Availability and Current Uses; OECD Series on Risk Management of Chemicals; OECD: Paris, France, 2020. [Google Scholar] [CrossRef]
- Lo Faro, E.; Bonofiglio, A.; Barbi, S.; Montorsi, M.; Fava, P. Polycaprolactone/Starch/Agar Coatings for Food-Packaging Paper: Statistical Correlation of the Formulations’ Effect on Diffusion, Grease Resistance, and Mechanical Properties. Polymers 2023, 15, 3921. [Google Scholar] [CrossRef] [PubMed]
- Sellman, F.A.; Benselfelt, T.; Larsson, P.T.; Wågberg, L. Hornification of Cellulose-Rich Materials - A Kinetically Trapped State. Carbohydr. Polym. 2023, 318, 121132. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Youngblood, J.P. Cellulose Nanofibril-Based Hybrid Coatings with Enhanced Moisture Barrier Properties. Mater. Adv. 2025, 6, 2833–2844. [Google Scholar] [CrossRef]
- Heinze, T.; Liebert, T. Unconventional Methods in Cellulose Functionalization. Prog. Polym. Sci. 2001, 26, 1689–1762. [Google Scholar] [CrossRef]
- Isogai, A. Wood Nanocelluloses: Fundamentals and Applications as New Bio-Based Nanomaterials. J. Wood Sci. 2013, 59, 449–459. [Google Scholar] [CrossRef]
- Habibi, Y.; Lucia, L.A.; Rojas, O.J. Cellulose Nanocrystals: Chemistry, Self-Assembly, and Applications. Chem. Rev. 2010, 110, 3479–3500. [Google Scholar] [CrossRef] [PubMed]
- Fox, S.C.; Li, B.; Xu, D.; Edgar, K.J. Regioselective Esterification and Etherification of Cellulose: A Review. Biomacromolecules 2011, 12, 1956–1972. [Google Scholar] [CrossRef] [PubMed]
- Isogai, A.; Saito, T.; Fukuzumi, H. TEMPO-Oxidized Cellulose Nanofibers. Nanoscale 2011, 3, 71–85. [Google Scholar] [CrossRef]
- National Center for Biotechnology Information. PubChem Compound Summary for CID 5793, D-Glucose. 2026. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/D-Glucose (accessed on 11 January 2026).
- National Center for Biotechnology Information. PubChem Compound Summary for CID 439178, Cellobiose. 2026. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Cellobiose (accessed on 11 January 2026).
- National Center for Biotechnology Information. PubChem Compound Summary for Cellulose. 2026. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/cellulose (accessed on 11 January 2026).
- Edgar, K.J.; Buchanan, C.M.; Debenham, J.S.; Rundquist, P.A.; Seiler, B.D.; Shelton, M.C.; Tindall, D. Advances in Cellulose Ester Performance and Application. Prog. Polym. Sci. 2001, 26, 1605–1688. [Google Scholar] [CrossRef]
- Johansson, L.S.; Campbell, J.M. Reproducible XPS on Biopolymers: Cellulose Studies. Surf. Interface Anal. 2004, 36, 1018–1022. [Google Scholar] [CrossRef]
- Kokol, V.; Božič, M.; Vogrinčič, R.; Mathew, A.P. Characterisation and Properties of Homo- and Heterogenously Phosphorylated Nanocellulose. Carbohydr. Polym. 2015, 125, 301–313. [Google Scholar] [CrossRef]
- Popescu, C.M.; Larsson, P.T.; Olaru, N.; Vasile, C. Spectroscopic Study of Acetylated Kraft Pulp Fibers. Carbohydr. Polym. 2012, 88, 530–536. [Google Scholar] [CrossRef]
- Rodionova, G.; Lenes, M.; Eriksen, Ø.; Gregersen, Ø. Surface Chemical Modification of Microfibrillated Cellulose: Improvement of Barrier Properties for Packaging Applications. Cellulose 2010, 18, 127–134. [Google Scholar] [CrossRef]
- Li, Z.; Rabnawaz, M. Oil- and Water-Resistant Coatings for Porous Cellulosic Substrates. ACS Appl. Polym. Mater. 2018, 1, 103–111. [Google Scholar] [CrossRef]
- Muncke, J. Exposure to Endocrine Disrupting Compounds via the Food Chain: Is Packaging a Relevant Source? Sci. Total Environ. 2009, 407, 4549–4559. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Li, R.M.; Runge, T.; Feng, J.; Feng, J.; Hu, S.; Shi, Q.S. Solvent-Free Acetylation of Cellulose by 1-Ethyl-3-Methylimidazolium Acetate-Catalyzed Transesterification. ACS Sustain. Chem. Eng. 2019, 7, 16971–16978. [Google Scholar] [CrossRef]
- Heinze, T.; Pfeiffer, K. Studies on the Synthesis and Characterization of Carboxymethylcellulose. Angew. Makromol. Chem. 1999, 266, 37–45. [Google Scholar] [CrossRef]
- Heinze, T.; Koschella, A. Carboxymethyl Ethers of Cellulose and Starch—A Review. Macromol. Symp. 2005, 223, 13–40. [Google Scholar] [CrossRef]
- Kontturi, K.S.; Tammelin, T.; Johansson, L.S.; Stenius, P. Adsorption of Cationic Starch on Cellulose Studied by QCM-D. Langmuir 2008, 24, 4743–4749. [Google Scholar] [CrossRef]
- Naserifar, S.; Kuijpers, P.F.; Wojno, S.; Kádár, R.; Bernin, D.; Hasani, M. In Situ Monitoring of Cellulose Etherification in Solution: Probing the Impact of Solvent Composition on the Synthesis of 3-Allyloxy-2-Hydroxypropyl-Cellulose in Aqueous Hydroxide Systems. Polym. Chem. 2022, 13, 4111–4123. [Google Scholar] [CrossRef]
- Benselfelt, T.; Nordenström, M.; Lindström, S.B.; Wågberg, L. Explaining the Exceptional Wet Integrity of Transparent Cellulose Nanofibril Films in the Presence of Multivalent Ions-Suitable Substrates for Biointerfaces. Adv. Mater. Interfaces 2019, 6, 1900333. [Google Scholar] [CrossRef]
- Katz, A.K.; Glusker, J.P.; Beebe, S.A.; Bock, C.W. Calcium Ion Coordination: A Comparison with That of Beryllium, Magnesium, and Zinc. J. Am. Chem. Soc. 1996, 118, 5752–5763. [Google Scholar] [CrossRef]
- Kudzin, M.H.; Mrozińska, Z.; Urbaniak, P. Vapor Phosphorylation of Cellulose by Phosphorus Trichlo-Ride: Selective Phosphorylation of 6-Hydroxyl Function-The Synthesis of New Antimicrobial Cellulose 6-Phosphate(III)-Copper Complexes. Antibiotics 2021, 10, 203. [Google Scholar] [CrossRef]
- Hubbe, M.A.; Venditti, R.A.; Rojas, O.J. What Happens to Cellulosic Fibers during Papermaking and Recycling? A Review. BioResources 2007, 2, 739–788. Available online: https://bioresources.cnr.ncsu.edu/resources/what-happens-to-cellulosic-fibers-during-papermaking-and-recycling-a-review/ (accessed on 4 January 2026).
- Kudzin, M.H.; Mrozińska, Z.; Giełdowska, M.; Krata, A.A.; Urbaniak, P.; Drabowicz, J. Phosphorylation of the Cellulose Surface with PCl3 and P(O)Cl3. Phosphorus Sulfur Silicon Relat. Elem. 2022, 197, 634–638. [Google Scholar] [CrossRef]
- Asaadi, S.; Kakko, T.; King, A.W.T.; Kilpeläinen, I.; Hummel, M.; Sixta, H. High-Performance Acetylated Ioncell-F Fibers with Low Degree of Substitution. ACS Sustain. Chem. Eng. 2018, 6, 9418–9426. [Google Scholar] [CrossRef]
- Cunha, A.G.; Zhou, Q.; Larsson, P.T.; Berglund, L.A. Topochemical Acetylation of Cellulose Nanopaper Structures for Biocomposites: Mechanisms for Reduced Water Vapour Sorption. Cellulose 2014, 21, 2773–2787. [Google Scholar] [CrossRef]
- Kulomaa, T.; Matikainen, J.; Karhunen, P.; Heikkilä, M.; Fiskari, J.; Kilpeläinen, I. Cellulose Fatty Acid Esters as Sustainable Film Materials-Effect of Side Chain Structure on Barrier and Mechanical Properties. RSC Adv. 2015, 5, 80702–80708. [Google Scholar] [CrossRef]
- Willberg-Keyriläinen, P.; Vartiainen, J.; Harlin, A.; Ropponen, J. The Effect of Side-Chain Length of Cellulose Fatty Acid Esters on Their Thermal, Barrier and Mechanical Properties. Cellulose 2016, 24, 505–517. [Google Scholar] [CrossRef]
- Blomstedt, M.; Vuorinen, T. Modification of Softwood Kraft Pulp with Carboxymethyl Cellulose and Cationic Surfactants. J. Wood Sci. 2007, 53, 223–228. [Google Scholar] [CrossRef]
- Schimper, C.B.; Pachschwoell, P.S.; Hettegger, H.; Neouze, M.A.; Nedelec, J.M.; Wendland, M.; Rosenau, T.; Liebner, F. Aerogels from Cellulose Phosphates of Low Degree of Substitution: A TBAF·H2O/DMSO Based Approach. Molecules 2020, 25, 1695. [Google Scholar] [CrossRef] [PubMed]
- Khakalo, A.; Jaiswal, A.K.; Kumar, V.; Gestranius, M.; Kangas, H.; Tammelin, T. Production of High-Solid-Content Fire-Retardant Phosphorylated Cellulose Microfibrils. ACS Sustain. Chem. Eng. 2021, 9, 12365–12375. [Google Scholar] [CrossRef]
- Wang, J.; Gardner, D.J.; Stark, N.M.; Bousfield, D.W.; Tajvidi, M.; Cai, Z. Moisture and Oxygen Barrier Properties of Cellulose Nanomaterial-Based Films. ACS Sustain. Chem. Eng. 2017, 6, 49–70. [Google Scholar] [CrossRef]
- Hubbe, M.A. Bonding between Cellulosic Fibers. BioResources 2006, 1, 281–318. [Google Scholar] [CrossRef]
- Tarrés, Q.; Delgado-Aguilar, M.; Pèlach, M.A.; González, I.; Boufi, S.; Mutjé, P. Remarkable Increase of Paper Strength by Combining Enzymatic Cellulose Nanofibers in Bulk and TEMPO-Oxidized Nanofibers as Coating. Cellulose 2016, 23, 3939–3950. [Google Scholar] [CrossRef]
- Nair, S.S.; Zhu, J.Y.; Deng, Y.; Ragauskas, A.J. High Performance Green Barriers Based on Nanocellulose. Sustain. Chem. Process. 2014, 2, 23. [Google Scholar] [CrossRef]
- Herrera, M.A.; Sirviö, J.A.; Mathew, A.P.; Oksman, K. Environmental Friendly and Sustainable Gas Barrier on Porous Materials: Nanocellulose Coatings Prepared Using Spin- and Dip-Coating. Mater. Des. 2016, 93, 19–25. [Google Scholar] [CrossRef]
- Guivier, M.; Almeida, G.; Domenek, S.; Chevigny, C. Resilient High Oxygen Barrier Multilayer Films of Nanocellulose and Polylactide. Carbohydr. Polym. 2023, 312, 120761. [Google Scholar] [CrossRef] [PubMed]
- Muncke, J.; Myers, J.P.; Scheringer, M.; Porta, M. Food Packaging and Migration of Food Contact Materials: Will Epidemiologists Rise to the Neotoxic Challenge? J. Epidemiol. Community Heal. 2014, 68, 592–594. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Food Contact Materials-Food Safety-European Commission. Available online: https://food.ec.europa.eu/food-safety/chemical-safety/food-contact-materials_en (accessed on 4 January 2026).
- European Union. Commission Regulation (EU) No 10/2011 on Plastic Materials and Articles Intended to Come into Contact with Food. 2011. Available online: https://eur-lex.europa.eu/eli/reg/2011/10/oj (accessed on 4 January 2026).
- Nerin, C.; Alfaro, P.; Aznar, M.; Domeño, C. The Challenge of Identifying Non-Intentionally Added Substances from Food Packaging Materials: A Review. Anal. Chim. Acta 2013, 775, 14–24. [Google Scholar] [CrossRef]
- Simoneau, C.; European Commission. Guidelines on Testing Conditions for Articles in Contact with Foodstuffs (with a Focus on Kitchenware). Available online: https://publications.jrc.ec.europa.eu/repository/handle/JRC51601 (accessed on 3 January 2026).
- Lorenzini, R.; Biedermann, M.; Grob, K.; Garbini, D.; Barbanera, M.; Braschi, I. Migration Kinetics of Mineral Oil Hydrocarbons from Recycled Paperboard to Dry Food: Monitoring of Two Real Cases. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2013, 30, 760–770. [Google Scholar] [CrossRef]
- Biedermann, M.; Ingenhoff, J.E.; Zurfluh, M.; Richter, L.; Simat, T.; Harling, A.; Altkofer, W.; Helling, R.; Grob, K. Migration of Mineral Oil, Photoinitiators and Plasticisers from Recycled Paperboard into Dry Foods: A Study under Controlled Conditions. Food Addit. Contam.-Part A 2013, 30, 885–898. [Google Scholar] [CrossRef]
- Grob, K. Mineral Oil Hydrocarbons in Food: A Review. Food Addit. Contam. Part A Chem. Anal. Control. Expo. Risk Assess. 2018, 35, 1845–1860. [Google Scholar] [CrossRef]
- German Federal Institute for Risk Assessment. XXXVI. Paper and Board for Food Contact As of 01.02.2023. Available online: https://www.bfr.bund.de/cm/349/XXXVI-Paper-and-Board-for-Food-Contact.pdf (accessed on 3 January 2026).
- Nerín, C.; Bourdoux, S.; Faust, B.; Gude, T.; Lesueur, C.; Simat, T.; Stoermer, A.; Van Hoek, E.; Oldring, P. Guidance in Selecting Analytical Techniques for Identification and Quantification of Non-Intentionally Added Substances (NIAS) in Food Contact Materials (FCMS). Food Addit. Contam. Part A 2022, 39, 620–643. [Google Scholar] [CrossRef]
- Peters, R.J.B.; Groeneveld, I.; Sanchez, P.L.; Gebbink, W.; Gersen, A.; de Nijs, M.; van Leeuwen, S.P.J. Review of Analytical Approaches for the Identification of Non-Intentionally Added Substances in Paper and Board Food Contact Materials. Trends Food Sci. Technol. 2019, 85, 44–54. [Google Scholar] [CrossRef]
- Ávila Ramírez, J.A.; Gómez Hoyos, C.; Arroyo, S.; Cerrutti, P.; Foresti, M.L. Acetylation of Bacterial Cellulose Catalyzed by Citric Acid: Use of Reaction Conditions for Tailoring the Esterification Extent. Carbohydr. Polym. 2016, 153, 686–695. [Google Scholar] [CrossRef]
- Chen, M.J.; Li, R.M.; Zhang, X.Q.; Feng, J.; Feng, J.; Liu, C.F.; Shi, Q.S. Homogeneous Transesterification of Sugar Cane Bagasse toward Sustainable Plastics. ACS Sustain. Chem. Eng. 2016, 5, 360–366. [Google Scholar] [CrossRef]
- Saito, T.; Kimura, S.; Nishiyama, Y.; Isogai, A. Cellulose Nanofibers Prepared by TEMPO-Mediated Oxidation of Native Cellulose. Biomacromolecules 2007, 8, 2485–2491. [Google Scholar] [CrossRef] [PubMed]
- Blilid, S.; Kȩdzierska, M.; Miłowska, K.; Wrońska, N.; El Achaby, M.; Katir, N.; Belamie, E.; Alonso, B.; Lisowska, K.; Lahcini, M.; et al. Phosphorylated Micro- and Nanocellulose-Filled Chitosan Nanocomposites as Fully Sustainable, Biologically Active Bioplastics. ACS Sustain. Chem. Eng. 2020, 8, 18354–18365. [Google Scholar] [CrossRef]
- Saedi, S.; Garcia, C.V.; Kim, J.T.; Shin, G.H. Physical and Chemical Modifications of Cellulose Fibers for Food Packaging Applications. Cellulose 2021, 28, 8877–8897. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, X.; Xie, Y.; Zhang, K. Functional Nanomaterials through Esterification of Cellulose: A Review of Chemistry and Application. Cellulose 2018, 25, 3703–3731. [Google Scholar] [CrossRef]
- Syverud, K.; Stenius, P. Strength and Barrier Properties of MFC Films. Cellulose 2008, 16, 75–85. [Google Scholar] [CrossRef]
- Aulin, C.; Gällstedt, M.; Lindström, T. Oxygen and Oil Barrier Properties of Microfibrillated Cellulose Films and Coatings. Cellulose 2010, 17, 559–574. [Google Scholar] [CrossRef]
- Lavoine, N.; Desloges, I.; Dufresne, A.; Bras, J. Microfibrillated Cellulose - Its Barrier Properties and Applications in Cellulosic Materials: A Review. Carbohydr. Polym. 2012, 90, 735–764. [Google Scholar] [CrossRef]
- Ferrer, A.; Pal, L.; Hubbe, M. Nanocellulose in Packaging: Advances in Barrier Layer Technologies. Ind. Crops Prod. 2017, 95, 574–582. [Google Scholar] [CrossRef]
- Tayeb, A.H.; Tajvidi, M. Sustainable Barrier System via Self-Assembly of Colloidal Montmorillonite and Cross-Linking Resins on Nanocellulose Interfaces. ACS Appl. Mater. Interfaces 2018, 11, 1604–1615. [Google Scholar] [CrossRef]
- Singh, S.; Sögut, E.; Uysal-Unalan, I.; Karlovits, I.; Coma, V.; Corredig, M.; Poças, F. Current Usage of Paperboard Packaging: A Case Study of the European Landscape on Tomato Packaging. Food Packag. Shelf Life 2025, 51, 101572. [Google Scholar] [CrossRef]
- Cheeseman, M.A.; Machuga, E.J.; Bailey, A.B. A Tiered Approach to Threshold of Regulation. Food Chem. Toxicol. 1999, 37, 387–412. [Google Scholar] [CrossRef]
- Muncke, J.; Backhaus, T.; Geueke, B.; Maffini, M.V.; Martin, O.V.; Myers, J.P.; Soto, A.M.; Trasande, L.; Trier, X.; Scheringer, M. Scientific Challenges in the Risk Assessment of Food Contact Materials. Environ. Health Perspect. 2017, 125, 095001. [Google Scholar] [CrossRef]
- Koster, S.; Bani-Estivals, M.H.; Bonuomo, M.; Bradley, E.; Chagnon, M.C.; Garcia, M.L.; Godts, F.; Gude, T.; Helling, R.; Paseiro-Losada, P.; et al. Guidance on Best Practices on the Risk Assessment of Non Intentionally Added Substances (NIAS) in Food Contact Materials and Articles. ILSI Europe Report Series, 16/07/2015. Available online: https://ilsi.eu/publication/guidance-on-best-practices-on-the-risk-assessment-of-non-intentionally-added-substances-nias-in-food-contact-materials-and-articles/ (accessed on 4 January 2026).
- Groh, K.J.; Muncke, J. In Vitro Toxicity Testing of Food Contact Materials: State-of-the-Art and Future Challenges. Compr. Rev. Food Sci. Food Saf. 2017, 16, 1123–1150. [Google Scholar] [CrossRef] [PubMed]
- Foroughi, F.; Ghomi, E.R.; Dehaghi, F.M.; Borayek, R.; Ramakrishna, S. A Review on the Life Cycle Assessment of Cellulose: From Properties to the Potential of Making It a Low Carbon Material. Materials 2021, 14, 714. [Google Scholar] [CrossRef]
- Nadeem, H.; Dehghani, M.; Garnier, G.; Batchelor, W. Life Cycle Assessment of Cellulose Nanofibril Films via Spray Deposition and Vacuum Filtration Pathways for Small Scale Production. J. Clean. Prod. 2022, 342, 130890. [Google Scholar] [CrossRef]
- Arvidsson, R.; Nguyen, D.; Svanström, M. Life Cycle Assessment of Cellulose Nanofibrils Production by Mechanical Treatment and Two Different Pretreatment Processes. Environ. Sci. Technol. 2015, 49, 6881–6890. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Packaging Waste. Available online: https://environment.ec.europa.eu/topics/waste-and-recycling/packaging-waste_en (accessed on 4 January 2026).
- CEPI. Paper-Based Packaging Recyclability Guidelines; CEPI: Oslo, Norway, 2020. [Google Scholar]
- Fibre-Based Packaging Recyclability Evaluation Protocol; CEPI: Oslo, Norway, 2022.
- Circularity by Design Guideline for Fibre-Based Packaging; RadTech International North America: Chevy Chase, MD, USA, 2024.
- Jahangiri, F.; Mohanty, A.K.; Misra, M. Sustainable Biodegradable Coatings for Food Packaging: Challenges and Opportunities. Green Chem. 2024, 26, 4934–4974. [Google Scholar] [CrossRef]
- Mousania, Z.; Atkinson, J.D. A Cradle-to-Grave Life Cycle Assessment of Multilayer Plastic Film Food Packaging Materials, Comparing to a Paper-Based Alternative. Waste Manag. 2025, 200, 114747. [Google Scholar] [CrossRef]
- Bremenkamp, I.; Sousa Gallagher, M.J. Life Cycle Assessment Methods for Investigating Novel Food Packaging Systems. Environ. Sci. Adv. 2024, 3, 1337–1350. [Google Scholar] [CrossRef]
- Grease Resistance Test for Paper and Paperboard, Test Method T 559 Cm-22. Available online: https://imisrise.tappi.org/TAPPI/Products/01/T/0104T559.aspx (accessed on 4 January 2026).
- ASTM F1249; Test Method for Water Vapor Transmission Rate Through Plastic Film and Sheeting Using a Modulated Infrared Sensor. ASTM: West Conshohocken, PA, USA, 2020. [CrossRef]
- ISO 535:2023; Paper and Board-Determination of Water Absorptiveness-Cobb Method. ISO: Geneva, Switzerland, 2023. Available online: https://www.iso.org/standard/80320.html (accessed on 4 January 2026).
- ASTM D3985-17; Test Method for Oxygen Gas Transmission Rate Through Plastic Film and Sheeting Using a Coulometric Sensor. ASTM: West Conshohocken, PA, USA, 2017. [CrossRef]
- Danielewicz, D. Manufacturing of Form-Molded Pulp Products (FMPPs) in the Papermaking Industry—A Review. BioResources 2025, 20, 5114–5156. [Google Scholar] [CrossRef]
- Eyley, S.; Thielemans, W. Surface Modification of Cellulose Nanocrystals. Nanoscale 2014, 6, 7764–7779. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.; Liu, W.; Ye, S.; Batista, L. Packaging Design for the Circular Economy: A Systematic Review. Sustain. Prod. Consum. 2022, 32, 817–832. [Google Scholar] [CrossRef]
- Fukuzumi, H.; Saito, T.; Isogai, A. Influence of TEMPO-Oxidized Cellulose Nanofibril Length on Film Properties. Carbohydr. Polym. 2013, 93, 172–177. [Google Scholar] [CrossRef] [PubMed]
- Tayeb, A.H.; Tajvidi, M.; Bousfield, D.; Tayeb, A.H.; Tajvidi, M.; Bousfield, D. Enhancing the Oxygen Barrier Properties of Nanocellulose at High Humidity: Numerical and Experimental Assessment. Sustain. Chem. 2020, 1, 198–208. [Google Scholar] [CrossRef]
- Hubbe, M.A.; Ferrer, A.; Tyagi, P.; Yin, Y.; Salas, C.; Pal, L.; Rojas, O.J. Nanocellulose in Thin Films, Coatings, and Plies for Packaging Applications: A Review. BioResources 2017, 12, 2143–2233. [Google Scholar] [CrossRef]
- Brennan, L.; Langley, S.; Verghese, K.; Lockrey, S.; Ryder, M.; Francis, C.; Phan-Le, N.T.; Hill, A. The Role of Packaging in Fighting Food Waste: A Systematised Review of Consumer Perceptions of Packaging. J. Clean. Prod. 2021, 281, 125276. [Google Scholar] [CrossRef]
- Wikström, F.; Williams, H.; Trischler, J.; Rowe, Z. The Importance of Packaging Functions for Food Waste of Different Products in Households. Sustainability 2019, 11, 2641. [Google Scholar] [CrossRef]
- Klemm, D.; Kramer, F.; Moritz, S.; Lindström, T.; Ankerfors, M.; Gray, D.; Dorris, A. Nanocelluloses: A New Family of Nature-Based Materials. Angew. Chem. Int. Ed. 2011, 50, 5438–5466. [Google Scholar] [CrossRef]
- Fukuzumi, H.; Saito, T.; Iwata, T.; Kumamoto, Y.; Isogai, A. Transparent and High Gas Barrier Films of Cellulose Nanofibers Prepared by TEMPO-Mediated Oxidation. Biomacromolecules 2008, 10, 162–165. [Google Scholar] [CrossRef] [PubMed]


| Modification Chemistry | Typical DS Range | Localization | Key Functional Effect | Packaging Relevance | Ref. |
|---|---|---|---|---|---|
| Acetylation | 0.05–0.30 | surface/ near-surface | ↓* moisture uptake, ↓ WVTR | moisture barrier | [40,54,55] |
| Fatty acid esterification | 0.02–0.15 | surface-dominant | ↑ KIT grease resistance (KIT 7–10) | grease barrier | [56,57] |
| Etherification (CMC) | 0.05–0.20 | bulk/surface | ↑ bonding, ↑ swelling | strength, formation | [58] |
| Phosphorylation (P(V)) | <0.10 | surface-selective | ionic crosslinking, ↑ wet stability | molded fiber | [39,59,60] |
| TEMPO oxidation | 0.05–0.15 | surface | ↑ bonded area, ↑ O2 barrier (dry) | gas-barrier layers | [33,61] |
| Modification Strategy/Nanocellulose Systems | OTR (23 °C, 50% RH) (cm3·m−2·day−1·bar−1) | WVTR (23 °C, 50% RH) (g·m−2·day−1) | Grease Resistance (KIT) | RH Sensitivity | Ref. |
|---|---|---|---|---|---|
| Unmodified paper/board | >>1000 | ~800–1200 | 0–1 | very high | [5,21,22] |
| CNF coating (dry or low RH) | <10 | >500 | 1–2 | extreme | [28,61,65] |
| Acetylated fibers (low DS) | 50–300 | 300–600 | 2–4 | moderate | [41,55] |
| Fatty-acid-modified fibers | 200–600 | 150–400 | 7–10 | low-moderate | [56,57] |
| Cellulose Modification Strategies | Typical Source of Potential Migrants/NIAS | Qualitative Migration Concern | Ref. |
|---|---|---|---|
| Acetylation (low DS) | Residual acetic species; low-molecular-weight esters; hydrolysis products | Generally low at controlled DS; increases with aging | [78,79,80] |
| Long-chain fatty-acid esterification | Fatty acid fragments; residual catalysts or solvents | Relevant mainly for fatty foods and long contact times | [81,82] |
| Etherification (CMC-type) | Unreacted etherifying agents; counter-ions; oligomeric residues | Elevated if grafted; lower when used as additive | [83,84] |
| TEMPO-mediated oxidation | Oxidation by-products; residual salts | Moderate; sensitive to humidity and aging | [85,86] |
| Phosphorylation (P(V)) | Phosphate species; counter-ion exchange products | Generally moderate; chemistry- and ion-dependent | [73,87,88] |
| Multivalent-ion complexation | Leachable metal ions (Ca2+, Mg2+, Al3+) | Low when food-approved ions are used | [77] |
| Recycled fibre background | MOSH/MOAH; printing-ink-related NIASs | High unless functional barriers are applied | [76,89] |
| Cellulose Modification Strategies | Reaction Efficiency | Relative Cost | Energy Consumption | Process Characteristics | Ref. |
|---|---|---|---|---|---|
| Etherification (e.g., CMC) | High (70–90%) | Medium | Low/Medium | Water–alcohol processes; easy solvent regeneration | [40,54,55,78,79,80] |
| Esterification (e.g., CA) | Medium (50–75%) | High | High | Requires anhydrides and acids; energy-intensive distillation and acid recovery | [56,57,81,82] |
| TEMPO oxidation | High (up to 100%) | Very High | Medium High | High catalyst cost; precise pH control required | [33,61,83,84,85,86] |
| Enzymatic modification | Very High | High (enzymes) | Very Low | Green process but limited to low DS values | [83,85,98,100] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Kudzin, M.H.; Mrozińska, Z.; Chruściel, J.J.; Olczyk, J.; Sikora, M.; Sulak, E.; Walawska, A. Chemical Modification of Cellulose Fibers for Sustainable Food Packaging: Structure–Property–Sustainability Relationships. Materials 2026, 19, 1124. https://doi.org/10.3390/ma19061124
Kudzin MH, Mrozińska Z, Chruściel JJ, Olczyk J, Sikora M, Sulak E, Walawska A. Chemical Modification of Cellulose Fibers for Sustainable Food Packaging: Structure–Property–Sustainability Relationships. Materials. 2026; 19(6):1124. https://doi.org/10.3390/ma19061124
Chicago/Turabian StyleKudzin, Marcin H., Zdzisława Mrozińska, Jerzy J. Chruściel, Joanna Olczyk, Monika Sikora, Edyta Sulak, and Anetta Walawska. 2026. "Chemical Modification of Cellulose Fibers for Sustainable Food Packaging: Structure–Property–Sustainability Relationships" Materials 19, no. 6: 1124. https://doi.org/10.3390/ma19061124
APA StyleKudzin, M. H., Mrozińska, Z., Chruściel, J. J., Olczyk, J., Sikora, M., Sulak, E., & Walawska, A. (2026). Chemical Modification of Cellulose Fibers for Sustainable Food Packaging: Structure–Property–Sustainability Relationships. Materials, 19(6), 1124. https://doi.org/10.3390/ma19061124







