Bone-Healing Enhancement Using Particulate Biomaterials and Fibrin-Based Compounds: A Narrative Literature Review of Evidence in Animal Models
Abstract
1. Introduction
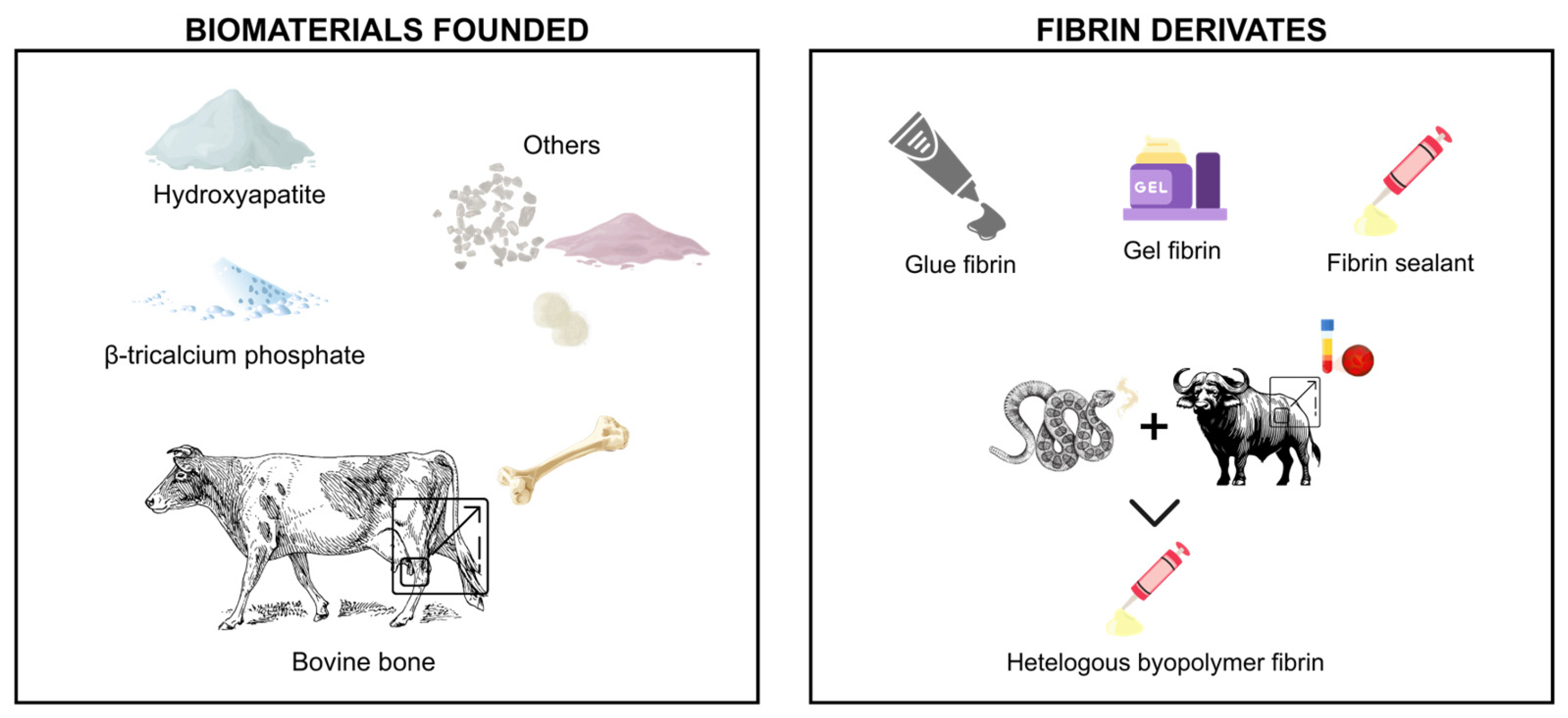
2. Particulate Biomaterials
2.1. Bioceramics
2.2. Hydroxyapatite
2.3. β-Tricalcium Phosphate (β-TCP)
2.4. Bioglass
2.5. Bovine Bone
3. Fibrin-Based Biomaterials
3.1. Fibrin Glue
3.2. Fibrin Sealant
3.3. Platelet-Rich-Fibrin (PRF)
4. Association of Biomaterials with Fibrin Derivates
5. Comparative Analysis of the Studies Summarized in Table 1
6. Conclusions
Future Research Roadmap
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| HA | Hydroxyapatite |
| β-TCP | Beta-Tricalcium Phosphate |
| BCP | Biphasic Calcium Phosphate |
| Ca/P | Calcium/Phosphate ratio |
| CaCO3 | Calcium Carbonate |
| ACP | Amorphous Calcium Phosphate |
| OCP | Octacalcium Phosphate |
| DBBM | Deproteinized Bovine Bone Mineral |
| DBB | Deproteinized Bovine Bone |
| PRF | Platelet-Rich Fibrin |
| PRP | Platelet-Rich Plasma |
| FG | Fibrin Glue |
| FS | Fibrin Sealant |
| HFB | Heterologous Fibrin Biopolymer |
| MBG | Mesoporous Bioactive Glass |
| MSCs | Mesenchymal Stem Cells |
| PBM | Photobiomodulation |
| CEVAP | Centro de Estudos de Venenos e Animais Peçonhentos (UNESP) |
| ECP | Experimental Calcium Phosphate |
| nHA | Nano-Hydroxyapatite |
| TG2 | Transglutaminase Type 2 |
| EMSCs | Ectomesenchymal Stem Cells |
| BMP-2/rhBMP-2 | Bone Morphogenetic Protein-2/Recombinant Human BMP-2 |
| PHF/PHFL | Groups with Deproteinized Bovine Bone + Fibrin + Photobiomodulation (Laser) |
References
- Maciel, G.B.M.; Maciel, R.M.; Danesi, C.C. Bone Cells and Their Role in Physiological Remodeling. Mol. Biol. Rep. 2023, 50, 2857–2863. [Google Scholar] [CrossRef]
- Xu, J.; Yu, L.; Liu, F.; Wan, L.; Deng, Z. The Effect of Cytokines on Osteoblasts and Osteoclasts in Bone Remodeling in Osteoporosis: A Review. Front. Immunol. 2023, 14, 1222129. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.; Kim, J.H.; Shim, J.H.; Hwang, N.S.; Heo, C.Y. Bioactive Calcium Phosphate Materials and Applications in Bone Regeneration. Biomater. Res. 2019, 23, 4. [Google Scholar] [CrossRef] [PubMed]
- De Pace, R.; Molinari, S.; Mazzoni, E.; Perale, G. Bone Regeneration: A Review of Current Treatment Strategies. J. Clin. Med. 2025, 14, 1838. [Google Scholar] [CrossRef] [PubMed]
- Tang, G.; Liu, Z.; Liu, Y.; Yu, J.; Wang, X.; Tan, Z.; Ye, X. Recent Trends in the Development of Bone Regenerative Biomaterials. Front. Cell Dev. Biol. 2021, 9, 665813. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, W.; Yue, W.; Qin, W.; Zhao, Y.; Xu, G. Research Progress of Bone Grafting: A Comprehensive Review. Int. J. Nanomed. 2025, 20, 4729–4757. [Google Scholar] [CrossRef]
- Kohli, N.; Ho, S.; Brown, S.J.; Sawadkar, P.; Sharma, V.; Snow, M.; García-Gareta, E. Bone Remodelling In Vitro: Where Are We Headed?:—A Review on the Current Understanding of Physiological Bone Remodelling and Inflammation and the Strategies for Testing Biomaterials In Vitro. Bone 2018, 110, 38–46. [Google Scholar] [CrossRef]
- Iaquinta, M.R.; Mazzoni, E.; Manfrini, M.; D’Agostino, A.; Trevisiol, L.; Nocini, R.; Trombelli, L.; Barbanti-Brodano, G.; Martini, F.; Tognon, M. Innovative Biomaterials for Bone Regrowth. Int. J. Mol. Sci. 2019, 20, 618. [Google Scholar] [CrossRef]
- Martin, V.; Bettencourt, A. Bone Regeneration: Biomaterials as Local Delivery Systems with Improved Osteoinductive Properties. Mater. Sci. Eng. C 2018, 82, 363–371. [Google Scholar] [CrossRef]
- Azi, M.L.; Aprato, A.; Santi, I.; Kfuri, M., Jr.; Masse, A.; Joeris, A. Autologous Bone Graft in the Treatment of Post-Traumatic Bone Defects: A Systematic Review and Meta-Analysis. BMC Musculoskelet. Disord. 2016, 17, 465. [Google Scholar] [CrossRef]
- Schmidt, A.H. Autologous Bone Graft: Is It Still the Gold Standard? Injury 2021, 52, S18–S22. [Google Scholar] [CrossRef] [PubMed]
- Sohn, H.S.; Oh, J.K. Review of Bone Graft and Bone Substitutes with an Emphasis on Fracture Surgeries. Biomater. Res. 2019, 23, 9. [Google Scholar] [CrossRef] [PubMed]
- Chang, D.G.; Park, J.B.; Han, Y. Surgical Outcomes of Two Kinds of Demineralized Bone Matrix Putties/Local Autograft Composites in Instrumented Posterolateral Lumbar Fusion. BMC Musculoskelet. Disord. 2021, 22, 200. [Google Scholar] [CrossRef] [PubMed]
- Pagani, B.T.; De Oliveira Rosso, M.P.; Moscatel, M.B.M.; De Moraes Trazzi, B.F.; Da Cunha, M.R.; Issa, J.P.M.; Buchaim, D.V.; Buchaim, R.L. Update on Synthetic Biomaterials Combined with Fibrin Derivatives for Regenerative Medicine: Applications in Bone Defect Treatment: Systematic Review. World J. Orthop. 2025, 16, 106181. [Google Scholar] [CrossRef]
- Łuczak, J.W.; Palusińska, M.; Matak, D.; Pietrzak, D.; Nakielski, P.; Lewicki, S.; Grodzik, M.; Szymański, Ł. The Future of Bone Repair: Emerging Technologies and Biomaterials in Bone Regeneration. Int. J. Mol. Sci. 2024, 25, 12766. [Google Scholar] [CrossRef]
- Xing, Y.; Qiu, L.; Liu, D.; Dai, S.; Sheu, C.L. The Role of Smart Polymeric Biomaterials in Bone Regeneration: A Review. Front. Bioeng. Biotechnol. 2023, 11, 1240861. [Google Scholar] [CrossRef]
- Dallabrida, A.L.; Camargo, N.H.A.; Moraes, A.N.; Gava, A.; Dalmônico, G.M.L.; Costa, B.D.; Oleskovicz, N. Bioceramic Characterization of Calcium Phosphates Microstructured in Different Composition in Sheep. Pesqui. Vet. Bras. 2018, 38, 1327–1336. [Google Scholar] [CrossRef]
- Pabst, A.; Becker, P.; Götz, W.; Heimes, D.; Thiem, D.G.E.; Blatt, S.; Kämmerer, P.W. A Comparative Analysis of Particulate Bovine Bone Substitutes for Oral Regeneration: A Narrative Review. Int. J. Implant. Dent. 2024, 10, 26. [Google Scholar] [CrossRef]
- Sleman, N.; Khalil, A. A Comprehensive Review of Biomaterials for Maxillary Sinus Floor Augmentation: Exploring Diverse Bone Graft Options. Open Dent. J. 2025, 19, e18742106378788. [Google Scholar] [CrossRef]
- Ge, L.; Chen, S. Recent Advances in Tissue Adhesives for Clinical Medicine. Polymers 2020, 12, 939. [Google Scholar] [CrossRef]
- Boursier, M.; Bayon, Y.; Negrell, C.; Pinaud, J.; Caillol, S. Biocompatible Glues: Recent Progress and Emerging Frontiers in Surgical Adhesion. Polymers 2025, 17, 1749. [Google Scholar] [CrossRef] [PubMed]
- Amirhekmat, A.; Brown, W.E.; Salinas, E.Y.; Hu, J.C.; Athanasiou, K.A.; Wang, D. Mechanical Evaluation of Commercially Available Fibrin Sealants for Cartilage Repair. Cartilage 2024, 15, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi-Mobarakeh, L.; Kolahreez, D.; Ramakrishna, S.; Williams, D. Key Terminology in Biomaterials and Biocompatibility. Curr. Opin. Biomed. Eng. 2019, 10, 45–50. [Google Scholar] [CrossRef]
- Sheikh, Z.; Hamdan, N.; Ikeda, Y.; Grynpas, M.; Ganss, B.; Glogauer, M. Natural Graft Tissues and Synthetic Biomaterials for Periodontal and Alveolar Bone Reconstructive Applications: A Review. Biomater. Res. 2017, 21, 9. [Google Scholar] [CrossRef]
- Elgali, I.; Omar, O.; Dahlin, C.; Thomsen, P. Guided Bone Regeneration: Materials and Biological Mechanisms Revisited. Eur. J. Oral. Sci. 2017, 125, 315–337. [Google Scholar] [CrossRef]
- Tahmasebi, E.; Alam, M.; Yazdanian, M.; Tebyanian, H.; Yazdanian, A.; Seifalian, A.; Mosaddad, S.A. Current Biocompatible Materials in Oral Regeneration: A Comprehensive Overview of Composite Materials. J. Mater. Res. Technol. 2020, 9, 11731–11755. [Google Scholar] [CrossRef]
- Bohner, M.; Galea, L.; Doebelin, N. Calcium Phosphate Bone Graft Substitutes: Failures and Hopes. J. Eur. Ceram. Soc. 2012, 32, 2663–2671. [Google Scholar] [CrossRef]
- Haugen, H.J.; Lyngstadaas, S.P.; Rossi, F.; Perale, G. Bone Grafts: Which Is the Ideal Biomaterial? J. Clin. Periodontol. 2019, 46, 92–102. [Google Scholar] [CrossRef]
- Dorozhkin, S.V. Calcium Orthophosphates (CaPO4): Occurrence and Properties. Prog. Biomater. 2016, 5, 9–70. [Google Scholar] [CrossRef]
- Eliaz, N.; Metoki, N. Calcium Phosphate Bioceramics: A Review of Their History, Structure, Properties, Coating Technologies and Biomedical Applications. Materials 2017, 10, 334. [Google Scholar] [CrossRef]
- Ginebra, M.P.; Espanol, M.; Maazouz, Y.; Bergez, V.; Pastorino, D. Bioceramics and Bone Healing. EFORT Open Rev. 2018, 3, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Xia, D.; Ni, Z.; Ou, T.; Wang, Y.; Zhang, H.; Mao, L.; Lin, K.; Xu, S.; Liu, J. Calcium Silicate Bioactive Ceramics Induce Osteogenesis through Oncostatin M. Bioact. Mater. 2021, 6, 810–822. [Google Scholar] [CrossRef] [PubMed]
- Bai, L.; Song, P.; Su, J. Bioactive Elements Manipulate Bone Regeneration. Biomater. Transl. 2023, 4, 248–269. [Google Scholar] [PubMed]
- Brunello, G.; Panda, S.; Schiavon, L.; Sivolella, S.; Biasetto, L.; Del Fabbro, M. The Impact of Bioceramic Scaffolds on Bone Regeneration in Preclinical in Vivo Studies: A Systematic Review. Materials 2020, 13, 1500. [Google Scholar] [CrossRef]
- Brochu, B.M.; Sturm, S.R.; Kawase De Queiroz Goncalves, J.A.; Mirsky, N.A.; Sandino, A.I.; Panthaki, K.Z.; Panthaki, K.Z.; Nayak, V.V.; Daunert, S.; Witek, L.; et al. Advances in Bioceramics for Bone Regeneration: A Narrative Review. Biomimetics 2024, 9, 690. [Google Scholar] [CrossRef]
- Yamahara, S.; Montenegro Raudales, J.L.; Akiyama, Y.; Ito, M.; Chimedtseren, I.; Arai, Y.; Wakita, T.; Hiratsuka, T.; Miyazawa, K.; Goto, S.; et al. Appropriate Pore Size for Bone Formation Potential of Porous Collagen Type I-Based Recombinant Peptide. Regen. Ther. 2022, 21, 294–306. [Google Scholar] [CrossRef]
- Zhang, K.; Fan, Y.; Dunne, N.; Li, X. Effect of Microporosity on Scaffolds for Bone Tissue Engineering. Regen. Biomater. 2018, 5, 115–124. [Google Scholar] [CrossRef]
- Usseglio, J.; Dumur, A.; Pagès, E.; Renaudie, É.; Abélanet, A.; Brie, J.; Champion, É.; Magnaudeix, A. Microporous Hydroxyapatite-Based Ceramics Alter the Physiology of Endothelial Cells through Physical and Chemical Cues. J. Funct. Biomater. 2023, 14, 460. [Google Scholar] [CrossRef]
- Ghayor, C.; Bhattacharya, I.; Guerrero, J.; Özcan, M.; Weber, F.E. 3D-Printed HA-Based Scaffolds for Bone Regeneration: Microporosity, Osteoconduction and Osteoclastic Resorption. Materials 2022, 15, 1433. [Google Scholar] [CrossRef]
- Liu, W.; Cheong, N.; He, Z.; Zhang, T. Application of Hydroxyapatite Composites in Bone Tissue Engineering: A Review. J. Funct. Biomater. 2025, 16, 127. [Google Scholar] [CrossRef]
- Wang, M.; Xu, Y.; Cao, L.; Xiong, L.; Shang, D.; Cong, Y.; Zhao, D.; Wei, X.; Li, J.; Fu, D.; et al. Mechanical and Biological Properties of 3D Printed Bone Tissue Engineering Scaffolds. Front. Bioeng. Biotechnol. 2025, 13, 1545693. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Liu, X.; Zhang, Q.; Jin, Y.; Zhao, G. Meta-Analysis of the Effects of Calcium Phosphate Bone Tissue Engineering Scaffolds on Orthodontic Bone Augmentation and Tooth Movement. Front. Bioeng. Biotechnol. 2025, 13, 1553822. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Kim, S.; Song, I. Octacalcium Phosphate, a Promising Bone Substitute Material: A Narrative Review. J. Yeungnam Med. Sci. 2024, 41, 4–12. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.F. There Is No Such Thing as a Biocompatible Material. Biomaterials 2014, 35, 10009–10014. [Google Scholar] [CrossRef]
- Liu, Q.; Zhu, B.; Yang, H.; Liu, C.; Chen, Y.; Wu, X.; Duan, W.; Feng, L.; Wang, B.; Shao, L.; et al. Engineering Injectable Bone/Bioadhesive Grafts Delivery System with Self-Healing Properties for Bone Regeneration. Bioact. Mater. 2025, 54, 47–70. [Google Scholar] [CrossRef]
- Choi, Y.J.; Chang, H.J.; Kim, M.J.; Lee, J.H.; Lee, B.K. Efficacy of Pure Beta Tricalcium Phosphate Graft in Dentoalveolar Surgery: A Retrospective Evaluation Based on Serial Radiographic Images. Maxillofac. Plast. Reconstr. Surg. 2023, 45, 25. [Google Scholar] [CrossRef]
- Garcia, D.C.; Mingrone, L.E.; Pinotti, F.E.; Seade, L.; Melo, R.d.; Lugão, A.B.; Bezerra, J.A.B.; Sá, M.J.C.d. Assessment of the Osseointegration of Pure-Phase β-Tricalcium Phosphate (β-TCP) Ceramic Cylinder Implants in Critical Segmental Radial Bone Defects in Rabbits. Vet. Sci. 2025, 12, 200. [Google Scholar] [CrossRef]
- Mishchenko, O.; Yanovska, A.; Kosinov, O.; Maksymov, D.; Moskalenko, R.; Ramanavicius, A.; Pogorielov, M. Synthetic Calcium–Phosphate Materials for Bone Grafting. Polymers 2023, 15, 3822. [Google Scholar] [CrossRef]
- Nugraha, A.P.; Yang, H.; Chen, J.; Yang, K.; Kraisintu, P.; Zaww, K.; Ma, A.; Wang, R.; Alhadi, N.E.A.M.; Vanegas Sáenz, J.R.; et al. β-Tricalcium Phosphate as Alveolar Bone Grafting in Cleft Lip/Palate: A Systematic Review. Dent. J. 2023, 11, 234. [Google Scholar] [CrossRef]
- Somngam, C.; Samartkit, S.; Kanchanasurakit, S.; Strietzel, F.P.; Khongkhunthian, P. New Bone Formation of Biphasic Calcium Phosphate Bone Substitute Material: A Systematic Review and Network Meta-Analysis of Randomized Controlled Trials (RCTs). Int. J. Implant. Dent. 2025, 11, 47. [Google Scholar] [CrossRef]
- Brum, I.d.S.; Frigo, L.; Ribeiro da Silva, J.F.; Ciambarella, B.T.; Nascimento, A.L.R.; Pereira, M.J.d.S.; Elias, C.N.; de Carvalho, J.J. Comparison Between Nano-Hydroxyapatite/Beta-Tricalcium Phosphate Composite and Autogenous Bone Graft in Bone Regeneration Applications: Biochemical Mechanisms and Morphological Analysis. Int. J. Mol. Sci. 2025, 26, 52. [Google Scholar] [CrossRef] [PubMed]
- Medeiros Savi, F. Histodynamics of Calcium Phosphate Coating on the Osseointegration of Medical-Grade Polycaprolactone β-Tricalcium Phosphate Scaffolds. Front. Biomater. Sci. 2024, 3, 1448902. [Google Scholar] [CrossRef]
- Borden, M.; Westerlund, L.E.; Lovric, V.; Walsh, W. Controlling the Bone Regeneration Properties of Bioactive Glass: Effect of Particle Shape and Size. J. Biomed. Mater. Res. B Appl. Biomater. 2022, 110, 910–922. [Google Scholar] [CrossRef] [PubMed]
- Al-Harbi, N.; Mohammed, H.; Al-Hadeethi, Y.; Bakry, A.S.; Umar, A.; Hussein, M.A.; Abbassy, M.A.; Vaidya, K.G.; Al Berakdar, G.; Mkawi, E.M.; et al. Silica-Based Bioactive Glasses and Their Applications in Hard Tissue Regeneration: A Review. Pharmaceuticals 2021, 14, 75. [Google Scholar] [CrossRef]
- Rahaman, M.N.; Day, D.E.; Sonny Bal, B.; Fu, Q.; Jung, S.B.; Bonewald, L.F.; Tomsia, A.P. Bioactive Glass in Tissue Engineering. Acta Biomater. 2011, 7, 2355–2373. [Google Scholar] [CrossRef]
- Hum, J.; Boccaccini, A.R. Collagen as Coating Material for 45S5 Bioactive Glass-Based Scaffolds for Bone Tissue Engineering. Int. J. Mol. Sci. 2018, 19, 1807. [Google Scholar] [CrossRef]
- Renno, A.C.M.; Bossini, P.S.; Crovace, M.C.; Rodrigues, A.C.M.; Zanotto, E.D.; Parizotto, N.A. Characterization and in Vivo Biological Performance of Biosilicate. Biomed. Res. Int. 2013, 2013, 141427. [Google Scholar] [CrossRef]
- Ha, S.W.; Viggeswarapu, M.; Habib, M.M.; Beck, G.R. Bioactive Effects of Silica Nanoparticles on Bone Cells Are Size, Surface, and Composition Dependent. Acta Biomater. 2018, 82, 184–196. [Google Scholar] [CrossRef]
- Skallevold, H.E.; Rokaya, D.; Khurshid, Z.; Zafar, M.S. Bioactive Glass Applications in Dentistry. Int. J. Mol. Sci. 2019, 20, 5960. [Google Scholar] [CrossRef]
- Salinas, A.J.; Esbrit, P. Mesoporous Bioglasses Enriched with Bioactive Agents for Bone Repair, with a Special Highlight of María Vallet-Regí’s Contribution. Pharmaceutics 2022, 14, 202. [Google Scholar] [CrossRef]
- Arcos, D.; Vallet-Regí, M. Sol-Gel Silica-Based Biomaterials and Bone Tissue Regeneration. Acta Biomater. 2010, 6, 2874–2888. [Google Scholar] [CrossRef] [PubMed]
- Kamadjaja, D.B.; Abidin, Z.Z.; Diana, R.; Kharis, I.; Mira Sumarta, N.P.; Amir, M.S.; Rizqiawan, A.; Danudiningrat, C.P.; Nakamura, N. In Vivo Analyses of Osteogenic Activity and Bone Regeneration Capacity of Demineralized Freeze-Dried Bovine Bone Xenograft: A Potential Candidate for Alveolar Bone Fillers. Int. J. Dent. 2021, 2021, 1724374. [Google Scholar] [CrossRef] [PubMed]
- Soares, M.Q.S.; Van Dessel, J.; Jacobs, R.; Yaedú, R.Y.F.; Sant’Ana, E.; da Silva Corrêa, D.; Madeira, M.F.C.; Duarte, M.A.H.; Rubira-Bullen, I.R.F. Morphometric Evaluation of Bone Regeneration in Segmental Mandibular Bone Defects Filled with Bovine Bone Xenografts in a Split-Mouth Rabbit Model. Int. J. Implant. Dent. 2019, 5, 32. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.; Yang, R.; Cooper, P.R.; Khurshid, Z.; Shavandi, A.; Ratnayake, J. Bone Grafts and Substitutes in Dentistry: A Review of Current Trends and Developments. Molecules 2021, 26, 3007. [Google Scholar] [CrossRef]
- Li, X.; Lin, S.; Duan, S. The Impact of Deproteinized Bovine Bone Particle Size on Histological Outcomes in Sinus Floor Elevation: A Systematic Review and Meta-Analysis. Int. J. Implant. Dent. 2023, 9, 35. [Google Scholar] [CrossRef]
- Noori, A.; Ashrafi, S.J.; Vaez-Ghaemi, R.; Hatamian-Zaremi, A.; Webster, T.J. A Review of Fibrin and Fibrin Composites for Bone Tissue Engineering. Int. J. Nanomed. 2017, 12, 4937–4961. [Google Scholar] [CrossRef]
- Li, S.; Dan, X.; Chen, H.; Li, T.; Liu, B.; Ju, Y.; Li, Y.; Lei, L.; Fan, X. Developing Fibrin-Based Biomaterials/Scaffolds in Tissue Engineering. Bioact. Mater. 2024, 40, 597–623. [Google Scholar] [CrossRef]
- Paiva Lobato, L.L.; Laís Cristo Teixeira, H.; Miranda Barata, K.; Salles Ferreira, C.R.; Pereira da Silva Pena, F.; De Souza Tavares, W. MAPEAMENTO DE DIAGNÓSTICOS DE ENFERMAGEM EM PESSOAS COM FERIDAS DE DIFÍCIL CICATRIZAÇÃO. Rev. Enferm. Atual. Derme 2025, 99, e025022. [Google Scholar] [CrossRef]
- Shi, W.; Que, Y.; Zhang, X.; Bian, L.; Yu, X.; Tang, X.; Yang, G.; Dai, Y.; Bi, S.; Lv, D.; et al. Functional Tissue-Engineered Bone-like Graft Made of a Fibrin Scaffold and TG2 Gene-Modified EMSCs for Bone Defect Repair. NPG Asia Mater. 2021, 13, 28. [Google Scholar] [CrossRef]
- Rojas-Murillo, J.A.; Simental-Mendía, M.A.; Moncada-Saucedo, N.K.; Delgado-Gonzalez, P.; Islas, J.F.; Roacho-Pérez, J.A.; Garza-Treviño, E.N. Physical, Mechanical, and Biological Properties of Fibrin Scaffolds for Cartilage Repair. Int. J. Mol. Sci. 2022, 23, 9879. [Google Scholar] [CrossRef]
- Sanz-Horta, R.; Matesanz, A.; Gallardo, A.; Reinecke, H.; Jorcano, J.L.; Acedo, P.; Velasco, D.; Elvira, C. Technological Advances in Fibrin for Tissue Engineering. J. Tissue Eng. 2023, 14, 20417314231190288. [Google Scholar] [CrossRef] [PubMed]
- Matesanz, A.; Sanz-Horta, R.; Gallardo, A.; Quílez, C.; Reinecke, H.; Acedo, P.; Velasco, D.; Martínez-Campos, E.; Jorcano, J.L.; Elvira, C. Succinimidyl Alginate-Modified Fibrin Hydrogels from Human Plasma for Skin Tissue Engineering. Gels 2025, 11, 540. [Google Scholar] [CrossRef] [PubMed]
- Gamboa-Martínez, T.C.; Luque-Guillén, V.; González-García, C.; Gõmez Ribelles, J.L.; Gallego-Ferrer, G. Crosslinked Fibrin Gels for Tissue Engineering: Two Approaches to Improve Their Properties. J. Biomed. Mater. Res. A 2015, 103, 614–621. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, A.d.C.; Fideles, S.O.M.; Pomini, K.T.; Reis, C.H.B.; Bueno, C.R.d.S.; Pereira, E.d.S.B.M.; Rossi, J.d.O.; Novais, P.C.; Pilon, J.P.G.; Rosa, G.M., Jr.; et al. Effects of Therapy with Fibrin Glue Combined with Mesenchymal Stem Cells (Mscs) on Bone Regeneration: A Systematic Review. Cells 2021, 10, 2323. [Google Scholar] [CrossRef]
- Wodzig, M.H.H.; Peters, M.J.M.; Emanuel, K.S.; Van Hugten, P.P.W.; Wijnen, W.; Jutten, L.M.; Boymans, T.A.; Loeffen, D.V.; Emans, P.J. Minced Autologous Chondral Fragments with Fibrin Glue as a Simple Promising One-Step Cartilage Repair Procedure: A Clinical and MRI Study at 12-Month Follow-Up. Cartilage 2022, 13, 19–31. [Google Scholar] [CrossRef]
- Gasparotto, V.P.O.; Landim-Alvarenga, F.C.; Oliveira, A.L.R.; Simões, G.F.; Lima-Neto, J.F.; Barraviera, B.; Ferreira, R.S. A New Fibrin Sealant as a Three-Dimensional Scaffold Candidate for Mesenchymal Stem Cells. Stem Cell Res. Ther. 2014, 5, 78. [Google Scholar] [CrossRef]
- Frauz, K.; Teodoro, L.F.R.; Carneiro, G.D.; Da Veiga, F.C.; Ferrucci, D.L.; Bombeiro, A.L.; Simões, P.W.; Alvares, L.E.; De Oliveira, A.L.R.; Vicente, C.P.; et al. Transected Tendon Treated with a New Fibrin Sealant Alone or Associated with Adipose-Derived Stem Cells. Cells 2019, 8, 56. [Google Scholar] [CrossRef]
- Biscola, N.P.; Cartarozzi, L.P.; Ulian-Benitez, S.; Barbizan, R.; Castro, M.V.; Spejo, A.B.; Ferreira, R.S.; Barraviera, B.; Oliveira, A.L.R. Multiple Uses of Fibrin Sealant for Nervous System Treatment Following Injury and Disease. J. Venom. Anim. Toxins Incl. Trop. Dis. 2017, 23, 13. [Google Scholar] [CrossRef]
- Rosso, M.P.d.O.; Oyadomari, A.T.; Pomini, K.T.; Della Coletta, B.B.; Shindo, J.V.T.C.; Ferreira Júnior, R.S.; Barraviera, B.; Cassaro, C.V.; Buchaim, D.V.; Teixeira, D.d.B.; et al. Photobiomodulation Therapy Associated with Heterologous Fibrin Biopolymer and Bovine Bone Matrix Helps to Reconstruct Long Bones. Biomolecules 2020, 10, 383. [Google Scholar] [CrossRef]
- Abbade, L.P.F.; Barraviera, S.R.C.S.; Silvares, M.R.C.; Lima, A.B.B.d.C.O.; Haddad, G.R.; Gatti, M.A.N.; Medolago, N.B.; Rigotto Carneiro, M.T.; dos Santos, L.D.; Ferreira, R.S.; et al. Treatment of Chronic Venous Ulcers with Heterologous Fibrin Sealant: A Phase I/II Clinical Trial. Front. Immunol. 2021, 12, 627541. [Google Scholar] [CrossRef]
- Barros, L.C.; Ferreira, R.S.; Barraviera, S.R.C.S.; Stolf, H.O.; Thomazini-Santos, I.A.; Mendes-Giannini, M.J.S.; Toscano, E.; Barraviera, B. A New Fibrin Sealant from Crotalus durissus terrificus Venom: Applications in Medicine. J. Toxicol. Environ. Health B Crit. Rev. 2009, 12, 553–571. [Google Scholar] [CrossRef] [PubMed]
- Venante, H.S.; Chappuis-Chocano, A.P.; Marcillo-Toala, O.O.; Da Silva, R.A.; Da Costa, R.M.B.; Pordeus, M.D.; Barraviera, B.; Junior, R.S.F.; Lara, V.S.; Neppelenbroek, K.H.; et al. Fibrin Biopolymer Incorporated with Antimicrobial Agents: A Proposal for Coating Denture Bases. Materials 2021, 14, 1618. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Liu, P.; Xue, X.; Zhang, Z.; Wang, L.; Jiang, Y.; Zhang, C.; Zhou, H.; Lv, S.; Shen, W.; et al. The Role of Platelet-Rich Plasma in Biomedicine: A Comprehensive Overview. iScience 2025, 28, 111705. [Google Scholar] [CrossRef] [PubMed]
- You, J.S.; Jung, G.W.; Oh, J.S.; Moon, S.Y.; Lee, W.P.; Jo, H.H. Volumetric Evaluation of Effects of Platelet-Rich Fibrin and Concentrated Growth Factor on Early Bone Healing after Endodontic Microsurgery: A Randomized Controlled Trial. BMC Oral Health 2023, 23, 821. [Google Scholar] [CrossRef]
- He, L.; Lin, Y.; Hu, X.; Zhang, Y.; Wu, H. A Comparative Study of Platelet-Rich Fibrin (PRF) and Platelet-Rich Plasma (PRP) on the Effect of Proliferation and Differentiation of Rat Osteoblasts in Vitro. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontology 2009, 108, 707–713. [Google Scholar] [CrossRef]
- Dohan, D.M.; Choukroun, J.; Diss, A.; Dohan, S.L.; Dohan, A.J.J.; Mouhyi, J.; Gogly, B. Platelet-Rich Fibrin (PRF): A Second-Generation Platelet Concentrate. Part I: Technological Concepts and Evolution. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontology 2006, 101, e37–e44. [Google Scholar] [CrossRef]
- Jia, K.; You, J.; Zhu, Y.; Li, M.; Chen, S.; Ren, S.; Chen, S.; Zhang, J.; Wang, H.; Zhou, Y. Platelet-Rich Fibrin as an Autologous Biomaterial for Bone Regeneration: Mechanisms, Applications, Optimization. Front. Bioeng. Biotechnol. 2024, 12, 1286035. [Google Scholar] [CrossRef]
- Chou, L.H.; Yang, T.S.; Wong, P.C.; Chen, Y.C. Synergistic Effects of Platelet-Rich Fibrin and Photobiomodulation on Bone Regeneration in MC3T3-E1 Preosteoblasts. Photodiagnosis Photodyn. Ther. 2025, 51, 104436. [Google Scholar] [CrossRef]
- Diab, N.A.F.; Ibrahim, A.S.M.; Abdallah, A.M. Fluid Platelet-Rich Fibrin (PRF) Versus Platelet-Rich Plasma (PRP) in the Treatment of Atrophic Acne Scars: A Comparative Study. Arch. Dermatol. Res. 2023, 315, 1249–1255. [Google Scholar] [CrossRef]
- Silva, F.F.V.E.; Chauca-Bajaña, L.; Caponio, V.C.A.; Cueva, K.A.S.; Velasquez-Ron, B.; Padín-Iruegas, M.E.; Almeida, L.L.; Lorenzo-Pouso, A.I.; Suárez-Peñaranda, J.M.; Pérez-Sayáns, M. Regeneration of Periodontal Intrabony Defects Using Platelet-Rich Fibrin (PRF): A Systematic Review and Network Meta-Analysis. Odontology 2024, 112, 1047–1068. [Google Scholar] [CrossRef]
- Chao, S.C.; Wang, M.J.; Pai, N.S.; Yen, S.K. Preparation and Characterization of Gelatin-Hydroxyapatite Composite Microspheres for Hard Tissue Repair. Mater. Sci. Eng. C 2015, 57, 113–122. [Google Scholar] [CrossRef]
- Nam, J.W.; Kim, H.J. Stepwise Verification of Bone Regeneration Using Recombinant Human Bone Morphogenetic Protein-2 in Rat Fibula Model. J. Korean Assoc. Oral. Maxillofac. Surg. 2017, 43, 373–387. [Google Scholar] [CrossRef] [PubMed]
- Süloğlu, A.K.; Karacaoğlu, E.; Bilgic, H.A.; Selmanoğlu, G.; Koçkaya, E.A.; Karaaslan, C. Osteogenic Differentiation of Adipose Tissue-Derived Mesenchymal Stem Cells on Fibrin Glue- or Fibronectin-Coated Ceraform®. J. Biomater. Appl. 2019, 34, 375–385. [Google Scholar] [CrossRef] [PubMed]
- Cassaro, C.V.; Justulin, L.A.; De Lima, P.R.; De Assis Golim, M.; Biscola, N.P.; De Castro, M.V.; De Oliveira, A.L.R.; Doiche, D.P.; Pereira, E.J.; Ferreira, R.S.; et al. Fibrin Biopolymer as Scaffold Candidate to Treat Bone Defects in Rats. J. Venom. Anim. Toxins Incl. Trop. Dis. 2019, 25, e20190027. [Google Scholar] [CrossRef]
- Sadeghinia, A.; Davaran, S.; Salehi, R.; Jamalpoor, Z. Nano-Hydroxy Apatite/Chitosan/Gelatin Scaffolds Enriched by a Combination of Platelet-Rich Plasma and Fibrin Glue Enhance Proliferation and Differentiation of Seeded Human Dental Pulp Stem Cells. Biomed. Pharmacother. 2019, 109, 1924–1931. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.; Zhang, Y.; Cao, Z.; Ye, F.; Lin, Z.; Li, Y. Development of CaCO3 Microsphere-Based Composite Hydrogel for Dual Delivery of Growth Factor and Ca to Enhance Bone Regeneration. Biomater. Sci. 2019, 7, 3614–3626. [Google Scholar] [CrossRef]
- Tan, M.H.; Chanchareonsook, N.; Jansen, J.A.; Yu, N.; Liu, E.W.Y.; Goh, B.T. The Use of Biphasic Calcium Phosphate with or without Bone Morphogenic Protein-2 for Bone Regeneration during Immediate Implant Placement in a Micropig Model: A Pilot Study. J. Oral. Maxillofac. Surg. Med. Pathol. 2020, 32, 19–25. [Google Scholar] [CrossRef]
- de Oliveira, C.T.B.; Leonel, B.C.; de Oliveira, A.C.; de Brito Paiva, M.; Ramos, J.; Barraviera, B.; Ferreira Junior, R.S.; Shimano, A.C. Effects of Fibrin Sealant and Bone Fragments on Defect Regeneration Performed on Rat Tibiae: An Experimental Study. J. Mech. Behav. Biomed. Mater. 2020, 104, 103662. [Google Scholar] [CrossRef]
- Zhao, H.; Wang, X.; Jin, A.; Wang, M.; Wang, Z.; Huang, X.; Dai, J.; Wang, X.; Lin, D.; Shen, S.G. Reducing Relapse and Accelerating Osteogenesis in Rapid Maxillary Expansion Using an Injectable Mesoporous Bioactive Glass/Fibrin Glue Composite Hydrogel. Bioact. Mater. 2022, 18, 507–525. [Google Scholar] [CrossRef]
- Guastaldi, F.P.S.; Matheus, H.R.; Faloni, A.P.d.S.; de Almeida-Filho, E.; Cominotte, M.A.; Moretti, L.A.C.; Verzola, M.H.A.; Marcantonio, E.; de Almeida, J.M.; Guastaldi, A.C.; et al. A New Multiphase Calcium Phosphate Graft Material Improves Bone Healing—An in Vitro and in Vivo Analysis. J. Biomed. Mater. Res. B Appl. Biomater. 2022, 110, 2686–2704. [Google Scholar] [CrossRef]
- Buchaim, D.V.; Andreo, J.C.; Pomini, K.T.; Barraviera, B.; Ferreira, R.S.; Duarte, M.A.H.; Alcalde, M.P.; Reis, C.H.B.; de Bortoli Teixeira, D.; de Souza Bueno, C.R.; et al. A Biocomplex to Repair Experimental Critical Size Defects Associated with Photobiomodulation Therapy. J. Venom. Anim. Toxins Incl. Trop. Dis. 2022, 28, e20210056. [Google Scholar] [CrossRef] [PubMed]
- Bojan, A.J.; Stadelmann, V.A.; Wu, D.; Pujari-Palmer, M.; Insley, G.; Sundh, D.; Persson, C.; Engqvist, H.; Procter, P. A New Bone Adhesive Candidate-Does It Work in Human Bone? An Ex-Vivo Preclinical Evaluation in Fresh Human Osteoporotic Femoral Head Bone. Injury 2022, 53, 1858–1866. [Google Scholar] [CrossRef] [PubMed]
- Kageyama, T.; Akieda, H.; Sonoyama, Y.; Sato, K.; Yoshikawa, H.; Isono, H.; Hirota, M.; Kitajima, H.; Chun, Y.S.; Maruo, S.; et al. Bone Beads Enveloped with Vascular Endothelial Cells for Bone Regenerative Medicine. Acta Biomater. 2023, 165, 168–179. [Google Scholar] [CrossRef]
- Pomini, K.T.; Buchaim, D.V.; Bighetti, A.C.C.; Hamzé, A.L.; Reis, C.H.B.; Duarte, M.A.H.; Alcalde, M.P.; Barraviera, B.; Júnior, R.S.F.; de Souza, A.T.; et al. Tissue Bioengineering with Fibrin Scaffolds and Deproteinized Bone Matrix Associated or Not with the Transoperative Laser Photobiomodulation Protocol. Molecules 2023, 28, 407. [Google Scholar] [CrossRef] [PubMed]
- Vigliar, M.F.R.; Marega, L.F.; Duarte, M.A.H.; Alcalde, M.P.; Rosso, M.P.d.O.; Ferreira Junior, R.S.; Barraviera, B.; Reis, C.H.B.; Buchaim, D.V.; Buchaim, R.L. Photobiomodulation Therapy Improves Repair of Bone Defects Filled by Inorganic Bone Matrix and Fibrin Heterologous Biopolymer. Bioengineering 2024, 11, 78. [Google Scholar] [CrossRef]
- Rossi, J.d.O.; Araujo, E.M.C.; Camargo, M.E.C.; Ferreira Junior, R.S.; Barraviera, B.; Miglino, M.A.; Nogueira, D.M.B.; Reis, C.H.B.; Gil, G.E.; Vinholo, T.R.; et al. Effectiveness of the Association of Fibrin Scaffolds, Nanohydroxyapatite, and Photobiomodulation with Simultaneous Low-Level Red and Infrared Lasers in Bone Repair. Materials 2024, 17, 4351. [Google Scholar] [CrossRef]
- Yamada, Y.; Boo, J.S.; Ozawa, R.; Nagasaka, T.; Okazaki, Y.; Hata, K.I.; Ueda, M. Bone Regeneration Following Injection of Mesenchymal Stem Cells and Fibrin Glue with a Biodegradable Scaffold. J. Cranio-Maxillofac. Surg. 2003, 31, 27–33. [Google Scholar] [CrossRef]
- Dhopte, A.; Devi, K.B.; Pandaw, K.; Rani, A.; Chand, S.; Bagde, H. The Effect of Platelet-Rich Fibrin (PRF) in Accelerating Periodontal Regeneration: A Randomized Controlled Clinical Trial. J. Pharm. Bioallied Sci. 2024, 16, S3831–S3833. [Google Scholar] [CrossRef]
- Aldommari, E.A.; Omair, A.; Qasem, T. Titanium-Prepared Platelet-Rich Fibrin Enhances Alveolar Ridge Preservation: A Randomized Controlled Clinical and Radiographic Study. Sci. Rep. 2025, 15, 24065. [Google Scholar] [CrossRef]
- Molina-Barahona, M.; Castillo, J.; Freire-Meza, E.; Vásquez-Palacios, A.C.; Morales-Navarro, D.; Avecillas-Rodas, R. Radiographic Evaluation in Alveolar Preservation Using Platelet-Rich Fibrin: A Randomized Controlled Trial. Dent. J. 2025, 13, 231. [Google Scholar] [CrossRef]


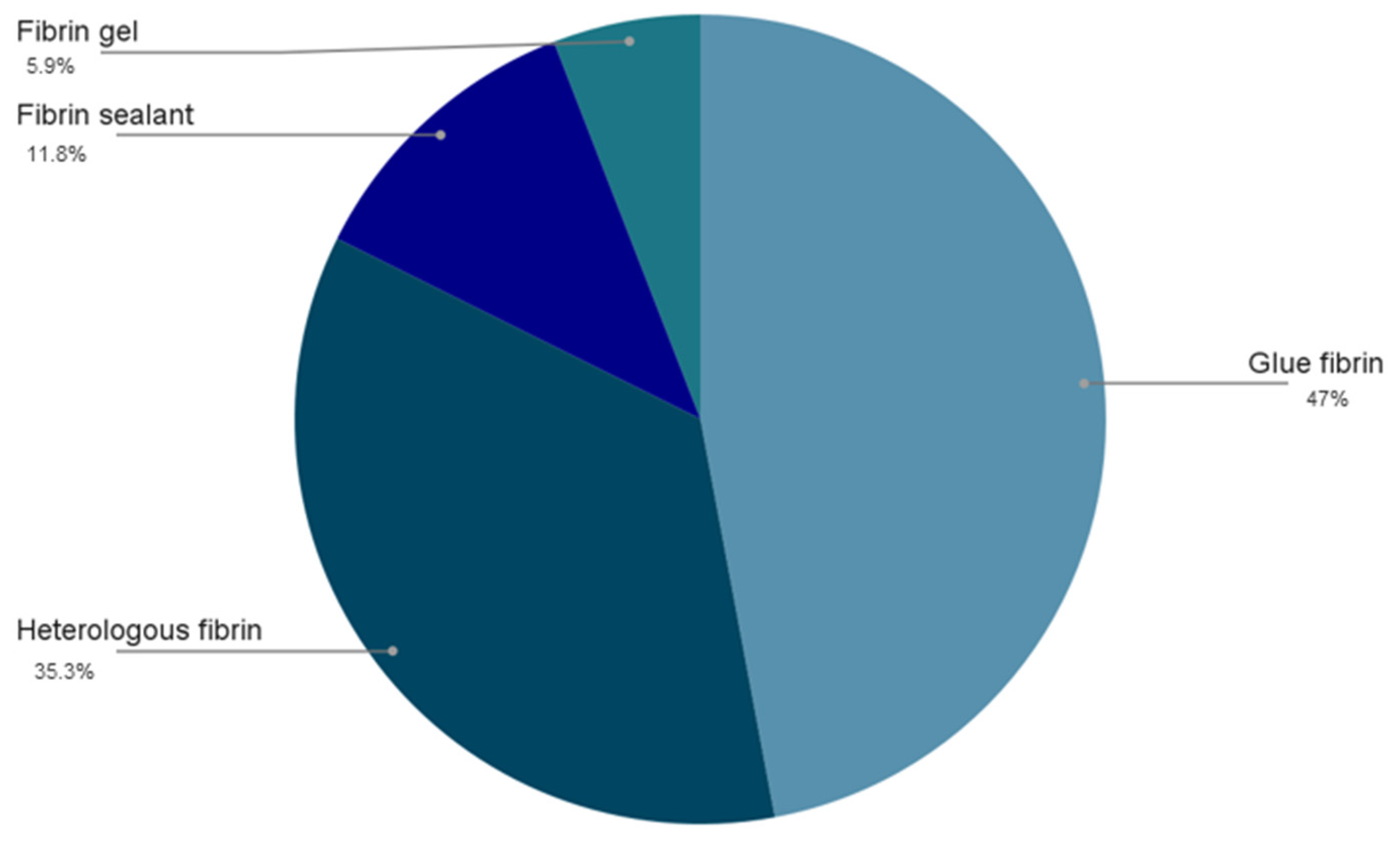

| Study/Year | Type of Biomaterial | Type of Fibrin Derivate | Experimental Model | Outcomes | Limitations |
|---|---|---|---|---|---|
| Chao et al. (2015) [91] | Hydroxyapatite (HA) composite microspheres (HA + gelatin) | Fibrin glue (FG) | Rat calvarial defect model (Sprague Dawley rats) | HA microspheres improved bone regeneration vs. FG; high osteo-conductivity | Small sample; short follow-up; no clinical validation |
| Nam et al. (2017) [92] | Xenogenic bone (CollaOss) | Fibrin glue (FG) | Rat fibula critical-sized defect model | rhBMP-2 + FG enhanced bone formation; early ectopic bone; later remodeling | FG alone minimal effect; limited bone volume long term |
| Suloglu et al. (2019) [93] | Hydroxyapatite Granules (HA) | Fibrin sealant (bovine) | Rat calvarial critical-sided defect model | HA + FS increased bone and vessel formation vs. controls | Short-term; small model; no mechanical testing |
| Cassaro et al. (2019) [94] | Biphasic calcium phosphate (BCP: Hydroxyapatite (HA) + β-Tricalcium phosphate (+ β-TCP)) | Fibrin biopolymer (snake venom + buffalo fibrinogen) | Rat femoral defect (Wistar rats) | BCP + fibrin biopolymer improved handling, integration, and osteo-conduction | Incomplete bone fill; short duration; rat model |
| Sadeghinia et al. (2019) [95] | Xenogenic bovine bone mineral (Bio-Oss) | Fibrin glue (FG) | Rat calvarial critical-size defect | Bio-Oss + FG promoted greater bone formation and integration | Partial regeneration; short-term; small model |
| Gong et al. (2019) [96] | Calcium barbonate (CaCO3) microspheres | Fibrin glue hydrogel | Rabbit tibia defect + in vitro nBMSCs | CaCO3 + FG hydrogel increased strength, sustained BMP-2 release, faster healing | Complex design; growth factor dependent; preclinical only |
| Tan et al. (2020) [97] | β-tricalcium phosphate (β-TCP) scaffold | Fibrin Sealant (FS) | Rat calvarial critical-sized defect | β-TCP + FS improved trabeculae thickness and defect filling | Short-term; no clinical data |
| De Oliveira et al. (2020) [98] | Autologous bone fragments (particulate graft) | Fibrin Sealant (FS) (snake venom + buffalo fibrinogen) | Rat tibial defect (Wistar rats) | FS + autologous bone raised rigidity and mineral density vs. control | Small defect; not load-bearing; limited model |
| Zhao et al. (2022) [99] | Mesoporous bioactive glass (MBG) | Fibrin glue (FG) | Rat model of rapid maxillary expansion | MBG + FG improved osteogenesis, reduced relapse, and balanced remodeling | Animal model only; no human data |
| Guastaldi et al. (2022) [100] | Experimental calcium phosphate - ECP (ACP + OCP + HA) compared with DBB and β-TCP | Fibrin sealant (snake venom enzyme + buffalo fibrinogen) | Rabbit calvarial critical-sided defects | ECP + FS increased resorption and bone deposition; comparable to DBB, β-TCP | Animal study only; long-term results unknown |
| Buchaim et al. (2022) [101] | Xenogenic bone graft (Bio-Oss–bovine hydroxyapatite) | Fibrin sealant (snake venom enzyme + buffalo fibrinogen) | Rat calvarial critical-sized defects + photobiomodulation therapy | Bio-Oss + FS + PBM enhanced bone and collagen deposition | Short follow-up; rat model; limited translation |
| Bojan et al. (2022) [102] | Phosphoserin e-modified α-tricalcium phosphate (OsStic® bone adhesive) | Fibrin tissue adhesive (TisseelTM) as control | Ex vivo human (osteoporotic bone, hip arthroplasty patients) | OsStic® stronger adhesion vs. FG (123 N vs. 5 N); suitable for fixation | Ex vivo only; no healing evaluation |
| Kageyamaet et al. (2023) [103] | β-tricalcium phosphate (β-TCP) + collagen microgels with MSCs (bone beads) | Fibrin gel (fibrinogen + thrombin) covering the transplant in rat model | Nude mice (cranial defects) and nude rats (cranial defects) | β-TCP + fibrin gel improved vascularization and bone regeneration | Short-term; small model; scalability uncertain |
| Pomini et al. (2023) [104] | Deproteinized bovine bone (DBBM) particles | Fibrin sealant (venom snake enzyme + buffalo fibrinogen) | Rat calvarial critical-sized defects + photobiomodulation (PBM) | DBBM + FS + PBM increased bone volume and collagen integration | Incomplete defect repair; experimental proportions |
| Vigliar et al. (2024) [105] | Hydroxyapatite and collagen composite granules | Autologous fibrin glue (fibrin-based matrix) | Rat cranial critical-sized defects | HA/collagen + fibrin glue accelerated repair and vascularization | Short-term; small animals; no human trials |
| Rossi et al. (2024) [106] | Nanohydroxyapatite (nHA) | Fibrin sealant (venom snake enzyme + buffalo fibrinogen) | Critical-sized calvarial defect in rats | nHA + FS + PBM enhanced bone regeneration and organization | 42-day follow-up only; preclinical model |
| Liu et al. (2025) [45] | Calf bone granules (bovine bone) | Fibrin glue (used as control) | Rat femoral artery hemorrhage, rabbit femoral condyle defect, rat skull defect | PEG/gelatin hydrogel with bone granules improved retention and healing | ≤8 weeks follow-up; no clinical trials |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Bueno, L.M.M.; dos Santos, C.P.C.; Cruel, P.T.E.; Romanini, G.; Padula, L.R.C.; Oliveira, C.R.d.S.; Buchaim, D.V.; Buchaim, R.L. Bone-Healing Enhancement Using Particulate Biomaterials and Fibrin-Based Compounds: A Narrative Literature Review of Evidence in Animal Models. Materials 2026, 19, 224. https://doi.org/10.3390/ma19020224
Bueno LMM, dos Santos CPC, Cruel PTE, Romanini G, Padula LRC, Oliveira CRdS, Buchaim DV, Buchaim RL. Bone-Healing Enhancement Using Particulate Biomaterials and Fibrin-Based Compounds: A Narrative Literature Review of Evidence in Animal Models. Materials. 2026; 19(2):224. https://doi.org/10.3390/ma19020224
Chicago/Turabian StyleBueno, Lívia Maluf Menegazzo, Camila Pascoal Correia dos Santos, Paola Tatiana Espinosa Cruel, Gabriela Romanini, Lithiene Ribeiro Castilho Padula, Cindel Regina dos Santos Oliveira, Daniela Vieira Buchaim, and Rogerio Leone Buchaim. 2026. "Bone-Healing Enhancement Using Particulate Biomaterials and Fibrin-Based Compounds: A Narrative Literature Review of Evidence in Animal Models" Materials 19, no. 2: 224. https://doi.org/10.3390/ma19020224
APA StyleBueno, L. M. M., dos Santos, C. P. C., Cruel, P. T. E., Romanini, G., Padula, L. R. C., Oliveira, C. R. d. S., Buchaim, D. V., & Buchaim, R. L. (2026). Bone-Healing Enhancement Using Particulate Biomaterials and Fibrin-Based Compounds: A Narrative Literature Review of Evidence in Animal Models. Materials, 19(2), 224. https://doi.org/10.3390/ma19020224









