Fabrication of Copper Nanowires Highly Conductive and Flexible Circuits by Direct Ink Writing
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of Copper Nanowires
2.3. Preparation of Copper Conductive Ink and 3D Printing of Copper Circuits
2.4. Characterization
2.5. Fluid Mechanics Simulation
3. Results and Discussion
3.1. Synthesis of Copper Nanowires
3.2. Preparation of Copper Conductive Ink and 3D Printing of Copper Circuits
3.3. Electrical Properties of Copper Nanowires-Based Flexible Circuits
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Yang, W.; Cheng, X.; Guo, Z.; Sun, Q.; Wang, J.; Wang, C. Design, Fabrication and Applications of Flexible RFID Antennas Based on Printed Electronic Materials and Technologies. J. Mater. Chem. C 2023, 11, 406–425. [Google Scholar] [CrossRef]
- Khan, Y.; Thielens, A.; Muin, S.; Ting, J.; Baumbauer, C.; Arias, A.C. A New Frontier of Printed Electronics: Flexible Hybrid Electronics. Adv. Mater. 2020, 32, 1905279. [Google Scholar] [CrossRef] [PubMed]
- Rivadeneyra, A.; Loghin, F.C.; Falco, A. Technological Integration in Printed Electronics. In Flexible Electronics; IntechOpen: London, UK, 2018; pp. 93–111. ISBN 1-78923-456-5. [Google Scholar]
- Cruz, S.M.F.; Rocha, L.A.; Viana, J.C. Printing Technologies on Flexible Substrates for Printed Electronics. In Flexible Electronics; IntechOpen: London, UK, 2018; ISBN 1-78923-457-3. [Google Scholar]
- Huang, Z.; Tang, Y.; Guo, H.; Feng, X.; Zhang, T.; Li, P.; Qian, B.; Xie, Y. 3D Printing of Ceramics and Graphene Circuits-on-Ceramics by Thermal Bubble Inkjet Technology and High Temperature Sintering. Ceram. Int. 2020, 46, 10096–10104. [Google Scholar] [CrossRef]
- Lee, H.-H.; Chou, K.-S.; Huang, K.-C. Inkjet Printing of Nanosized Silver Colloids. Nanotechnology 2005, 16, 2436. [Google Scholar] [CrossRef]
- Saadi, M.; Maguire, A.; Pottackal, N.T.; Thakur, M.S.H.; Ikram, M.M.; Hart, A.J.; Ajayan, P.M.; Rahman, M.M. Direct Ink Writing: A 3D Printing Technology for Diverse Materials. Adv. Mater. 2022, 34, 2108855. [Google Scholar] [CrossRef]
- Shahzad, A.; Lazoglu, I. Direct Ink Writing (DIW) of Structural and Functional Ceramics: Recent Achievements and Future Challenges. Compos. Part B Eng. 2021, 225, 109249. [Google Scholar] [CrossRef]
- Choi, H.W.; Zhou, T.; Singh, M.; Jabbour, G.E. Recent Developments and Directions in Printed Nanomaterials. Nanoscale 2015, 7, 3338–3355. [Google Scholar] [CrossRef]
- Htwe, Y.Z.N.; Mariatti, M. Printed Graphene and Hybrid Conductive Inks for Flexible, Stretchable, and Wearable Electronics: Progress, Opportunities, and Challenges. J. Sci. Adv. Mater. Devices 2022, 7, 100435. [Google Scholar] [CrossRef]
- Sharma, V.K.; Yngard, R.A.; Lin, Y. Silver Nanoparticles: Green Synthesis and Their Antimicrobial Activities. Adv. Colloid Interface Sci. 2009, 145, 83–96. [Google Scholar] [CrossRef]
- Li, X.; Wang, Y.; Yin, C.; Yin, Z. Copper Nanowires in Recent Electronic Applications: Progress and Perspectives. J. Mater. Chem. C 2020, 8, 849–872. [Google Scholar] [CrossRef]
- Nam, V.B.; Lee, D. Copper Nanowires and Their Applications for Flexible, Transparent Conducting Films: A Review. Nanomaterials 2016, 6, 47. [Google Scholar] [CrossRef]
- Xiong, J.; Wang, Y.; Xue, Q.; Wu, X. Synthesis of Highly Stable Dispersions of Nanosized Copper Particles Using L-Ascorbic Acid. Green Chem. 2011, 13, 900–904. [Google Scholar] [CrossRef]
- Scardaci, V. Copper Nanowires for Transparent Electrodes: Properties, Challenges and Applications. Appl. Sci. 2021, 11, 8035. [Google Scholar] [CrossRef]
- Mardiansyah, D.; Badloe, T.; Triyana, K.; Mehmood, M.Q.; Raeis-Hosseini, N.; Lee, Y.; Sabarman, H.; Kim, K.; Rho, J. Effect of Temperature on the Oxidation of Cu Nanowires and Development of an Easy to Produce, Oxidation-Resistant Transparent Conducting Electrode Using a PEDOT: PSS Coating. Sci. Rep. 2018, 8, 10639. [Google Scholar] [CrossRef]
- Mock, J.; Bobinger, M.; Bogner, C.; Lugli, P.; Becherer, M. Aqueous Synthesis, Degradation, and Encapsulation of Copper Nanowires for Transparent Electrodes. Nanomaterials 2018, 8, 767. [Google Scholar] [CrossRef]
- Liu, B.; Dong, B.; Li, H.; Lou, R.; Chen, Y. 3D Printing Finite Element Analysis of Continuous Fiber Reinforced Composite Materials Considering Printing Pressure. Compos. Part B Eng. 2024, 277, 111397. [Google Scholar] [CrossRef]
- Abbot, D.W.; Kallon, D.V.V.; Anghel, C.; Dube, P. Finite Element Analysis of 3D Printed Model via Compression Tests. Procedia Manuf. 2019, 35, 164–173. [Google Scholar] [CrossRef]
- Li, G.; Jinn, J.T.; Wu, W.T.; Oh, S.I. Recent Development and Applications of Three-Dimensional Finite Element Modeling in Bulk Forming Processes. J. Mater. Process. Technol. 2001, 113, 40–45. [Google Scholar] [CrossRef]
- Yu, C.; Malakpoor, K.; Huyghe, J.M. A Mixed Hybrid Finite Element Framework for the Simulation of Swelling Ionized Hydrogels. Comput. Mech. 2019, 63, 835–852. [Google Scholar] [CrossRef]
- Zheng, Q.; Xie, B.; Xu, Z.; Wu, H. A Systematic Printability Study of Direct Ink Writing towards High-Resolution Rapid Manufacturing. Int. J. Extreme Manuf. 2023, 5, 035002. [Google Scholar] [CrossRef]
- Zhang, H.; Ye, F.; Chen, F.; Yuan, W.; Yan, W. Numerical Investigation on the Viscoelastic Polymer Flow in Material Extrusion Additive Manufacturing. Addit. Manuf. 2024, 81, 103992. [Google Scholar] [CrossRef]
- Sourov, M.A.; Emu, S.I.; Thakur, M.S.H.; Ikram, M.M.; Morshed, A.; Rahman, M.M. A General Simulation-Based Study on Printability of Inks in Direct Ink Writing. Sci. Rep. 2025, 15, 9842. [Google Scholar] [CrossRef]
- Xu, D.; Agassant, J.-F.; Pigeonneau, F. Dimensions of the Deposited Strand in the Material Extrusion Process: Experimental and Numerical Investigations. Addit. Manuf. 2022, 59, 103107. [Google Scholar] [CrossRef]
- Tabakova, S.; Nikolova, E.; Radev, S. Carreau Model for Oscillatory Blood Flow in a Tube. AIP Conf. Proc. 2014, 1629, 336–343. [Google Scholar]
- Lavrov, A. Numerical Modeling of Steady-State Flow of a Non-Newtonian Power-Law Fluid in a Rough-Walled Fracture. Comput. Geotech. 2013, 50, 101–109. [Google Scholar] [CrossRef]
- Huang, X.; Garcia, M.H. A Herschel–Bulkley Model for Mud Flow down a Slope. J. Fluid Mech. 1998, 374, 305–333. [Google Scholar] [CrossRef]
- Ferrer, M.; Pham, A.N.; Waite, T.D. Kinetic Modeling Assisted Analysis of Vitamin C-Mediated Copper Redox Transformations in Aqueous Solutions. J. Phys. Chem. A 2023, 127, 10663–10680. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Sun, X.; Ye, H.; You, T.; Song, X.; Sun, S. Selective Synthesis of Copper Nanoplates and Nanowires via a Surfactant-Assisted Hydrothermal Process. Mater. Chem. Phys. 2010, 120, 1–5. [Google Scholar] [CrossRef]
- Biçer, M.; Şişman, İ. Controlled Synthesis of Copper Nano/Microstructures Using Ascorbic Acid in Aqueous CTAB Solution. Powder Technol. 2010, 198, 279–284. [Google Scholar] [CrossRef]
- Saikia, B.; Suryanarayana, I.; Saikia, B.K.; Haque, I. Hydrogen Bond Formation Between Aliphatic Alcohols and Tertiary Amines. Spectrochim. Acta Part Mol. Spectrosc. 1991, 47, 791–798. [Google Scholar] [CrossRef]
- Zhang, P.; Sun, Q.; Fang, S.; Guo, H.; Liu, K.; Zhang, L.; Zhu, Q.; Wang, M. Fabrication of Nano Copper Highly Conductive and Flexible Printed Electronics by Direct Ink Writing. ACS Appl. Mater. Interfaces 2024, 17, 1847–1860. [Google Scholar] [CrossRef] [PubMed]
- McKennell, R. Cone-Plate Viscometer. Anal. Chem. 1956, 28, 1710–1714. [Google Scholar] [CrossRef]
- Dzisah, P.; Ravindra, N.M. Modeling of Rheological Properties of Metal Nanoparticle Conductive Inks for Printed Electronics. In TMS 2021 150th Annual Meeting & Exhibition Supplemental Proceedings; Springer: Berlin/Heidelberg, Germany, 2021; pp. 964–979. [Google Scholar]
- Liu, K.; Zhang, Y.; Huang, L.; Feng, C.; Li, Y.; Zhang, S.; Jin, X.; Jiang, H.; Zhu, Q.; Zhang, P. Enhanced Printability of High-Viscosity Chitosan/Acrylamide Inks via Aluminum Ions Coordination for Precision 3D Bioprinting of Scaffolds. Carbohydr. Polym. 2025, 355, 123359. [Google Scholar] [CrossRef] [PubMed]
- Fang, S.; Liu, K.; Zhou, X.; Jin, X.; Feng, C.; Zhu, Q.; Jiang, H.; Zhang, P. Design and Evaluation of High-Viscosity Gelatin/Polyacrylamide/Sodium Alginate Triple-Network Hydrogels for 3D Printing: Fluid Dynamics Simulation and Experimental Method. Int. J. Biol. Macromol. 2025, 318, 144886. [Google Scholar] [CrossRef]
- Tang, C.Y.; Yang, Z. Transmission Electron Microscopy (TEM). In Membrane Characterization; Elsevier: Amsterdam, The Netherlands, 2017; pp. 145–159. [Google Scholar]
- Wirth, R. Focused Ion Beam (FIB) Combined with SEM and TEM: Advanced Analytical Tools for Studies of Chemical Composition, Microstructure and Crystal Structure in Geomaterials on a Nanometre Scale. Chem. Geol. 2009, 261, 217–229. [Google Scholar] [CrossRef]
- Fereshteh, Z.; Salavati-Niasari, M.; Saberyan, K.; Hosseinpour-Mashkani, S.M.; Tavakoli, F. Synthesis of Nickel Oxide Nanoparticles from Thermal Decomposition of a New Precursor. J. Clust. Sci. 2012, 23, 577–583. [Google Scholar] [CrossRef]
- Wang, F.; Javaid, S.; Chen, W.; Wang, A.; Buntine, M.A.; Jia, G. Synthesis of Atomically Thin CdTe Nanoplatelets by Using Polytelluride Tellurium Precursors. Aust. J. Chem. 2020, 74, 179–185. [Google Scholar] [CrossRef]
- Bobinger, M.; Mock, J.; La Torraca, P.; Becherer, M.; Lugli, P.; Larcher, L. Tailoring the Aqueous Synthesis and Deposition of Copper Nanowires for Transparent Electrodes and Heaters. Adv. Mater. Interfaces 2017, 4, 1700568. [Google Scholar] [CrossRef]
- Yang, H.-J.; He, S.-Y.; Tuan, H.-Y. Self-Seeded Growth of Five-Fold Twinned Copper Nanowires: Mechanistic Study, Characterization, and SERS Applications. Langmuir 2014, 30, 602–610. [Google Scholar] [CrossRef] [PubMed]
- Bom, S.; Ribeiro, R.; Ribeiro, H.M.; Santos, C.; Marto, J. On the Progress of Hydrogel-Based 3D Printing: Correlating Rheological Properties with Printing Behaviour. Int. J. Pharm. 2022, 615, 121506. [Google Scholar] [CrossRef]
- Kim, J.H.; Lee, S.; Wajahat, M.; Ahn, J.; Pyo, J.; Chang, W.S.; Seol, S.K. 3D Printing of Highly Conductive Silver Architectures Enabled to Sinter at Low Temperatures. Nanoscale 2019, 11, 17682–17688. [Google Scholar] [CrossRef]
- Ogazi, A.C.; Osifo, P.O. Dynamics of Drop Formation and Characterization of Additive Manufactured CS/AgNP/PVA Composite Membranes. J. Appl. Polym. Sci. 2023, 140, e54472. [Google Scholar] [CrossRef]
- Deshmukh, R.; Calvo, M.; Schreck, M.; Tervoort, E.; Sologubenko, A.S.; Niederberger, M. Synthesis, Spray Deposition, and Hot-Press Transfer of Copper Nanowires for Flexible Transparent Electrodes. ACS Appl. Mater. Interfaces 2018, 10, 20748–20754. [Google Scholar] [CrossRef]
- Li, J.; Song, Z.; Liu, Z.; Xie, X.; Guan, P.; Zhu, Y. Exploring Surface-Driven Mechanisms for Low-Temperature Sintering of Nanoscale Copper. Appl. Sci. 2025, 15, 476. [Google Scholar] [CrossRef]
- Lin, B.; Gelves, G.A.; Haber, J.A.; Sundararaj, U. Electrical, Rheological, and Mechanical Properties of Polystyrene/Copper Nanowire Nanocomposites. Ind. Eng. Chem. Res. 2007, 46, 2481–2487. [Google Scholar] [CrossRef]
- Cheng, B.; Ngan, A.H. The Sintering and Densification Behaviour of Many Copper Nanoparticles: A Molecular Dynamics Study. Comput. Mater. Sci. 2013, 74, 1–11. [Google Scholar] [CrossRef]
- Yin, Z.; Cho, S.; You, D.-J.; Ahn, Y.; Yoo, J.; Kim, Y.S. Copper Nanowire/Multi-Walled Carbon Nanotube Composites as All-Nanowire Flexible Electrode for Fast-Charging/Discharging Lithium-Ion Battery. Nano Res. 2018, 11, 769–779. [Google Scholar] [CrossRef]
- Chiguma, J.; Jones Junior, W.E. Template-Free Synthesis of Aligned Polyaniline Nanorods/Tubes and Copper/Copper Hydroxide Nanowires for Application as Fillers in Polymer Nanocomposites. Adv. Mater. Phys. Chem. 2018, 8, 71–88. [Google Scholar] [CrossRef]
- Zhang, B.; Yang, G.; Li, C.; Huang, K.; Wu, J.; Hao, S.; Huang, Y. Electrochemical Behaviors of Hierarchical Copper Nano-Dendrites in Alkaline Media. Nano Res. 2018, 11, 4225–4231. [Google Scholar] [CrossRef]
- Cheng, Y.; Wang, S.; Wang, R.; Sun, J.; Gao, L. Copper Nanowire Based Transparent Conductive Films with High Stability and Superior Stretchability. J. Mater. Chem. C 2014, 2, 5309–5316. [Google Scholar] [CrossRef]
- Ye, E.; Zhang, S.-Y.; Liu, S.; Han, M.-Y. Disproportionation for Growing Copper Nanowires and Their Controlled Self-Assembly Facilitated by Ligand Exchange. Chem. Weinh. Bergstr. Ger. 2011, 17, 3074–3077. [Google Scholar] [CrossRef]
- Jin, M.; He, G.; Zhang, H.; Zeng, J.; Xie, Z.; Xia, Y. Shape-controlled Synthesis of Copper Nanocrystals in an Aqueous Solution with Glucose as a Reducing Agent and Hexadecylamine as a Capping Agent. Angew. Chem. Int. Ed. 2011, 50, 10560–10564. [Google Scholar] [CrossRef]
- Shi, Y.; Li, H.; Chen, L.; Huang, X. Obtaining Ultra-Long Copper Nanowires via a Hydrothermal Process. Sci. Technol. Adv. Mater. 2005, 6, 761–765. [Google Scholar] [CrossRef]
- Huang, X.; Chen, Y.; Chiu, C.-Y.; Zhang, H.; Xu, Y.; Duan, X.; Huang, Y. A Versatile Strategy to the Selective Synthesis of Cu Nanocrystals and the In Situ Conversion to CuRu Nanotubes. Nanoscale 2013, 5, 6284–6290. [Google Scholar] [CrossRef]
- Rathmell, A.R.; Bergin, S.M.; Hua, Y.-L.; Li, Z.-Y.; Wiley, B.J. The Growth Mechanism of Copper Nanowires and Their Properties in Flexible, Transparent Conducting Films. Adv. Mater. 2010, 22, 3558–3563. [Google Scholar] [CrossRef] [PubMed]
- Cui, F.; Yu, Y.; Dou, L.; Sun, J.; Yang, Q.; Schildknecht, C.; Schierle-Arndt, K.; Yang, P. Synthesis of Ultrathin Copper Nanowires Using Tris (Trimethylsilyl) Silane for High-Performance and Low-Haze Transparent Conductors. Nano Lett. 2015, 15, 7610–7615. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.; Lye, M.L.; Zeng, H.C. Large-Scale Synthesis of High-Quality Ultralong Copper Nanowires. Langmuir 2005, 21, 3746–3748. [Google Scholar] [CrossRef]
- Ye, S.; Rathmell, A.R.; Stewart, I.E.; Ha, Y.-C.; Wilson, A.R.; Chen, Z.; Wiley, B.J. A Rapid Synthesis of High Aspect Ratio Copper Nanowires for High-Performance Transparent Conducting Films. Chem. Commun. 2014, 50, 2562–2564. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.I.; Park, I.-J.; Choi, S.-Y. Synthesis of Ultrathin Metal Nanowires with Chemically Exfoliated Tungsten Disulfide Nanosheets. Nano Lett. 2020, 20, 3740–3746. [Google Scholar] [CrossRef]
- Liu, Z.; Yang, Y.; Liang, J.; Hu, Z.; Li, S.; Peng, S.; Qian, Y. Synthesis of Copper Nanowires via a Complex-Surfactant-Assisted Hydrothermal Reduction Process. J. Phys. Chem. B 2003, 107, 12658–12661. [Google Scholar] [CrossRef]
- Mohl, M.; Pusztai, P.; Kukovecz, A.; Konya, Z.; Kukkola, J.; Kordas, K.; Vajtai, R.; Ajayan, P.M. Low-Temperature Large-Scale Synthesis and Electrical Testing of Ultralong Copper Nanowires. Langmuir 2010, 26, 16496–16502. [Google Scholar] [CrossRef] [PubMed]

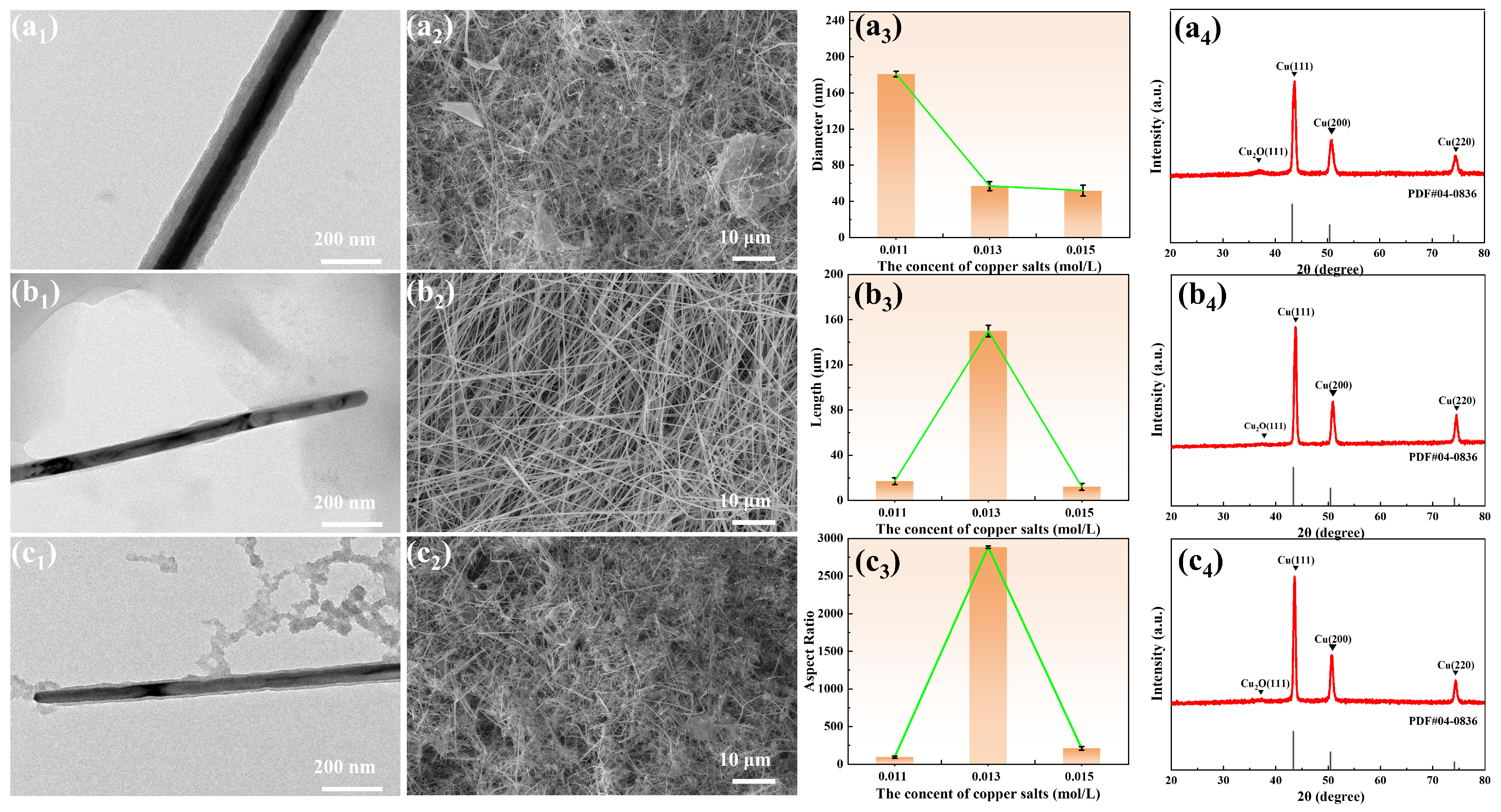


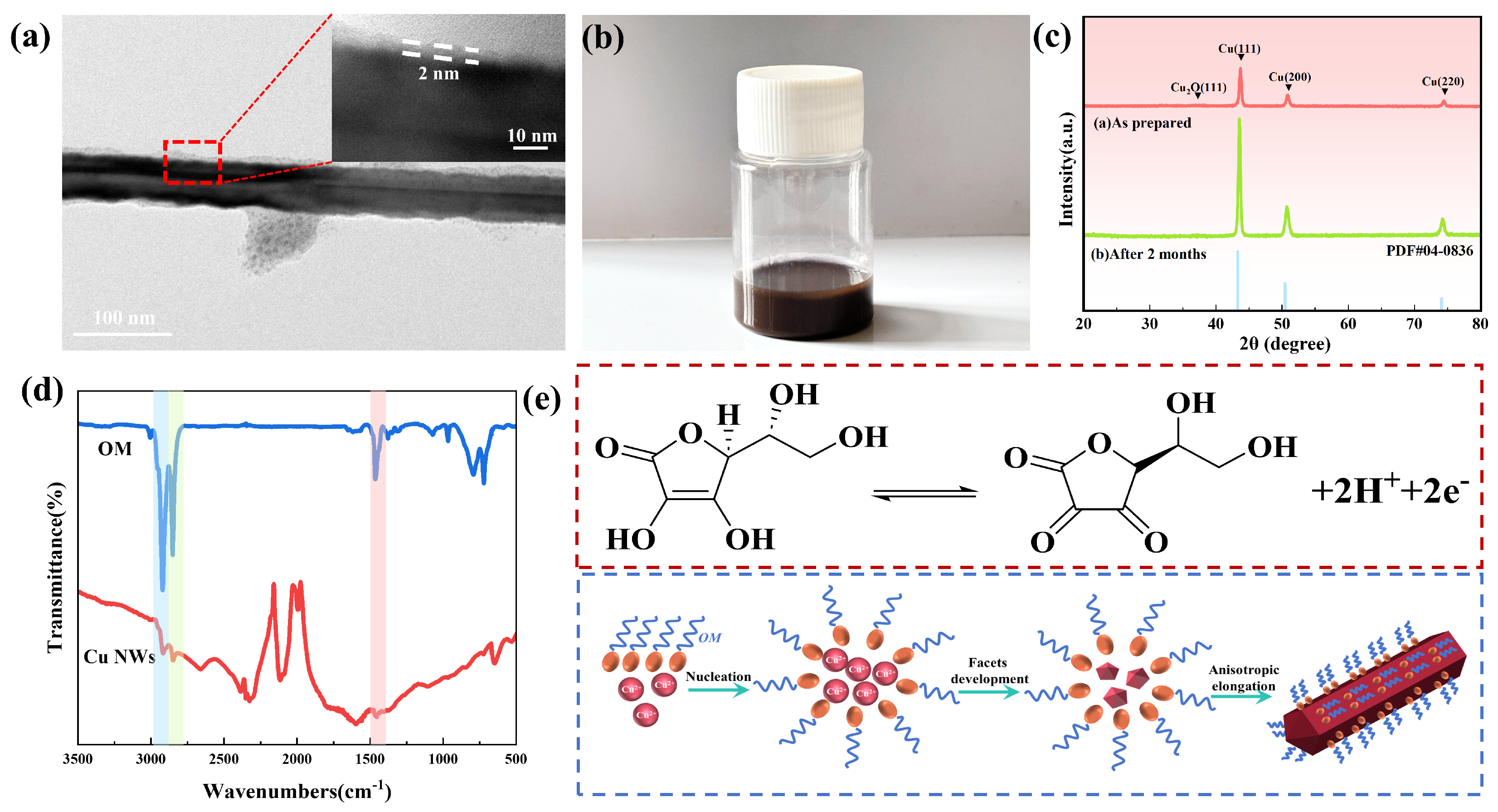





| Sample | Cupric Chloride (mol/L) | Ascorbic Acid (mol/L) | Oleylamine (mol) |
|---|---|---|---|
| 1 | 0.011 | 0.199 | 0.0058 |
| 2 | 0.013 | 0.199 | 0.0058 |
| 3 | 0.015 | 0.199 | 0.0058 |
| 4 | 0.013 | 0.142 | 0.0058 |
| 5 | 0.013 | 0.256 | 0.0058 |
| 6 | 0.013 | 0.199 | 0.0072 |
| 7 | 0.013 | 0.199 | 0.0086 |
| Copper Nanowires Content | Yield Stress (τ0, Pa) | Consistency Coefficient (k, Pa·sⁿ) | Flow Behavior Index (n, Dimensionless) |
|---|---|---|---|
| 10% | 0.00537 | 0.02746 | 0.98635 |
| 20% | 0.02645 | 0.02196 | 1.01918 |
| 30% | 0.0474 | 0.03931 | 0.93381 |
| 40% | 0.16863 | 0.02842 | 1.0707 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Guo, H.; Huang, H.; Shi, S.; Sun, Q.; Sun, J.; Liu, K.; Zhu, Q.; Zhang, P. Fabrication of Copper Nanowires Highly Conductive and Flexible Circuits by Direct Ink Writing. Materials 2026, 19, 618. https://doi.org/10.3390/ma19030618
Guo H, Huang H, Shi S, Sun Q, Sun J, Liu K, Zhu Q, Zhang P. Fabrication of Copper Nanowires Highly Conductive and Flexible Circuits by Direct Ink Writing. Materials. 2026; 19(3):618. https://doi.org/10.3390/ma19030618
Chicago/Turabian StyleGuo, Hui, Haoting Huang, Shijian Shi, Qinghua Sun, Jinping Sun, Kang Liu, Qiang Zhu, and Peng Zhang. 2026. "Fabrication of Copper Nanowires Highly Conductive and Flexible Circuits by Direct Ink Writing" Materials 19, no. 3: 618. https://doi.org/10.3390/ma19030618
APA StyleGuo, H., Huang, H., Shi, S., Sun, Q., Sun, J., Liu, K., Zhu, Q., & Zhang, P. (2026). Fabrication of Copper Nanowires Highly Conductive and Flexible Circuits by Direct Ink Writing. Materials, 19(3), 618. https://doi.org/10.3390/ma19030618








