Polyurethane Recycling: Sustainable Development Perspectives and Innovative Approaches
Highlights
- Recent advances in catalytic depolymerization, bio-based polyols and NIPU chemistry support transition to circular life cycles.
- Hybrid strategies show promise for improving material recovery and reducing environmental impact.
- Mechanical recycling remains feasible but reduces mechanical and insulation performance.
- Chemical recycling enables recovery of polyols suitable for new polyurethane systems.
- Biological routes show potential for selective cleavage of urethane and ester linkages.
Abstract
1. Introduction
2. Chemistry of Polyurethanes
2.1. Chemical Characteristics of Polyols
2.1.1. Types of Polyols
- (1)
- anionic catalysts, such as NaOH, KOH, Ba(OH)2, Sr(OH)2, C16H30CaO4 or naphthenates;
- (2)
- cationic catalysts, including BF3, CF3SO3H, PF5 or SbF5;
- (3)
- coordination catalysts, such as Al(OR)3, Zn(OR)2, Ti(OR)4 or Zn3[Co(CN)6]3.
2.1.2. Bio-Based Polyols
2.1.3. Natural-Oil Polyols (NOPs)
- (1)
- hydrolysis, which increases the concentration of polar species such as glycerol and free fatty acids and leads to darkening;
- (2)
- oxidation, which alters the content of conjugated dienes and trienes;
- (3)
2.2. Characteristics of Isocyanates
2.2.1. Non-Isocyanate Polyurethanes (NIPUs)
2.2.2. Bio-Based Isocyanates: Sources and Industrial Availability
- Dimethyl nonadecanedioate and dimethyl tricosanedioate were synthesized via somerizing alkoxycarbonylation (Figure 11).
- These products were reduced to the corresponding diols.
- The diols were converted to bromides via an Appel II reaction. Finally, nucleophilic substitution yielded the target diisocyanates.
- The overall process yield reached up to 40%.
2.2.3. Lignin as a Renewable Source for Isocyanate Production
2.2.4. Amino-Acid-Based Isocyanates
2.2.5. Algae-Derived Isocyanates
2.2.6. Saccharide-Based Isocyanates
2.2.7. Cashew Nut Shell Liquid as an Alternative Feedstock for Isocyanate Synthesis
2.3. Additives for Polyurethanes
2.3.1. Blowing Agents
- (A)
- Physical Blowing Agents
- (B)
- Chemical Blowing Agents
2.3.2. Catalysts
Amine Catalysts
Metal Catalysts
2.3.3. Surfactants
2.3.4. Flame Retardants
| Flame Retardant | Chemical Name | Chemical Formula | Literature Reference |
|---|---|---|---|
| TDCPP | Tris(1,3-dichloro-2-propyl) phosphate | C9H15Cl6O4P | [108] |
| DMMP | Dimethyl methyl phosphonate | C3H9O3P | [109] |
| APP | Ammonium polyphosphate | (NH4PO3)n | [110] |
| EG | Expandable graphite | C (or C42(HSO4)(H2SO4)2) | [110] |
| AlPi | Aluminum diethyl phosphinate | C8H20O4P2Al | [111] |
| Flame Retardant | Chemical Name/Source | Reference |
|---|---|---|
| Alginic acid coating with hydroxyapatite (HAP) | Sodium alginate + hydroxyapatite | [112] |
| Lignin + sugarcane bagasse ash | Kraft lignin + bagasse ash | [113] |
| Kraft lignin | Kraft-type lignin | [114] |
| Bio-based melamine–formaldehyde resin | Glycerol/lignosulfonate-modified MF resin | [115] |
| Bio-polyol from rapeseed oil + phosphorus FR | Rapeseed-oil-based bio-polyol + DMMP | [116] |
Hydrogel Coatings
Halogenated Flame Retardants
Phosphorus-Based Flame Retardants
Additives for Flame Retardant Systems
2.4. Adhesion Promoters
- tack—instantaneous bonding ability enabling rapid adhesion and substrate wetting,
- shear adhesion—resistance to deformation and creep under shear stress,
- peel strength—the ability to be cleanly removed without leaving residues.
- (1)
- soft segments with low glass-transition temperatures (responsible for tack),
- (2)
- hard segments with defined melting transitions (providing cohesive strength),
- (3)
- crosslinking components, enabling structural integrity and controlled adhesion.
2.5. Thermal and UV Stabilizers
2.5.1. Thermal Stabilizers
2.5.2. UV Stabilizers
2.6. Plasticizers
2.7. Solvents
3. Applications of Polyurethane Materials
3.1. Clothing, Household Appliances and Automotive Applications
3.2. Civil Engineering
3.3. Electronics
3.4. Marine Sector Applications
3.5. Biomedical Applications
4. Polyurethanes—Environmental Aspects
Aging and Degradation of Polyurethane Materials
5. Recycling of Polyurethanes
5.1. Mechanical Recycling
- Rebonding—Flexible polyurethane foam is shredded into small particles and bonded to produce products such as sports mats and carpet underlays.
- Grinding or powdering—Finely ground PU waste is mixed with one of the original reactants, typically a polyol (up to approximately 30 wt%), to manufacture new polyurethane materials.
- Compression molding—Powdered PU waste is processed under elevated temperature and pressure, allowing the production of components containing up to nearly 100% recycled material [138].
5.1.1. Grinding and Reprocessing
5.1.2. Rebonding
5.1.3. Adhesive Pressing
5.1.4. Mechanical Processing Without Adhesives
5.1.5. Reaction Injection Molding (RIM)
5.1.6. Compression Molding
5.2. Energy Recovery
5.3. Thermochemical Recycling
5.3.1. Pyrolysis
5.3.2. Hydrogenation
5.4. Chemical Recycling of Polyurethanes
5.4.1. Glycolysis

5.4.2. Carbamate Aminolysis in Polyurethanes—Linear Carbonates
5.4.3. Hydrolysis
5.4.4. Hydro-Glycolysis
Acidolysis
5.5. Biological Degradation of Polyurethanes
5.6. Upcycling of Polyurethanes
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Franz, A.W.; Buchholz, S.; Albach, R.W.; Schmid, R. Towards Greener Polymers: Trends in the German Chemical Industry. Green Carbon 2024, 2, 33–44. [Google Scholar] [CrossRef]
- Kolluru, S.; Thakur, A.; Tamakuwala, D.; Kumar, V.V.; Ramakrishna, S.; Chandran, S. Sustainable Recycling of Polymers: A Comprehensive Review. Polym. Bull. 2024, 81, 9569–9610. [Google Scholar] [CrossRef]
- Porobic Katnic, S.; de Souza, F.M.; Gupta, R.K. Recent Progress in Enzymatic Degradation and Recycling of Polyurethanes. Biochem. Eng. J. 2024, 208, 109363. [Google Scholar] [CrossRef]
- Rossignolo, G.; Malucelli, G.; Lorenzetti, A. Recycling of Polyurethanes: Where We Are and Where We Are Going. Green Chem. 2024, 26, 1132–1152. [Google Scholar] [CrossRef]
- Simonini, L.; Sorze, A.; Maddalena, L.; Carosio, F.; Dorigato, A. Mechanical Reprocessing of Polyurethane and Phenolic Foams to Increase the Sustainability of Thermal Insulation Materials. Polym. Test. 2024, 138, 108539. [Google Scholar] [CrossRef]
- Kemona, A.; Piotrowska, M. Polyurethane Recycling and Disposal: Methods and Prospects. Polymers 2020, 12, 1752. [Google Scholar] [CrossRef]
- Gama, N.; Godinho, B.; Marques, G.; Silva, R.; Barros-Timmons, A.; Ferreira, A. Recycling of Polyurethane Scraps via Acidolysis. Chem. Eng. J. 2020, 395, 125102. [Google Scholar] [CrossRef]
- Fonseca, L.P.; Duval, A.; Luna, E.; Ximenis, M.; De Meester, S.; Avérous, L.; Sardon, H. Reducing the Carbon Footprint of Polyurethanes by Chemical and Biological Depolymerization: Fact or Fiction? Curr. Opin. Green Sustain. Chem. 2023, 41, 100802. [Google Scholar] [CrossRef]
- Gupta, R.K.; Kahol, P.K. Polyurethane Chemistry: Renewable Polyols and Isocyanates; ACS Symposium Series 1380; American Chemical Society: Washington, DC, USA, 2021. [Google Scholar] [CrossRef]
- Mehta, P.; Chidambaram, S.P.; Selwyn, W.T.; Borgohain, A. Technical Comparisons of Foams Using Various Blowing Agent Blends. In Proceedings of the Polyurethanes Technical Conference 2014, Dallas, TX, USA, 22–24 September 2014; pp. 162–171. Available online: https://www.proceedings.com/content/025/025500webtoc.pdf (accessed on 24 October 2025).
- Madbouly, S.A. Novel Recycling Processes for Thermoset Polyurethane Foams. Curr. Opin. Green Sustain. Chem. 2023, 42, 100835. [Google Scholar] [CrossRef]
- Silva, A.L.; Bordado, J.C. Recent Developments in Polyurethane Catalysis: Catalytic Mechanisms Review. Catal. Rev. 2004, 46, 31–51. [Google Scholar] [CrossRef]
- Zhu, M.; Ma, Z.; Liu, L.; Zhang, J.; Huo, S.; Song, P. Recent Advances in Fire-Retardant Rigid Polyurethane Foam. J. Mater. Sci. Technol. 2022, 112, 315–328. [Google Scholar] [CrossRef]
- Sanda, H.A.; Abubakar, M.A.; Abubakar, A.M.; Bashir, M.; Stojchevski, M. Utilization of Neem Seed Oil as Surfactant in the Production of Flexible and Rigid Polyurethane Foams. Emerg. Technol. Eng. J. 2024, 1, 40–52. [Google Scholar] [CrossRef]
- Ionescu, M. Chemistry and Technology of Polyols for Polyurethanes; Rapra Technology Limited: Shawbury, UK, 2005; Available online: https://books.google.pl/books?hl=pl&lr=&id=TnUGLC604XgC&oi=fnd&pg=PA1&dq=Ionescu,+M.+Chemistry+and+Technology+of+Polyols+for+Polyurethanes%3B+Rapra+Technology+Limited:+Shawbury,+Shrewsbury,+UK,+2005.&ots=jJGLWVcL6U&sig=_9FOMiLd5EQsVlphEi1coEL7QyQ&redir_esc=y#v=onepage&q=Ionescu%2C%20M.%20Chemistry%20and%20Technology%20of%20Polyols%20for%20Polyurethanes%3B%20Rapra%20Technology%20Limited%3A%20Shawbury%2C%20Shrewsbury%2C%20UK%2C%202005.&f=false (accessed on 24 October 2025).
- Domanska, A.; Boczkowska, A. Biodegradable Polyurethanes from Crystalline Prepolymers. Polym. Degrad. Stab. 2014, 108, 175–181. [Google Scholar] [CrossRef]
- Cregut, M.; Bedas, M.; Durand, M.J.; Thouand, G. New insights into polyurethane biodegradation and realistic prospects for the development of a sustainable waste recycling process. Biotechnol. Adv. 2013, 31, 1634–1647. [Google Scholar] [CrossRef] [PubMed]
- Akindoyo, J.O.; Beg, M.H.; Ghazali, S.; Islam, M.R.; Jeyaratnam, N.; Yuvaraj, A.R. Polyurethane Types, Synthesis and Applications—A Review. RSC Adv. 2016, 6, 114453–114482. [Google Scholar] [CrossRef]
- Chundawat, T.S.; Verma, N.; Vaya, D. Development in Synthesis and Coating Applications of Polyurethane. J. Chil. Chem. Soc. 2021, 66, 5142–5148. [Google Scholar] [CrossRef]
- Prociak, A.; Kurańska, M.; Malewska, E. Porous polyurethane plastics synthetized using bio-polyols from renewable raw materials. Polimery 2021, 62, 353–363. [Google Scholar] [CrossRef]
- Thomas, S.; Rane, A.V.; Kanny, K.; Abitha, V.K.; Thomas, M.G. Recycling of Polyurethane Foams ISBN: 978-0-323-51133-9 William Andrew, Applied Science Publishers. 2018. Available online: https://books.google.pl/books?hl=pl&lr=&id=8aRBDwAAQBAJ&oi=fnd&pg=PP1&dq=Recycling+of+Polyurethanes:+&ots=8e5_pV2UxK&sig=WsyWW0kbcpG3-dlPEeiVBJXgsxo&redir_esc=y#v=onepage&q=Recycling%20of%20Polyurethanes%3A&f=false (accessed on 9 February 2026).
- Szycher, M. Szycher’s Handbook of Polyurethanes; CRC Press: Boca Raton, FL, USA, 2013. [Google Scholar] [CrossRef]
- Muscat, A.; de Olde, E.M.; de Boer, I.J.M.; Ripoll-Bosch, R. The Battle for Biomass: A Systematic Review of Food–Feed–Fuel Competition. Glob. Food Secur. 2020, 25, 100330. [Google Scholar] [CrossRef]
- Hai, T.A.P.; de Backer, L.J.S.; Cosford, N.D.P.; Burkart, M.D. Renewable Polyurethanes from Sustainable Biological Precursors. Biomacromolecules 2021, 22, 1770–1794. [Google Scholar] [CrossRef]
- Hai, T.A.P.; Neelakantan, N.; Tessman, M.; Sherman, S.D.; Griffin, G.; Pomeroy, R.; Burkart, M.D. Flexible Polyurethanes, Renewable Fuels, and Flavorings from a Microalgae oil waste stream. Green Chem. 2020, 22, 3088–3094. [Google Scholar] [CrossRef]
- Saalah, S.; Abdullah, L.C.; Aung, M.M.; Salleh, M.Z.; Awang Biak, D.R.; Basri, M.; Osman Al Edrus, S.S. Chemical and Thermo-Mechanical Properties of Waterborne Polyurethane Dispersion derived from Jatropha Oil. Polymers 2021, 13, 795. [Google Scholar] [CrossRef] [PubMed]
- Lligadas, G.; Ronda, J.C.; Galia, M.; Cadiz, V. Renewable Polymeric Materials from Vegetable Oils: A Perspective. Mater. Today 2013, 16, 337–343. [Google Scholar] [CrossRef]
- Patil, C.K.; Jung, D.W.; Jirimali, H.D.; Baik, J.H.; Gite, V.V.; Hong, S.C. Nonedible Vegetable Oil-Based Polyols in Anticorrosive and Antimicrobial Polyurethane Coatings. Polymers 2021, 13, 3149. [Google Scholar] [CrossRef] [PubMed]
- Sharmin, E.; Ashraf, S.M.; Ahmad, S. Synthesis, characterization, antibacterial and corrosion protective properties of epoxies, epoxy-polyols and epoxy-polyurethane coatings from linseed and Pongamia glabra seed oils. Int. J. Biol. Macromol. 2007, 40, 407–422. [Google Scholar] [CrossRef]
- Mouren, A.; Avérous, L. Sustainable Cycloaliphatic Polyurethanes: From Synthesis to Applications. Chem. Soc. Rev. 2023, 52, 277–317. [Google Scholar] [CrossRef]
- Llevot, A.; Grau, E.; Carlotti, S.; Grelier, S.; Cramail, H. From Lignin-derived Aromatic Compounds to Novel Biobased Polymers. Macromol. Rapid Commun. 2016, 37, 9–28. [Google Scholar] [CrossRef]
- Kaur, R.; Singh, P.; Tanwar, S.; Varshney, G.; Yadav, S. Assessment of bio-based polyurethanes: Perspective on applications and bio-degradation. Macromol 2022, 2, 284–314. [Google Scholar] [CrossRef]
- Gallezot, P. Conversion of biomass to selected chemical products. Chem. Soc. Rev. 2012, 41, 1538–1558. [Google Scholar] [CrossRef]
- de Souza, F.M.; Kahol, P.K.; Gupta, R.K. Biomass-Derived Polyurethanes for Sustainable Future. In Advances in Green Synthesis: Avenues and Sustainability; Springer International Publishing: Cham, Switzerland. [CrossRef]
- Yang, S.K.; Mu, Y.X.; Li, D.; Li, T.; Lin, H.L.; Zhang, A.P.; Bian, J.; Chen, D.Q.; Yang, K.C. Degradable Thermoplastic Polyurethane Achieved through Dynamic Imine Bond-Containing Biobased Chain Extenders Incorporating to Regulate Microstructures. ACS Appl. Polym. Mater. 2025, 7, 2633–2643. [Google Scholar] [CrossRef]
- Santos, M.; Mariz, M.; Tiago, I.; Alarico, S.; Ferreira, P. Bio-Based Polyurethane Foams: Feedstocks, Synthesis, and Applications. Biomolecules 2025, 15, 680. [Google Scholar] [CrossRef]
- Prociak, A.; Kurańska, M.; Cabulis, U.; Ryszkowska, J.; Leszczyńska, M.; Uram, K.; Kirpluks, M. Effect of bio-polyols with different chemical structures on foaming of polyurethane systems and foam properties. Ind. Crop. Prod. 2018, 120, 262–270. [Google Scholar] [CrossRef]
- Kurańska, M.; Prociak, A.; Cabulis, U.; Kirpluks, M. Water-blown polyurethane-polyisocyanurate foams based on bio-polyols with wood fibers. Polimery 2015, 60, 705–712. [Google Scholar] [CrossRef]
- Amri, M.R.; Al-Edrus, S.S.O.; Guan, C.T.; Yasin, F.M.; Hua, L.S. Jatropha Oil as a Substituent for Palm Oil in Biobased Polyurethane. Int. J. Polym. Sci. 2021, 2021, 6655936. [Google Scholar] [CrossRef]
- Sołtysiński, M.; Piszczek, K.; Romecki, D.; Narożniak, S.; Tomaszewska, J.; Skórczewska, K. Conversion of polyurethane technological foam waste and post-consumer polyurethane mattresses into polyols—Industrial applications. Polimery 2018, 63, 234–238. [Google Scholar] [CrossRef]
- Onn, M.; Jalil, M.J.; Mohd Yusoff, N.I.S.; Edward, E.B.; Wahit, M.U. A Comprehensive Review on Chemical Route to Convert Waste Cooking Oils to Renewable Polymeric Materials. Ind. Crop. Prod. 2024, 211, 118194. [Google Scholar] [CrossRef]
- Dworakowska, S.; Bogdal, D.; Prociak, A. Microwave-Assisted Synthesis of Polyols from Rapeseed Oil and Properties of Flexible Polyurethane Foams. Polymers 2012, 4, 1462–1477. [Google Scholar] [CrossRef]
- Akdogan, E.; Erdem, M. A comprehensive research of low-density bio-based rigid polyurethane foams from sugar beet pulp-based biopolyol: From closed-cell towards open-cell structure. Ind. Crop. Prod. 2023, 200, 116809. [Google Scholar] [CrossRef]
- Rajput, B.S.; Forman, A.; Halloran, M.W.; Phung Hai, T.A.; Scofield, G.B.; Burkart, M.D. Variation of Aliphatic Diisocyanates in Bio-Based TPUs. Macromolecules 2023, 56, 8813 8822. [Google Scholar] [CrossRef]
- Petrović, Z.S.; Wan, X.; Bilić, O.; Zlatanić, A.; Hong, J.; Javni, I.; Degruson, D. Polyols and Polyurethanes from Crude Algal Oils. J. Am. Oil Chem. Soc. 2013, 90, 1073–1078. [Google Scholar] [CrossRef]
- Staroń, A. Composite Materials Based on Waste Cooking Oil for Construction Applications. Buildings 2023, 13, 994. [Google Scholar] [CrossRef]
- Gama, N.; Ferreira, A.; Barros-Timmons, A. Polyurethane Foams: Past, Present, and Future. Materials 2018, 11, 1841. [Google Scholar] [CrossRef] [PubMed]
- Gaikwad, M.S.; Gite, V.V.; Mahulikar, P.P.; Hundiwale, D.G.; Yemul, O.S. Eco-friendly polyurethane coatings from cottonseed and karanja oil. Prog. Org. Coat. 2015, 86, 16. [Google Scholar] [CrossRef]
- American Chemistry Council. Polyurethane Applications and Benefits. Available online: https://www.americanchemistry.com (accessed on 24 October 2025).
- Delavarde, A.; Savin, G.; Derkenne, P.; Boursier, M.; Morales-Cerrada, R.; Nottelet, B. Sustainable polyurethanes: Toward new cutting-edge opportunities. Prog. Polym. Sci. 2024, 151, 101805. [Google Scholar] [CrossRef]
- Kausar, A. Polyurethane Nanocomposite Coatings: State of the Art and Perspectives. Polym. Int. 2018, 67, 1470–1477. [Google Scholar] [CrossRef]
- Camadanli, S.; Hisir, A.; Dural, S. Synthesis and Performance of Moisture-Curable Solvent-Free Silane-Terminated Polyurethanes for Coating and Sealant Applications. J. Appl. Polym. Sci. 2022, 139, 51722. [Google Scholar] [CrossRef]
- Agrawal, A.; Kaur, R.; Walia, R.S. PU foam derived from renewable sources: Perspective on properties enhancement: An overview. Eur. Polym. J. 2017, 95, 255–274. [Google Scholar] [CrossRef]
- National Institute for Occupational Safety and Health (NIOSH). Safer Choice Program. Available online: https://www.epa.gov/saferchoice (accessed on 24 October 2025).
- Stachak, P.; Łukaszewska, I.; Hebda, E.; Pielichowski, K. Recent Advances in Fabrication of Non-Isocyanate Polyurethane-Based Composite Materials. Materials 2021, 14, 3497. [Google Scholar] [CrossRef]
- Engels, H.W.; Pirkl, H.G.; Albers, R.; Albach, R.W.; Krause, J.; Hoffmann, A.; Dormish, J. Polyurethanes: Versatile materials and sustainable problem solvers for today’s challenges. Angew. Chem. Int. Ed. 2013, 52, 9422–9441. [Google Scholar] [CrossRef]
- Bähr, M.; Bitto, A.; Mülhaupt, R. Cyclic limonene dicarbonate as a new monomer for non-isocyanate oligo-and polyurethanes (NIPU) based upon terpenes. Green Chem. 2012, 14, 1447–1454. [Google Scholar] [CrossRef]
- Vanaraj, R.; Suresh Kumar, S.M.; Kim, S.C.; Santhamoorthy, M. A Review on Sustainable Upcycling of Plastic Waste through Depolymerization into High-Value Monomer. Processes 2025, 13, 2431. [Google Scholar] [CrossRef]
- Grand View Research. Polyurethane Market Size, Share and Trends Analysis Report By Product (Rigid Foam, Flexible Foam), By Application (Construction, Furniture and Interiors), By Region, And Segment Forecasts, 2022–2030. Available online: https://www.grandviewresearch.com/industry-analysis/polyurethane-pu-market (accessed on 24 October 2025).
- Xia, L.; Gui, T.; Wang, J.; Tian, H.; Wang, Y.; Ning, L.; Wu, L. Bio-Based Coatings: Progress, Challenges and Future Perspectives. Polymers 2025, 17, 3266. [Google Scholar] [CrossRef] [PubMed]
- Vencorex. Tolonate™ X FLO 100 Product Information. Available online: https://www.vencorex.com (accessed on 24 October 2025).
- Grdadolnik, M.; Zdovc, B.; Drinčić, A.; Onder, O.C.; Utroša, P.; Ramos, S.G. Chemical Recycling of Flexible Polyurethane Foams by Aminolysis to Recover High-Quality Polyols. ACS Sustain. Chem. Eng. 2023, 11, 10864–10873. [Google Scholar] [CrossRef] [PubMed]
- ICE-Belgium. STABIO™ Polyisocyanates—Technical Data Sheet. Available online: https://cms.ice.be/files/249/tds-stabio-presentation-.pdf (accessed on 24 October 2025).
- Desai, Y.; Jariwala, S.; Gupta, R.K. Polyurethanes: Preparation, Properties, and Applications Volume 2: Advanced Applications. American Chemical Society; ACS Publications: Washington, DC, USA, 2023; Volume 1453. [Google Scholar] [CrossRef]
- Acik, G.; Karabulut, H.R.F.; Altinkok, C.; Karatavuk, A.O. Synthesis and characterization of biodegradable polyurethanes made from cholic acid and l-lysine diisocyanate ethyl ester. Polym. Degrad. Stab. 2019, 165, 43–48. [Google Scholar] [CrossRef]
- Niesiobędzka, J.; Datta, J. Challenges and recent advances in bio-based isocyanate production. Green Chem. 2023, 25, 2482–2504. [Google Scholar] [CrossRef]
- Fu, C.; Zheng, Z.; Yang, Z.; Chen, Y.; Shen, L. A fully bio-based waterborne polyurethane dispersion from vegetable oils: From synthesis of precursors by thiol-ene reaction to study of final material. Prog. Org. Coat. 2014, 77, 53–60. [Google Scholar] [CrossRef]
- Srivastava, A.; Maity, S.; Das, B.R. A review on multi-functional polyurethane (PU) coatings for fabric applications: Materials, processes and recent developments. Prog. Org. Coat. 2025, 207, 109377. [Google Scholar] [CrossRef]
- Hojabri, L.; Kong, X.; Narine, S.S. Functional thermoplastics from linear diols and diisocyanates produced entirely from renewable lipid sources. Biomacromolecules 2010, 11, 911–918. [Google Scholar] [CrossRef]
- Cifarelli, A.; Boggioni, L.; Vignali, A.; Tritto, I.; Bertini, F.; Losio, S. Flexible polyurethane foams from epoxidized vegetable oils and a bio-based diisocyanate. Polymers 2021, 13, 612. [Google Scholar] [CrossRef]
- Akay, O.; Altinkok, C.; Acik, G.; Yuce, H.; Ege, G.K. A bio-based and non-toxic polyurethane film derived from Luffa cylindrica cellulose and L-Lysine diisocyanate ethyl ester. Eur. Polym. J. 2021, 161, 110856. [Google Scholar] [CrossRef]
- Laurichesse, S.; Avérous, L. Chemical modification of lignins: Towards biobased polymers. Prog. Polym. Sci. 2014, 39, 1266–1290. [Google Scholar] [CrossRef]
- Chaoqun, Z.; Madbouly, S.A.; Kessler, M.R. Biobased Polyurethanes Prepared from Different Vegetable Oils. ACS Appl. Mater. Interfaces 2015, 7, 1226–1233. [Google Scholar] [CrossRef]
- Das, A.; Mahanwar, P. A brief discussion on advances in polyurethane applications. Adv. Ind. Eng. Polym. Res. 2020, 3, 93–101. [Google Scholar] [CrossRef]
- SI Group. Antioxidant Solutions for Polyurethanes and Polyols. Available online: https://discover.univarsolutions.com (accessed on 29 October 2025).
- Bhutra, K.; Datta, S.; More, A.P. A Comprehensive Review on Biobased Hyperbranched Polymers. Polym. Bull. 2024, 81, 12287–12345. [Google Scholar] [CrossRef]
- Makaveckas, T.; Šimonėlienė, A.; Šipailaitė-Ramoškienė, V. Lignin Valorization from Lignocellulosic Biomass. Extraction, Depolymerization, and Applications in the Circular Bioeconomy. Sustainability 2025, 17, 9913. [Google Scholar] [CrossRef]
- Georgs, V.; Piili, H.; Gustafsson, J.; Xu, C. A critical review on lignin structure, chemistry, and modification towards utilisation in additive manufacturing of lignin-based composites. Ind. Crop. Prod. 2025, 233, 121416. [Google Scholar] [CrossRef]
- Dandash, A.A.; Abu-Jdayil, B.; Tannous, J.H. Lignin Extraction from Various Biomass Sources: A Comprehensive Review of Characteristics, Applications, and Future Prospects. Chem. Rec. 2025, 25, 2500045. [Google Scholar] [CrossRef]
- El Bouhali, A.; Gnanasekar, P.; Habibi, Y. Chemical modifications of lignin. In Lignin-Based Materials for Biomedical Applications: Preparation, Characterization, and Implementation; Elsevier: Amsterdam, The Netherlands, 2021; pp. 159–194. [Google Scholar] [CrossRef]
- Glasser, W.G.; Hsu, O.H.; Reed, D.L.; Forte, R.C.; Wu, L.F. Lignin-Derived Polyols, Polyisocyanates, and Polyurethanes. In Urethane Chemistry and Applications; ACS Symposium Series 172; ACS Publications: Washington, DC, USA, 1981; pp. 311–338. [Google Scholar] [CrossRef]
- Watkins, D.; Nuruddin, M.; Hosur, M.; Tcherbi-Narteh, A.; Jeelani, S. Extraction and characterization of lignin from different biomass resources. J. Mater. Res. Technol. 2015, 4, 26–32. [Google Scholar] [CrossRef]
- Kuhire, S.S.; Nagane, S.S.; Wadgaonkar, P.P. Poly(ether urethane)s from aromatic diisocyanates based on lignin-derived phenolic acids. Polym. Int. 2017, 66, 892–899. [Google Scholar] [CrossRef]
- Thakur, V.K.; Thakur, M.K. Recent advances in green hydrogels from lignin: A review. Int. J. Biol. Macromol. 2015, 72, 834–847. [Google Scholar] [CrossRef]
- Sun, R.C. Lignin source and structural characterization. ChemSusChem 2020, 13, 4385–4393. [Google Scholar] [CrossRef]
- Konieczny, J.; Loos, K. Green Polyurethanes from Renewable Isocyanates and Biobased White Dextrins. Polymers 2019, 11, 256. [Google Scholar] [CrossRef]
- Emrani, J.; Benrashid, R.; Mohtarami, S.; Fini, E.; Abu-Lebdeh, T. Synthesis and Characterization of Bio-based Polyurethane Polymers. Am. J. Eng. Appl. Sci. 2018, 11, 1298–1309. [Google Scholar] [CrossRef]
- Guan, J.; Sacks, M.S.; Beckman, E.J.; Wagner, W.R. Synthesis, Characterization, and Cytocompatibility of Elastomeric, Biodegradable Poly(ester-urethane)ureas based on poly(caprolactone) and putrescine. J. Biomed. Mater. Res. 2002, 61, 493–503. [Google Scholar] [CrossRef] [PubMed]
- Atmaca, U. A novel approach for the synthesis of β-keto esters: One-pot reaction of carboxylic acids with chlorosulfonyl isocyanate. Arkivoc 2020, 6, 220–227. [Google Scholar] [CrossRef]
- Błażek, K.; Datta, J. Renewable natural resources as green alternative substrates to obtain bio-based non-isocyanate polyurethanes-review. Crit. Rev. Environ. Sci. Technol. 2019, 49, 173–211. [Google Scholar] [CrossRef]
- Belgacem, M.N.; Gandini, A. Chapter 18—Surface Modification of Cellulose Fibres. In Monomers, Polymers and Composites from Renewable Resources; Elsevier Science: Amsterdam, The Netherlands, 2008. [Google Scholar] [CrossRef]
- Gandini, A. Polymers from Renewable Resources: A Challenge for the Future of Macromolecular Materials. Macromolecules 2008, 41, 9491–9504. [Google Scholar] [CrossRef]
- Mosiewicki, M.A.; Aranguren, M.I. A short review on novel biocomposites based on plant oil precursors. Eur. Polym. J. 2013, 49, 1243–1256. [Google Scholar] [CrossRef]
- Garcon, R.; Clerk, C.; Gesson, J.P.; Bordado, J.; Nunes, T.; Caroco, S.; Rauter, A.P. Synthesis of novel polyurethanes from sugars and 1, 6-hexamethylene diisocyanate. Carbohydr. Polym. 2001, 45, 123–127. [Google Scholar] [CrossRef]
- Zenner, M.D.; Xia, Y.; Chen, J.S.; Kessler, M.R. Polyurethanes from isosorbide-based diisocyanates. ChemSusChem 2013, 6, 1182–1185. [Google Scholar] [CrossRef]
- Brzoska, J.; Smorawska, J.; Głowińska, E.; Datta, J. A green route for high-performance bio-based polyurethanes synthesized from modified bio-based isocyanates. Ind. Crop. Prod. 2024, 222, 1182. [Google Scholar] [CrossRef]
- Jia, P.Y.; Hu, L.H.; Shang, Q.Q.; Wang, R.; Zhang, M.; Zhou, Y. Self-Plasticization of PVC materials viachemical modification of mannich base of cardanol butyl ether. ACS Sustain. Chem. Eng. 2017, 5, 6665–6673. [Google Scholar] [CrossRef]
- Nair, C.P.R.; Bindu, R.L.; Joseph, V.C. Cyanate esters based on cardanol modified-phenol-formaldehyde resins: Syntheses and thermal characteristics. J. Polym. Sci. Part A Polym. Chem. 1995, 33, 621–627. [Google Scholar] [CrossRef]
- Sandhya, T.E. Synthesis and Characterization of Aliphatic-Aromatic Polyesters. Ph.D. Thesis, University of Pune, Pune, India, 2003. Available online: https://www.swaminathansivaram.in/publications/phdthesis/th1380SandhyaTE.pdf (accessed on 24 October 2025).
- Baron, A.; Rodriguez-Hernandez, J.; Ibarboure, E.; Derail, C.; Papon, E. Adhesives based on polyurethane graft multiblock copolymers: Tack, rheology and first morphological analyses. Int. J. Adhes. Adhes. 2009, 29, 1–8. [Google Scholar] [CrossRef]
- Brzeska, J.; Piotrowska-Kirschling, A. A Brief Introduction to the Polyurethanes According to the Principles of Green Chemistry. Processes 2021, 9, 1929. [Google Scholar] [CrossRef]
- Godinho, B.; Gama, N.; Barros-Timmons, A.; Ferreira, A. Recycling of different types of polyurethane foam wastes via acidolysis to produce polyurethane coatings. Sustain. Mater. Technol. 2021, 29, e00330. [Google Scholar] [CrossRef]
- Azobuild. Blowing Agents for Polyurethane Foams. Available online: https://www.azobuild.com (accessed on 24 October 2025).
- Pantone, V.; Annese, C.; Fusco, C.; Fini, P.; Nacci, A.; Russo, A.; D’Accolti, L. One-Pot Conversion of Epoxidized Soybean Oil (ESO) into Soy-Based Polyurethanes by MoCl2O2 Catalysis. Molecules 2017, 22, 333. [Google Scholar] [CrossRef]
- Hebda, E.; Bukowczan, A.; Michałowski, S.; Pielichowski, K. Flexible Polyurethane Foams Reinforced by Functionalized Polyhedral Oligomeric Silsesquioxanes: Structural Characteristics and Evaluation of Thermal/Flammability Properties. Polymers 2022, 14, 4743. [Google Scholar] [CrossRef]
- Silva, R.; Barros-Timmons, A.; Quinteiro, P. Life cycle assessment of fossil-and bio-based polyurethane foams: A review. J. Clean. Prod. 2023, 430, 139697. [Google Scholar] [CrossRef]
- Luo, Y.; Geng, Z.; Zhang, W.; He, J.; Yang, R. Strategy for constructing phosphorus-based flame-retarded polyure-thane elastomers for advanced performance in long-term. Polymers 2023, 15, 3711. [Google Scholar] [CrossRef]
- Wang, X.; Song, L.; Hu, Y. Phosphorus-based flame retardants for polyurethanes: Synthesis and mechanistic studies. In Materials and Chemistry of Flame-Retardant Polyurethanes Volume 2: Green Flame Retardants; American Chemical Society: Washington, DC, USA, 2021; pp. 161–187. [Google Scholar] [CrossRef]
- Tang, G.; Liu, X.; Zhou, L.; Zhang, P.; Deng, D.; Jiang, H. Steel slag waste combined with melamine pyrophosphate as a flame retardant for rigid polyurethane foams. Adv. Powder Technol. 2020, 31, 279–286. [Google Scholar] [CrossRef]
- Ma, Z.; Zhang, J.; Liu, L.; Zheng, H.; Dai, J.; Tang, L.C.; Song, P. A highly fire-retardant rigid polyurethane foam capable of fire-warning. Compos. Commun. 2022, 29, 101046. [Google Scholar] [CrossRef]
- Li, J.-L.; Gao, C.T.; Sun, X.; Peng, S.G.; Wang, Y.W.; Qin, S.H. Synergistic flame-retardant effects of aluminum diethyl phosphinate in PP/IFR system and the Flame-Retardant Mechanism. Int. Polym. Process. 2023, 36, 519–528. [Google Scholar] [CrossRef]
- Nabipour, H.; Wang, X.; Song, L.; Hu, Y. A fully bio-based coating made from alginate, chitosan and hydroxyapatite for protecting flexible polyurethane foam from fire. Carbohydr. Polym. 2020, 246, 116641. [Google Scholar] [CrossRef]
- Vega-Baudrit, J.; Delgado-Montero, K.; Madrigal-Carballo, S. Biodegradable polyurethanes from sugar cane biowastes. Cellul. Chem. Technol. 2012, 45, 507–514. [Google Scholar]
- Xiao, T.; Wu, L.; Xu, Q.; Yu, X.; Fu, Q.; Zhang, F.; Li, Y.; Yin, G.; Huang, L.; Fatehi, P.; et al. Highly-Flame-Retardant Performance and Sustainable Polyurethane Foams from Industrial Kraft Lignin via Exploiting Lignin Demethylation. Biomacromolecules 2025, 26(10), 6444–6457. [Google Scholar] [CrossRef]
- Wang, Y.; Zheng, X.; Jiang, K.; Han, D.; Zhang, Q. Bio-based melamine formaldehyde resins for flame-retardant polyurethane foams. Int. J. Biol. Macromol. 2024, 273, 132836. [Google Scholar] [CrossRef]
- Zemła, M.; Prociak, A.; Michałowski, S. Bio-Based Rigid Polyurethane Foams Modified with Phosphorus Flame Retardants. Polymers 2022, 14, 102. [Google Scholar] [CrossRef]
- Vahabi, H.; Gholami, F.; Tomas, M.; Movahedifar, E.; Yazdi, M.K.; Saeb, M.R. Hydrogel and aerogel-based flame-retardant polymeric materials: A review. J. Vinyl Addit. Technol. 2024, 30, 5–25. [Google Scholar] [CrossRef]
- Heidarian, M.; Shishesaz, M.R.; Kassiriha, S.M.; Nematollahi, M. Characterization of structure and corrosion resistivity of polyurethane/organoclay nanocomposite coatings prepared through an ultrasonication assisted process. Prog. Org. Coat. 2010, 68, 180–188. [Google Scholar] [CrossRef]
- Fuensanta, M.; Martín-Martínez, J.M. Thermoplastic polyurethane pressure sensitive adhesives made with mixtures of polypropylene glycols of different molecular weights. Int. J. Adhes. Adhes. 2019, 88, 81–90. [Google Scholar] [CrossRef]
- Akram, N.; Gurney, R.S.; Zuber, M.; Ishaq, M.; Keddie, J.L. Influence of polyol molecular weight and type on the tack and peel properties of waterborne polyurethane pressure-sensitive adhesives. Macromol. React. Eng. 2013, 7, 493–503. [Google Scholar] [CrossRef]
- Yousif, E.; Haddad, R. Photodegradation and photostabilization of polymers, especially polystyrene. SpringerPlus 2013, 2, 398. [Google Scholar] [CrossRef] [PubMed]
- Wellt Chemicals. Hindered Amine Light Stabilizer (HALS): Protecting Polymers from Degradation. Available online: https://welltchemicals.com (accessed on 24 October 2025).
- Wypych, G. Handbook of UV Degradation and Stabilization, 3rd ed.; ChemTec Publishing: Toronto, ON, Canada, 2020; Available online: https://books.google.pl/books?hl=pl&lr=&id=NL_VDwAAQBAJ&oi=fnd&pg=PA1&dq=Wypych,+G.+Handbook+of+UV+Degradation+and+Stabilization,+2nd+ed.%3B+ChemTec+Publishing:+Toronto,+Canada,+&ots=xUz7Q8URut&sig=IT1_q9NoqwKIIapCQRE5hNjByPo&redir_esc=y#v=onepage&q&f=false (accessed on 24 October 2025).
- Rahman, M.; Brazel, C.S. The plasticizer market: An assessment of traditional plasticizers and research trends to meet new challenges. Prog. Polym. Sci. 2004, 29, 1223–1248. [Google Scholar] [CrossRef]
- Hepburn, C. Polyurethane Elastomers; Elsevier Science: Amsterdam, The Netherlands, 1992. [Google Scholar] [CrossRef]
- Cognard, P. Handbook of Adhesives and Sealants: Basic Concepts and High Tech Bonding; Elsevier: Amsterdam, The Netherlands, 2005; Volume 1, Available online: https://books.google.pl/books?hl=pl&lr=&id=9XFD8ZsZzAsC&oi=fnd&pg=PR1&dq=Handbook+of+Adhesives+and+Sealants&ots=-rOhTUMCBl&sig=L1foLTEGSdR27MUteV4byVkmrG4&redir_esc=y#v=onepage&q=Handbook%20of%20Adhesives%20and%20Sealants&f=false (accessed on 24 October 2025).
- Nguyễn, T. Organic Coatings. In Handbook of Materials Selection; Myer, K., Ed.; John Wiley & Sons: New York, NY, USA, 2002; pp. 915–942. Available online: https://www.academia.edu/37888258/Organic_Coatings (accessed on 24 October 2025).
- Wang, C.; Murugadoss, V.; Kong, J.; He, Z.; Mai, X.; Shao, Q.; Guo, Z. Overview of carbon nanostructures and nano-composites for electromagnetic wave shielding. Carbon 2018, 140, 696–733. [Google Scholar] [CrossRef]
- Cui, J.; Xu, J.; Li, J.; Qiu, H.; Zheng, S.; Yang, J. A crosslinkable graphene oxide in waterborne polyurethane anticorrosive coatings: Experiments and simulation. Compos. Part B Eng. 2020, 188, 107889. [Google Scholar] [CrossRef]
- Chattopadhyay, D.K.; Raju, K.V.S.N. Structural Engineering of Polyurethane coatings for high performance applica-tions. Prog. Polym. Sci. 2007, 32, 352–418. [Google Scholar] [CrossRef]
- Li, Y.; Luo, X.; Hu, S. Bio-Based Polyols and Polyurethanes; Springer: Cham, Switzerland, 2015. [Google Scholar] [CrossRef]
- Chaudhary, M.L.; Gupta, R.K. Environmental and Health Concerns in Polyurethane. In Non-Isocyanate Polyurethanes: Chemistry, Progress, and Challenges; American Chemical Society: Washington, DC, USA, 2025; pp. 15–30. [Google Scholar] [CrossRef]
- Morales-Cerrada, R.; Tavernier, R.; Caillol, S. Fully Bio-Based Thermosetting Polyurethanes from Bio-Based Polyols and Isocyanates. Polymers 2021, 13, 1255. [Google Scholar] [CrossRef]
- Simon, J.; Barla, F.; Kelemen-Haller, A.; Farkas, F.; Kraxner, M. Thermal stability of polyurethanes. Chromatographia 1988, 25, 99–106. [Google Scholar] [CrossRef]
- Simón, D.; Borreguero, A.M.; De Lucas, A.; Rodríguez, J.F. Recycling of Polyurethanes from Laboratory to Industry, a Journey towards the Sutainability. Waste Manag. 2018, 76, 147–171. [Google Scholar] [CrossRef]
- Datta, J.; Głowińska, E.; Włoch, M. Mechanical Recycling via Regrinding, Rebonding, Adhesive Pressing and Mold-ing. In Recycling of Polyurethane Foams; William Andrew Publishing: Norwich, NY, USA, 2018; pp. 57–65. [Google Scholar] [CrossRef]
- Guo, L.; Wang, W.; Guo, X. Recycling of flexible polyurethane foams by regrinding scraps into powder to replace polyol for re-foaming. Materials 2022, 15, 6047. [Google Scholar] [CrossRef]
- de la Cruz-Martínez, F.; Martínez de Sarasa Buchaca, M.; Martínez, J.; Fernández-Baeza, J.; Sánchez-Barba, L.F.; Rodríguez-Diéguez, A.; Lara-Sánchez, A. Synthesis of bio-derived cyclic carbonates from renewable resources. ACS Sustain. Chem. Eng. 2019, 7, 20126–20138. [Google Scholar] [CrossRef]
- North, M.; Pasquale, R.; Young, C. Synthesis of cyclic carbonates from epoxides and carbon dioxide. Green Chem. 2010, 12, 1514–1539. [Google Scholar] [CrossRef]
- Šooš, Ľ.; Matúš, M.; Legutko, S.; Bábics, J. Research into Efficient Technology for Material Recovery of Waste Polyu-rethane Foams. Recycling 2025, 10, 107. [Google Scholar] [CrossRef]
- Shaikh, A.A.G.; Sivaram, S. Organic carbonates. Chem. Rev. 1996, 96, 951–976. [Google Scholar] [CrossRef]
- Pęczek, E.; Pamuła, R.; Białowiec, A. Recycled Waste as Polyurethane Additives or Fillers. Materials 2024, 17, 1013. [Google Scholar] [CrossRef]
- Deng, Y.; Dewil, R.; Appels, L.; Ansart, R.; Baeyens, J.; Kang, Q. Reviewing the thermo-chemical recycling of waste polyurethane foam. J. Environ. Manag. 2021, 278, 111527. [Google Scholar] [CrossRef]
- Peti, D.; Dobránsky, J.; Michalík, P. Recent Advances in Polymer Recycling: A Review of Chemical and Biological Processes for sustainable solutions. Polymers 2025, 17, 603. [Google Scholar] [CrossRef]
- Horváth, T.; Kecskés, K.; Jordán Csábrádiné, A.; Szőri-Dorogházi, E.; Viskolcz, B.; Szőri, M. Searching for the Achil-les’ Heel of Urethane Linkage—An Energetic Perspective. Polymers 2024, 16, 1126. [Google Scholar] [CrossRef]
- Zhou, W.; Neumann, P.; Al Batal, M.; Rominger, F.; Hashmi, A.S.K.; Schaub, T. Depolymerization of Technical Grade Polyamide 66 and Polyurethane Materials via Hydrogenation. ChemSusChem 2020, 14, 4176–4180. [Google Scholar] [CrossRef]
- Ragaert, K.; Delva, L.; Van Geem, K. Mechanical and Chemical Recycling of Solid Plastic Waste. Waste Manag. 2017, 69, 24–58. [Google Scholar] [CrossRef]
- Wieczorek, K.; Bukowski, P.; Stawiński, K.; Ryłko, I. Recycling of Polyurethane Foams via Glycolysis: A Review. Materials 2024, 17, 4617. [Google Scholar] [CrossRef]
- Siddiqui, M.N.; Redhwi, H.H.; Achilias, D.S. Recycling of poly(ethylene terephthalate) waste through methanolic pyrolysis in a microwave reactor. J. Anal. Appl. Pyrolysis 2012, 98, 214–220. [Google Scholar] [CrossRef]
- Lalhmangaihzuala, S.; Laldinpuii, Z.; Lalmuanpuia, C.; Vanlaldinpuia, K. Glycolysis of poly(ethylene terephthalate) using biomass-waste derived recyclable heterogeneous catalyst. Polymers 2020, 13, 37. [Google Scholar] [CrossRef] [PubMed]
- Ghaderian, A.; Haghighi, A.H.; Taromi, F.A.; Abdeen, Z.; Boroomand, A.; Taheri, S.M.R. Characterization of Rigid Polyurethane Foam Prepared from Recycling of PET Waste. Period. Polytech. Chem. Eng. 2015, 59, 296–305. [Google Scholar] [CrossRef]
- Senra, E.M.; Silva, A.L.; Pacheco, E.B. A review of waterborne polyurethane coatings and adhesives with polyester polyol from poly(ethylene terephthalate) waste. J. Polym. Environ. 2023, 31, 3719–3739. [Google Scholar] [CrossRef]
- Al-Sabagh, A.M.; Yehia, F.Z.; Eshaq, G.; Rabie, A.M.; ElMetwally, A.E. Greener Routes for Recycling of Poly(ethylene terephthalate). Egypt. J. Pet. 2016, 25, 53–64. [Google Scholar] [CrossRef]
- Grignard, B.; Thomassin, J.M.; Gennen, S.; Poussard, L.; Bonnaud, L.; Raquez, J.M.; Detrembleur, C. CO2-Blown Microcellular Non-Isocyanate Polyurethane (NIPU) Foams from bio-and CO2-sourced monomers to potentially thermal insulating materials. Green Chem. 2016, 18, 2206–2215. [Google Scholar] [CrossRef]
- Bakkali-Hassani, C.; Berne, D.; Ladmiral, V.; Caillol, S. Transcarbamoylation in polyurethanes: Underestimated exchange reactions. Macromolecules 2022, 55, 7974–7991. [Google Scholar] [CrossRef]
- Kathalewar, M.S.; Joshi, P.B.; Sabnis, A.S.; Malshe, V.C. Non-Isocyanate Polyurethanes: From Chemistry to Applications. Rsc Adv. 2013, 3, 4110–4129. [Google Scholar] [CrossRef]
- Das, S.; Klinedinst, D.B.; Yilgor, I.; Beyer, F.L.; Toki, S.; Hsiao, B.S.; Wilkes, G.L. Structure-Property Relationships of Segmented Polyurethanes and Polyureas Based on Single Isocyanate Molecules as Hard Segments. Polym. Prepr. 2007, 48, 849. Available online: https://www.researchgate.net/profile/Iskender-Yilgor/publication/296098818_Structure-property_relationships_of_segment-ed_polyurethanes_and_polyureas_based_on_single_isocyanate_molecules_as_hard_segments/links/56e7e69308aeb6f93f54d343/Structure-property-relationships-of-segmented-polyurethanes-and-polyureas-based-on-single-isocyanate-molecules-as-hard-segments.pdf (accessed on 24 October 2025).
- Kirchberg, A.; Khabazian Esfahani, M.; Röpert, M.C.; Wilhelm, M.; Meier, M.A. Sustainable Synthesis of Non-Isocyanate Polyurethanes Based on Renewable 2,3-Butanediol. Macromol. Chem. Phys. 2022, 223, 2200010. [Google Scholar] [CrossRef]
- El Mejjatti, A.; Harit, T.; Riahi, A.; Khiari, R.; Bouabdallah, I.; Malek, F. Chemical Recycling of Polyethylene Terephthalate: Application to the Synthesis of Multiblock Copolyesters. Express Polym. Lett. 2014, 8, 544–553. [Google Scholar] [CrossRef]
- Warsahartana, H.; Bashir, A.; Keyworth, A.; Davies, R.; Falkowska, M.; Asuquo, E.; Garforth, A. Catalytic Steam Hydrolysis of Polyethylene Terephthalate to Terephthalic Acid Followed by Repolymerisation. Chem. Eng. Trans. 2023, 100, 445–450. [Google Scholar] [CrossRef]
- Jia, Z.; Gao, L.; Qin, L.; Yin, J. Chemical recycling of PET to value-added products. RSC Sustain. 2023, 1, 2135–2147. [Google Scholar] [CrossRef]
- Mudondo, J.; Lee, H.S.; Jeong, Y.; Kim, T.H.; Kim, S.; Sung, B.H.; Kim, H.T. Recent advances in the chemobiological upcycling of polyethylene terephthalate (PET) into value-added chemicals. J. Microbiol. Biotechnol. 2022, 33, 1–14. [Google Scholar] [CrossRef]
- Salas, R.; Villa, R.; Velasco, F.; Macia, M.; Navarro, V.; Dupont, J.; Lozano, P. On the Hydrolytic Depolymerization of Polyurethane Foam Waste by Ionic Liquids. Molecules 2025, 30, 3523. [Google Scholar] [CrossRef]
- Nikje, M.M.A.; Nikrah, M.; Mohammadi, F.H.A. Microwave-assisted Polyurethane Bond Cleavage via Hydroglycolysis Process at atmospheric pressure. J. Cell. Plast. 2008, 44, 367–380. [Google Scholar] [CrossRef]
- He, H.W.; Hu, H.; Du, K.M.; Lu, M.; Yang, F.; Cui, L.X.; Wang, X. Prospects of high-value recycling methods for polyurethane based on the selective cleavage of C–O/C–N bonds. Green Chem. 2025, 27, 8467–8491. [Google Scholar] [CrossRef]
- Aksu, Y.; Haykiri-Acma, H.; Yaman, S. Recycle of Flexible Polyurethane Foam by Acidolysis and Reuse of Recovered Polyol. J. Polym. Environ. 2025, 33, 1147–1158. [Google Scholar] [CrossRef]
- Pu, M.; Fang, C.; Zhou, X.; Wang, D.; Lin, Y.; Lei, W.; Li, L. Recent Advances in Environment-Friendly Polyurethanes from Polyols Recovered from the Recycling and Renewable Resources: A Review. Polymers 2024, 16, 1889. [Google Scholar] [CrossRef]
- Gama, N.; Godinho, B.; Madureira, P.; Marques, G.; Barros-Timmons, A.; Ferreira, A. Polyurethane Recycling Through Acidolysis: Current Status and Prospects for the Future. J Polym Environ 2024, 32, 4777–4793. [Google Scholar] [CrossRef]
- Gama, N.; Godinho, B.; Marques, G.; Silva, R.; Barros-Timmons, A.; Ferreira, A. Recycling of polyurethane by aci-dolysis: The effect of reaction conditions on the properties of the recovered polyol. Polymer 2021, 219, 123561. [Google Scholar] [CrossRef]
- Liu, B.; Westman, Z.; Richardson, K.; Lim, D.; Stottlemyer, A.L.; Farmer, T.; Gillis, P.; Hooshyar, N.; Vlcek, V.; Christopher, P.; et al. Polyurethane foam chemical recycling: Fast acidolysis with maleic acid and full recovery of polyol. ACS Sustain. Chem. Eng. 2024, 12, 4435–4443. [Google Scholar] [CrossRef] [PubMed]
- Dżerzdżon, K.; Datta, J. Advances in the degradation and recycling of polyurethanes: Biodegradation strategies, MALDI applications, and environmental implications. Waste Manag. 2025, 198, 21–45. [Google Scholar] [CrossRef] [PubMed]
- Rajan, A.; Ameen, F.; Jambulingam, R.; Shankar, V. Biodegradation of polyurethane by fungi isolated from industrial wastewater—A sustainable approach to plastic waste management. Polymers 2024, 16, 1411. [Google Scholar] [CrossRef]
- Magnin, A.; Pollet, E.; Perrin, R.; Ullmann, C.; Persillon, C.; Phalip, V.; Avérous, L. Enzymatic recycling of thermoplastic polyurethanes: Synergistic effect of an esterase and an amidase and recovery of building blocks. Waste Manag. 2019, 85, 141–150. [Google Scholar] [CrossRef]
- Tan, T.; Wang, W.; Zhang, K.; Zhan, Z.; Deng, W.; Zhang, Q.; Wang, Y. Upcycling plastic wastes into value-added products by heterogeneous catalysis. ChemSusChem 2022, 15, e202200522. [Google Scholar] [CrossRef]
- Hartmann, D.; Rahman, T.; Carias, L.; Auad, M.L.; Adhikari, S. Upcycling Polyurethane Plastics via Thermochemi-cal Conversion Pathways: A Comparison of Hydrothermal Liquefaction and Pyrolysis Processes. ACS Sustain. Chem. Eng. 2024, 12, 15515–15527. [Google Scholar] [CrossRef]
- Sun, B.; Zou, J.; Qiu, W.; Tian, S.; Wang, M.; Tang, H.; Ma, D. Chemical transformation of polyurethane into valuable polymers. Natl. Sci. Rev. 2025, 12, nwae393. [Google Scholar] [CrossRef]
- Balu, R.; Dutta, N.K.; Roy Choudhury, N. Plastic Waste Upcycling: A Sustainable Solution for Waste Management, Product Development, and Circular Economy. Polymers 2022, 14, 4788. [Google Scholar] [CrossRef]



















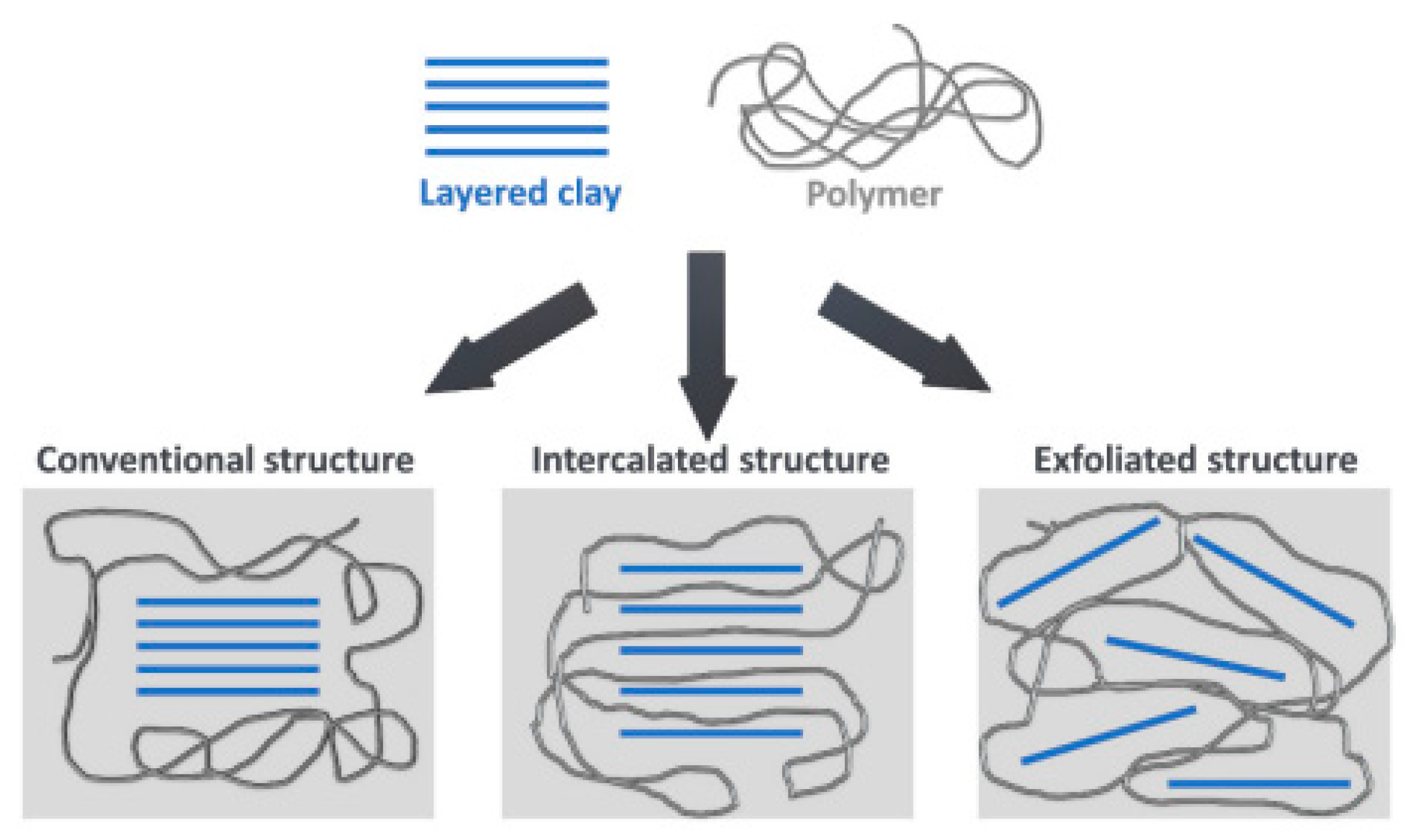






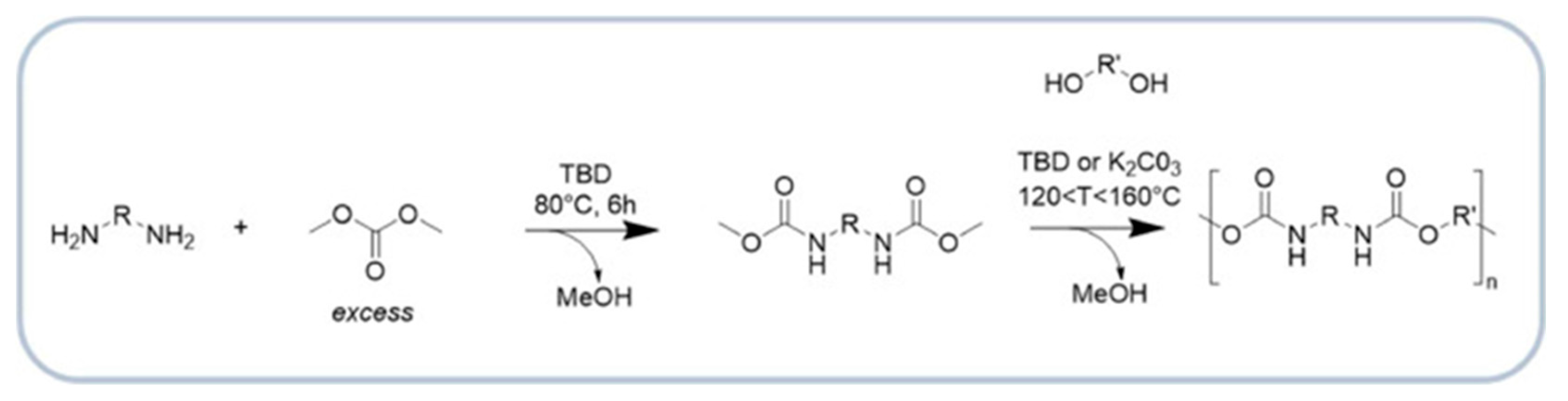






| Type of Raw Material | Synthesis Method | Characteristics of the Obtained Polyol | Physicochemical Properties | Thermal/Mechanical Properties | Main Application Areas |
|---|---|---|---|---|---|
| Vegetable oils (soybean, castor, palm) | Epoxidation followed by ring-opening with an alcohol or polyhydroxy compound | Moderate hydroxyl value, high functionality, adjustable viscosity | Hydroxyl value: 150–350 mg KOH/g; viscosity: 500–2500 mPa·s; acid value < 5 mg KOH g−1 | Tg: −40 °C to −10 °C; good flexibility; moderate thermal stability | Flexible/semi-rigid polyurethane foams; coatings |
| Lignin and cellulose derivatives | Chemical depolymerization, reduction or etherification | Aromatic structures; high thermal stability; variable reactivity | Hydroxyl value: 300–600 mg KOH g−1; viscosity > 3000 mPa·s | Tg: 50–120 °C; high modulus; dimensional stability | Rigid foams; composites; insulation materials |
| Glycerol and sugar alcohols | Esterification or etherification with organic acids or anhydrides | Low molecular weight; high hydroxyl functionality; high reactivity | Hydroxyl value: 400–900 mg KOH g−1; viscosity < 1000 mPa·s | Tg: −30 °C to −5 °C; high flexibility; low thermal resistance | Adhesives; coatings; flexible foams |
| Polyurethane waste and PET polymers | Chemical glycolysis or transesterification | Polyols of varied functionality; secondary hydroxyl groups | Hydroxyl value: 200–500 mg KOH g−1; residual ester content | Tg: 10–70 °C; moderate mechanical strength | Polyurethane recycling; circular-economy applications |
| Fatty-acid esters and microbial oils | Hydroformylation and hydrogenation | Linear/branched polyols; controlled polarity and chain length | Hydroxyl value: 100–250 mg KOH g−1; viscosity 400–1500 mPa·s | Tg: −50 °C to 0 °C; flexibility; oxidative stability | Coatings; elastomers; bio-based lubricants |
| Patent No. | Type | Description | Category |
|---|---|---|---|
| US9950996B2 | Bio-MDI/bio-aromatic isocyanate | Method for producing bio-based aromatic isocyanates from renewable feedstocks. | Bio-isocyanate |
| CN113461894A | PU foam from bio-TDI | Production of bio-based TDI and polyurethane foams derived from it. | Bio-isocyanate |
| EP3819259A1 | Sustainable isocyanate synthesis | Isocyanate production using RWGS, CO2 and renewable energy inputs. | Sustainable process |
| WO2014147142A1 | Allophanates for coatings | Low-toxicity allophanate compositions for polyurethane coating systems. | Green chemistry |
| US20200345678A1 | Bio-IPDI from fatty acids | Production of aliphatic isocyanates from vegetable-oil-derived fatty acids. | Bio-isocyanate |
| WO2019123456A1 | Bio-HDI from biomass | Synthesis of hexamethylene diisocyanate using bio-derived diamines obtained from agricultural waste. | Bio-isocyanate |
| Class | Function | Typical Use Level (%) |
|---|---|---|
| UV Absorbers (UVA) | Absorb harmful UV radiation, prevent yellowing and polymer degradation | 0.2–1.0 |
| HALS (Hindered Amine Light Stabilizers) | Neutralize free radicals, prevent surface degradation | 0.1–1.0 |
| Quenchers | Deactivate excited molecules, reduce photodegradation | 0.1–0.5 |
| Solvent Class | Examples | Typical Applications |
|---|---|---|
| Aromatic hydrocarbons | Toluene, Xylene | PU coatings, adhesives |
| Ketones | Acetone, Methyl ethyl ketone (MEK) Methyl isobutyl ketone (MIBK) | Viscosity control, cleaning |
| Esters | Ethyl acetate, Butyl acetate | High-quality PU coatings |
| Alcohols | Isopropanol, n-Butanol | Solvent blends, reactivity control |
| Glycol ethers | Propylene glycol monomethyl ether (PM) Dipropylene glycol monomethyl ether (DPM) Propylene glycol monomethyl ether acetate (PGMEA) | PU dispersions, coatings |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Polecki, K.; Paciorek-Sadowska, J.; Borowicz, M.; Isbrandt, M.; Zarzyka, I. Polyurethane Recycling: Sustainable Development Perspectives and Innovative Approaches. Materials 2026, 19, 805. https://doi.org/10.3390/ma19040805
Polecki K, Paciorek-Sadowska J, Borowicz M, Isbrandt M, Zarzyka I. Polyurethane Recycling: Sustainable Development Perspectives and Innovative Approaches. Materials. 2026; 19(4):805. https://doi.org/10.3390/ma19040805
Chicago/Turabian StylePolecki, Konrad, Joanna Paciorek-Sadowska, Marcin Borowicz, Marek Isbrandt, and Iwona Zarzyka. 2026. "Polyurethane Recycling: Sustainable Development Perspectives and Innovative Approaches" Materials 19, no. 4: 805. https://doi.org/10.3390/ma19040805
APA StylePolecki, K., Paciorek-Sadowska, J., Borowicz, M., Isbrandt, M., & Zarzyka, I. (2026). Polyurethane Recycling: Sustainable Development Perspectives and Innovative Approaches. Materials, 19(4), 805. https://doi.org/10.3390/ma19040805















