Low-Temperature Plasma Activation of Biomaterials and Its Stability over Time and Post-Sterilisation Effects
Abstract
1. Introduction
2. Materials and Methods
2.1. Material and Sample Preparation
- The first group consisted of Ti6Al4V sheet material with a thickness of 2 mm, rectangular specimens measuring 40 × 20 mm were fabricated from the sheet using abrasive waterjet cutting to minimise thermal influence on the material surface.
- The second group comprised circular Ti6Al4V specimens with a diameter of 15 mm, manufactured using selective laser melting (SLM) technology from metallic powder. The printing process used a layer thickness of 0.04 mm. After fabrication, the samples underwent heat treatment in accordance with the powder manufacturer’s recommended post-processing protocol.
- The third group consisted of circular CoCr alloy specimens with a diameter of 15 mm, also produced using SLM technology from metallic powder with a layer thickness of 0.04 mm. These samples were subjected to post-print heat treatment in accordance with the manufacturer’s specifications for the applied powder.
2.2. Plasma Activation Process
2.3. Sterilisation Procedure
- LTP-activated and stored samples to assess the degradation of LTP effects over time (LTPA),
- LTP-activated and then steam-sterilised samples to evaluate the influence of autoclaving (LTPA&S),
- non-activated but sterilised samples serving as controls (S).
2.4. Wettability Testing Using Canine Blood
2.5. Statistical Analysis
3. Results
3.1. Contact Angle of Three Non-Treated Materials (Group NN)
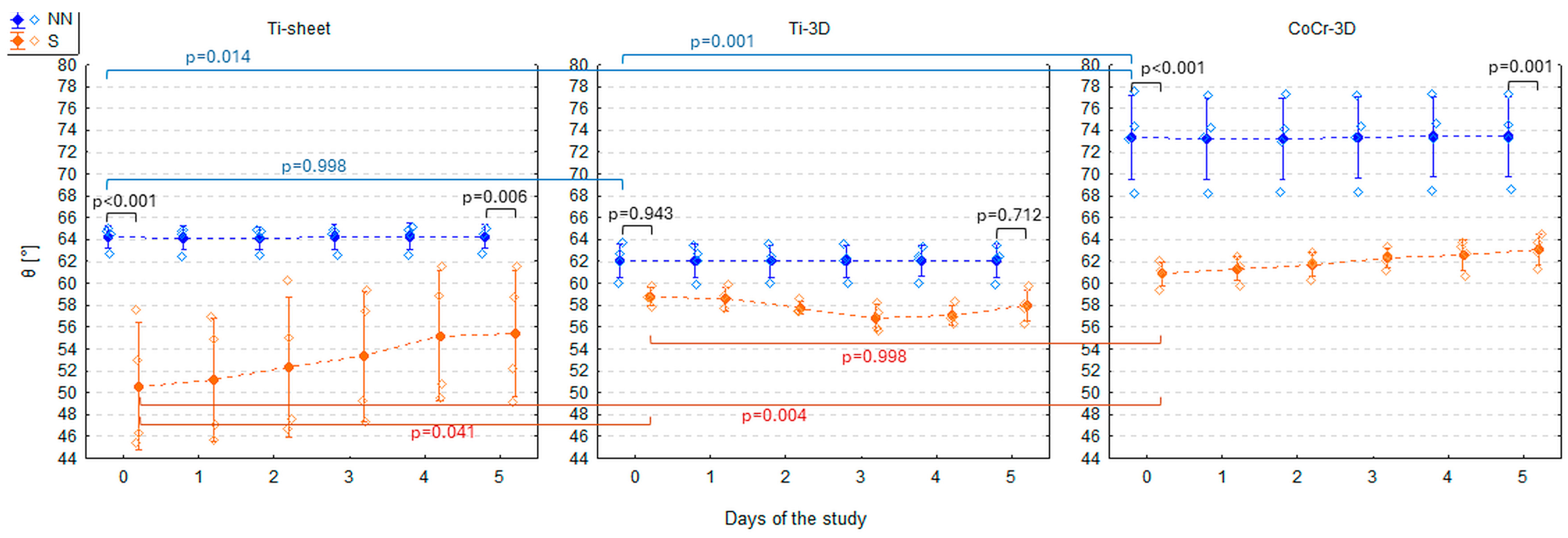
3.2. Contact Angle of Three Materials After Steam Sterilisation (Group S)
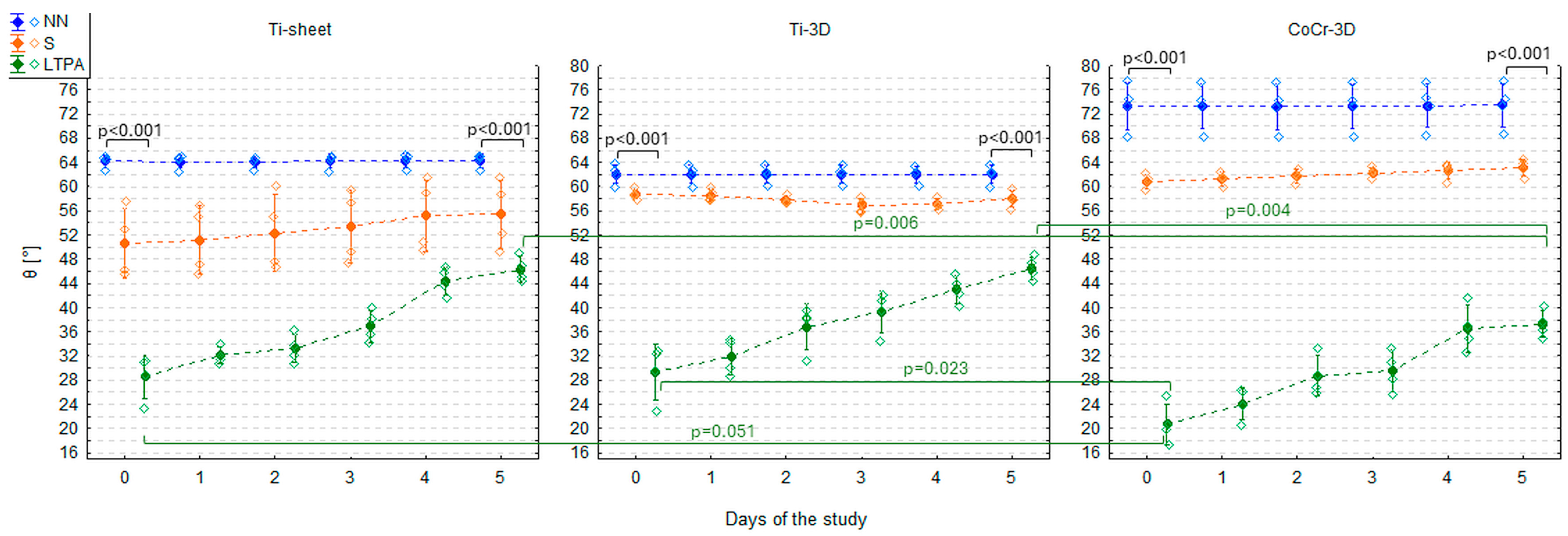
3.3. Contact Angle of Three Materials After LTP Activation
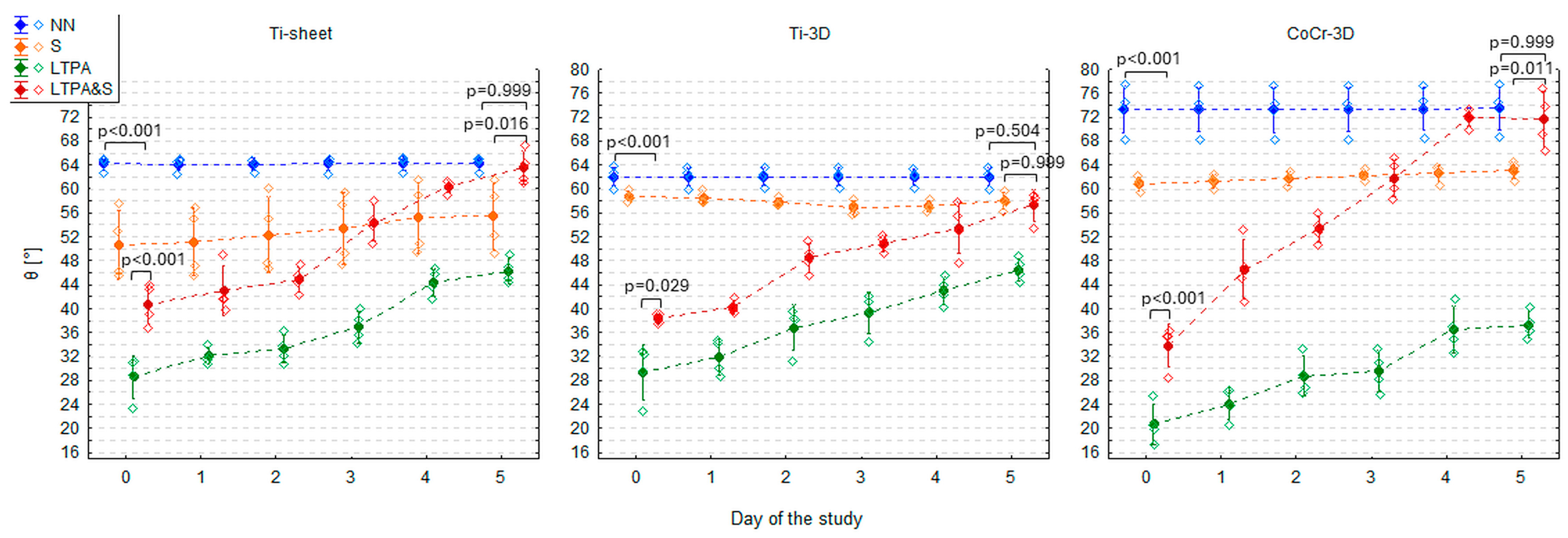
3.4. An Influence of Steam Sterilisation on the Contact Angle of Three LTP Activated Materials
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| SSIs | Surgical Site Infection |
| LTP | Low-Temperature Plasma |
| LTPA | Low-Temperature Plasma Activated |
| LTPA&S | Low-Temperature Plasma Activated and Steam-Sterilized |
| PDD | Piezoelectric Direct Discharge |
| SLM | Selective Laser Melting |
| Ti6Al4V | Titanium–Aluminum–Vanadium alloy |
| CoCr | Cobalt–Chromium alloy |
| NN | Non-Treated |
| S | Steam-Sterilized |
| GLM | General Linear Model |
| HSD | Honest Significant Difference |
| CI | Confidence Interval |
| SD | Standard Deviation |
References
- Verwilghen, D.R.; Pelosi, A.; Abbas, M.; Allerton, F.; Archer, D.; Baxter, G.; Brehm, W.; Burgess, B.A.; Dallap-Schaer, B.; Ferreira, J.; et al. Surgical site infection definitions consensus: A first step toward improving prevention in veterinary medicine. Am. J. Vet. Res. 2025, 1, 1–14. [Google Scholar] [CrossRef]
- Bandyopadhyay, A.; Mitra, I.; Goodman, S.B.; Kumar, M.; Bose, S. Improving biocompatibility for next generation of metallic implants. Prog. Mater. Sci. 2023, 133, 101053. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Ao, X.; Xie, P.; Wu, J.; Dong, Y.; Yu, D.; Wang, J.; Zhu, Z.; Xu, H.H.K.; Chen, W. Effects of novel non-thermal atmospheric plasma treatment of titanium on physical and biological improvements and in vivo osseointegration in rats. Sci. Rep. 2020, 10, 10637. [Google Scholar] [CrossRef] [PubMed]
- Long, S.; Zhu, J.; Jing, Y.; He, S.; Cheng, L.; Shi, Z. A Comprehensive Review of Surface Modification Techniques for Enhancing the Biocompatibility of 3D-Printed Titanium Implants. Coatings 2023, 13, 1917. [Google Scholar] [CrossRef]
- Sarvaiya, B.B.; Kumar, S.; Pathan, M.S.H.; Patel, S.; Gupta, V.; Haque, M. The Impact of Implant Surface Modifications on the Osseointegration Process: An Overview. Cureus 2025, 17, e81576. [Google Scholar] [CrossRef]
- Nemani, S.K.; Annavarapu, R.K.; Mohammadian, B.; Raiyan, A.; Heil, J.; Haque, M.A.; Sojoudi, H. Surface modification of polymers: Methods and applications. Adv. Mater. Interfaces 2018, 5, 1801247. [Google Scholar] [CrossRef]
- Jarad, N.A.; Chami, A.; Weitz, J.I.; Didar, T.F. Advancements in surface modification strategies of vascular grafts to improve biocompatibility and tissue integration. Explor. Biomater. 2024, 1, 241–265. [Google Scholar] [CrossRef]
- Laroussi, M. Cold Plasma in Medicine and Healthcare: The New Frontier in Low Temperature Plasma Applications. Front. Phys. 2020, 8, 74. [Google Scholar] [CrossRef]
- Karthik, C.; Sarngadharan, S.C.; Thomas, V. Low-Temperature Plasma Techniques in Biomedical Applications and Therapeutics: An Overview. Int. J. Mol. Sci. 2024, 25, 524. [Google Scholar] [CrossRef]
- Tabares, F.L.; Junkar, I. Cold plasma systems and their application in surface treatments for medicine. Molecules 2021, 26, 1903. [Google Scholar] [CrossRef]
- Scholtz, V.; Pazlarova, J.; Souskova, H.; Khun, J.; Julak, J. Nonthermal plasma—A tool for decontamination and disinfection. Biotechnol. Adv. 2015, 33, 1108–1119. [Google Scholar] [CrossRef]
- Pandiyaraj, K.N.; Kumar, A.A.; Ramkumar, M.C.; Sachdev, A.; Gopinath, P.; Cools, P.; De Geyter, N.; Morent, R.; Deshmukh, R.R. Influence of non-thermal TiCl4/Ar + O2 plasma-assisted TiOx based coatings on the surface of polypropylene films for tailoring of surface properties and cytocompatibility. Mater. Sci. Eng. C 2016, 62, 908–918. [Google Scholar] [CrossRef]
- Minati, L.; Migliaresi, C.; Speranza, G. Plasma assisted surface treatments of biomaterials. Biophys. Chem. 2017, 229, 151–164. [Google Scholar] [CrossRef]
- Kunrath, M.F.; Vargas, A.L.M.; Sesterheim, P.; Teixeira, E.R.; Hubler, R. Extension of hydrophilicity stability by reactive plasma treatment and wet storage on TiO2 nanotube surfaces for biomedical implant applications. J. R. Soc. Interface 2020, 17, 20200650. [Google Scholar] [CrossRef]
- von Woedtke, T.; Reuter, S.; Masur, K.; Weltmann, K.-D. Plasmas for medicine. Phys. Rep. 2013, 530, 291–320. [Google Scholar] [CrossRef]
- Karthik, C.; Rajalakshmi, S.; Thomas, S.; Thomas, V. Intelligent polymeric biomaterials surface driven by plasma processing. Curr. Opin. Biomed. Eng. 2023, 26, 100440. [Google Scholar] [CrossRef]
- Booth, J.P.; Mozetič, M.; Nikiforov, A.; Oehr, C. Foundations of plasma surface functionalization of polymers for industrial and biological applications. Plasma Sources Sci. Technol. 2022, 31, 103001. [Google Scholar] [CrossRef]
- Huhtamäki, T.; Tian, X.; Korhonen, J.T.; Ras, R.H.A. Surface-wetting characterization using contact-angle measurements. Nat. Protoc. 2018, 13, 1521–1538. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Chu, L.; Chen, J.; Qi, F.; Li, X.; Chen, X.; Yu, D.G. Asymmetric wettability fibrous membranes: Preparation and biologic applications. Compos. Part B Eng. 2024, 269, 111095. [Google Scholar] [CrossRef]
- Cai, S.; Wu, C.; Yang, W.; Liang, W.; Yu, H.; Liu, L. Recent advance in surface modification for regulating cell adhesion and behaviors. Nanotechnol. Rev. 2020, 9, 971–989. [Google Scholar] [CrossRef]
- Yu, J.; Zhou, M.; Zhang, L.; Wei, H. Antibacterial adhesion strategy for dental titanium implant surfaces: From mechanisms to application. J. Funct. Biomater. 2022, 13, 169. [Google Scholar] [CrossRef]
- Straumann® SLActive®. Product Information. Straumann Holding AG. 2017. Available online: http://slactive.straumann.com (accessed on 13 August 2025).
- Cheadle, W.G. Risk factors for surgical site infection. Surg. Infect. 2006, 7, S7–S11. [Google Scholar] [CrossRef] [PubMed]
- Akyalcin, S.; McIver, H.P.; English, J.D.; Ontiveros, J.C.; Gallerano, R.L. Effects of repeated sterilization cycles on primary stability of orthodontic mini-screws. Angle Orthod. 2013, 83, 674–679. [Google Scholar] [CrossRef] [PubMed]
- Mahshid, M.; Saboury, A.; Fayaz, A.; Sadr, S.J.; Lampert, F.; Mir, M. The effect of steam sterilization on the accuracy of spring-style mechanical torque devices for dental implants. Clin. Cosmet. Investig. Dent. 2012, 4, 29–35. [Google Scholar] [PubMed]
- King, J.B.; Roberts, H.W.; Bergeron, B.E.; Mayerchak, M.J. The effect of autoclaving on torsional moment of two nickel titanium endodontic files. Int. Endod. J. 2012, 45, 156–161. [Google Scholar] [CrossRef]
- Porto, A.N.; Borges, A.H.; Semenoff-Segundo, A.; A Raslan, S.; Pedro, F.L.M.; Jorge, A.O.C.; Bandeca, M.C. Effect of repeated sterilization cycles on the physical properties of scaling instruments: A scanning electron microscopy study. J. Int. Oral Health 2015, 7, 1–4. [Google Scholar]
- Yniguez, F.M.; Grimes, J.A.; Hodgson, M.M.; Trenta, M.; Chin-Chi, L.; Riggs, L.M. Effect of repeated steam sterilizations on insertional torque, torque to failure, and axial pullout strength of 3.5-mm and 2.0-mm cortical bone screws. Am. J. Vet. Res. 2024, 85, 1–6. [Google Scholar] [CrossRef]
- Kunrath, M.F.; Hubler, R.; Dahlin, C. Adverse effects of sterilization processes on the fundamental topographic properties of modified dental implant surfaces. J. Mater. Sci. Mater. Med. 2024, 35, 44. [Google Scholar] [CrossRef]
- Ghobeira, R.; Philips, C.; Declercq, H.; Cools, P.; De Geyter, N.; Cornelissen, R.; Morent, R. Effects of different sterilization methods on the physico-chemical and bioresponsive properties of plasma-treated polycaprolactone films. Biomed. Mater. 2017, 12, 015017. [Google Scholar] [CrossRef]
- Junkar, I.; Kulkarni, M.; Drašler, B.; Rugelj, N.; Mazare, A.; Flašker, A.; Drobne, D.; Humpolíček, P.; Resnik, M.; Schmuki, P.; et al. Influence of various sterilization procedures on TiO2 nanotubes used for biomedical devices. Bioelectrochemistry 2016, 109, 79–86. [Google Scholar] [CrossRef]
- Kido, D.; Komatsu, K.; Suzumura, T.; Matsuura, T.; Cheng, J.; Kim, J.; Park, W.; Ogawa, T. Influence of surface contaminants and hydrocarbon pellicle on the results of wettability measurements of titanium. Int. J. Mol. Sci. 2023, 24, 14688. [Google Scholar] [CrossRef] [PubMed]
- Alayan, J.; Vaquette, C.; Saifzadeh, S.; Hutmacher, D.; Ivanovski, S. Comparison of early osseointegration of SLA® and SLActive® implants in maxillary sinus augmentation: A pilot study. Clin. Oral Implant. Res. 2017, 28, 1325–1333. [Google Scholar] [CrossRef]
- Chouirfa, H.; Bouloussa, H.; Migonney, V.; Falentin-Daudré, C. Review of titanium surface modification techniques and coatings for antibacterial applications. Acta Biomater. 2019, 83, 37–54. [Google Scholar] [CrossRef]
- Sakudo, A.; Yagyu, Y.; Onodera, T. Disinfection and sterilization using plasma technology: Fundamentals and future perspectives for biological applications. Int. J. Mol. Sci. 2019, 20, 5216. [Google Scholar] [CrossRef]
- Relyon Plasma GmbH. Piezobrush® PZ3. Available online: https://www.relyon-plasma.com/piezobrush-pz3/ (accessed on 13 August 2025).
- Rakoczy, B. Act on the Protection of Animals Used for Scientific or Educational Purposes—Legal regulation review. Pol. Yearb. Environ. Law 2016, 5, 79–88. [Google Scholar] [CrossRef]
- Duske, K.; Koban, I.; Kindel, E.; Schröder, K.; Nebe, B.; Holtfreter, B.; Jablonowski, L.; Weltmann, K.D.; Kocher, T. Atmospheric plasma enhances wettability and cell spreading on dental implant metals. J. Clin. Periodontol. 2012, 39, 400–407. [Google Scholar] [CrossRef]
- Giro, G.; Tovar, N.; Witek, L.; Marin, C.; Silva, N.R.F.; Bonfante, E.A.; Coelho, P.G. Osseointegration assessment of chairside argon-based nonthermal plasma-treated Ca-P coated dental implants. J. Biomed. Mater. Res. Part A 2013, 101, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Taddei, E.B.; Henriques, V.A.R.; Silva, C.R.M.; Cairo, C.A.A. Production of new titanium alloy for orthopedic implants. Mater. Sci. Eng. C Biomim. Mater. Sens. Syst. 2004, 24, 683–687. [Google Scholar] [CrossRef]
- Lorenzetti, M.; Dogša, I.; Stošicki, T.; Stopar, D.; Kalin, M.; Kobe, S.; Novak, S. The Influence of Surface Modification on Bacterial Adhesion to Titanium-Based Substrates. ACS Appl. Mater. Interfaces 2015, 7, 1644–1651. [Google Scholar] [CrossRef]
- Harada, A.; Sasaki, H.; Asami, Y.; Hanazawa, K.; Miyazaki, S.; Sekine, H.; Yajima, Y. Effects of the application of low-temperature atmospheric plasma on titanium implants on wound healing in peri-implant connective tissue in rats. Int. J. Implant. Dent. 2024, 10, 15. [Google Scholar] [CrossRef] [PubMed]
- Bevilacqua, L.; Faccioni, F.; Porrelli, D.; Faccioni, P.; Rusin, F.; Frassetto, A.; Maglione, M. Blood Wettability of Different Dental Implant Surfaces after Different Pre-Treatments: Ultrasonic Instrumentation, Platelet-Rich Fibrin Coating, and Acid Etching. An In Vitro Study. Appl. Sci. 2021, 11, 1433. [Google Scholar] [CrossRef]
- Rupp, F.; Gittens, R.A.; Scheideler, L.; Marmur, A.; Boyan, B.D.; Schwartz, Z.; Geis-Gerstorfer, J. A review on the wettability of dental implant surfaces I: Theoretical and experimental aspects. Acta Biomater. 2014, 10, 2894–2906. [Google Scholar] [CrossRef] [PubMed]
- de Mel, A.; Cousins, B.G.; Seifalian, A.M. Surface modification of biomaterials: A quest for blood compatibility. Int. J. Biomater. 2012, 2012, 707863. [Google Scholar] [CrossRef] [PubMed]
- Wilson, C.J.; Clegg, R.E.; Leavesley, D.I.; Pearcy, M.J. Mediation of biomaterial-cell interactions by adsorbed proteins: A review. Tissue Eng. 2005, 11, 1–18. [Google Scholar] [CrossRef]
- Barjasteh, A.; Kaushik, N.; Choi, E.H.; Kaushik, N.K. Cold Atmospheric Pressure Plasma: A Growing Paradigm in Diabetic Wound Healing—Mechanism and Clinical Significance. Int. J. Mol. Sci. 2023, 24, 16657. [Google Scholar] [CrossRef] [PubMed]
- Vitoriano, J.d.O.; de Moura, C.E.B.; Braz, J.F.S.; Martins, G.M.; Barros, P.P.; Santos, C.S.; Melo, R.L.F.; Carmo, F.R.; Pessoa, R.S.; Alves-Junior, C. Aging behavior of titanium surfaces treated with non-thermal atmospheric pressure plasma at different temperatures. Appl. Surf. Sci. 2026, 715, 164537. [Google Scholar] [CrossRef]
- Wu, Y.; Wan, K.; Lu, J.; Yuan, C.; Cui, Y.; Duan, R.; Yu, J. Research Progress on Surface Modification of Titanium Implants. Coatings 2025, 15, 229. [Google Scholar] [CrossRef]



| Day of the Study | |||||||
|---|---|---|---|---|---|---|---|
| Material | Treatment * | 0 | 1 | 2 | 3 | 4 | 5 |
| Ti-sheet | NN | 64.24 ± 1.02 | 64.17 ± 1.12 | 64.14 ± 1.04 | 64.22 ± 1.12 | 64.29 ± 1.18 | 64.31 ± 1.13 |
| Ti-sheet | S | 50.58 ± 5.80 | 51.16 ± 5.62 | 52.36 ± 6.43 | 53.34 ± 5.97 | 55.19 ± 5.93 | 55.41 ± 5.73 |
| Ti-sheet | LTPA | 28.61 ± 3.61 | 32.08 ± 1.39 | 33.29 ± 2.33 | 36.93 ± 2.62 | 44.41 ± 2.33 | 46.33 ± 2.11 |
| Ti-sheet | LTPA&S | 40.77 ± 3.39 | 42.98 ± 4.07 | 44.89 ± 2.14 | 54.32 ± 2.94 | 60.40 ± 1.05 | 63.58 ± 2.78 |
| Ti-3D | NN | 62.09 ± 1.57 | 62.04 ± 1.54 | 62.04 ± 1.48 | 62.04 ± 1.46 | 62.04 ± 1.39 | 62.04 ± 1.56 |
| Ti-3D | S | 58.76 ± 0.82 | 58.56 ± 1.05 | 57.74 ± 0.61 | 56.81 ± 1.22 | 57.09 ± 0.89 | 57.96 ± 1.42 |
| Ti-3D | LTPA | 29.42 ± 4.62 | 31.85 ± 2.97 | 36.81 ± 3.85 | 39.31 ± 3.38 | 42.98 ± 2.30 | 46.56 ± 1.86 |
| Ti-3D | LTPA&S | 38.28 ± 0.81 | 40.26 ± 1.15 | 48.48 ± 2.39 | 50.89 ± 1.33 | 53.51 ± 4.38 | 57.30 ± 2.64 |
| CoCr-3D | NN | 73.35 ± 3.88 | 73.24 ± 3.73 | 73.20 ± 3.74 | 73.32 ± 3.71 | 73.42 ± 3.66 | 73.45 ± 3.64 |
| CoCr-3D | S | 60.88 ± 1.12 | 61.35 ± 1.13 | 61.75 ± 1.08 | 62.30 ± 0.88 | 62.60 ± 1.40 | 63.13 ± 1.38 |
| CoCr-3D | LTPA | 20.70 ± 3.38 | 24.14 ± 2.70 | 28.72 ± 3.31 | 29.56 ± 3.38 | 36.52 ± 3.86 | 37.33 ± 2.27 |
| CoCr-3D | LTPA&S | 33.84 ± 3.66 | 46.55 ± 4.97 | 53.34 ± 2.23 | 61.81 ± 3.16 | 71.82 ± 1.40 | 71.61 ± 4.66 |
| Intercept Coefficient | Slope Coefficient | |||
|---|---|---|---|---|
| Material | b0 (CI 95%) | b1 (CI 95%) | p-Value | R2 |
| Ti-sheet | 50.3° (46.3°, 54.4°) | 1.01° (−0.27°, 2.39°) | 0.114 | 0.111 |
| CoCr-3D | 60.9° (60.1°, 61.7°) | 0.44° (0.18°, 0.71°) | 0.002 | 0.356 |
| Intercept Coefficient | Slope Coefficient | |||
|---|---|---|---|---|
| Material | b0 (CI 95%) | b1 (CI 95%) | p-Value | R2 |
| Ti-sheet | 27.7° (25.7°, 29.7°) | 3.7° (3.0°, 4.4°) | <0.001 | 0.858 |
| Ti-3D | 29.1° (26.9°, 31.4°) | 3.5° (2.7°, 4.2°) | <0.001 | 0.808 |
| CoCr-3D | 20.8° (18.5°, 23.2°) | 3.5° (2.7°, 4.2°) | <0.001 | 0.796 |
| Intercept Coefficient | Slope Coefficient | |||
|---|---|---|---|---|
| Material | b0 (CI 95%) | b1 (CI 95%) | p-Value | R2 |
| Ti-sheet | 38.6° (36.1°, 41.1°) | 5.0° (4.2°, 5.8°) | <0.001 | 0.880 |
| Ti-3D | 38.3° (36.4°, 40.3°) | 3.9° (3.3°, 4.6°) | <0.001 | 0.878 |
| CoCr-3D | 37.0° (33.7°, 40.3°) | 7.8° (6.7°, 8.9°) | <0.001 | 0.910 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Trębacz, P.; Pawlik, M.; Kurkowska, A.; Wilk, K.; Piątek, A.; Czopowicz, M. Low-Temperature Plasma Activation of Biomaterials and Its Stability over Time and Post-Sterilisation Effects. Materials 2026, 19, 643. https://doi.org/10.3390/ma19030643
Trębacz P, Pawlik M, Kurkowska A, Wilk K, Piątek A, Czopowicz M. Low-Temperature Plasma Activation of Biomaterials and Its Stability over Time and Post-Sterilisation Effects. Materials. 2026; 19(3):643. https://doi.org/10.3390/ma19030643
Chicago/Turabian StyleTrębacz, Piotr, Mateusz Pawlik, Aleksandra Kurkowska, Karolina Wilk, Agata Piątek, and Michał Czopowicz. 2026. "Low-Temperature Plasma Activation of Biomaterials and Its Stability over Time and Post-Sterilisation Effects" Materials 19, no. 3: 643. https://doi.org/10.3390/ma19030643
APA StyleTrębacz, P., Pawlik, M., Kurkowska, A., Wilk, K., Piątek, A., & Czopowicz, M. (2026). Low-Temperature Plasma Activation of Biomaterials and Its Stability over Time and Post-Sterilisation Effects. Materials, 19(3), 643. https://doi.org/10.3390/ma19030643







