Fabrication of Highly Conductive Inkjet Printing Silver Nanoparticle Ink via a Synergistic Strategy Combining Centrifugal Classification and Dispersant Optimization
Highlights
- A synergistic strategy integrating centrifugal classification, high-pressure homogenization, and dispersant optimization was successfully developed.
- The resulting optimized Ag NP ink demonstrated excellent jetting stability (clogging-free) and suitable rheology for reliable inkjet printing.
- A high electrical conductivity of 1.506 × 107 S/m was achieved after low-temperature sintering at only 260 °C.
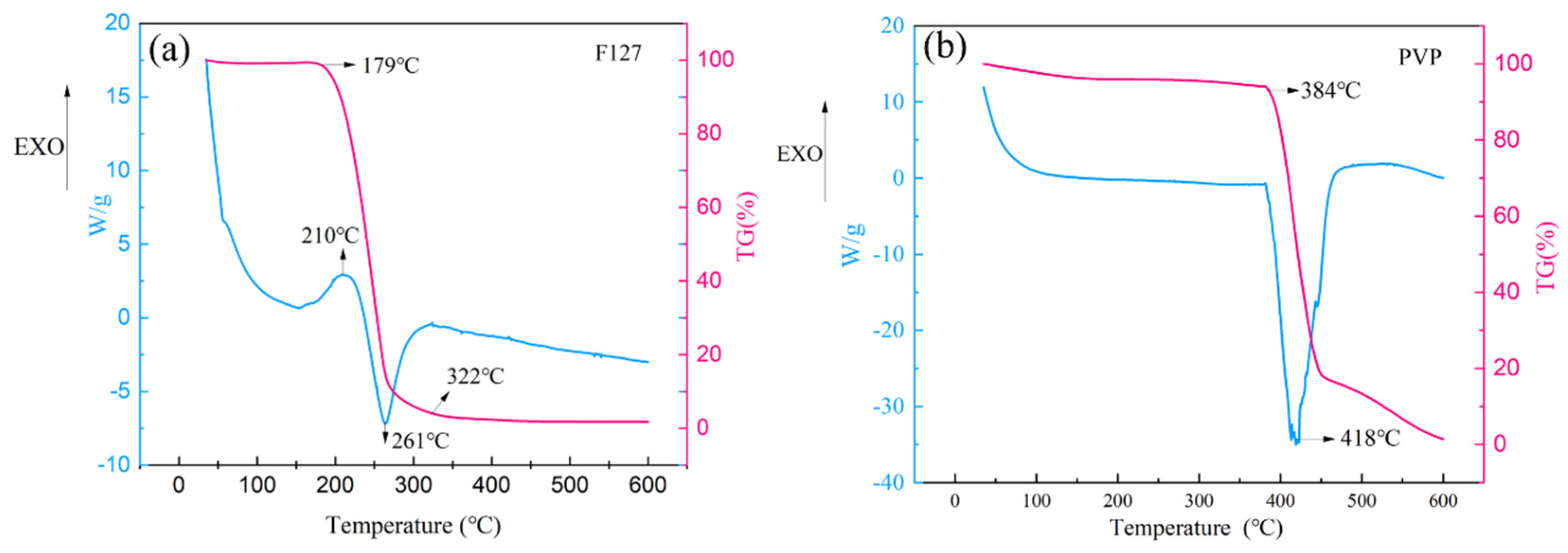
- Using Pluronic F127 as the sole dispersant allowed its complete removal during sintering, promoting the formation of a dense conductive network.
- The strategy provides a scalable and practical manufacturing pathway for producing high-performance printed electronics.
- The low-temperature sintering capability (260 °C) makes the ink compatible with heat-sensitive flexible substrates.
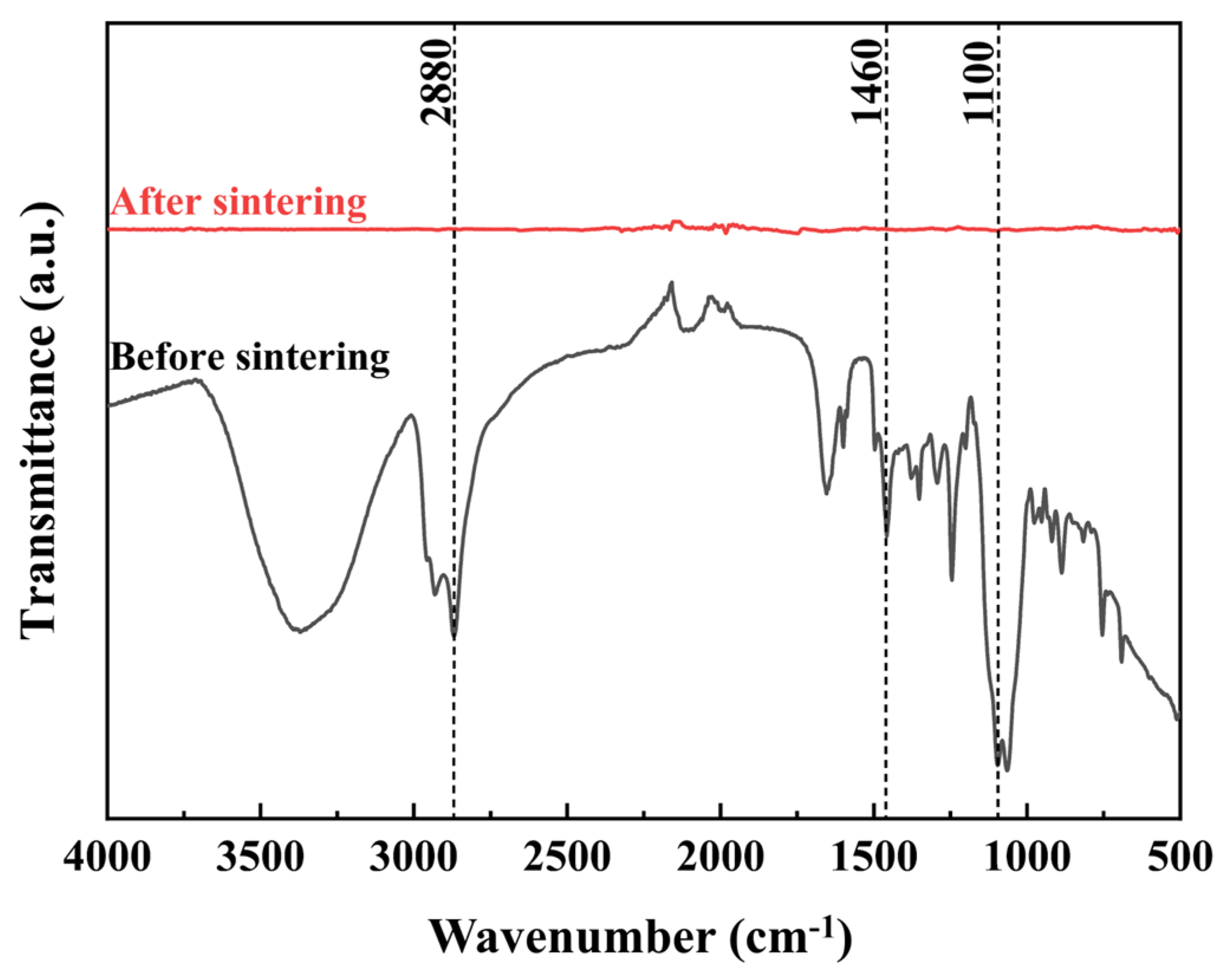
- The complete removal of the dispersant eliminates insulating residues, which is crucial for achieving high conductivity in printed metal features.
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Ag NP Ink Formulation and Optimization Strategy
2.2.1. Centrifugal Classification and Surface Modification
2.2.2. Ink Preparation and Dispersion Optimization
2.2.3. Dispersant Concentration Optimization
2.3. Inkjet Printing Process
2.4. Characterization
3. Results and Discussion
3.1. Optimization of Ag NPs Selection via Centrifugal Classification
3.2. Comparison of the Dispersion Methods
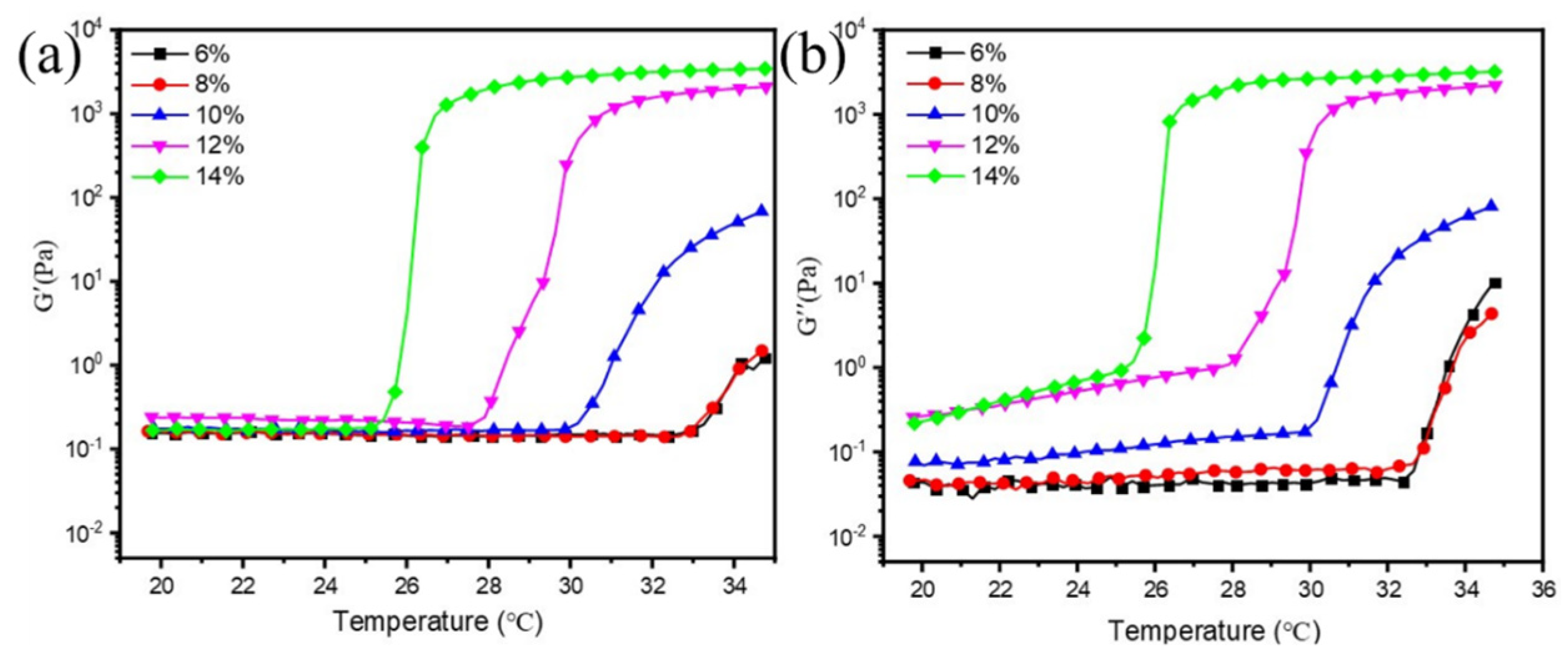
3.3. Effect of F127 Concentration on Dispersion and Rheology
3.4. Electrical Performance: Influence of Dispersant System and Sintering Temperature
3.5. Inkjet Printing Verification
3.5.1. Quantitative Printability Assessment
3.5.2. Printing Demonstration and Performance
4. Conclusions
- Centrifugal classification at 1500 r/min effectively selected Ag NPs (Brofos) with a narrow size distribution (>70% <50 nm), providing the foundational step for enhancing ink stability by reducing sedimentation driving forces.
- High-pressure homogenization proved superior to acoustic resonance dispersion, generating stronger shear forces to achieve a more stable dispersion with lower and more stable viscosity, which is crucial for printability.
- The key to achieving high conductivity lies in the selection of Pluronic F127 as the sole dispersant. It provided exceptional steric stability, and crucially, its relatively low decomposition temperature (~322 °C) allowed for near-complete removal during the 260 °C sintering process. This enabled extensive particle fusion into a dense network, yielding a high conductivity of (1.506 ± 0.08) × 107 S/m (on glass/PI substrates) with 10 wt.% F127.
- The synergistic integration of these optimized steps resulted in an ink with excellent overall performance under the specific conditions studied: appropriate rheology (viscosity = 6.83 mPa·s, surface tension = 19.60 mN/m), reliable inkjet printing stability under the optimized printing parameters, and high conductivity after low-temperature sintering, demonstrating significant potential for practical applications in printed electronics within this framework.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Al-Halhouli, A.; Qitouqa, H.; Alashqar, A.; Abu-Khalaf, J. Inkjet printing for the fabrication of flexible/stretchable wearable electronic devices and sensors. Sens. Rev. 2018, 38, 438–452. [Google Scholar] [CrossRef]
- Gao, M.; Li, L.; Song, Y. Inkjet printing wearable electronic devices. J. Mater. Chem. C 2017, 5, 2971–2993. [Google Scholar] [CrossRef]
- Nunes, D.; Pimentel, A.; Barquinha, P.; Mendes, M.; Coelho, J.; Almeida, H.; Fortunato, E.; Martins, R. Printing technology in metal–oxide-based flexible devices. In Flexible Devices Based on Metal Oxides; Elsevier: Amsterdam, The Netherlands, 2025; pp. 143–181. [Google Scholar]
- Nayak, L.; Mohanty, S.; Nayak, S.K.; Ramadoss, A. A review on inkjet printing of nanoparticle inks for flexible electronics. J. Mater. Chem. C 2019, 7, 8771–8795. [Google Scholar] [CrossRef]
- Nabi, S.; Isaev, A.; Chiolerio, A. Inkjet Printing of Functional Materials for Low-Temperature Electronics: A Review of Materials and Strategies. ACS Appl. Electron. Mater. 2024, 6, 7679–7719. [Google Scholar] [CrossRef]
- Cao, T.; Yang, Z.; Zhang, H.; Wang, Y. Inkjet printing quality improvement research progress: A review. Heliyon 2024, 10, e30163. [Google Scholar] [CrossRef]
- Sanchez-Duenas, L.; Gómez, E.; Larraaga, M.; Blanco, M.; Goitandia, A.M.; Aranzabe, E.; Vilas-Vilela, J.L. A Review on Sustainable Inks for Printed Electronics: Materials for Conductive, Dielectric and Piezoelectric Sustainable Inks. Materials 2023, 16, 3940. [Google Scholar] [CrossRef] [PubMed]
- Hima Priya, K.N.; Meghana, C.S.; Raju, N.K. Current developments in conductive nano-inks for flexible and wearable electronics. ECS Trans. 2022, 107, 11261. [Google Scholar] [CrossRef]
- Ibrahim, N.; Akindoyo, J.O.; Mariatti, M. Recent development in silver-based ink for flexible electronics. J. Sci. Adv. Mater. Devices 2022, 7, 100395. [Google Scholar] [CrossRef]
- Zhang, J.; Ahmadi, M.; Fargas, G.; Perinka, N.; Reguera, J.; Lanceros-Méndez, S.; Llanes, L.; Metals, E.J.-P.J. Silver Nanoparticles for Conductive Inks: From Synthesis and Ink Formulation to Their Use in Printing Technologies. Metals 2022, 12, 234. [Google Scholar] [CrossRef]
- Vaseem, M.; Mckerricher, G.; Shamim, A. Interfaces, Robust Design of a Particle-Free Silver-Organo-Complex Ink with High Conductivity and Inkjet Stability for Flexible Electronics. ACS Appl. Mater. Interfaces 2015, 8, 177. [Google Scholar] [CrossRef]
- Ibrahim, N.; Mustapha, M. Electrical conductivity of silver conductive ink synthesized using chemical reduction method. In Proceedings of the 3rd International Postgraduate Conference on Materials, Minerals & Polymer (Mamip) 2019, Penang, Malaysia, 31 October–1 November 2019. [Google Scholar]
- Chen, Z.; Gengenbach, U.; Koker, L.; Huang, L.; Mach, T.P.; Reichert, K.-M.; Thelen, R.; Ungerer, M. Systematic Investigation of Novel, Controlled Low-Temperature Sintering Processes for Inkjet Printed Silver Nanoparticle Ink. Small 2024, 20, e2306865. [Google Scholar] [CrossRef]
- Wagner, K.; Paquet, C.; Martinez-Rubi, Y.; Genest, M.; Lessard, B.H. Boron Nitride Nanotube Coatings for Thermal Management of Printed Silver Inks on Temperature Sensitive Substrates. Adv. Electron. Mater. 2021, 7, 2001035. [Google Scholar] [CrossRef]
- Yang, H.; Zhan, Z. Progress on low temperature sintering of nano-silver. In Proceedings of the 2021 IEEE 23rd Electronics Packaging Technology Conference (EPTC), Singapore, 7–9 December 2021; pp. 302–308. [Google Scholar]
- Nishchev, K.; Novopoltsev, M.; Savrasov, K.V.; Mishkin, V.P.; Eliseev, V.V.; Martynenko, V.A.; Grishanin, A.V. Investigation of the process of low-temperature sintering of silver-pastes by the scanning electron microscopy. Appl. Phys. 2015, 3, 10–14. [Google Scholar]
- Luo, S.Y.; Chen, J.; Xu, W.C.; Zhang, X.L.; Huo, L.X. Nano-Silver Paste with a Low Sintering Temperature. Adv. Mater. Res. 2012, 391–392, 745–748. [Google Scholar] [CrossRef]
- Lee, H.H.; Chou, K.S.; Huang, K.C. Inkjet printing of nanosized silver colloids. Nanotechnology 2005, 16, 2436–2441. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Wang, C.; Arrighi, V. An organic silver complex conductive ink using both decomposition and self-reduction mechanisms in film formation. J. Mater. Sci. Mater. Electron. 2018, 29, 2771–2783. [Google Scholar] [CrossRef]
- Roudenko, J.; Schirmer, J.; Reichenberger, M.; Neermann, S.; Franke, J. Influence of convection sintering parameters on electrical conductivity and adhesion of inkjet-printed silver nanoparticle inks on flexible substrates. In Proceedings of the 2018 13th International Congress Molded Interconnect Devices (MID), Würzburg, Germany, 25–26 September 2018. [Google Scholar]
- Titkov, A.I.; Bukhanets, O.G.; Gadirov, R.M.; Yukhin, Y.M.; Lyakhov, N.Z. Conductive inks for inkjet printing based on composition of nanoparticles and organic silver salt. Inorg. Mater. Appl. Res. 2015, 6, 375–381. [Google Scholar] [CrossRef]
- Ning, H.; Tao, R.; Rihui, Y.; Chen, J.Q.; Yongsheng, S. A Review on Green and Flexible Silver Nano-ink for Inkjet Printing. Mater. Rev. 2018, 32, 2959–2968. [Google Scholar]
- Jeong, S.; Song, H.C.; Lee, W.W.; Choi, Y.; Ryu, B.H. Preparation of aqueous Ag Ink with long-term dispersion stability and its inkjet printing for fabricating conductive tracks on a polyimide film. J. Appl. Phys. 2010, 108, 102805. [Google Scholar] [CrossRef]
- Perelaer, B.; Delaat, A.M.; Hendriks, C.; Schubert, U. Inkjet-printed silver tracks: Low temperature curing and thermal stability investigation. J. Mater. Chem. 2008, 18, 3209–3215. [Google Scholar] [CrossRef]
- Tai, Y.L.; Wang, Y.X.; Yang, Z.G.; Chai, Z.Q. Green approach to prepare silver nanoink with potentially high conductivity for printed electronics. Surf. Interface Anal. 2011, 43, 1480–1485. [Google Scholar] [CrossRef]
- Chiolerio, A.; Cotto, M.; Pandolfi, P.; Martino, P.; Camarchia, V.; Pirola, M.; Ghione, G. Ag nanoparticle-based inkjet printed planar transmission lines for RF and microwave applications: Considerations on ink composition, nanoparticle size distribution and sintering time. Microelectron. Eng. 2012, 97, 8–15. [Google Scholar] [CrossRef]
- Tai, Y.-L.; Yang, Z.-G. Facile and Scalable Preparation of Solid Silver Nanoparticles (<10 nm) for Flexible Electronics. ACS Appl. Mater. Interfaces 2015, 7, 17104–17111. [Google Scholar] [CrossRef]
- Trindade, G.F.; Wang, F.; Im, J.; He, Y.; Balogh, A.; Scurr, D.; Gilmore, I.; Tiddia, M.; Saleh, E.; Pervan, D.; et al. Residual polymer stabiliser causes anisotropic electrical conductivity during inkjet printing of metal nanoparticles. Commun. Mater. 2021, 2, 47. [Google Scholar]
- Khondoker, M.A.H.; Mun, S.C.; Kim, J. Synthesis and characterization of conductive silver ink for electrode printing on cellulose film. Appl. Phys. A 2013, 112, 411–418. [Google Scholar] [CrossRef]
- Derby, B. Inkjet Printing of Functional and Structural Materials: Fluid Property Requirements, Feature Stability, and Resolution. Annu. Rev. Mater. Res. 2010, 40, 395–414. [Google Scholar] [CrossRef]
- Grouchko, M.; Roitman, P.; Zhu, X.; Popov, I.; Kamyshny, A.; Su, H.; Magdassi, S. Merging of metal nanoparticles driven by selective wettability of silver nanostructures. Nat. Commun. 2014, 5, 2994. [Google Scholar] [CrossRef]
- Li, D.; Yu, X.; Hu, Y.; Xi, T.; Zhang, Z. Preparation and Characterization of Chitosan-Poloxamer-based Antibacterial Hydrogel Containing Silver Nanoparticles. J. Biomed. Eng. 2016, 33, 1124–1132. [Google Scholar]
- Bonaccorso, F.; Zerbetto, M.; Ferrari, A.C.; Amendola, V. Sorting Nanoparticles by Centrifugal Fields in Clean Media. J. Phys. Chem. C 2013, 117, 13217–13229. [Google Scholar] [CrossRef]
- Jing, X.; Zhao, W.; Lan, L. The effect of particle size on electric conducting percolation threshold in polymer/conducting particle composites. J. Mater. Sci. Lett. 2000, 19, 377–379. [Google Scholar] [CrossRef]
- Hwang, Y.; Lee, J.K.; Lee, J.K.; Jeong, Y.M.; Cheong, S.I.; Ahn, Y.C.; Kim, S.H. Production and dispersion stability of nanoparticles in nanofluids. Powder Technol. 2008, 186, 145–153. [Google Scholar] [CrossRef]
- Decharat, A.; Wagle, S.; Jacobsen, S.; Melandsø, F. Using Silver Nano-Particle Ink in Electrode Fabrication of High Frequency Copolymer Ultrasonic Transducers: Modeling and Experimental Investigation. Sensors 2015, 15, 9210–9227. [Google Scholar] [CrossRef] [PubMed]
- Nakach, M.; Authelin, J.-R.; Perrin, M.-A.; Lakkireddy, H.R. Comparison of high pressure homogenization and stirred bead milling for the production of nano-crystalline suspensions. Int. J. Pharm. 2018, 547, 61–71. [Google Scholar] [CrossRef]
- Alexandridis, P.; Hatton, T.A.J.C.; Physicochemical, S.A.; Aspects, E. Poly(ethylene oxide)-poly(propylene oxide)-poly (ethylene oxide) block copolymer surfactants in aqueous solutions and at interfaces: Thermodynamics, structure, dynamics, and modeling. Colloids Surf. A Physicochem. Eng. Asp. 1995, 96, 1–46. [Google Scholar] [CrossRef]
- Tuinier, R.; Rieger, J.; de Kruif, C. Depletion-induced phase separation in colloid-polymer mixtures. Adv. Colloid Interface Sci. 2003, 103, 1–31. [Google Scholar] [CrossRef]
- Hu, Y.; Yin, G.; Pu, X. Pluronic F127 Regulated Coprecipitation Preparation and Characterization of MnFe2O4 Nanoparticles. Mater. Sci. 2018, 5, 119–125. [Google Scholar]
- Kamyshny, A.; Magdassi, S. Conductive Nanomaterials for Printed Electronics. Small 2015, 10, 3515–3535. [Google Scholar] [CrossRef]
- Curreli, N.; Dessi, C.; Lodi, M.B.; Melis, A.; Simone, M.; Melis, N.; Pilia, L.; Guarnera, D.; Di Donato, L.; Fanti, A.; et al. Cost-Effective Conductive Paste for Radiofrequency Devices Using Carbon-Based Materials. Small Sci. 2024, 4, 2400282. [Google Scholar] [CrossRef] [PubMed]
- Capasso, A.; Castillo, A.D.R.; Sun, H.; Ansaldo, A.; Pellegrini, V.; Bonaccorso, F. Ink-jet printing of graphene for flexible electronics: An environmentally-friendly approach. Solid State Commun. 2015, 224, 53–63. [Google Scholar] [CrossRef]
- Torrisi, F.; Hasan, T.; Wu, W.; Sun, Z.; Lombardo, A.; Kulmala, T.S.; Hsieh, G.W.; Jung, S.; Bonaccorso, F.; Paul, P.J. Inkjet-Printed Graphene Electronics. ACS Nano 2012, 6, 2992–3006. [Google Scholar] [CrossRef]
- Gierczak, M.; Markowski, P.; Zaluk, Z.; Dziedzic, A.; Jankowski-Mihulowicz, P. Ink-jet printed conductive films—Geometrical and electrical characterization. In Proceedings of the 2016 39th International Spring Seminar on Electronics Technology (ISSE), Pilsen, Czech Republic, 18–22 May 2016; pp. 392–397. [Google Scholar]
- Ershov, B.; Ershov, V. Electrochemical Mechanism of Oxidative Dissolution of Silver Nanoparticles in Water: Effect of Size on Electrode Potential and Solubility. Nanomaterials 2023, 13, 12. [Google Scholar] [CrossRef] [PubMed]











| Component | Base Ink (with PVP) | Optimized Ink (F127 Only) | F127/PVP Composite Ink |
|---|---|---|---|
| Ag NPs (Brofos, modified) | 30 | 30 | 30 |
| PVP (K30) | 4.5 | 0 | 2.25 |
| Pluronic F127 | 0 | 10 | 5 |
| TEGME | 42.7 | 42.7 | 42.7 |
| DI water | 15 | 10 | 12.5 |
| IPA | 4.5 | 3 | 3.75 |
| Ethanol | 0.5 | 0.5 | 0.5 |
| Glycerol | 2.5 | 2.5 | 2.5 |
| 2-(2-ethoxyethoxy)ethyl acrylate | 0.1 | 0.1 | 0.1 |
| PVB | 0.1 | 0.1 | 0.1 |
| Defoamer K155 | 0.2 | 0.2 | 0.2 |
| Total | 100.0 | 100.0 | 100.0 |
| Parameter | Value (Mean ± SD) |
|---|---|
| Line width (mm) | 1.20 ± 0.01 |
| Thickness (μm) | 1.87 ± 0.06 |
| Sheet resistance (Ω/sq) | 0.0355 ± 0.002 |
| Conductivity (S/m) | (1.506 ± 0.08) × 107 |
| Coffee-ring effect | Not observed |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Zhou, G.-X.; Wang, Y.; Zhou, X.-P.; Zhang, K.; Yang, Z.-H.; Jia, D.-C.; Zhou, Y. Fabrication of Highly Conductive Inkjet Printing Silver Nanoparticle Ink via a Synergistic Strategy Combining Centrifugal Classification and Dispersant Optimization. Materials 2026, 19, 628. https://doi.org/10.3390/ma19030628
Zhou G-X, Wang Y, Zhou X-P, Zhang K, Yang Z-H, Jia D-C, Zhou Y. Fabrication of Highly Conductive Inkjet Printing Silver Nanoparticle Ink via a Synergistic Strategy Combining Centrifugal Classification and Dispersant Optimization. Materials. 2026; 19(3):628. https://doi.org/10.3390/ma19030628
Chicago/Turabian StyleZhou, Guo-Xiang, Yan Wang, Xing-Ping Zhou, Kuang Zhang, Zhi-Hua Yang, De-Chang Jia, and Yu Zhou. 2026. "Fabrication of Highly Conductive Inkjet Printing Silver Nanoparticle Ink via a Synergistic Strategy Combining Centrifugal Classification and Dispersant Optimization" Materials 19, no. 3: 628. https://doi.org/10.3390/ma19030628
APA StyleZhou, G.-X., Wang, Y., Zhou, X.-P., Zhang, K., Yang, Z.-H., Jia, D.-C., & Zhou, Y. (2026). Fabrication of Highly Conductive Inkjet Printing Silver Nanoparticle Ink via a Synergistic Strategy Combining Centrifugal Classification and Dispersant Optimization. Materials, 19(3), 628. https://doi.org/10.3390/ma19030628








