In recent years, considerable scholarly attention has been directed toward the development of cementitious materials derived from phosphogypsum. Notable studies by Chen et al. [
4], Deng et al. [
5], You et al. [
6], Liu et al. [
7], Wang et al. [
8], Ren et al. [
9], and Wang et al. [
10] have reported the synthesis of phosphogypsum-based cementitious composites through the incorporation of additives such as slag, fly ash, and other supplementary materials. These innovations not only enabled the stabilization and effective utilization of phosphogypsum but also advanced sustainable, low-carbon practices within the construction industry. Nonetheless, the use of phosphogypsum as a precursor in alkali-activated materials might decrease the initial mechanical strength due to the generation of expansive degradation products. Furthermore, the long-term durability of these materials under extreme environmental conditions has yet to be comprehensively evaluated [
11]. Qu et al. [
12] conducted a comprehensive evaluation of phosphogypsum pretreatment techniques combined with auxiliary cementitious materials, examining their effect on workability, mechanical properties, shrinkage behavior, and transport characteristics of the resulting composites. Li et al. [
13] proposed an environmentally benign ultra-low-alkalinity cementitious material composed of phosphogypsum, granulated fine slag, and sulfoaluminate cement, which demonstrates rapid setting, reduced hydration heat, improved mortar strength, and effective immobilization of hazardous constituents inherent in phosphogypsum. Tang et al. [
14] developed a cementitious material derived from solid waste by blending phosphogypsum with granulated blast furnace slag and fly ash, systematically investigating its mechanical performance, hydration mechanisms, and pollutant stabilization capabilities. In a similar vein, Meng et al. [
15] engineered a high-performance composite cementitious material entirely from solid wastes, offering an innovative strategy for the co-disposal and resource recovery of diverse solid waste streams. Hua et al. [
16] examined a cementitious system primarily composed of phosphogypsum and finely ground blast furnace slag, assessing its effect resistance, flexural strength, freeze-thaw durability, and microstructural features. Shi et al. [
17] prepared environmentally friendly cementitious materials with high proportions of phosphogypsum and hydrated lime, analyzing the effects of raw material ratios on mechanical strength. Furthermore, Yang et al. [
18] formulated backfilling cementitious materials by combining phosphogypsum with slag, utilizing orthogonal experimental designs to optimize material strength across varying proportions and fineness levels, thereby identifying the optimal composition for phosphogypsum-based cementitious applications. These studies found that alkaline activation constitutes a pivotal factor affecting the performance of phosphogypsum-based cementitious materials [
19]. Zhang et al. [
20] incorporated slag and fly ash into hemihydrate phosphogypsum and employed NaOH as an alkaline activator to examine its effect on the mechanical properties of the resulting cementitious composites. Similarly, Ouyang et al. [
21] investigated the effect of steel slag powder as an alkaline activator on the hydration behavior and mechanical strength of sulfate phosphogypsum slag cement. Ma et al. [
22] developed a steel fiber-reinforced super-sulfate cement by combining phosphogypsum, finely ground blast furnace slag, and steel slag, supplemented with alkaline activators such as NaOH, to evaluate its mechanical performance. Lin et al. [
23] formulated composite cementitious materials based on phosphogypsum and slag powder, augmented with quicklime, NaOH, and NaHCO
3, to assess the effects of various admixtures on fluidity, setting time, and compressive strength. Peng et al. [
24] explored the effect of different alkalinity levels on the strength and other properties of hemihydrate phosphogypsum-based cementitious materials. In a related investigation, Lin et al. [
25] examined the effect of varying lime content on the mechanical properties and volumetric expansion of phosphogypsum-based cementitious materials, emphasizing the critical role of alkaline environments in the formation and distribution of alunite. Pan et al. [
26] prepared phosphogypsum-based cementitious slurry backfill materials using phosphogypsum and fly ash as primary raw materials, with red mud serving as an alkaline activator, and analyzed their mechanical characteristics. Sun et al. [
27] assessed the effects of different types and dosages of alkaline materials on impurity content and expansion behavior within phosphogypsum-based cementitious systems. Wang et al. [
28] evaluated the effect of five distinct alkaline activator combinations on the strength of a novel phosphogypsum-based geopolymer cementitious material. Peng et al. [
29] proposed an innovative all-solid-waste cementitious material by combining modified phosphogypsum with finely ground blast furnace slag and steel slag, utilizing sodium hydroxide as the alkaline activator to investigate its strength development. Although these studies have clarified the effects of varying alkali dosages on mechanical properties, there remains a paucity of comprehensive and systematic research specifically addressing the role of alkaline modulus in these materials.
Among the multiple factors affecting the performance of phosphogypsum-based cementitious materials, the alkali modulus of the alkali activator emerges as a critical parameter governing their behavior [
30,
31]. However, most existing studies primarily focus on the effects of individual alkali activator parameters, lacking a comprehensive evaluation of the synergistic interactions between alkali dosage and alkali modulus [
32]. This gap impedes a thorough understanding of the interactive mechanisms through which these factors affect hydration kinetics and microstructural evolution. For instance, in the development of geopolymer formulations utilizing the Design of Experiments methodology, it has been demonstrated that interactions between alkali-related parameters and other variables—such as precursor ratios and curing regimes—significantly affect material performance. This finding underscores the necessity for systematic optimization of these parameters. Additionally, distinct precursor combinations demand specific alkali modulus values, emphasizing the need for targeted investigations tailored to particular mix designs [
33]. Moreover, the current literature reveals a notable gap regarding the adaptability of alkali parameters within diverse slag–phosphogypsum–fly ash blended systems, with limited attention given to the interactive effects arising from variations in substrate proportions and alkali activator characteristics [
34].
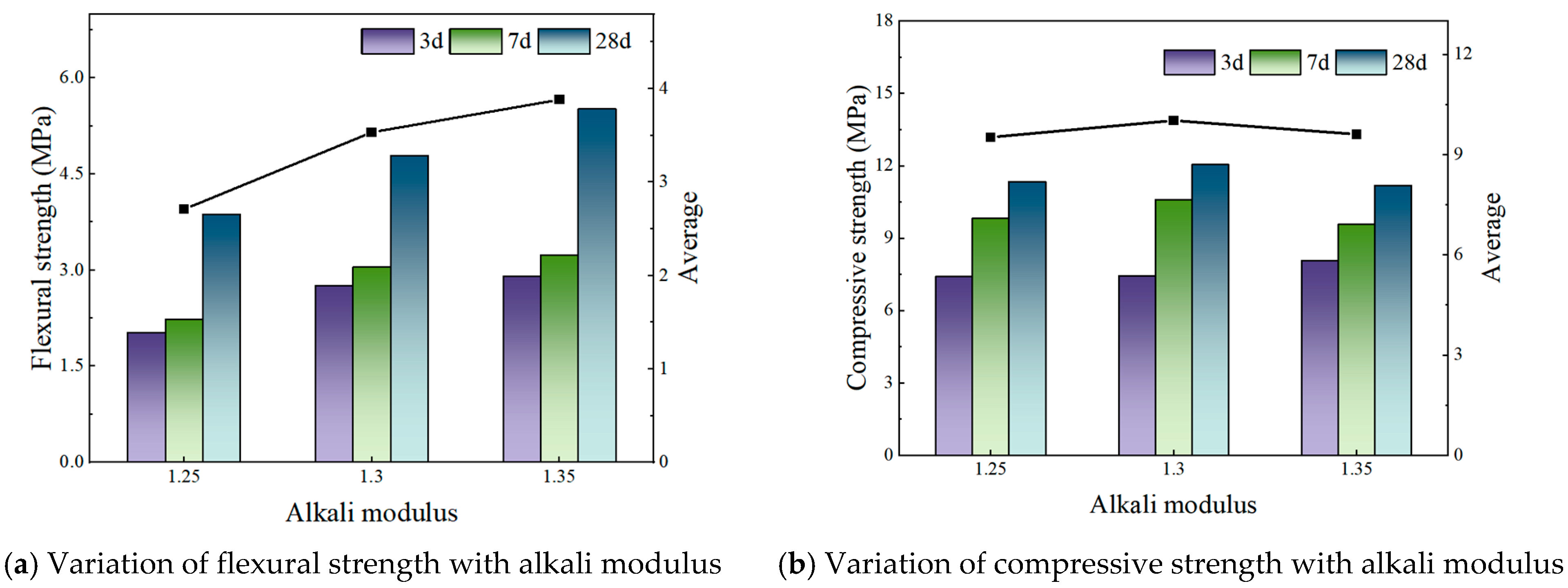
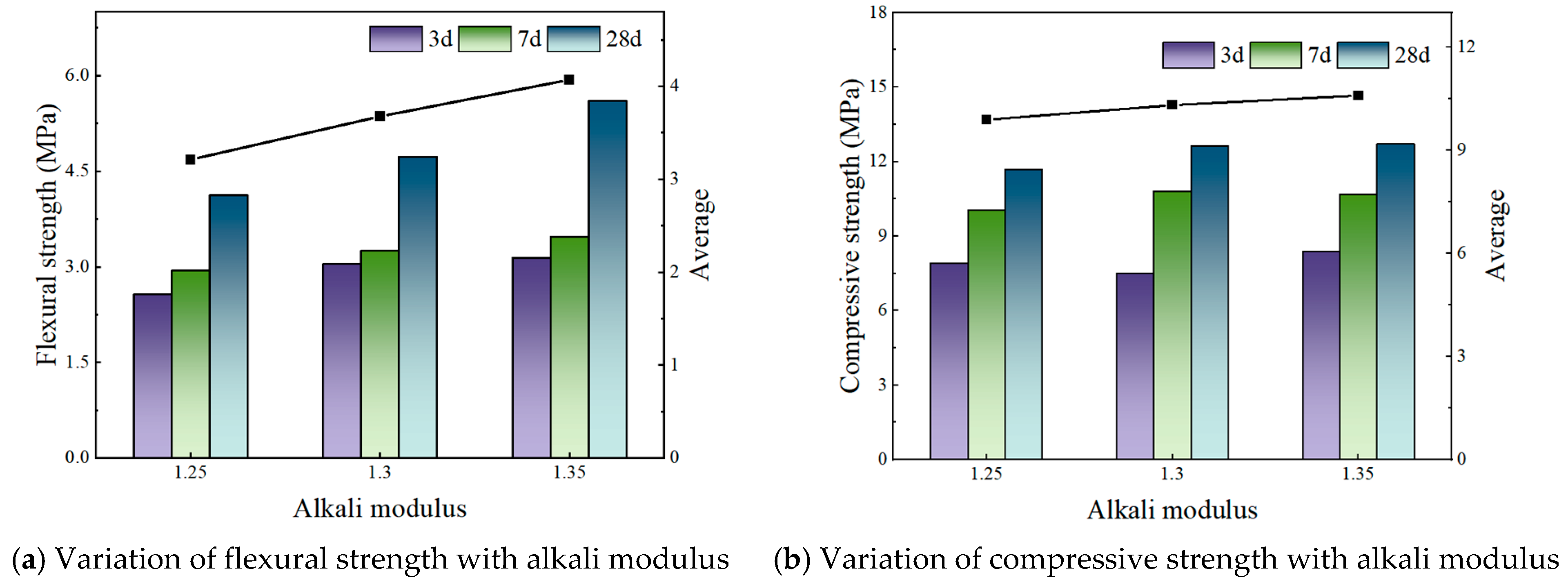
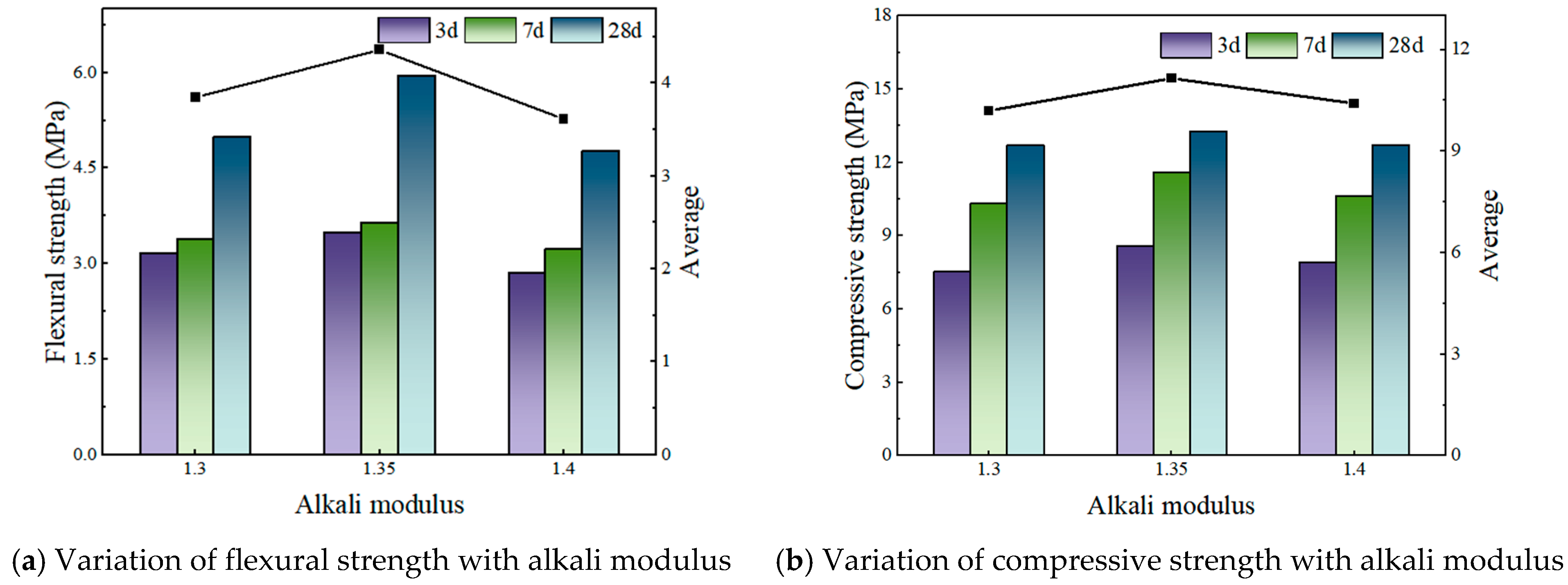
Addressing these challenges, this study focuses on the alkaline modulus as a key regulatory factor, systematically evaluating its effect on the early-age properties of slag–phosphogypsum composite alkali-activated materials (HSFP) across various slag–phosphogypsum–fly ash composite systems. The synergistic effect of the alkaline modulus on the formation of hydration products is also investigated by microscopic characterization methods, such as X-ray diffraction (XRD), scanning electron microscopy (SEM), and thermogravimetric-differential scanning calorimetry (TG-DSC). The results identify optimal conditions for enhancing mechanical properties, thereby providing both a theoretical framework and practical recommendations for the optimization of mix designs and facilitating the large-scale utilization of phosphogypsum-based cementitious materials.