A Comparative Analysis of the Properties of Coal Liquefaction Residues and Limestone Fine Aggregates
Abstract
1. Introduction
2. Physical Properties
2.1. Basic Performance
- (1)
- Particle Composition
- (2)
- Technical Indicators
- (1)
- Table 1 shows that ICLR and limestone fine aggregate have similar apparent relative density and solidity. Both have low mud content, though limestone fine aggregate is slightly higher. ICLR has a higher sand equivalent, indicating better cleanliness, as limestone fine aggregate may contain impurities like soil and dust, while ICLR mainly contains unreacted catalysts with relatively simple impurities. ICLR is slightly less angular than limestone due to its smoother surface and lower inter-particle friction.
- (2)
- DCLR exhibits an apparent relative density approximately half that of limestone fine aggregate, accompanied by slightly lower solidity. In terms of mud content and sand equivalent, however, the differences between the two materials are negligible. Notably, DCLR demonstrates higher angularity than limestone fine aggregate; this is primarily attributed to its low density, light particle mass, and poor fluidity. Regarding water absorption, limestone fine aggregate shows a value of 1.2%, compared to 1.5% for ICLR and 1.9% for DCLR. The elevated water absorption of DCLR is mainly due to its high organic content and porous structure. Consequently, in practical applications, this characteristic necessitates appropriate adjustments to the asphalt content or the implementation of pre-mixing treatments.
- (3)
- Compared with ICLR and limestone fine aggregate, DCLR melts at a relatively high temperature. Therefore, according to the technical standards related to asphalt performance in “Test Methods for Asphalt and Asphalt Mixtures for Highway Engineering” (JTG E20—2011) [18], basic performance tests were carried out on DCLR, and the technical standards of 90# asphalt were selected for comparison, as shown in Table 2. All key performance indicators are presented as averages based on the results of at least three parallel tests to ensure the reliability of the data.
2.2. Mechanical Properties
- (1)
- Table 3 illustrates that the Vickers hardness of ICLR is 2.5 times greater than that of limestone fine aggregate, and its modulus of elasticity is 1.2 times greater. Despite having a slightly lower density compared to limestone fine aggregate, ICLR emerges as a lightweight, high-strength material. It exhibits significantly higher hardness and modulus, as well as superior compressive and destructive resistance compared to limestone fine aggregate.
- (2)
- DCLR exhibits Vickers hardness and elastic modulus values that are merely one-tenth those of limestone fine aggregate, indicating its intrinsically inferior hardness and deformation resistance. This marked discrepancy stems from the disordered arrangement and weak cohesion among DCLR’s organic components. In contrast, limestone fine aggregate possesses an ordered, tightly bonded molecular structure, typically characterized by strong metallic or ionic bonds that underpin its superior hardness and deformation resistance.
2.3. Microscopic Morphology
- (1)
- Under 500× magnification (Figure 3), limestone fine aggregate shows pronounced angularity, unevenness, and a distinct pore structure with continuous, uniform peaks and valleys. ICLR and DCLR particles mainly adhere to irregular fine particle clusters of relatively uniform shape and size. ICLR is flatter than DCLR, with less surface undulation and a knitted flocculent structure; much of its surface consists of amorphous residual carbon, with numerous amorphous spherical glass phases clustered on it. In contrast, DCLR has a corrugated shape with consistent corrugation direction, densely packed granular material, and a denser melt from particle clusters than ICLR. Most small DCLR particles form plaque clusters with inconspicuous surface pores and contain uniformly attached residual carbon.
- (2)
- At 6000× magnification, limestone fine aggregate, ICLR, and DCLR all exhibit similar continuous laminar scale-like structures. Limestone and ICLR show greater similarity in densification, forming a uniform, highly dense, cross-layered patchy lamellar structure, with higher fragmentation and arrangement density than DCLR. In contrast, DCLR possesses a denser surface than limestone but features a relatively sparse scale-like structure dominated by small lamellae that differ markedly from the larger lamellae and are unevenly distributed across them. Overall, the micro-morphologies of ICLR and DCLR are not significantly different from those of limestone; all exhibit a degree of surface roughness, which enhances asphalt adsorption and contributes to higher strength. ICLR and DCLR thus share certain micromorphological similarities with limestone.
2.4. Pore Size Distribution
- (1)
- According to Figure 4, the most probable pore diameter of ICLR is larger than that of limestone fine aggregate. Across the three particle sizes, both materials exhibit similar pore size distributions, primarily consisting of mesopores in the 2–50 nm range, along with minor amounts of micropores (<2 nm) and macropores (>50 nm). In contrast, DCLR shows a larger most probable pore diameter than both limestone fine aggregate and ICLR, indicating a more developed pore structure in the larger size range.
- (2)
- Across three ICLR particle sizes, micropore volume and number vary little, with maximum pore volume concentrated between 0.000025 and 0.000045 cm3/g·nm. Mesopore and macropore volumes for the 1.18–2.36 mm and 2.36–4.75 mm sizes are greater than for the 0.6–1.18 mm sizes, and generally increase with particle size, implying greater asphalt accommodation. In contrast, limestone fine aggregate has a micropore void volume of 0.002–0.004 cm3/g·nm; its meso- and macropore structures for 0.6–1.18 mm and 1.18–2.36 mm are similar and higher than for 2.36–4.95 mm. Overall, limestone’s total pore volume exceeds that of ICLR.
- (3)
- The pore diameter distributions for the three particle sizes of DCLR are all above 2 nm, indicating the absence of micropore structures and the presence of only mesopores and macropores. The maximum pore volume of DCLR’s pore diameter distribution is similar to that of ICLR, concentrated between 0.000025 and 0.000045 cm3/g·nm. For mesopores of 2 to 50 nm and macropores larger than 50 nm, the pore diameter distributions and corresponding void volumes of the 1.18 to 2.36 mm and 2.36 to 4.75 mm particle sizes of DCLR are nearly identical, suggesting minimal differences in the pore diameter distribution across the three particle sizes. Comparatively, the overall pore volume of limestone fine aggregate remains larger than that of DCLR from a quantitative standpoint.
3. Chemical Characteristics
3.1. Elemental Composition
- (1)
- As shown in Table 4, limestone fine aggregate consists of four main elements. Carbon (C), oxygen (O), and calcium (Ca) are the dominant components, together making up more than 98% of the total composition, while silicon (Si) is present only in a trace amount.
- (2)
- ICLR mainly consists of O (49.43%), Si (14.84%), C (11.44%), and S (7.63%), along with smaller proportions of Al (6.11%) and Ca (5.17%). Other elements detected include non-metals H and N, as well as metals Fe, Ti, and Mg. Relative to limestone fine aggregate, ICLR exhibits a much richer variety of both non-metal and metal elements, indicating good potential for use as an aggregate material.
- (3)
- The elemental composition of DCLR is dominated by carbon, hydrogen, nitrogen, and sulfur, with carbon and hydrogen occurring in relatively high proportions. The elevated C/H atomic ratio points to an abundance of aromatic rings in its molecular structure. Within DCLR, polar heteroatom-containing groups (S, N, and O) are embedded in condensed aromatic matrices through non-covalent interactions such as hydrogen bonding, π-H bonding, and π-π stacking [20]. Carbon atoms are present mainly as benzene rings, methyl groups, and cyclic methylene units; oxygen occurs chiefly as carbonyl and ester functionalities, while nitrogen is largely incorporated in pyrrole rings [21].
3.2. Molecular Weight Distribution
3.3. Chemical Composition
- (1)
- Table 5, Table 6 and Table 7 indicate that the primary constituents of ICLR are SiO2, Al2O3, and CaO, with significant amounts of Fe2O3, TiO2, and MgO also present. In comparison to limestone fine aggregate, ICLR has a higher SiO2 content, whereas limestone fine aggregate contains only 2.4% SiO2. Chemically, SiO2, being an atomic crystal with covalent bonds, requires significantly more energy to break its bonds compared to the ionic bonds in CaCO3, an ionic crystal. Consequently, SiO2 is harder than CaCO3, implying that ICLR is harder than limestone fine aggregate.
- (2)
- DCLR can be fractionated into four components through sequential extraction with distinct solvents (n-hexane, toluene, and tetrahydrofuran): heavy oil, asphaltene, pre-asphaltene, and tetrahydrofuran-insoluble substances. The mass fractions are as follows: heavy oil (20–30%), primarily naphthalene derivatives with alkyl substitutions; asphaltene (20–40%), mainly condensed aromatic hydrocarbons with six-membered rings; pre-asphaltene (15–30%), composed of multiple condensed aromatic hydrocarbons linked by bridge bonds and hydrogenated aromatic hydrocarbons; and THFIS, primarily unreacted coal, quartz, calcium sulfate, pyrite, and other minerals, with a mass fraction of about 45%. According to the classification of asphalt into four components, DCLR can also be categorized into saturates, aromatics, asphaltenes, and resins. The low content of saturates and aromatics contributes to DCLR’s high viscosity, while the low resin content results in poor adhesion and ductility in macroscopic performance. The high asphaltene content leads to a high softening point and increased viscosity, endowing DCLR with hard, brittle, and viscous characteristics.
- (3)
- The primary component of DCLR is organic matter, predominantly asphaltenes at 66.20%, followed by resins at 13.91%, with trace amounts of saturates and aromatics. Additionally, DCLR contains about 15% ash, which is inorganic matter. This indicates that DCLR is composed of approximately 85% organic matter and 15% inorganic matter. Although DCLR is not primarily composed of oxides like ICLR or limestone fine aggregate, the organic compounds it contains provide a favorable material basis for enhancing the adhesion between asphalt and aggregates, increasing the bonding strength of asphalt binders, improving the elasticity, and reducing the cracking of the mixture.
3.4. Crystal Phase
- (1)
- Figure 6 reveals that, in accordance with the Powder Diffraction File (PDF) standards, the limestone fine aggregate contains various crystalline phases. Specifically, a diffraction peak at 9.3° suggests the presence of trace laumontite crystals (Ca[AlSi2O6]2·4H2O). Peaks at 26.58° and 27.92° are attributed to quartz (SiO2) and sphene (CaTi[SiO4]O) crystals, respectively, while the peak at 29.32° is indicative of calcite (CaCO3). The predominant phase in the limestone fine aggregate is calcite (CaCO3), with quartz (SiO2) as a secondary phase. Elements such as magnesium (Mg), aluminum (Al), and iron (Fe) are typically found in the form of oxides within the calcium carbonate matrix. Traces of anorthite (CaSiO3) and wollastonite (CaSiO3) are also detected [24].
- (2)
- The X-ray diffraction (XRD) pattern of ICLR exhibits a broad, upward-protruding peak between 20° and 30°, colloquially referred to as a “steamed bun peak,” which is indicative of non-crystalline compound aggregation. Specific peaks are identified at 26.56° for quartz (SiO2) and at 29.36° for calcite. ICLR is characterized by a scarcity of distinct diffraction peaks, with the predominant phase being an amorphous glassy matrix, predominantly aluminosilicate, accompanied by minor crystalline phases such as quartz (SiO2), mullite (3Al2O3·2SiO2), calcite, and hematite (Fe2O3). The amorphous components include glass, slag wax, and amorphous carbon [25]. The original crystalline structure of inorganic minerals is altered to a glassy state following intense high-temperature oxidation, resulting in small and flat crystal peaks in the XRD pattern [26]. Quartz, due to its high melting point, retains some crystalline structure in parts that have not melted. ICLR thus primarily presents an irregular glassy structure, composed predominantly of amorphous materials with a minor presence of crystalline substances.
- (3)
- In the small-angle region (2θ ≤ 13°), the diffraction peaks of DCLR exhibit a monotonically increasing trend. As documented in references [27,28], the peaks within the range of 2θ ≤ 35° are predominantly a result of the superposition of two distinct microcrystalline diffraction peaks: the 002 peak and the γ peak. The 002 peak, which occurs between 20° and 35°, arises from carbon microcrystals formed by the stacking of aromatic layers, also referred to as aromatic carbon microcrystals. This peak reflects the parallel and azimuthal alignment of aromatic carbon layers in three-dimensional space; a taller and narrower peak suggests a more ordered arrangement of these layers. Conversely, a broader 002 peak is indicative of the intercalation of small-sized microcrystals between the aromatic layers. The γ peak, observed at angles less than or equal to 20° (2θ ≤ 20°), is attributed to aliphatic hydrocarbon side chains, diverse functional groups, and carbon microcrystals associated with the aliphatic hydrocarbons that are linked to the aromatic layer structure, collectively termed aliphatic carbon microcrystals.
- (4)
- The XRD peak intensities for limestone fine aggregate range from 5000 to 22,500, for ICLR from 2500 to 3500, and for DCLR from 2000 to 7000. Relative to DCLR and ICLR, the limestone fine aggregate exhibits higher peak intensities and sharper peak shapes, signifying superior phase crystallinity and greater crystal content. DCLR demonstrates peak sizes and sharpness that exceed those of ICLR, suggesting that DCLR possesses higher crystallinity and a greater quantity of crystalline material compared to ICLR.
- (5)
- Collectively, the concentrations of Ca, Mg, and Fe oxides in ICLR and DCLR are lower than those found in limestone fine aggregate, resulting in a generally lower alkalinity for these aggregates when compared to limestone fine aggregate. Nonetheless, ICLR contains minor quantities of Ca, Mg, and Fe oxides and exhibits a higher alkalinity than DCLR. Consequently, considering the aspects of mineral crystal phases and material properties, ICLR is deemed more appropriate as a fine aggregate material.
3.5. Functional Groups
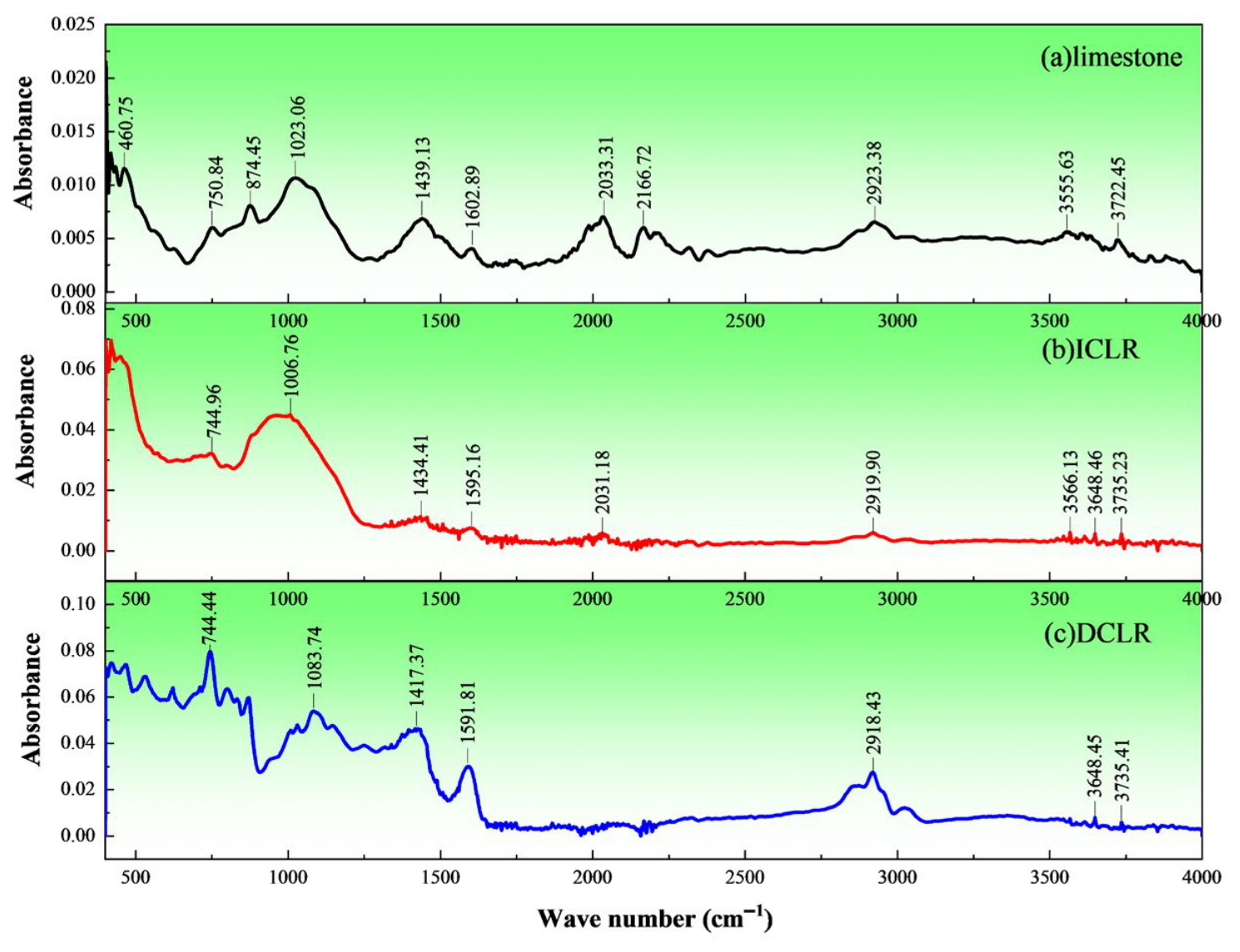
- (1)
- Figure 7 shows three main fingerprint-region peaks (400–1300 cm−1) for limestone fine aggregate: a minor one-substituted benzene ring peak at ~750.84 cm−1, a moderate para-disubstituted benzene ring peak at ~874.45 cm−1, and an intense carbonyl double peak at 1023.06 cm−1. In the functional group region (1300–4000 cm−1), 3–5 peaks of similar intensity appear: at 1439.13 cm−1 (-CH3), 2166.72 cm−1 (unsaturated R2C=CHR), and a broader, weaker peak at 2923.38 cm−1 (-CH2-). This indicates that the aggregate contains significant alkane substances.
- (2)
- In the fingerprint region, ICLR shows two main peaks: a one-substituted benzene peak at 744.96 cm−1 and an intense, broad C-OH hydroxyl peak at 1006.76 cm−1. In the functional group region, 3–5 peaks of comparably lower intensity appear: -CH3 bending at 1434.41 cm−1, C=C stretching near 1595.16 cm−1, a minor -CH2- peak near 2919.90 cm−1, and a medium free O-H hydroxyl stretch at 3648.46 cm−1. These features indicate ICLR contains alkanes, free secondary amines (amine compounds), alcohols and phenolics.
- (3)
- In the functional group region (1300–4000 cm−1), DCLR shows three main peaks: a medium C=C absorption near 1591.81 cm−1, stretching vibrations for -CH3 and -CH2- near 2918.43 and 2850 cm−1, and an O-H stretch between 3600 and 3700 cm−1. In the fingerprint region (400–1300 cm−1), DCLR has more characteristic peaks, which are broader and more dispersed, mainly between 1000 and 1300 cm−1 and 500–800 cm−1. A sharp, narrow peak at 744.44 cm−1 indicates a one-substituted benzene structure, and a medium broad peak at 1083.74 cm−1 indicates C-OH groups. Collectively, these features suggest DCLR is an unsaturated hydrocarbon compound, specifically an alkane-substituted benzene isomer. In the FTIR spectrum of DCLR, the absorption peak at 1591 cm−1 corresponds to the C=C stretching vibration of the aromatic ring; the sharp peak at 744 cm−1 corresponds to the monosubstituted benzene ring structure; and the broad peak at 1083 cm−1 corresponds to the C–OH hydroxyl stretching vibration. Among these functional groups, the aromatic ring structure forms π–π conjugation with the aromatic components in asphalt, serving as the primary chemical mechanism for enhancing asphalt–aggregate adhesion; meanwhile, polar groups such as the C–OH hydroxyl group can form hydrogen bonds or acid–base interactions with polar sites on the aggregate surface (e.g., Ca2+, Si–OH), further improving interfacial bonding.
- (4)
- Compared to ICLR and DCLR, limestone fine aggregate has more continuous and uniform characteristic peaks in both position and intensity. ICLR and DCLR show greater peak discontinuities, unevenness, and more pronounced intensity differences. Stronger, broader peaks are mainly in the fingerprint region, while weaker, narrower ones are in the functional group region. Absorbance values: limestone 0.005–0.01; ICLR strong peaks 0.03–0.05 (others ~0.005); DCLR strong peaks 0.03–0.08 (weak peaks 0.01–0.02). This indicates DCLR has the most diverse and concentrated functional groups, while ICLR and limestone show less significant overall differences.
3.6. Thermal Stability
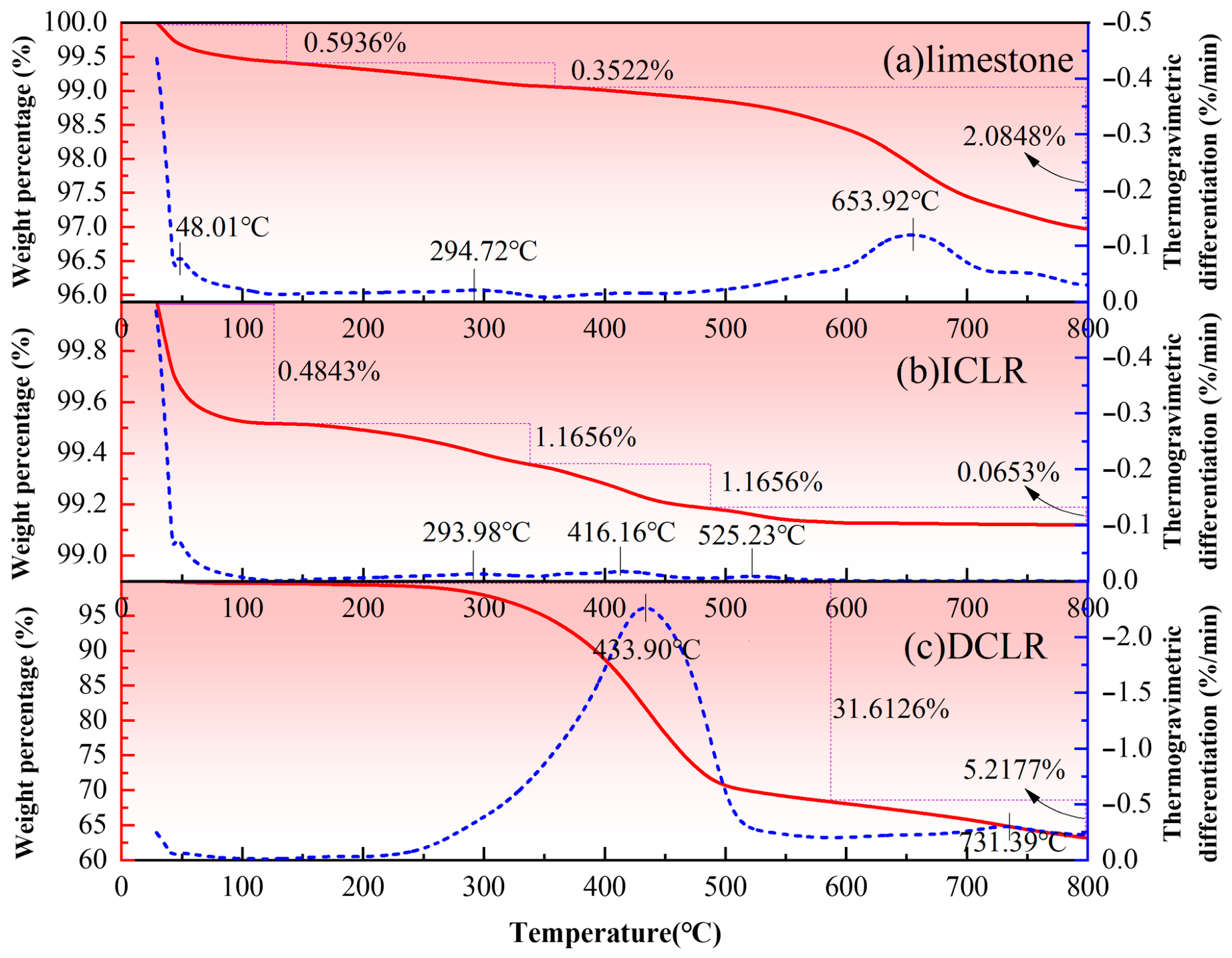
- (1)
- It was found in Figure 8 that the weight of the three materials under study decreases with rising temperature. The weight loss of limestone fine aggregate occurs in three distinct stages: 0~131 °C, 131~353 °C, and 353~800 °C. For ICLR, the weight loss is distributed across four stages: 0~132 °C, 132~338 °C, 33~480 °C, and 480~800 °C. DCLR exhibits weight loss in two stages: 0~588 °C and 588~800 °C. The weight percentage is calculated as the ratio of the material’s weight at any given temperature to its initial weight. It is observed that from the initial temperature to 800 °C, ICLR experiences the least weight loss among the three materials, followed by limestone fine aggregate, and then DCLR. Consequently, the thermal stability ranking is ICLR (with the highest stability), limestone fine aggregate (in the middle), and DCLR (with the lowest). This is attributed to the fact that ICLR is primarily composed of complex oxides with a stable crystal phase and excellent high-temperature resistance. The main component of limestone fine aggregate is CaCO3, which maintains good thermal stability at lower temperatures but decomposes into CaO and CO2 at higher temperatures, resulting in weight loss. The significant weight loss of DCLR is primarily due to the volatilization of components such as asphaltenes and resins at elevated temperatures.
- (2)
- Thermogravimetric differentiation, which involves taking the first derivative of the weight percentage curve with respect to time, characterizes the rate of weight change, with peak points indicating the temperatures at which the rate of weight change is most rapid during each stage. Analysis reveals that the limestone fine aggregate experiences its most rapid weight loss near 48 °C, 295 °C, and 654 °C. Prior to 500 °C, the limestone fine aggregate consistently exhibits a lower and more stable rate of weight loss, with an acceleration in mass loss beyond 500 °C. For ICLR, the most rapid weight loss occurs near 294 °C, 416 °C, and 525 °C, maintaining a lower and more stable rate of loss across the entire temperature range. DCLR’s most rapid weight loss is observed near 434 °C and 731 °C. Below 250 °C, DCLR shows a minimal weight loss rate, which begins to increase thereafter. The temperature range from 250 °C to 500 °C marks the interval of most rapid weight loss for DCLR, with the rate fluctuating little after 500 °C. However, compared to the initial range of 0–250 °C, DCLR still exhibits a higher rate of mass loss.
- (3)
- DCLR is prone to melting and decomposition at elevated temperatures. Thermogravimetric analysis indicates that DCLR maintains relatively good thermal stability up to 250 °C. In road engineering, the preparation temperature for asphalt mixtures typically ranges from 160 °C to 180 °C and seldom exceeds 200 °C; even at higher discharge temperatures, it is around 240 °C. To investigate whether DCLR undergoes melting and decomposition within the mixer during asphalt mixture preparation, and consequently, whether there is a change in the proportion of its constituent components, we selected a test temperature range of 160 °C to 240 °C. The specific procedure involved setting four temperatures: 170 °C, 190 °C, 210 °C, and 230 °C. DCLR was placed in a muffle furnace and heated for 2 h at each of these temperatures. Subsequently, samples were taken for an asphalt four-component test to assess the changes in the four-component content of DCLR at these temperatures. The findings are presented in Table 8.
- (1)
- Table 8 indicates that as the temperature is elevated from 170 °C to 230 °C, the loss on ignition of DCLR increases from 0.26% to 4.83%. This suggests that the degree of component loss in DCLR escalates with increasing temperature. The colloidal instability coefficient (Ic) serves as a metric for assessing the type of asphalt colloidal structure and determining if the four components are within an optimal range. A higher Ic value signifies a less stable colloidal structure type. The data in the table reveal that with the temperature increase, the Ic of DCLR progressively diminishes, suggesting that the colloidal structure of DCLR becomes more stable. In other words, higher temperatures favor the attainment of a more stable colloidal structure for DCLR.
- (2)
- With the increase in temperature, there is a gradual increment in the relative content of saturates, aromatics, and resins, while the relative content of asphaltenes decreases. This suggests that the reduction in asphaltenes is more pronounced than that of saturates, aromatics, and resins as the temperature rises. Specifically, within the temperature range of 170 °C to 230 °C, the loss of asphaltenes is 2.8%, whereas the losses for saturates, aromatics, and resins are 0.17%, 0.44%, and 1.16%, respectively. These findings imply that the incorporation of DCLR as a fine aggregate in mixture preparation may facilitate the penetration of its organic components into the asphalt, potentially inducing an asphalt modification effect.
3.7. Environmental Characteristics
4. Comparison of Physical and Chemical Properties
5. Discussion
5.1. Comparison with Existing Literature
5.2. Implications for Asphalt Pavement Applications
6. Conclusions
- (1)
- This study provides the first systematic comparative analysis of the physical and chemical properties of indirect coal liquefaction residue (ICLR) and direct coal liquefaction residue (DCLR) against limestone fine aggregate. It reveals that both ICLR and DCLR exhibit gradation compositions and microscopic morphological characteristics (uniformly distributed flaky surface structures) highly similar to those of limestone fine aggregate, with only minor differences in pore size distribution. These findings establish a fundamental material basis for substituting natural fine aggregates with coal liquefaction residues in asphalt mixtures.
- (2)
- A key scientific contribution is the demonstration that ICLR, primarily composed of SiO2 crystalline phases and trace metal oxides (Fe, Ti, Al, Mg), possesses significantly higher Vickers hardness (615 HV) and elastic modulus (98 GPa) than limestone, along with superior thermal stability. Its technical specifications (apparent relative density, sand equivalent, and angularity) are comparable to those of limestone, making ICLR a high-strength, lightweight alternative that can enhance the compressive and shear resistance of asphalt pavements without increasing structural weight.
- (3)
- For DCLR, the study reveals a unique “active filler” mechanism: rich in polycyclic aromatic hydrocarbons and asphaltenes (66.2%), DCLR exhibits lower mechanical strength but, within the typical asphalt mixing temperature range (170–230 °C), loses 2.8% of its asphaltene content along with partial saturates, aromatics, and resins. This controlled migration of organic components into the asphalt binder creates an in situ modification effect, improving asphalt–aggregate adhesion and mixture flexibility—a novel functionality not offered by conventional limestone fine aggregate.
- (4)
- From an engineering and environmental perspective, both ICLR and DCLR pose no leaching toxicity risks, with heavy metals and polycyclic aromatic hydrocarbons remaining below regulatory limits. Their use as fine aggregates in pavement construction thus enables 100% substitution of natural aggregates, mitigates the environmental burden of landfill disposal, and supports circular economy goals in road infrastructure. These findings provide a scientific basis and practical pathway for the sustainable utilization of coal liquefaction residues in asphalt mixtures.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Wang, B.; Li, L.; Xian, Y.; Yu, P.; Hao, W. Fundamental coal demand prediction under the goal of carbon neutrality in 2060. Clean Coal Technol. 2022, 28, 1–13. [Google Scholar]
- Wang, X.; Li, X. The Development Trend of China’s Coal Transportation in the Context of Energy Revolution and the Countermeasures. China Bus. Mark. 2019, 33, 67–75. [Google Scholar]
- Zhang, X.; Zhang, Y.; Shi, X. Evoluti onary game study on government and enterprise behavior strategy of coal green logistics under “double-carbon” goal: A study based on the system dynamics perspective. Coal Econ. Res. 2022, 42, 30–38. [Google Scholar]
- Ji, J.; Wang, Z.; Li, P.; Zheng, W.; Xu, X.; Wang, Z.; Han, B.; Wei, J.; Li, H. A review on direct coal liquefaction residue applied in asphalt pavements. J. Clean. Prod. 2023, 395, 136273. [Google Scholar] [CrossRef]
- Wang, X.; Hu, F.; Wang, G. Research status and prospect of coal indirect liquefaction synthetic oil technology. Clean Coal Technol. 2020, 26, 110–120. [Google Scholar]
- Wang, Z.; Ji, J.; Wang, Z.; Ma, T.; Li, P.; Han, B.; Xu, X. A review on indirect coal liquefaction residue used in asphalt pavement. Constr. Build. Mater. 2023, 364, 129755. [Google Scholar] [CrossRef]
- Hu, F.; Wang, X.; Mao, X.; Li, J.; Zhao, P. Research progress and prospect of direct liquefaction technology from coal to oil. Clean Coal Technol. 2020, 26, 99–109. [Google Scholar]
- Editorial Department of China Journal of Highway. Review on China’s Pavement Engineering Research·2020. China J. Highw. 2022, 33, 1–66. [Google Scholar]
- Zhang, L.; Wei, J.; Wang, J.; Bai, Y.; Song, X.; Su, W.; Lv, P.; Nai, G.; Yu, G. Deep insight into the ash fusibility and viscosity fluctuation behavior during co-gasification of coal and indirect coal liquefaction residue. Fuel 2021, 305, 121620. [Google Scholar] [CrossRef]
- Feng, Y.; Lu, J.; Wang, J.; Mi, J.; Zhang, M.; Ge, M.; Li, Y.; Zhang, Z.; Wang, W. Desulfurization sorbents for green and clean coal utilization and downstream toxics reduction: A review and perspectives. J. Clean. Prod. 2020, 273, 123080. [Google Scholar] [CrossRef]
- Lin, J.; Fridley, D.; Lu, H.; Price, L.; Zhou, N. Has coal use peaked in China: Near-term trends in China’s coal consumption. Energy Policy 2018, 123, 208–214. [Google Scholar] [CrossRef]
- Liu, Z.; Sun, Y.; Li, W.; Yang, Q.; Ma, Q. Research progress of gasification slag utilization from indirect coal liquefaction process. Clean Coal Technol. 2016, 22, 118–123. [Google Scholar]
- Zhang, L.; Wen, X.; Ma, S.; Pan, W.; Qu, H.; He, X. Study on Baking—Free Brick Preparation with Residue of Indirect Coal Liquefaction. Coal Eng. 2014, 46, 103–105. [Google Scholar]
- Gu, X.; Zhou, M.; Shi, S. Molecular structure of heavy oil fraction from the Shenhua coal direct liquefaction residue. J. China Coal Soc. 2006, 31, 76–80. [Google Scholar]
- Wang, Z.; Ji, J.; Han, B.; Li, P.; Wang, Z.; Li, H. Laboratory investigation on pavement performance of indirect coal liquefaction residue in asphalt mixture. J. Clean. Prod. 2022, 363, 132183. [Google Scholar] [CrossRef]
- JTG F40-2004; Technical Specification for Construction of Highway Asphalt Pavements. Renmin Communication Press: Beijing, China, 2004. (In Chinese)
- Wang, Z.; Wang, Z.; Sha, C.; Guo, S.; Zheng, W.; Cao, H.; Shan, X.; Cao, G.; Ji, J.; Li, P. Conduct an advanced numerical simulation investigation on the rutting performance of coal liquefaction residue asphalt mixture. Front. Mater. 2025, 12, 1638678. [Google Scholar] [CrossRef]
- JTG E20-2011; Standard Test Methods of Bituminous Mixture for Highway Engineering. Renmin Communication Press: Beijing, China, 2011. (In Chinese)
- Wang, Z.; Wang, Z.; Guo, S.; Sha, C.; Yang, Y.; Zhang, M.; Zhou, X.; Wang, P. Influence of coal liquefaction residues as fine aggregate substitution on asphalt mixture performance and adsorption characteristics. Sci. Rep. 2025, 15, 44152. [Google Scholar] [CrossRef]
- Fan, Y. Exploratory Study on Properties and Application of Residues from Direct Coal Liquefaction Residue. Master’s Thesis, East China University of Science and Technology, Shanghai, China, 2011. (In Chinese) [Google Scholar]
- Lin, H.; Li, K.; Zhang, X.; Wang, H.; Cheng, S. Analysis and structural model of coal liquefaction asphaltene. J. Fuel Chem. Technol. 2014, 42, 779–784. (In Chinese) [Google Scholar]
- Gu, X. Discussion on Structural Characteristics of Shenhua Direct Coal Liquefaction Residue. Master’s Thesis, China Coal Research Institute, Beijing, China, 2005. [Google Scholar]
- Xu, L.; Tang, M.; Duan, L.E.; Liu, B.; Ma, X.; Zhang, Y.; Argyle, M.D.; Fan, M. Pyrolysis Characteristics and Kinetics of Residue from China Shenhua Industrial Direct Coal Liquefaction Plant. Thermochem. Acta 2014, 589, 1–10. [Google Scholar] [CrossRef]
- Chang, S. Determination of Main Components of Limestone by Alkali Fusion ICP-OES. China Non-Met. Miner. Ind. 2022, 36, 65–66. [Google Scholar]
- Qu, J.; Zhang, J.; Sun, Z.; Yang, C.; Shi, D.; Li, S.; Li, H. Research progress on comprehensive utilization of coal gasification slag. Clean Coal Technol. 2020, 26, 184–193. [Google Scholar]
- Fan, N.; Zhang, Y.; Fan, P.; Fan, X.; Wang, J.; Wang, J.; Dong, L.; Fan, M.; Bao, W. Research progress on characteristic analysis and resource utilization of coal gasification slag. Clean Coal Technol. 2022, 28, 145–154. [Google Scholar]
- Xu, C. Characterization analysis of semicoke from direct coal liquefaction residue of Shenhua coal. Coal Qual. Technol. 2021, 36, 42–47. [Google Scholar]
- Zhou, C.; Song, Y.; Liu, Q.; Li, Y.; Zhi, K.; Teng, Y.; He, R.; Zhang, Y. Upgrading of Shengli lignite and the corresponding surface and morphology functional structure. J. Chin. Electron Microsc. Soc. 2013, 32, 237–243. [Google Scholar]
- NB/SH/T 0509-2010; Test Method for Separation of Asphalt into Four Fractions. China Communication Press: Beijing, China, 2010.
- HJ 557-2010; Leaching Method for Leaching Toxicity of Solid Waste. China Communications Press: Beijing, China, 2010.
- GB5085.3-2007; Identification Standards for Hazardous Wastes—Identification for Extraction Toxicity. Standards Press of China: Beijing, China, 2007.
- Li, J.; Li, Z.; Zhang, Y.; Wang, D.; Chen, X.; Fang, Z.; Zhang, W.; Chen, Z. Ash fusion and kinetic characteristics during co-gasification of petroleum coke and coal liquefaction residue. Fuel 2026, 406, 136824. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, Y.; Ru, Y.; Li, Y.; Li, Y.; Gao, Z. Rheological properties study of high-viscosity asphalt based on direct coal liquefaction residue. Front. Built Environ. 2025, 11, 1619524. [Google Scholar] [CrossRef]
- Gao, Y.; Zhang, X.; Jiang, Z.; Ding, W.; Miao, D.; Qiao, Q. Utilization of direct coal liquefaction residue (DCLR) in recycled emulsified asphalt mixtures: A solution as geopolymer binder and filler materials. Constr. Build. Mater. 2024, 438, 137278. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, X.; Liu, F.; Su, J.; Zhao, Q.; Liu, L. Effect of replacement of limestone mineral powder with fly ash and direct coal liquefaction residue on the rheological properties of asphalt mastic. Constr. Build. Mater. 2024, 412, 134803. [Google Scholar] [CrossRef]






| Item | Test Results | Technology Standard | ||
|---|---|---|---|---|
| Limestone Fine Aggregate | ICLR | DCLR | ||
| Apparent relative density | 2.783 | 2.612 | 1.208 | ≥2.60 |
| Solidity/(%) | 87 | 90 | 76 | ≥12 |
| Mud content/(%) | 0.7 | 0.1 | 0.3 | ≤3 |
| Sand equivalent/(%) | 74 | 93 | 81 | ≥60 |
| Angularity/s | 41.7 | 26.2 | 86.4 | ≥30 |
| Water absorption rate/% | 1.2 | 1.5 | 1.9 | ≤3 |
| Technical Indicators | Result | 90 Petroleum Asphalt Technical Indicators | Method |
|---|---|---|---|
| Penetration at 25 °C/(0.1 mm) | 2~5 | 80~100 | T 0604 |
| Ductility at 10 °C/(cm) | 1.9 | ≥20 | T 0605 |
| Softening Point/(°C) | 190 | ≥42 | T 0606 |
| Item | Result | ||
|---|---|---|---|
| Limestone Fine Aggregate | ICLR | DCLR | |
| Vickers hardness (HV) | 246 | 615 | 21 |
| Modulus of elasticity (GPa) | 81 | 98 | 6.83 |
| Index | C | H | O | N | S | Si | Ca | Fe | Ti | Al | Mg |
|---|---|---|---|---|---|---|---|---|---|---|---|
| ICLR (%) | 11.44 | 1.80 | 49.43 | 0.45 | 7.63 | 14.84 | 5.17 | 1.86 | 0.71 | 6.11 | 0.57 |
| DCLR (%) | 75.9 | 4.31 | — | 0.35 | 2.47 | — | — | — | — | — | — |
| limestone Fine aggregate (%) | 11.71 | — | 48.13 | — | — | 1.12 | 39.04 | — | — | — | — |
| Ingredient Category | SiO2 | Fe2O3 | TiO2 | Al2O3 | CaO | MgO | Other |
|---|---|---|---|---|---|---|---|
| ICLR (%) | 51.38 | 3.95 | 2.03 | 19.26 | 10.58 | 1.63 | 11.17 |
| Divided by Four Components of Asphalt | Saturation | Aromatic Components | Colloid | Asphaltene |
|---|---|---|---|---|
| Test value (%) | 0.8 | 4.4 | 13.91 | 66.2 |
| Divided by extracted components | Heavy oil | Asphaltene | Preasphaltene | Tetrahydrofuran insoluble matter |
| Test value (%) | 20~30 | 20~40 | 15~30 | 45 |
| Ingredient Category | CaCO3 | SiO2 |
|---|---|---|
| Test value (%) | 97.6 | 2.4 |
| Category | Sample Quality (g) | Quality After Combustion (g) | Burning Loss (%) | Saturation (%) | Aromatic Components (%) | Colloid (%) | Asphaltene (%) | Ic |
|---|---|---|---|---|---|---|---|---|
| 170 °C | 18.0884 | 18.0422 | 0.26 | 0.46 | 2.01 | 9.13 | 88.4 | 7.98 |
| 190 °C | 21.1102 | 20.7832 | 1.55 | 0.63 | 2.47 | 9.20 | 87.7 | 7.57 |
| 210 °C | 19.2810 | 18.6274 | 3.39 | 1.02 | 3.09 | 9.29 | 86.6 | 7.08 |
| 230 °C | 20.5633 | 19.5709 | 4.83 | 1.44 | 3.54 | 9.42 | 85.6 | 6.71 |
| Items | Result | Items | Result |
|---|---|---|---|
| Copper | Not exceeding the standard | Cadmium | Not exceeding the standard |
| Lead | Not exceeding the standard | Chromium | Not exceeding the standard |
| Zinc | Not exceeding the standard | Nickel | Not exceeding the standard |
| Arsenic | Not exceeding the standard | Mercury | Not exceeding the standard |
| Naphthalene | Not detected | Benzofluoranthene | Not detected |
| Dihydrophenanthrene | Not detected | Benzo [a] pyrene | Not detected |
| Peng | Not detected | Benzoanthracene | Not detected |
| Fluorene | Not detected | Dibenzoanthracene | Not detected |
| Phenanthrene | Not detected | Indene pyrene | Not detected |
| Anthracene | Not detected | Benzo [a] pyrene | Not detected |
| Fluoranthene | Not detected | Benzofluoranthene | Not detected |
| Pyrene | Not detected | Benzo [a] pyrene | Not detected |
| Benzoanthracene | Not detected | Total amount of polycyclic aromatic hydrocarbons | Not detected |
| Physical and Chemical Properties | Comparison Results | Physical and Chemical Properties | Comparison Results |
|---|---|---|---|
| Basic Performance | Similarly, the particle size distribution is concentrated in the range of 0~4.75 mm | Chemical composition | ICLR is similar, DCLR has certain differences |
| Mechanical Properties | ICLR is similar, with higher hardness and modulus; DCLR has certain differences, with lower hardness and modulus | Crystal Phase | ICLR is similar and contains a significant amount of SiO2 crystal structure and amorphous material; DCLR has certain differences, mainly containing aromatic carbon microcrystals and fatty carbon microcrystals |
| Microscopic Morphology | Similar, with uniformly distributed scale-like structures on the surface | Functional Groups | ICLR is similar to limestone fine aggregate, with high concentration of DCLR functional groups and rich aromatic and cyclic aromatic structures that are easily bound to asphalt |
| Pore Size Distribution | ICLR and DCLR are similar and have little difference from limestone fine aggregates | Thermal Stability | ICLR is similar to limestone fine aggregate, with little difference in DCLR |
| Element composition | ICLR is similar, DCLR has certain differences | Environmental characteristics | Similar, both do not pose a risk of leaching toxicity |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Wu, H.; Wang, Z.; Li, P.; Li, M.; Li, J.; Guo, S. A Comparative Analysis of the Properties of Coal Liquefaction Residues and Limestone Fine Aggregates. Materials 2026, 19, 1994. https://doi.org/10.3390/ma19101994
Wu H, Wang Z, Li P, Li M, Li J, Guo S. A Comparative Analysis of the Properties of Coal Liquefaction Residues and Limestone Fine Aggregates. Materials. 2026; 19(10):1994. https://doi.org/10.3390/ma19101994
Chicago/Turabian StyleWu, Hao, Zhe Wang, Pengfei Li, Mingliang Li, Jun Li, and Shuangfeng Guo. 2026. "A Comparative Analysis of the Properties of Coal Liquefaction Residues and Limestone Fine Aggregates" Materials 19, no. 10: 1994. https://doi.org/10.3390/ma19101994
APA StyleWu, H., Wang, Z., Li, P., Li, M., Li, J., & Guo, S. (2026). A Comparative Analysis of the Properties of Coal Liquefaction Residues and Limestone Fine Aggregates. Materials, 19(10), 1994. https://doi.org/10.3390/ma19101994







