Effect of Film Thickness and Defects on the Corrosion Behavior of Anodic Oxide Films on Aluminum Alloys
Abstract
1. Introduction
2. Experimental Materials and Methods
2.1. Materials and Sample Preparation
2.2. Experimental Methods
3. Results and Discussion
3.1. Microstructure of Anodized Films
3.2. Macroscopic Corrosion Morphology
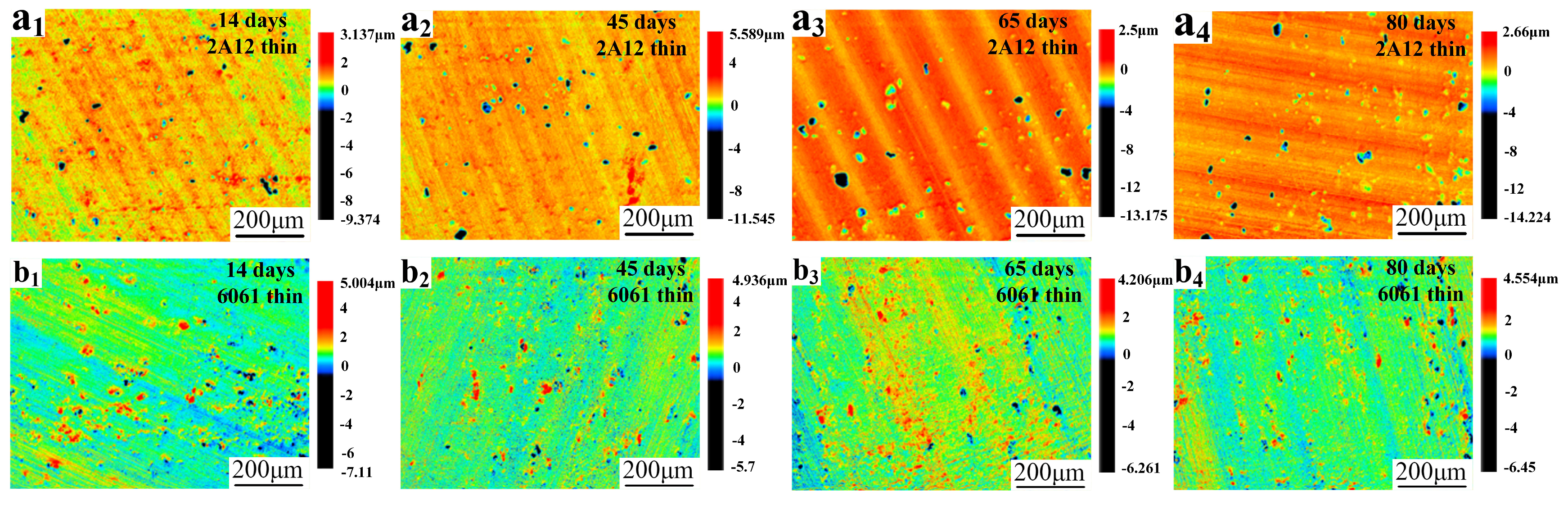
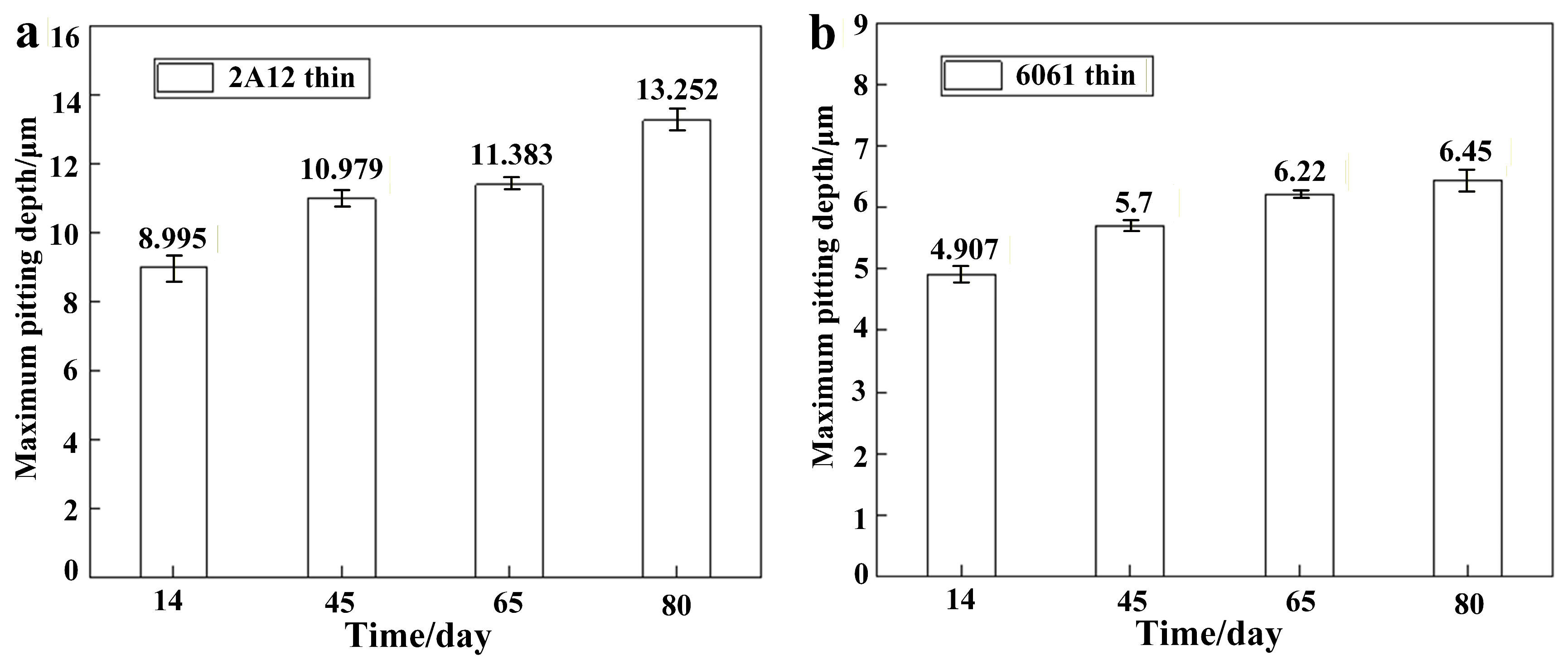
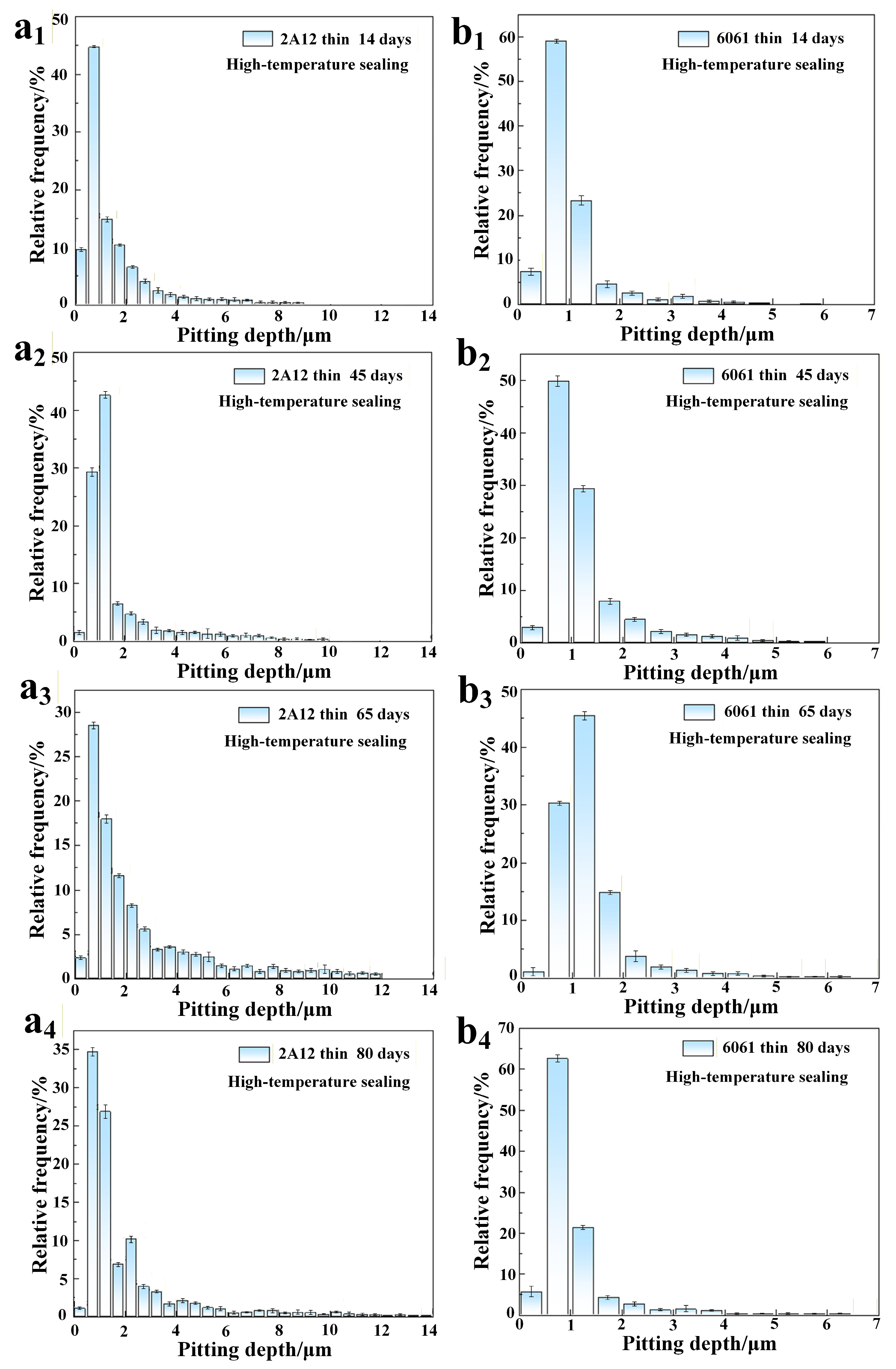
3.3. Microscopic Corrosion Morphology
3.4. Corrosion Product Analysis
4. Conclusions
- The corrosion resistance of both 2A12 and 6061 aluminum alloys increased with the thickness of the anodized film. The pit depth of 2A12 and 6061 thin anodized films (3–5 μm) significantly increased with the salt spray exposure period, whereas the pit development of the thick anodized films (15–20 μm) for both alloys was more gradual, and no corrosion penetrated through the film to the substrate.
- The 2A12 thick anodized film (15–20 μm) exhibited cracks and void defects, with poor adhesion between the film and the substrate, making cracks more prone to long-term corrosion initiation. The 6061 thick anodized film (15–20 μm) contained only a small number of void defects and exhibited superior uniformity and compactness. The film integrity remained good throughout, providing more stable protection to the substrate.
- The corrosion resistance of 6061 aluminum alloy is superior, and both sealing methods provided good protection to the 6061 alloy substrate. The corrosion products of both 2A12 and 6061 samples consisted of Al2O3 and AlO(OH), but the 6061 samples generated fewer corrosion products and exhibited overall better resistance to neutral salt spray corrosion.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Ning, G.; Fu, Y.L.; Wang, Y.Z.; Meng, Q.; Zhu, Y.X. Microstructure and mechanical properties in friction stir welded 5A06 aluminum alloy thick plate. Mater. Des. 2016, 113, 273–283. [Google Scholar]
- Zhang, Y.B.; Zhang, P.; Li, J.H.; Zeng, Q.; Li, M.J.; Hu, Y.F.; Peng, Y.H. Machine-learning assisted compositional optimization of 2xxx series aluminum alloys towards tensile strength. Mater. Res. Express 2023, 10, 016518. [Google Scholar] [CrossRef]
- Mol, S.K.; Sadiq, A. Influence of new AA 2xxx layers on the strength and damage responses of fibre metal laminates during soft body impact. Compos. Struct. 2024, 344, 118339. [Google Scholar] [CrossRef]
- Yang, J.J.; Yin, S.Y.; Fan, C.H.; Ou, L.; Wang, J.H.; Peng, H.; Wang, B.W.; Luo, D.; Dong, S.Y.; Zhang, Z.Y. Effect of voltage on structure and properties of 2024 aluminum alloy surface anodized aluminum oxide films. Surf. Coat. Technol. 2024, 479, 130508. [Google Scholar]
- Nelson, V.; Javier, R.; Claudia, A.; Roberto, S.; Fabiola, P.; Mamie, S.; Lisa, M.; Maritza, P. Alumoxane film for corrosion protection of 2024 aluminum alloy. J. Mater. Res. Technol. 2023, 26, 4942–4956. [Google Scholar] [CrossRef]
- Jiang, H.B.; Li, Q.; Xu, W.; Tian, Y.B.; Ji, P.G.; Tao, W.M.; Fang, F. Effect of Ce on the Microstructure and Corrosion Resistance of Zn-Al-Mg Alloy Coatings. Mater. Prot. 2024, 57, 14–20. [Google Scholar]
- Mendes, L.F.; Moraes, A.S.; Santos, J.S.; Leite, F.L.; Francis, T.S. Investigation of roughness and specular quality of commercial aluminum (6061 alloy) for fabrication of nanoporous anodic alumina films. Surf. Coat. Technol. 2017, 310, 199–206. [Google Scholar] [CrossRef]
- Shen, W.N.; Wang, Y.W.; Li, G.; Ge, Y.F.; Hu, M.Y.; Tang, Y.F. Effects of Coating Thickness on the Microstructure and Properties of Black Micro-Arc Oxidation Ceramic Coating on 6061 Aluminum Alloy. Mater. Prot. 2024, 57, 28–34. [Google Scholar]
- You, X.H.; Xing, Z.Q.; Jiang, S.W.; Zhu, Y.; Lin, Y.H.; Qiu, H.S.; Nie, R.J.; Yang, J.H.; Hui, D.; Chen, W.; et al. A review of research on aluminum alloy materials in structural engineering. Dev. Built Environ. 2024, 17, 100319. [Google Scholar] [CrossRef]
- Donatus, U.; Thompson, G.E.; Zhou, X. Effect of prior sputter deposition of pure aluminium on the corrosion behaviour of anodized friction stir weld of dissimilar aluminium alloys. Scr. Mater. 2016, 123, 126–129. [Google Scholar] [CrossRef]
- Wang, W.; Li, X.J.; Liu, W.J.; Xing, F.; Wang, J.; Zhang, K. Experimental study on hydrophobic properties and corrosivity of laser cleaned 7075 aluminum alloy anodized film surface. Opt. Laser Technol. 2023, 166, 109615. [Google Scholar] [CrossRef]
- Ding, Z. Mechanistic study of thin film sulfuric acid anodizing rate difference between Al2024 T3 and Al6061 T6. Surf. Coat. Technol. 2019, 357, 280–288. [Google Scholar] [CrossRef]
- Shen, X.M.; Wang, J.C. Study on mechanical properties of anodized films on 5052 aluminum alloy. Int. J. Electrochem. Sci. 2022, 17, 221022. [Google Scholar] [CrossRef]
- Li, Y.Z.; Zhu, J.; Wang, J.; Bai, J.D.; Fan, M.J.; Xu, Y.L. Dual-Phase Enabled Sinusoidal Current Anodizing for Efficient Preparation of High Specific Capacitance Anode Aluminum Foils. Adv. Eng. Mater. 2024, 26, 23. [Google Scholar] [CrossRef]
- Yang, Y.S.; Cheng, J.; Liu, S.L.; Wang, H.Y.; Dong, P. Effect of NaAlO2 sealing on corrosion resistance of 2024 aluminum allou anodized film. Mater. Corros. 2019, 70, 120–127. [Google Scholar] [CrossRef]
- Zhang, Y.G.; Chen, Y.L.; Bian, G.X.; Zhang, Y. Electrochemical behavior and corrosion mechanism of anodized 7B04 aluminum alloy in acid NaCl environments. J. Alloys Compd. 2021, 886, 161231. [Google Scholar] [CrossRef]
- Wang, Q.L.; Li, H.J. Study on anodic oxidation of 2099 aluminum lithium alloy and sealing treatment in environmental friendly solutions. Int. J. Electrochem. Sci. 2023, 18, 100186. [Google Scholar] [CrossRef]
- Yu, X.F.; Zhang, G.Q.; Zhang, Z.X.; Wang, Y.G. Research on corrosion resistance of anodized and sealed 6061 aluminum alloy in 3.5% sodium chloride solution. Int. J. Electrochem. Sci. 2023, 18, 5. [Google Scholar] [CrossRef]
- Shin, D.H.; Kim, S.J. Effects of hard anodizing on mechanical and electrochemical characteristics of aluminum alloys under tribocorrosion condition. Corros. Sci. 2024, 240, 112503. [Google Scholar] [CrossRef]
- Zhu, W.B.; Deng, Y.L.; Guo, X.B. Influence of adjusting the anodizing and aging sequences on the microstructure, fatigue property and corrosion resistance of anodized AA6082 alloys. Mater. Charact. 2022, 189, 111941. [Google Scholar] [CrossRef]
- Sulki, L.; Je, C.Y. Phosphoric acid anodizing effect on morphology and corrosion resistance of nanostructured anodic oxide layers on 6061 aluminum alloy. Met. Mater. Int. 2025, 31, 2598–2608. [Google Scholar] [CrossRef]
- Zhao, X.H.; Liu, W.H.; Zuo, Y.; Yang, L.F. The cracking behaviors of anodic films on 1050 and 2024 aluminum alloys after heating up to 300 °C. J. Alloys Compd. 2009, 479, 473–479. [Google Scholar] [CrossRef]
- Qian, J.C.; Xu, B.; Zou, H.Q.; Wu, H.C. Research on Corrosion Behavior of Typical Aluminum Alloy Bottom Layer in Neutral Salt Spray Environment. Mater. Prot. 2023, 56, 74–80. [Google Scholar]
- Liu, N.Z.; Jiang, B.; Ji, Z.S.; Cui, P.X.; Wang, Y.L.; Song, H. Salt spray corrosion and electrochemical corrosion property of anodic oxide films on ADC12 aluminum alloy. Mod. Phys. Lett. B 2022, 36, 04. [Google Scholar] [CrossRef]
- Delphine, V.R.; Najat, C.; Emmanuel, R. Anodizing of multiphase aluminium alloys in sulfuric acid: In-situ electrochemical behaviour and oxide properties. Electrochim. Acta 2016, 211, 1056–1065. [Google Scholar]
- ASTM G46-94; Standard Guide for Examination and Evaluation of Pitting Corrosion. ASTM: West Conshohocken, PA, USA, 2005.
- Liu, J.H.; Shi, H.B.; Yu, M.; Du, R.T.; Rong, G.; Li, S.M. Effect of divalent metal ions on durability and anticorrosion performance of layered double hydroxides on anodized 2A12 aluminum alloy. Surf. Coat. Technol. 2019, 373, 56–64. [Google Scholar] [CrossRef]
- Wan, Y.; Wang, H.; Zhang, Y.D.; Wang, X.M.; Li, Y.B. Study on Anodic Oxidation and Sealing of Aluminum Alloy. Int. J. Electrochem. Sci. 2018, 13, 2175–2185. [Google Scholar] [CrossRef]







| Grade | Si | Fe | Cu | Mn | Mg | Ni | Cr | Zn | Ti | Al |
|---|---|---|---|---|---|---|---|---|---|---|
| 2A12-T4 | 0.5 | 0.5 | 3.8~4.9 | 0.3~0.9 | 1.2~1.8 | 0.1 | - | 0.3 | 0.15 | Bal. |
| 6061-T6 | 0.4~0.8 | 0.7 | 0.15~0.4 | 0.15 | 0.8~1.2 | - | 0.04~0.35 | 0.25 | 0.15 | Bal. |
| Anodizing | Medium-Temperature Sealing | High-Temperature Sealing |
|---|---|---|
| H2SO4: 165–170 g/L Al3+: 10 g/L 18 V 19–21 °C 10–40 min | Ni2+: 1.0–1.1 g/L, NiSO4 pH = 5.45 60 °C 20 min | deionized water pH = 5.5–6.5 97–100 °C 60 min |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Wang, S.; Tao, H.; Zhuo, X.; He, L.; Liu, P.; Dong, K.; Zhang, B.; Xiao, K.; Wu, J. Effect of Film Thickness and Defects on the Corrosion Behavior of Anodic Oxide Films on Aluminum Alloys. Materials 2026, 19, 515. https://doi.org/10.3390/ma19030515
Wang S, Tao H, Zhuo X, He L, Liu P, Dong K, Zhang B, Xiao K, Wu J. Effect of Film Thickness and Defects on the Corrosion Behavior of Anodic Oxide Films on Aluminum Alloys. Materials. 2026; 19(3):515. https://doi.org/10.3390/ma19030515
Chicago/Turabian StyleWang, Song, Huwei Tao, Xianqin Zhuo, Linyue He, Pengfei Liu, Kai Dong, Bowei Zhang, Kui Xiao, and Junsheng Wu. 2026. "Effect of Film Thickness and Defects on the Corrosion Behavior of Anodic Oxide Films on Aluminum Alloys" Materials 19, no. 3: 515. https://doi.org/10.3390/ma19030515
APA StyleWang, S., Tao, H., Zhuo, X., He, L., Liu, P., Dong, K., Zhang, B., Xiao, K., & Wu, J. (2026). Effect of Film Thickness and Defects on the Corrosion Behavior of Anodic Oxide Films on Aluminum Alloys. Materials, 19(3), 515. https://doi.org/10.3390/ma19030515






