Toward Safer and Greener Insulation: Formaldehyde-Free, Flame-Retardant, and Bio-Based Phenolic Foams from Tannin and Modified-Lignin Combination
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Materials
2.2. EtOH-Induced Hydrogenolysis Thermal Degradation of Lignin
2.3. Foam Preparation Procedure
2.4. Characterization and Testing
3. Results
3.1. Lignin Degradation/Activation
3.1.1. Microscopic Characterization of Treated Lignin Powders
3.1.2. Functionality Evolution
3.1.3. Thermal Degradation
3.2. Foams Properties
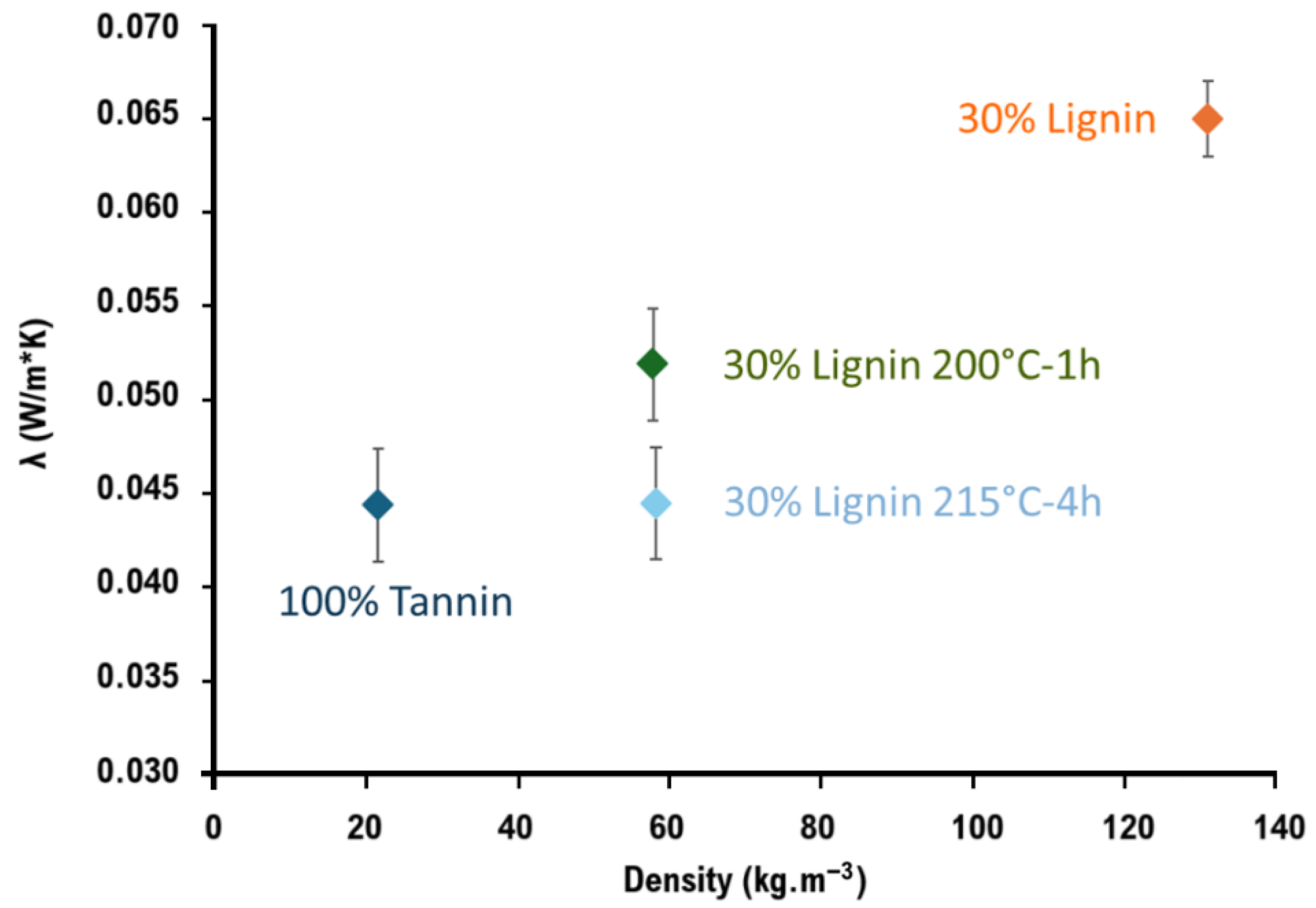
3.2.1. Foam Density and Thermal Conductivity
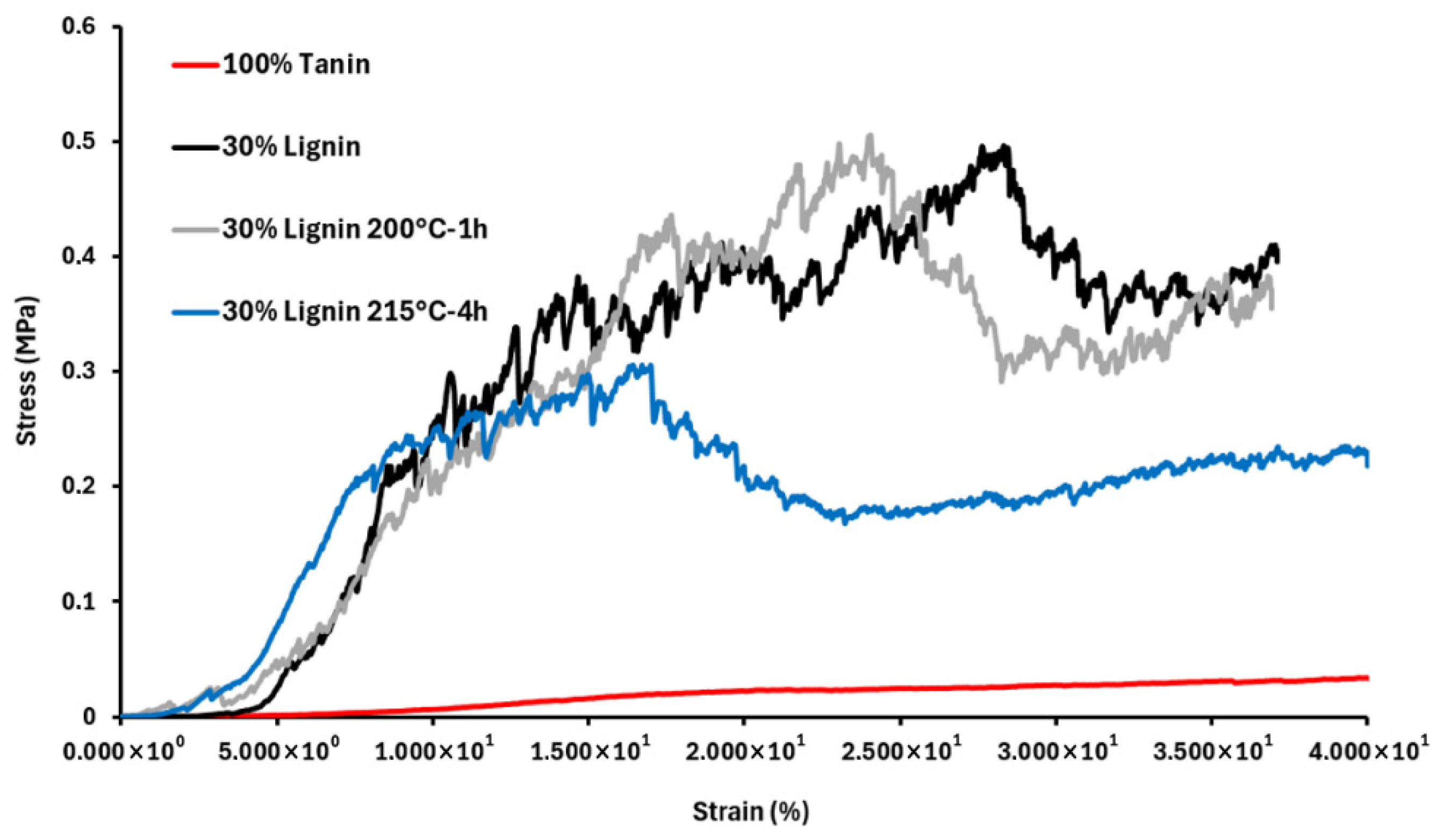
3.2.2. Foam Mechanical Performances
3.2.3. Foam Thermal Degradation
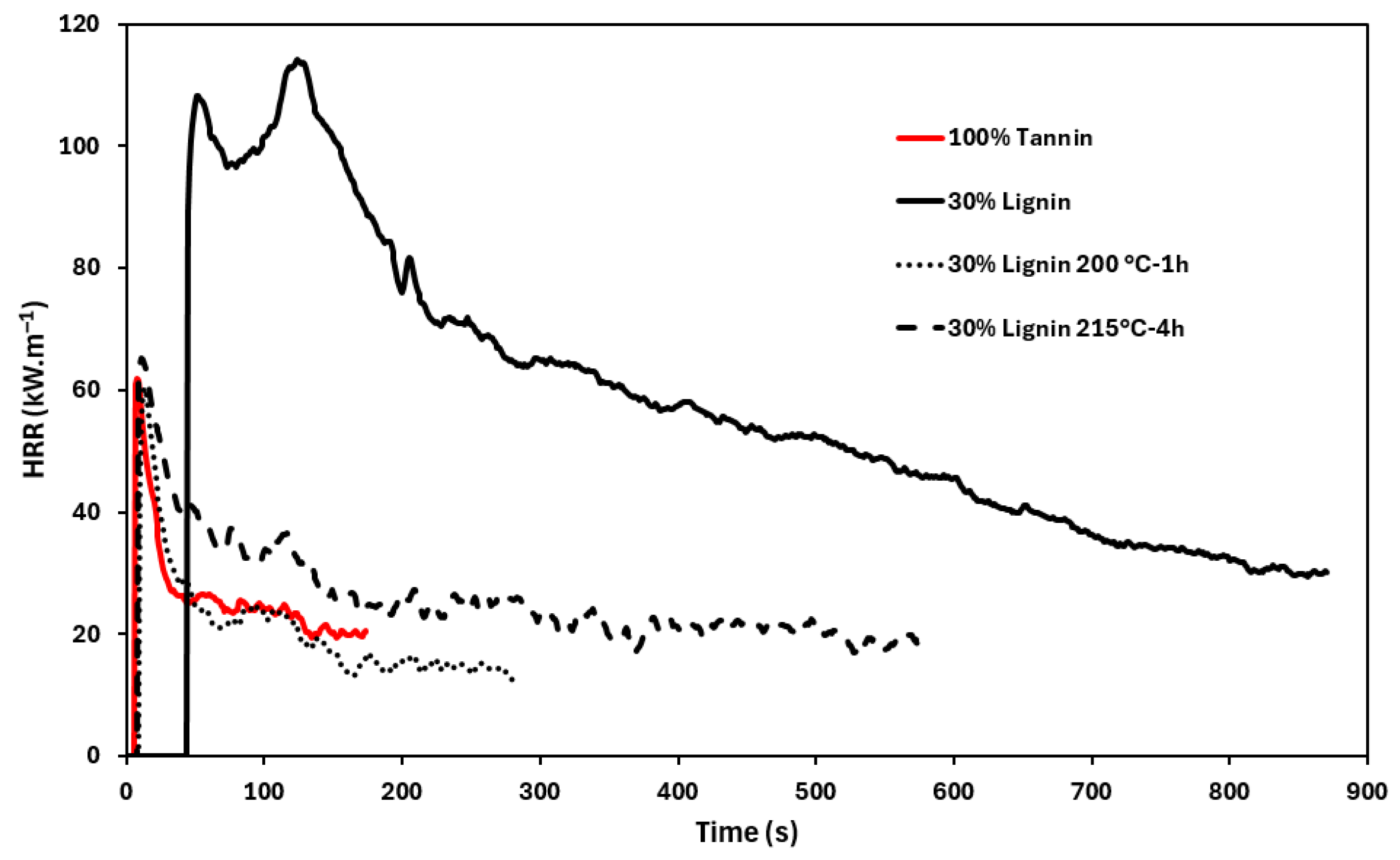
3.2.4. Foam Flame-Retardant Properties
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| TGA | Thermogravimetric analysis |
| MLC | Mass loss cone calorimeter |
| pHRR | Peak heat release rate |
| PIR | Polyisocyanurate |
| SEM | Scanning electron microscopy |
| PTSA | Para-toluene sulphonic acid |
| FTIR | Fourier transform–infrared spectroscopy |
| TTI | Time to ignition |
| THR | Total heat release |
| PU | Polyurethane |
References
- Bo, C.; Wei, S.; Hu, L.; Jia, P.; Liang, B.; Zhou, J.; Zhou, Y. Synthesis of a Cardanol-Based Phosphorus-Containing Polyurethane Prepolymer and Its Application in Phenolic Foams. RSC Adv. 2016, 6, 62999–63005. [Google Scholar] [CrossRef]
- Yu, Y.; Wang, Y.; Xu, P.; Chang, J. Preparation and Characterization of Phenolic Foam Modified with Bio-Oil. Materials 2018, 11, 2228. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Chen, X.; Fan, Z.; Ni, R.; Liu, X.; Tian, Y.; Zhou, B. Using Lignin Degraded to Synthesize Phenolic Foams with Excellent Flame Retardant Property. Colloids Surf. A Physicochem. Eng. Asp. 2023, 666, 131373. [Google Scholar] [CrossRef]
- Yang, C.; Zhuang, Z.; Yang, Z. Pulverized Polyurethane Foam Particles Reinforced Rigid Polyurethane Foam and Phenolic Foam. J. Appl. Polym. Sci. 2014, 131, app.39734. [Google Scholar] [CrossRef]
- Qiao, W.; Li, S.; Guo, G.; Han, S.; Ren, S.; Ma, Y. Synthesis and Characterization of Phenol-Formaldehyde Resin Using Enzymatic Hydrolysis Lignin. J. Ind. Eng. Chem. 2015, 21, 1417–1422. [Google Scholar] [CrossRef]
- Qu, J.; Han, Q.; Gao, F.; Qiu, J. Carbon Foams Produced from Lignin-Phenol-Formaldehyde Resin for Oil/Water Separation. New Carbon Mater. 2017, 32, 86–91. [Google Scholar] [CrossRef]
- Liang, B.; Li, X.; Hu, L.; Bo, C.; Zhou, J.; Zhou, Y. Foaming Resol Resin Modified with Polyhydroxylated Cardanol and Its Application to Phenolic Foams. Ind. Crops Prod. 2016, 80, 194–196. [Google Scholar] [CrossRef]
- Dunky, M. Wood Adhesives Based on Natural Resources: A Critical Review: Part III. Tannin- and Lignin-Based Adhesives. In Progress in Adhesion and Adhesives; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2021; pp. 383–529. [Google Scholar] [CrossRef]
- Sui, G.; Cheng, Y.; Yang, X.; Wang, X.; Wang, Z. Use of Sustainable Glucose and Furfural in the Synthesis of Formaldehyde-free Phenolic Resole Resins. J. Appl. Polym. Sci. 2019, 136, 47733. [Google Scholar] [CrossRef]
- Ballerini, A.; Despres, A.; Pizzi, A. Non-Toxic, Zero Emission Tannin-Glyoxal Adhesives for Wood Panels. Holz Roh Werkst. 2005, 63, 477–478. [Google Scholar] [CrossRef]
- Li, J.; Wang, W.; Zhang, S.; Gao, Q.; Zhang, W.; Li, J. Preparation and Characterization of Lignin Demethylated at Atmospheric Pressure and Its Application in Fast Curing Biobased Phenolic Resins. RSC Adv. 2016, 6, 67435–67443. [Google Scholar] [CrossRef]
- Londoño Zuluaga, C.; Du, J.; Chang, H.-M.; Jameel, H.; Gonzalez, R.W. Lignin Modifications and Perspectives towards Applications of Phenolic Foams: A Review. BioResources 2018, 13, 9158–9179. [Google Scholar] [CrossRef]
- Yang, S.; Wen, J.-L.; Yuan, T.-Q.; Sun, R.-C. Characterization and Phenolation of Biorefinery Technical Lignins for Lignin–Phenol–Formaldehyde Resin Adhesive Synthesis. RSC Adv. 2014, 4, 57996–58004. [Google Scholar] [CrossRef]
- Meikleham, N.E.; Pizzi, A. Acid- and Alkali-Catalyzed Tannin-Based Rigid Foams. J. Appl. Polym. Sci. 1994, 53, 1547–1556. [Google Scholar] [CrossRef]
- Tondi, G.; Pizzi, A.; Olives, R. Natural Tannin-Based Rigid Foams as Insulation for Doors and Wall Panels. Maderas Cienc. Tecnol. 2008, 10, 219–227. [Google Scholar] [CrossRef]
- Issaoui, H.; De Hoyos-Martinez, P.L.; Pellerin, V.; Dourges, M.-A.; Deleuze, H.; Bourbigo, S.; Charrier-El Bouhtoury, F. Effect of Catalysts and Curing Temperature on the Properties of Biosourced Phenolic Foams. ACS Sustain. Chem. Eng. 2021, 9, 6209–6223. [Google Scholar] [CrossRef]
- Merle, J.; Birot, M.; Deleuze, H.; Mitterer, C.; Carré, H.; Bouhtoury, F.C.-E. New Biobased Foams from Wood Byproducts. Mater. Des. 2016, 91, 186–192. [Google Scholar] [CrossRef]
- Kong, X.; Liu, C.; Fan, Y.; Li, M.; Xiao, R. Depolymerization of Technical Lignin to Valuable Platform Aromatics in Lower Alcohol without Added Catalyst and External Hydrogen. Fuel Process. Technol. 2023, 242, 107637. [Google Scholar] [CrossRef]
- ISO 17554; Reaction to Fire Tests—Mass Loss Measurement. ISO: Geneva, Switzerland, 2014.
- ASTM D3801; Standard Test Method for Measuring the Comparative Burning Characteristics of Solid Plastics in a Vertical Position. ASTM: West Conshohocken, PA, USA, 2020.
- Lee, S.; Teramoto, Y.; Shiraishi, N. Resol-type Phenolic Resin from Liquefied Phenolated Wood and Its Application to Phenolic Foam. J. Appl. Polym. Sci. 2002, 84, 468–472. [Google Scholar] [CrossRef]
- Auad, M.L.; Zhao, L.; Shen, H.; Nutt, S.R.; Sorathia, U. Flammability Properties and Mechanical Performance of Epoxy Modified Phenolic Foams. J. Appl. Polym. Sci. 2007, 104, 1399–1407. [Google Scholar] [CrossRef]
- Delgado-Sánchez, C.; Sarazin, J.; Santiago-Medina, F.J.; Fierro, V.; Pizzi, A.; Bourbigot, S.; Celzard, A. Impact of the Formulation of Biosourced Phenolic Foams on Their Fire Properties. Polym. Degrad. Stab. 2018, 153, 1–14. [Google Scholar] [CrossRef]









| Wave Number (cm−1) | Corresponding Group |
|---|---|
| 3335 | O-H bonds |
| 2932 | CH aliphatic |
| 1582 | C=O organic acid salt |
| 1367 | OH phenolic |
| 1262 | C-O aromatic ether (syringyl) |
| 1205 | C-O tertiary alcohol (guaiacyl) |
| 1125 | C-O aliphatic ether -O- linkage |
| 1033 | C-O primary alcohol or methoxy groups |
| 854 | CH aromatic |
| 744 | CH aromatic |
| 627 | CH deformation |
| mmol RCOOH/g | mmol OH/g | |
|---|---|---|
| Lignin | 2.91 | 4.55 |
| 200 °C-1 h | 3.06 | 2.85 |
| 215 °C-4 h | 3.24 | 1.18 |
| Sample | Density (kg/m3) | Thermal Conductivity λ [W·m−1·K−1] | Elastic Modulus (MPa) | Compression Strength (MPa) |
|---|---|---|---|---|
| Phenolic foam (standard) | 32–120 | 0.021–0.045 | NR | 0.10–0.12 |
| 100% Tannin | 21.5 | 0.044 ± 0.003 | 0.43 ± 0.2 | 0.04 ± 0.01 |
| 30% Lignin | 131.0 | 0.065 ± 0.002 | 19.5 ± 6.6 | 0.42 ± 0.10 |
| 30% Lignin 200 °C-1 h | 57.8 | 0.052 ± 0.003 | 7.45 ± 1.4 | 0.45 ± 0.07 |
| 30% Lignin 215 °C-4 h | 58.2 | 0.045 ± 0.003 | 7.3 ± 0.8 | 0.26 ± 0.06 |
| Sample | TTI (s | pHRR (kW·m−2) | THR (MJ.m−2) | Combustion Efficiency (MJ·m−2·g−1) | UL-94 |
|---|---|---|---|---|---|
| 100% Tannin | 8 ± 2 | 47 ± 13 | 2.5 ± 0.4 | 0.26 | V-0 |
| 30% Lignin | 43 ± 9 | 107 ± 7 | 31 ± 9 | 0.53 | V-0 |
| 30% Lignin 200 °C-1 h | 9 ± 3 | 52 ± 5 | 4.3 ± 0.7 | 0.22 | V-0 |
| 30% Lignin 215 °C-4 h | 11 ± 5 | 45 ± 15 | 4.8 ± 0.3 | 0.16 | V-0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Lazko, J.; Mariage, J.; Joyet, C.; Layachi, A.; Satha, H.; Dubois, P.; Laoutid, F. Toward Safer and Greener Insulation: Formaldehyde-Free, Flame-Retardant, and Bio-Based Phenolic Foams from Tannin and Modified-Lignin Combination. Materials 2026, 19, 334. https://doi.org/10.3390/ma19020334
Lazko J, Mariage J, Joyet C, Layachi A, Satha H, Dubois P, Laoutid F. Toward Safer and Greener Insulation: Formaldehyde-Free, Flame-Retardant, and Bio-Based Phenolic Foams from Tannin and Modified-Lignin Combination. Materials. 2026; 19(2):334. https://doi.org/10.3390/ma19020334
Chicago/Turabian StyleLazko, Jevgenij, Jérôme Mariage, Célia Joyet, Abdelheq Layachi, Hamid Satha, Philippe Dubois, and Fouad Laoutid. 2026. "Toward Safer and Greener Insulation: Formaldehyde-Free, Flame-Retardant, and Bio-Based Phenolic Foams from Tannin and Modified-Lignin Combination" Materials 19, no. 2: 334. https://doi.org/10.3390/ma19020334
APA StyleLazko, J., Mariage, J., Joyet, C., Layachi, A., Satha, H., Dubois, P., & Laoutid, F. (2026). Toward Safer and Greener Insulation: Formaldehyde-Free, Flame-Retardant, and Bio-Based Phenolic Foams from Tannin and Modified-Lignin Combination. Materials, 19(2), 334. https://doi.org/10.3390/ma19020334









