3.1. Surface Characterization of the Deposited Coatings
Diamond films were deposited by hot-filament chemical-vapor deposition in a CH
4 and H
2 atmosphere, where the chamber pressure, methane concentration, and deposition temperature play critical roles in determining the film quality and surface morphology. Using the experimental parameters listed in
Table 1, three diamond films with distinct surface morphologies were obtained. The surface morphologies of these representative deposited films were characterized by scanning electron microscopy.
As shown in the SEM image in
Figure 3a, the surface of sample 1 is dominated by globular particles with an overall spherical morphology. The SEM micrographs reveal distinct growth mechanisms. The spherical coating (sample 1) consists of agglomerated nanocrystallites forming clusters of 5–6 μm, induced by the high secondary nucleation rate, which explains the apparent non-uniformity. In contrast, the pyramidal (sample 2) and prismatic (sample 3) coatings exhibit well-faceted micro-crystals with larger, clearer grain boundaries, typical of a lower nucleation density growth mode. As shown in
Figure 3b, the surface morphology of sample 2 exhibits a well-defined pyramidal structure, with sharp facets oriented toward the outward viewing direction and a relatively uniform spatial distribution. As shown in
Figure 3c, sample 3 presents a morphology in which flat crystal planes are oriented toward the outward viewing direction. The grains display well-defined shapes with distinct grain boundaries.
Prior to analysis, all Raman spectra were baseline-corrected using OriginPro 2024 software to eliminate fluorescence background and allow for accurate comparison. ID/IG is also displayed in the figure. The Raman spectra of the three CVD diamond coatings are presented in
Figure 4. In the Raman spectrum of CVD diamond, the characteristic peak of natural diamond appears at approximately 1332 cm
−1 [
19]. The characteristic D band associated with sp
2 amorphous carbon is located near 1350 cm
−1. Peaks observed in the range of 1430 cm
−1 to 1470 cm
−1 are likely attributed to the trans-polyacetylene modes at grain boundaries. In addition, the characteristic G band of sp
2 amorphous carbon is observed in the range of 1520 cm
−1 to 1580 cm
−1.
The Raman spectral features of the spherical diamond coating are distinctly different from those of sample 2 and sample 3, as shown in
Figure 4a. In addition to the diamond characteristic peak located near 1332 cm
−1, sample 1 exhibits a reduced peak intensity and a relatively broad peak width at this position. A pronounced peak is also observed at approximately 1580 cm
−1, with a peak intensity lower than that of the 1332 cm
−1 peak and a comparatively broad linewidth. These spectral characteristics are attributed to the fabrication of sample 1 under conditions that promote a high secondary nucleation rate and the formation of dislocation twins, which inhibit grain growth. As the grain size decreases, the surface grain boundary area increases, facilitating the formation of non-diamond carbon phases at the grain boundaries in polycrystalline diamond films. Consequently, the increased content of non-diamond carbon leads to a broadening of the full width at half maximum of the diamond characteristic peak. In addition, a distinct peak is observed near 1140 cm
−1 for the spherical diamond coating, which originates from trans-polyacetylene. Because the spherical structures are composed of numerous fine crystallites, smaller grain sizes result in a larger grain boundary area and a higher concentration of trans-polyacetylene at the grain boundaries. There is an inverse relationship between grain size and sp2 content. The nanocrystalline spherical coating (small grains) exhibits a higher volume fraction of grain boundaries, which are the primary sites for sp2-bonded amorphous carbon accumulation.
The Raman spectrum of sample 2 is shown in
Figure 4b. The strongest peak appears near 1332 cm
−1, which is characteristic of the sp
3 bonding structure of diamond. The Raman intensity around 1330 cm
−1 is commonly used as an indicator for evaluating the quality of diamond films [
20], and under appropriate process parameters, micrometer-scale diamond films typically exhibit a pronounced peak at this position. In contrast, the relative intensities of the D band, G band, and trans-polyacetylene-related peaks are relatively weak, indicating that the sample consists predominantly of micrometer-grained diamond and that the deposited film exhibits high structural quality.
The Raman spectrum of sample 3 is shown in
Figure 4c. The most intense peak is located near the diamond characteristic peak at 1332 cm
−1, indicating that the sample prepared under these conditions consists predominantly of micrometer-grained diamond. The relative intensities of the D band, G band, and trans-polyacetylene-related peaks are comparatively weak, further confirming the high quality of the deposited diamond film.
To provide a rigorous assessment, the peak positions, shifts, and FWHM values were calculated (
Table 2). The broader FWHM (full width at half maximum) in sample 1 (12.4 cm
−1) quantitatively confirms its nanocrystalline nature and higher internal stress state. This morphology evolution aligns with the ‘Synthetic Growth Concept’ [
21], where high renucleation rates promote twin formation and modify the bonding state.
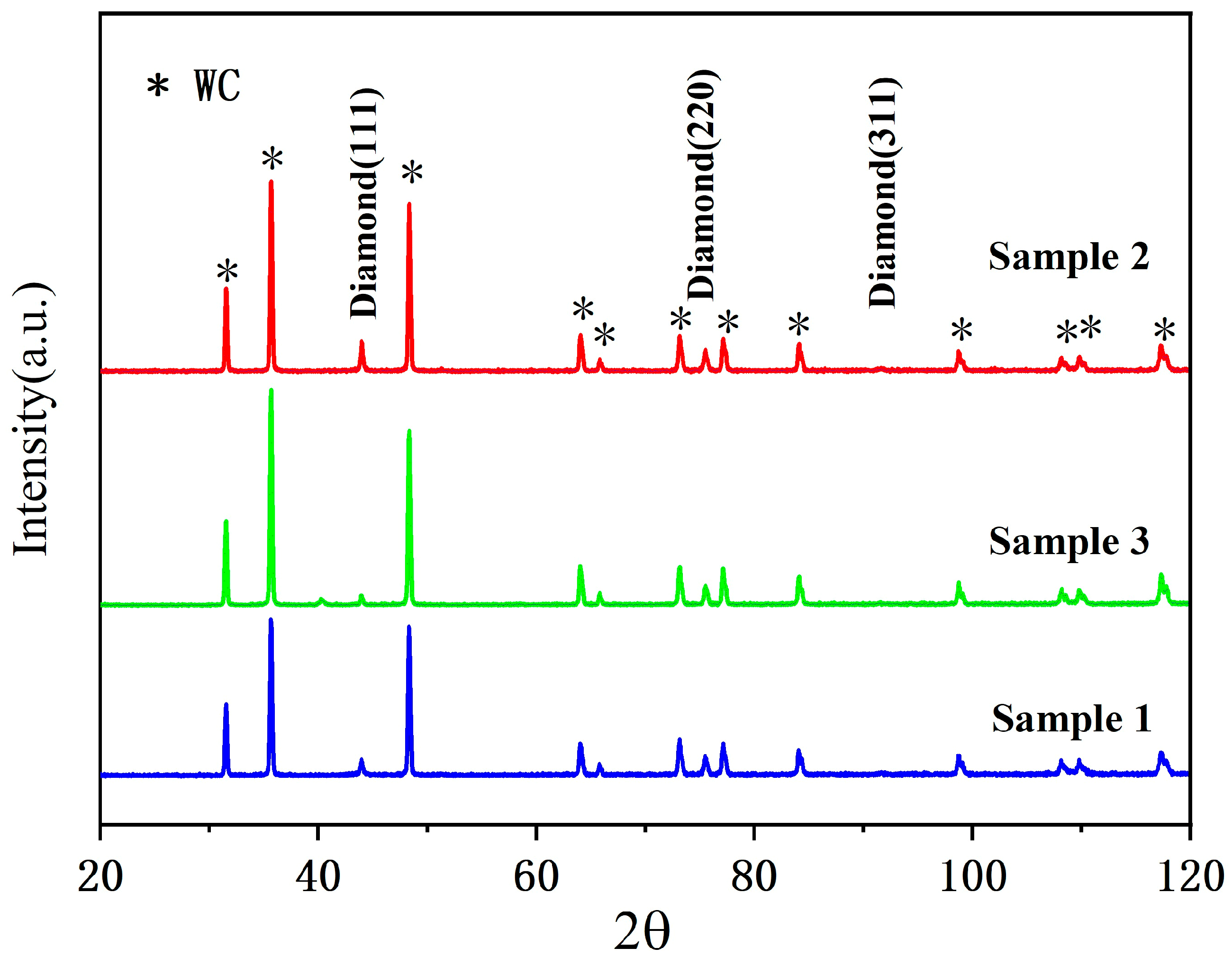
To further corroborate the structural findings from Raman spectroscopy, X-ray diffraction (XRD) patterns are shown in
Figure 5. All samples exhibit characteristic diffraction peaks corresponding to the (111) and (220) planes of cubic diamond, confirming high crystallinity. This structural verification complements the Raman analysis.
The surface roughness of the three diamond films with different morphologies was characterized using a VK-X100 laser scanning confocal microscopy system (Keyence, Neu-Isenburg, Germany), as shown in
Figure 6. Sample 2 exhibits surface roughness values of Rq 0.524 μm and Ra 0.423 μm, where Rq represents the root mean square surface roughness and Ra denotes the arithmetic mean surface roughness. Sample 3 shows surface roughness values of Rq 0.680 μm and Ra 0.515 μm, whereas sample 1 presents the highest roughness, with Rq 1.239 μm and Ra 0.809 μm. All samples were deposited on identical substrates and subjected to the same pretreatment procedures, allowing the influence of substrate roughness on the measured diamond film surface roughness to be neglected. Among the three morphologies, the pyramidal diamond film exhibits the lowest roughness. This film grows preferentially along the <111> crystallographic planes, forming inclined pyramidal facets with sharp features that overlap during growth, resulting in indistinct grain boundaries and a reduced surface roughness. The prismatic diamond film, in which flat planes are oriented toward the viewing direction, shows an intermediate roughness. It grows predominantly along the <110> crystallographic planes, leading to uniform vertical growth with clearly defined grain boundaries and pronounced hierarchical features. In contrast, the spherical diamond film exhibits the highest surface roughness. Although this film consists of nanometer-scale diamond grains, the aggregation and clustering of these nanocrystallites during growth result in micrometer-scale spherical agglomerates with relatively large particle sizes.
Figure 7 presents the SEM images of the diamond-coated tools after lapping sapphire substrates. As observed from the images, the original surface structures of the diamond coatings experienced wear during lapping, while the coatings remained well bonded to the substrates, with no evidence of delamination or coating spallation. In
Figure 7a, sample 1 exhibits a loss of structural regularity after lapping, and wear debris is observed adhering to the surface. In
Figure 7b, wear debris is found accumulated along the grain boundaries of sample 2, and the sharp pyramidal edges are noticeably blunted. In
Figure 7c, wear debris is present between the grains of sample 3, and the edges of the cubic or truncated pyramidal grains are either blunted or locally damaged.
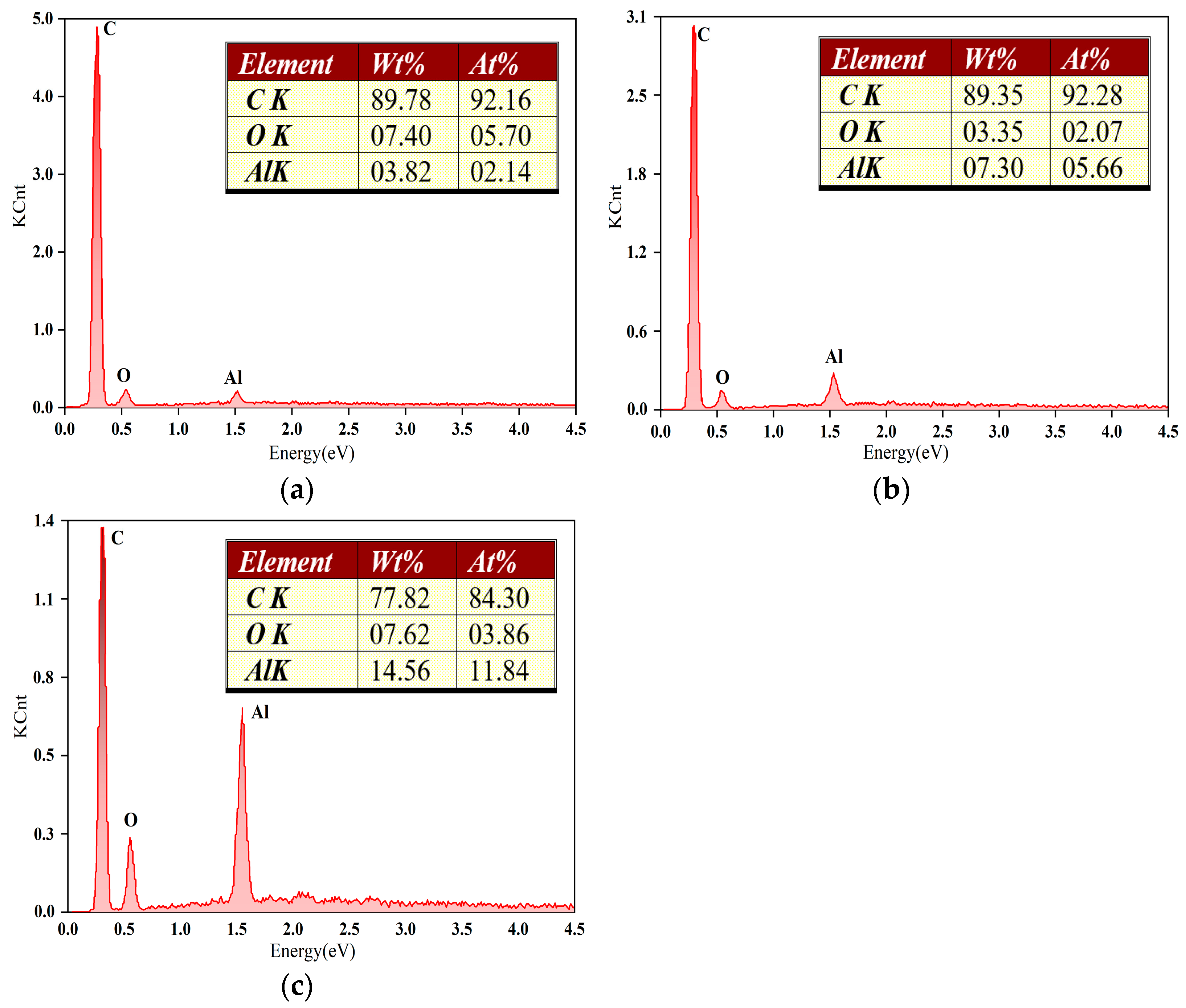
The elemental distribution on the surface of the CVD diamond coatings after lapping was analyzed using energy dispersive spectroscopy, as shown in
Figure 8. The carbon (C) content in the three diamond-coated surfaces is 92.16%, 92.28%, and 84.2%, respectively. The oxygen (O) content is 5.7%, 2.07%, and 3.86%, respectively. The aluminum (Al) content is 2.14%, 5.66%, and 11.84%, respectively. The dominant elements detected are C, O, and Al, indicating that the wear debris is primarily composed of sapphire (Al
2O
3). As observed in
Figure 8b, the wear debris generated after lapping is predominantly block-like in morphology, whereas the debris produced after lapping with the spherical diamond coating in
Figure 8a exhibits a strip-like morphology. This difference suggests that, during sapphire substrate lapping with the spherical diamond coating, sapphire does not undergo purely brittle fracture. Instead, under the embedding, extrusion, and cutting actions of the spherical diamond particles, the material experiences deformation behavior analogous to the lateral plastic flow and accumulation observed in metallic cutting processes. When sapphire is processed under conditions below the critical load for lateral crack initiation, dislocation slip occurs between sapphire grains under shear stress, leading to plastic deformation. Under such conditions, the brittle material can be removed through a plastic flow-dominated material removal mechanism.
Figure 9a,b presents the Raman spectra of sample 1 and sample 2 after lapping. The characteristic Raman peak of natural diamond is located at 1341 cm
−1. However, owing to the presence of residual stress, the Raman peak position of CVD diamond shifts relative to 1336 cm
−1. Based on the measured shift in the diamond characteristic peak, the direction and magnitude of residual stress in the diamond coatings can be semi-quantitatively estimated. The residual stress in the CVD diamond films was calculated using the following equation [
22,
23]:
In the equation, ρ represents the residual stress in the CVD diamond film, and k is a coefficient related to the deposition conditions and substrate material, which is generally taken as a negative value. Δv denotes the relative shift in the Raman peak position, where vs corresponds to the measured diamond peak position of the sample, and v0 represents the peak position of natural diamond. When the calculated value of ρ is positive, the residual stress is tensile in nature, whereas a negative value of ρ indicates compressive residual stress.
Before lapping, the Raman peak of sample 1 was located at 1341 cm−1. According to Equation (2), the calculated residual stress in the CVD diamond film was negative, indicating compressive stress. After lapping, the Raman peak shifted to 1338 cm−1, suggesting a reduction in the residual compressive stress within the diamond film. Similarly, for sample 2, the Raman peak was located at 1338 cm−1 before lapping and shifted to 1331 cm−1 after lapping, indicating a decrease in residual compressive stress. This behavior can be attributed to repeated collisions between the coating and the sapphire substrate under applied load during lapping, as well as substantial heat generation resulting from micro-asperity cutting interactions. These effects induce localized strain within the coating, and regions with higher stress concentrations undergo larger strain. As a result, stress redistribution occurs within the coating, leading to stress homogenization and an overall reduction in residual stress.
The full width at half maximum of the characteristic Raman peak of natural diamond is generally approximately 2.3 cm
−1. The FWHM value of diamond is closely related to the quality of diamond films, including factors such as the crystallinity of diamond grains, the defect density of the film, and the presence of impurities. Therefore, the FWHM of the diamond characteristic peak is regarded as an important parameter for evaluating diamond film quality. As shown in
Figure 9c, the FWHM value of sample 3 increases after lapping compared with that before lapping, indicating the formation of sp
2-bonded graphite, amorphous carbon, and other non-diamond carbon phases at the grain boundaries.
With an increasing content of sp
2-bonded graphite, amorphous carbon, and other carbon impurity phases in the diamond film, the Raman peak intensity ratio of non-diamond components to the diamond component (
In/
I1332) decreases accordingly [
24]. Compared with the Raman spectra before lapping, the relative intensity ratio of the non-diamond Raman peaks in the range of 1497 cm
−1 to 1597 cm
−1 to the diamond peak increases after lapping. This observation indicates that the bonding state of carbon on the surface of the CVD diamond coating changes after lapping, accompanied by an increased content of amorphous carbon at the grain boundaries. The substantial heat generated during lapping can lead to transiently elevated temperatures, which promote the transformation of diamond into non-diamond carbon phases. In contrast, the pronounced increase in the Raman intensity near the diamond peak at 1332 cm
−1 after testing is attributed to the removal of the growth surface during lapping, resulting in a diamond structure that more closely approaches the maximum structural quality near the substrate interface.
3.2. Tribological Characteristics and Lapping Mechanisms of CVD Diamond-Coated Tools
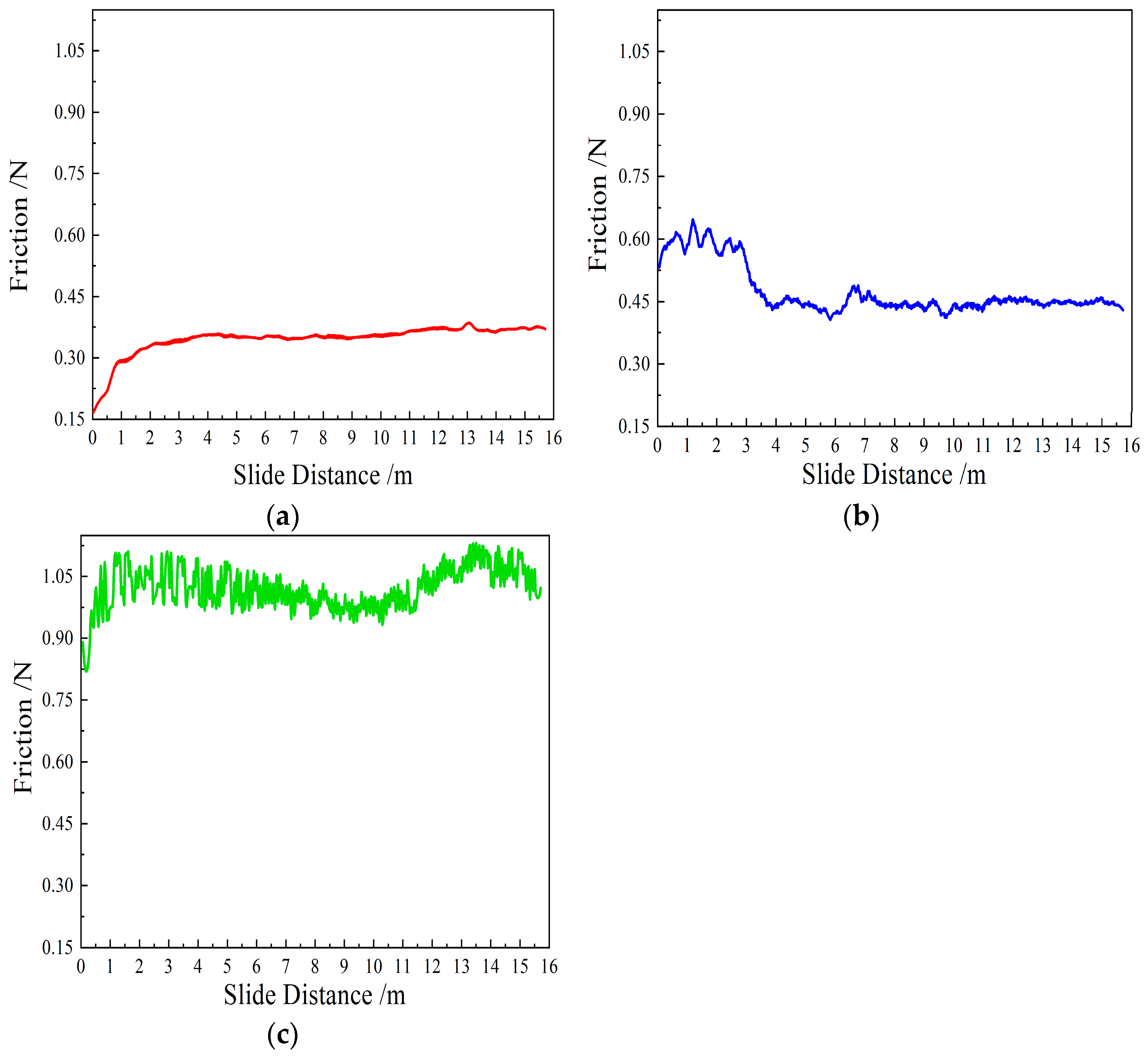
Figure 10 illustrates the variation in tangential force as a function of sliding distance for the three CVD diamond coatings. As can be seen from the figure, the evolution of tangential force differs markedly among the CVD diamond coatings with different surface morphologies.
As shown in
Figure 10a, during the initial stage of lapping, sample 1 forms a very limited real contact area with the uneven sapphire substrate, and most diamond particles within the coating are not subjected to direct normal loading. Owing to the high hardness of CVD diamond, the spherical particles freely abrade the surface asperities of the sapphire substrate, rapidly removing surface irregularities. As a result, the real contact area increases quickly, leading to a gradual rise in tangential force. After reaching a steady-state regime, the tangential force stabilizes at approximately 0.35 N. In comparison with the tangential force evolution of the pyramidal and prismatic diamond coatings, the spherical CVD diamond-coated tool exhibits a lower steady-state tangential force with minimal fluctuation.
As shown in
Figure 10b, sample 2 exhibits a pronounced running-in stage followed by a steady-state stage. During the initial stage, the sharp edges of the pyramidal diamond coating penetrate into the sapphire substrate, resulting in a relatively high tangential force. However, the excessive local stress combined with the intrinsically sharp geometry leads to progressive edge blunting with increasing sliding distance. Meanwhile, the pyramidal diamond coating provides a relatively limited chip accommodation capacity, causing the generated wear debris to gradually accumulate within the intergranular spaces and hinder the participation of newly exposed diamond grains in the lapping process. According to tribochemical theories [
25], the sharp edges are likely to promote the formation of sp
2-bonded graphite under high pressure and elevated temperature. Consequently, the tangential force decreases rapidly after the sliding distance reaches a critical value and subsequently stabilizes at approximately 0.45 N.
As shown in
Figure 10c, sample 3 exhibits an initial tangential force evolution similar to that of the spherical diamond coating, characterized by a gradual increase from a low value. However, unlike the spherical diamond coating, which reaches a steady state after a sliding distance of approximately 3 m, the prismatic diamond coating attains a stable regime after only about 0.5 m of sliding. This behavior is attributed to the flat rectangular surface morphology of the prismatic diamond coating, which contains edges and corners that enable rapid removal of asperities on the sapphire substrate during the initial stage. As the surface asperities on the sapphire substrate are progressively reduced, the real contact area increases substantially. Owing to its flat rectangular morphology, the prismatic diamond coating provides a larger contact area with the sapphire substrate than the spherical and pyramidal diamond coatings. According to Tabor’s contact friction theory, a larger real contact area results in a higher friction coefficient. Consequently, the prismatic diamond coating exhibits a higher steady-state tangential force, reaching approximately 1.0 N to 1.1 N. During lapping, hard wear debris is generated. Because of the relatively large intergranular spacing of the prismatic diamond coating, part of the debris falls into the intergranular gaps and is removed from the contact interface, while another part acts as abrasive particles that plow between the diamond crystal planes and the sapphire substrate, leading to pronounced fluctuations in tangential force. In addition, within the sliding distance range of 5–9 m, the tangential force gradually decreases, whereas in the range of 9–14 m, the tangential force increases progressively, indicating a certain degree of self-dressing behavior.
Throughout the stable lapping stage, the coefficient of friction (COF) for the spherical tool stabilized at 0.15 as shown in
Figure 11, with a low standard deviation, indicating excellent tribological stability compared to the fluctuating forces observed in the prismatic coating.
To study the lapping characteristics of different morphologies of diamond-coated tools on sapphire materials, a single-grain abrasive micromachining model is established. For HFCVD diamond-coated tools lapping sapphire crystals, the grain is embedded in the surface of the sapphire workpiece and is called an effective grain abrasive. The material removal rates of different shapes of diamond-grain abrasives are analyzed. In order to simplify the model, the following assumptions are made before establishing the model:
- (1)
During the lapping process, the abrasive grain is assumed to be a rigid body and does not undergo elastic deformation under the load.
- (2)
Diamond grains or particles are closely and uniformly distributed, with consistent size, and do not affect each other.
- (3)
The lapping pressure is shared by the diamond-coated tool and the workpiece, and the applied external force is borne by the grain abrasive.
- (4)
The chemical reaction between the coating tool, polishing fluid, and workpiece is not considered.
Figure 12 schematically shows a biaxial contact diagram of a spherical CVD diamond particle and a sapphire wafer.
Di is the diameter of the diamond particle,
dαj is the depth at which the particle is embedded in sapphire, and
Pj1 is the normal force exerted on the grain.
The lapping pressure is evenly borne by the particles in contact with the sapphire workpiece on the coating, so there is
In the formula,
N is the total number of particles on the coated tool,
Pj is the force applied to each particle,
P0 is the pressure acting on the workpiece, and
Aw is the surface area of the workpiece being machined. Let
Aj represent the area of the pit formed by the abrasive grain indenting into the workpiece surface; then we have
According to the definition of hardness, the normal force
Pj1 acting on a single diamond particle is as given in Equation (5) [
25,
26].
In the formula,
Hα is the hardness of the sapphire crystal, and λ is a constant representing the influence of factors other than the indenter geometry on hardness, which does not change with the geometry of the indenter. For uniform and dense CVD diamond coatings, it is assumed that the grain abrasive particles are uniformly distributed on the coating surface, and the distance between each grain abrasive particle and its adjacent grains is
l, so
l =
D. When the size of the coated tool is known, the number of grain abrasive particles on a single coated tool can be roughly calculated.
By substituting Equation (4) into Equation (5) and combining with Equation (6), the depth to which a single particle is pressed into the workpiece can be obtained as shown in Equation (7).
Figure 13 shows a schematic diagram of the two-body contact between a pyramid-shaped CVD diamond grit and a sapphire wafer. For the pyramid-shaped CVD diamond, the entire grit is equivalent to a regular octahedron.
If the abrasive grains of its crystal have an embedding depth of
dβj in the sapphire, then the area
Aj of the cross-section A
2E
2F
2C
2 of the pit formed by the crystal grains indenting the work:
The normal force
Pj2 acting on a single diamond grit particle is as follows:
The depth to which a single-grain abrasive is pressed into the sapphire workpiece is given by Equation (10):
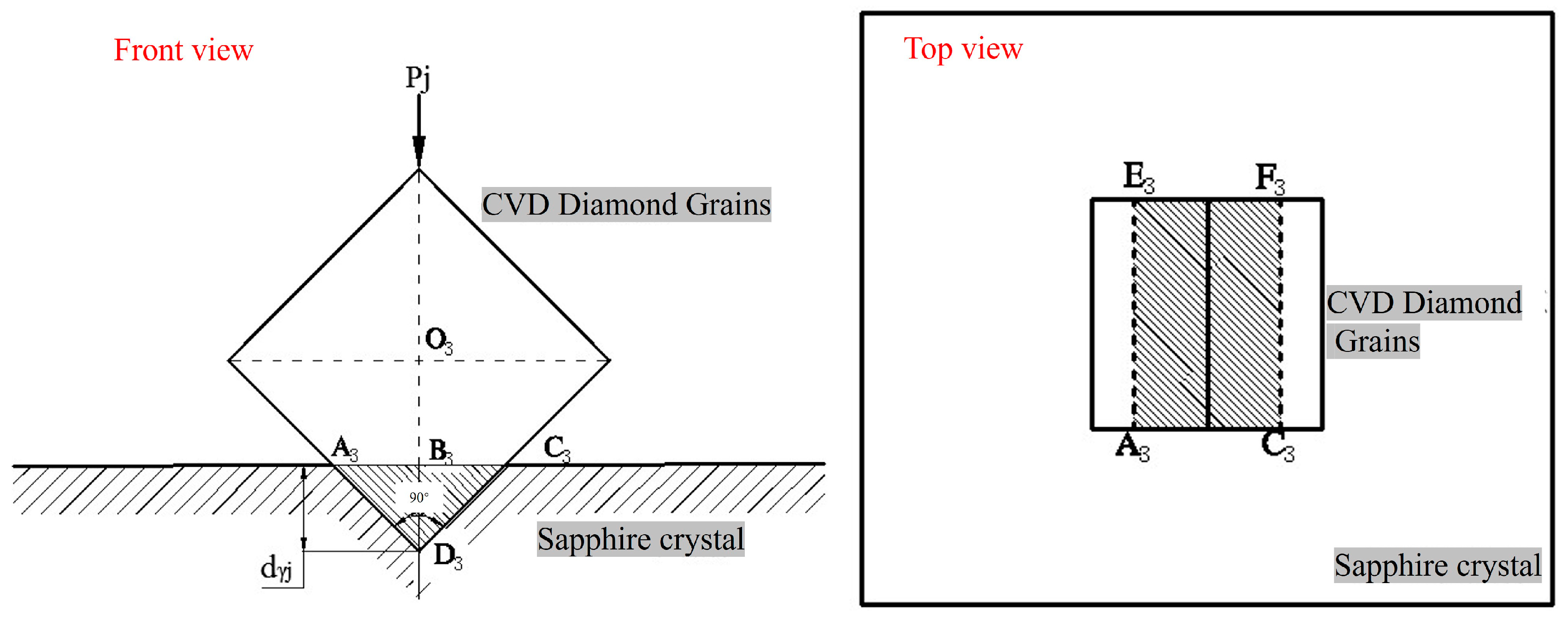
Figure 14 is a schematic diagram of the two-body contact between prism-shaped CVD diamond grits and a sapphire wafer. For the prism-shaped diamond grits, the pit formed on the workpiece surface by the indentation of the grit is a rectangle A
3C
3F
3E
3. The lapping grains of its crystal have an embedding depth of
dγj in sapphire, and the area
Aj of its cross-section A
3C
3F
3E
3 is:
The normal force
Pj3 acting on a single diamond grit particle is as follows:
The depth to which a single-grain abrasive is pressed into the sapphire workpiece is as given in Formula (13).
It can be seen from Equations (10)–(13) that for CVD diamond-coated tools, the depth of penetration of a single-grain abrasive
dγj is not only related to the normal force on the diamond-grain abrasive and the material of the workpiece but also to the shape of the grain abrasive, the size of the grain abrasive, and the diameter of different CVD diamonds. For the same diamond-grain abrasive, when conditions remain unchanged, the larger the equivalent diameter D of the CVD diamond-grain abrasive, the greater the depth of penetration
dj into sapphire. It can be seen from Equations (7), (10), and (13), that the penetration depth of prismatic CVD diamond > than that of the penetration depth of pyramidal > and that of spherical CVD diamond under the same effective particle size and pressure. This is also the reason for the difference in the tangential force of the three different types of diamond-coated tools, as shown in
Figure 10. The specific depth can be calculated through the model, which provides a theoretical model basis for further in-depth study of the mechanism.
Substituting the experimental parameters (normal load = 2 N, sapphire hardness ≈ 20 GPa) into Equation (13), the theoretical penetration depth for the prismatic coating is calculated to be 0.85 μm. This value is consistent with the measured surface roughness (Ra 0.809 μm), validating the proposed geometrical model.
Figure 15 presents the surface roughness map of the sapphire spherical surface after lapping using the spherical diamond-coated tool, obtained by a Keyence optical microscopy system. The surface roughness Ra values are 0.22 μm, 0.299 μm, and 0.469 um representing a substantial improvement in surface quality compared with that achieved by milling and grinding crown-forming processes and indicating that a favorable lapping performance has been preliminarily attained. However, further magnification reveals that a certain degree of disordered scratches is still present on the processed surface, as shown in the optical metallographic micrograph in
Figure 16. From a microscopic perspective, these scratches are primarily attributed to compressive and sliding interactions between diamond crystallites and the sapphire surface during lapping, which lead to localized abrasive scratching. Therefore, further processing of the sapphire surface is still required subsequently.
Figure 17 is a bar chart illustrating the material removal rate (MRR) of sapphire when three types of diamond coatings are paired with diamond for friction grinding. It can be observed that sample 3 (prismatic shape) exhibits the highest removal rate, sample 2 (pyramidal shape) shows a medium removal rate, and sample 1 (spherical shape) has the lowest removal rate, with the corresponding values being 11.2 μm/min, 17.6 μm/min, and 24.3 μm/min, respectively. This result is consistent with the single abrasive grain removal model established earlier. Under the same pressure, the prismatic shape achieves the maximum indentation depth, thereby yielding the highest removal rate, which is directly associated with its surface morphology. This further verifies the correctness of the established model, and the model can be applied to the machining of similar materials. Consequently, it is concluded that diamond-coated tools can serve as efficient grinding tools for sapphire materials, and the machining quality can be controlled by regulating the surface morphology of the coating.
The reason for the efficient machining is that the workpiece and tool are in two-body contact, rather than three-body contact with free abrasives. The traditional free abrasive grinding is a three-body contact process. During machining, the abrasive slurry flows between the workpiece and the processing tool (or substrate), with the abrasive serving as an intermediate medium to form a “tool-abrasive-workpiece” ternary contact system. The abrasive exerts a grinding effect on the workpiece surface through rolling, sliding, impact, and other means [
27].
Our innovative method adopts a two-body contact mode. The HFCVD diamond coating fixes the abrasive on the substrate surface, avoiding the problems of disordered distribution, mutual collision and wear, and loss of abrasives in the loose abrasive slurry. Each diamond particle on the coating surface can participate in the machining process efficiently, and the abrasive utilization rate is increased from less than 30% in the three-body contact to more than 80% [
28]. At the same time, the cutting angle and contact pressure of the fixed abrasive are easier to control. By optimizing the coating thickness, grain size, and surface morphology, the uniform distribution of cutting load can be achieved, which greatly reduces the energy loss during the machining process and improves the machining efficiency compared with the three-body contact mode. The contact position and contact load between the abrasive and the workpiece have clear controllability, which avoids the uneven cutting depth caused by the random rolling and sliding of loose abrasives in the three-body contact. In addition, the micro-slip phenomenon at the contact interface can be effectively suppressed by optimizing the structure of the fixed abrasive. Furthermore, the HFCVD diamond coating has extremely high hardness, wear resistance, and surface flatness; it is not susceptible to abrasion, shedding, or chipping during cutting and can effectively reduce scratches, microcracks, and surface roughness fluctuations on the workpiece surface, enabling stable acquisition of higher machining accuracy. It eliminates the processes of preparation, circulation, and recovery of abrasive slurry; reduces environmental pollution caused by abrasive waste and slurry leakage; lowers the process cost and environmental governance pressure; and conforms to the development trend of green machining.