Enhanced Aging Stability of Ordered Mesoporous Silica Materials Synthesized via True Liquid Crystal Templating—A Small-Angle X-Ray Scattering Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Material Synthesis
2.2. Gas Physisorption Analysis
2.3. X-Ray Diffraction Analysis

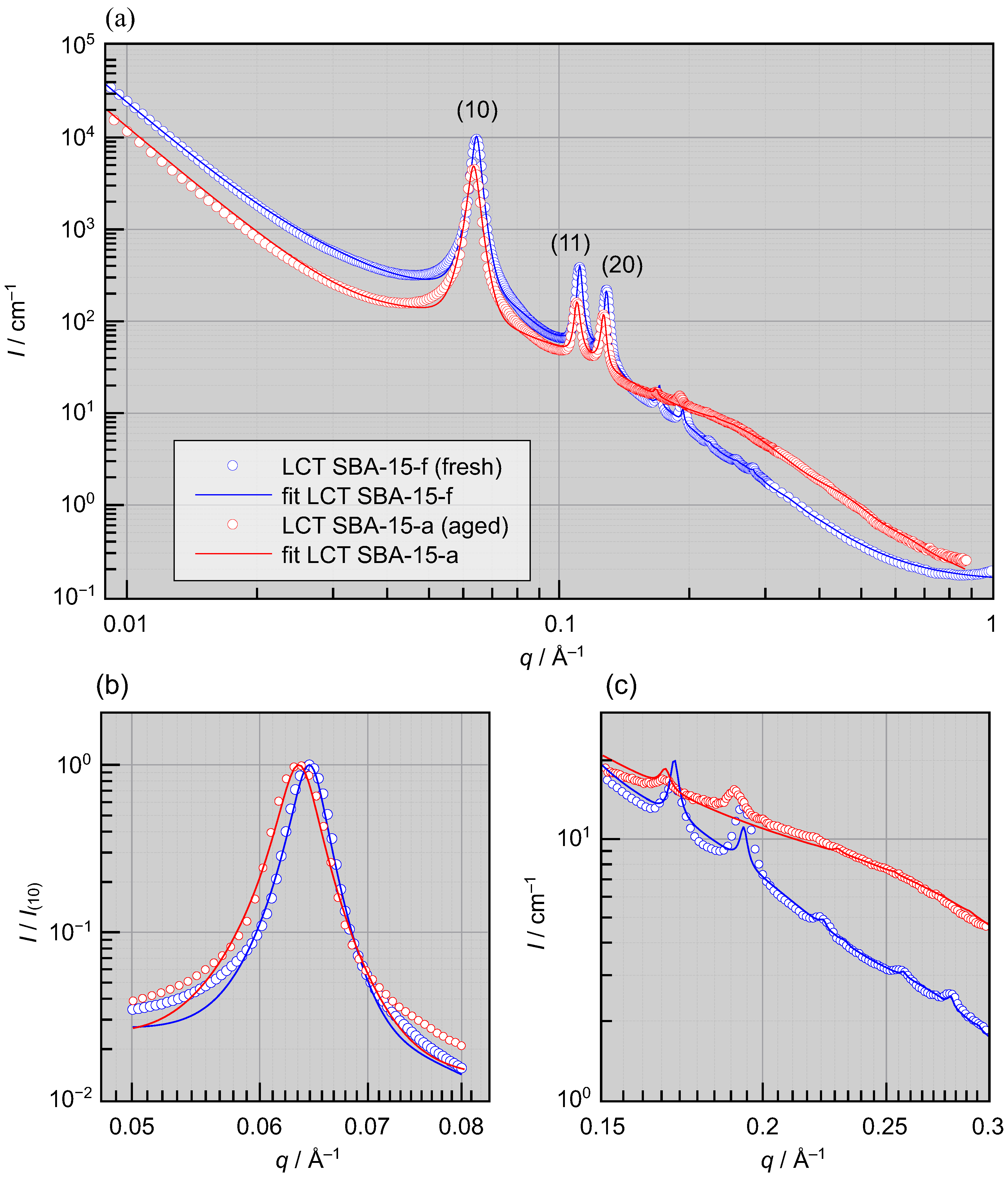

3. Results
3.1. Experimental Observations
- On the one hand, at higher values of q corresponding to the length scales of micropores, a strong increase in scattering intensity is observed. This observation suggests that, the microporosity increased during aging. As a result of this increase in micropore scattering, the weakest Bragg reflections are no longer seen in the aged sample.
- On the other hand, the Bragg peaks have slightly shifted to smaller values of the scattering vector q, which is particularly evident in the leading (10) peak seen in Figure 3b (as well as in the higher order peaks). This shift indicates an expansion of the 2D hexagonal lattice during aging.
3.2. Detailed Analysis and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| OMMs | Ordered mesoporous materials |
| OMS | Ordered mesoporous silica |
| LCT | Liquid crystal templating |
| TLCT | True liquid crystal templating |
| SAXS | Small angle X-ray scattering |
| BET | Brunauer-Emmett-Teller |
| DFT | Density functional theory |
| NLDFT | Nonlinear density functional theory |
References
- International Union of Pure and Applied Chemistry (IUPAC). Mesopore. In IUPAC Compendium of Chemical Terminology, 5th ed.; IUPAC: Málaga, Spain, 2025. [Google Scholar] [CrossRef]
- Wan, Y.; Zhao, D. On the controllable soft-templating approach to mesoporous silicates. Chem. Rev. 2007, 107, 2821–2860. [Google Scholar] [CrossRef] [PubMed]
- Impéror-Clerc, M.; Albouy, P.-A. Characterization of the Structure of Mesoporous Materials by X-ray Scattering. In Comprehensive Guide for Mesoporous Materials; Aliofkhazraei, M., Ed.; Nova Science Publishers: Hauppauge, NY, USA, 2015; Volume 1, Chapter 13; pp. 331–360. [Google Scholar]
- Losito, D.W.; de Araujo, D.R.; Bezzon, V.D.N.; Oseliero Filho, P.L.; Fonseca, F.L.A.; Chagas, C.d.S.; Barbosa, E.; Oliveira, C.L.P.; Fantini, M.C.d.A.; Ferreira, F.F.; et al. Mesoporous Silica–Fe3O4 Nanoparticle Composites as Potential Drug Carriers. ACS Appl. Nano Mater. 2021, 4, 13363–13378. [Google Scholar] [CrossRef]
- Tischler, I.; Schlaich, A.; Holm, C. Disentanglement of Surface and Confinement Effects for Diene Metathesis in Mesoporous Confinement. ACS Omega 2024, 9, 598–606. [Google Scholar] [CrossRef]
- Kresge, C.T.; Leonowicz, M.E.; Roth, W.J.; Vartuli, J.C.; Beck, J.S. Ordered Mesoporous Molecular-Sieves Synthesized by a Liquid-Crystal Template Mechanism. Nature 1992, 359, 710–712. [Google Scholar] [CrossRef]
- Zhao, X.S.; Audsley, F.; Lu, G.Q. Irreversible change of pore structure of MCM-41 upon hydration at room temperature. J. Phys. Chem. B 1998, 102, 4143–4146. [Google Scholar] [CrossRef]
- Bruckner, J.R.; Bauhof, J.; Gebhardt, J.; Beurer, A.K.; Traa, Y.; Giesselmann, F. Mechanisms and Intermediates in the True Liquid Crystal Templating Synthesis of Mesoporous Silica Materials. J. Phys. Chem. B 2021, 125, 3197–3207. [Google Scholar] [CrossRef]
- Burleigh, M.C.; Markowitz, M.A.; Jayasundera, S.; Spector, M.S.; Thomas, C.W.; Gaber, B.P. Mechanical and Hydrothermal Stabilities of Aged Periodic Mesoporous Organosilicas. J. Phys. Chem. B 2003, 107, 12628–12634. [Google Scholar] [CrossRef]
- Adeniran, B.; Mokaya, R. On the Shelf Life and Aging Stability of Mesoporous Silica: Insights on Thermodynamically Stable MCM-41 Structure from Assessment of 12-Year-Old Samples. Chem. Mater. 2012, 24, 4450–4458. [Google Scholar] [CrossRef]
- Zhang, F.; Yan, Y.; Yang, H.; Meng, Y.; Yu, C.; Tu, B.; Zhao, D. Understanding effect of wall structure on the hydrothermal stability of mesostructured silica SBA-15. J. Phys. Chem. B 2005, 109, 8723–8732. [Google Scholar] [CrossRef]
- Albouy, P.A.; Ayral, A. Coupling X-ray Scattering and Nitrogen Adsorption: An Interesting Approach for the Characterization of Ordered Mesoporous Materials. Application to Hexagonal Silica. Chem. Mater. 2002, 14, 3391–3397. [Google Scholar] [CrossRef]
- Zickler, G.A.; Jähnert, S.; Funari, S.S.; Findenegg, G.H.; Paris, O. Pore lattice deformation in ordered mesoporous silica studied by in situ small-angle X-ray diffraction. J. Appl. Crystallogr. 2007, 40, s522–s526. [Google Scholar] [CrossRef]
- Gor, G.Y.; Huber, P.; Bernstein, N. Adsorption-Induced Deformation of Nanoporous Materials—A Review. Appl. Phys. Rev. 2017, 4, 011303. [Google Scholar] [CrossRef]
- CRC 1333. Collaborative Research Center 1333: Molecular Heterogeneous Catalysis in Confined Geometries. Available online: https://www.crc1333.de/ (accessed on 27 November 2025).
- Beurer, A.K.; Dieterich, S.; Solodenko, H.; Kaya, E.; Merdanoǧlu, N.; Schmitz, G.; Traa, Y.; Bruckner, J.R. Comparative study of lattice parameter and pore size of ordered mesoporous silica materials using physisorption, SAXS measurements and transmission electron microscopy. Micropor. Mesopor. Mater. 2023, 354, 112508. [Google Scholar] [CrossRef]
- Sundblom, A.; Oliveira, C.L.P.; Palmqvist, A.E.C.; Pedersen, J.S. Modeling in Situ Small-Angle X-ray Scattering Measurements Following the Formation of Mesostructured Silica. J. Phys. Chem. C 2009, 113, 7706–7713. [Google Scholar] [CrossRef]
- Tan, X.; Gerbelli, B.B.; Fantini, M.C.d.A.; Oliveira, C.L.P.; Bordallo, H.N.; Oseliero Filho, P.L. Retrieving the size distribution of SBA-15 mesopores from small-angle X-ray scattering data using a Monte Carlo method. J. Appl. Crystallogr. 2023, 56, 1381–1391. [Google Scholar] [CrossRef] [PubMed]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S. Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef]
- Sarawade, P.B.; Kim, J.K.; Hilonga, A.; Kim, H.T. Influence of aging conditions on textural properties of water-glass-based silica aerogels prepared at ambient pressure. J. Chem. Eng. 2010, 27, 1301–1309. [Google Scholar] [CrossRef]
- Lai, W.; Yang, S.; Jiang, Y.; Zhao, F.; Li, Z.; Zaman, B.; Fayaz, M.; Li, X.; Chen, Y. Artefact peaks of pore size distributions caused by unclosed sorption isotherm and tensile strength effect. Adsorption 2020, 26, 633–644. [Google Scholar] [CrossRef]
- Reichenbach, C.; Kalies, G.; Enke, D.; Klank, D. Cavitation and pore blocking in nanoporous glasses. Langmuir 2011, 27, 10699–10704. [Google Scholar] [CrossRef] [PubMed]
- Michaux, F.; Baccile, N.; Impéror-Clerc, M.; Malfatti, L.; Folliet, N.; Gervais, C.; Manet, S.; Meneau, F.; Pedersen, J.S.; Babonneau, F. In situ time-resolved SAXS study of the formation of mesostructured organically modified silica through modeling of micelle evolution during surfactant-templated self-assembly. Langmuir 2012, 28, 17477–17493. [Google Scholar] [CrossRef]
- Prass, J.; Müter, D.; Fratzl, P.; Paris, O. Capillarity-driven deformation of ordered nanoporous silica. Appl. Phys. Lett. 2009, 95, 083121. [Google Scholar] [CrossRef]
- Ludescher, L.; Morak, R.; Braxmeier, S.; Putz, F.; Hüsing, N.; Reichenauer, G.; Paris, O. Hierarchically organized materials with ordered mesopores: Adsorption isotherm and adsorption-induced deformation from small-angle scattering. Phys. Chem. Chem. Phys. 2020, 22, 12713–12723. [Google Scholar] [CrossRef] [PubMed]
- Ciccotti, M.; George, M.; Ranieri, V.; Wondraczek, L.; Marlière, C. Dynamic condensation of water at crack tips in fused silica glass. J. Non-Cryst. Solids 2008, 354, 564–568. [Google Scholar] [CrossRef]
- Galarneau, A.; Nader, M.; Guenneau, F.; Di Renzo, F.; Gedeon, A. Understanding the stability in water of mesoporous SBA-15 and MCM-41. J. Phys. Chem. C 2007, 111, 8268–8277. [Google Scholar] [CrossRef]
- Leroch, S.; Wendland, M. Influence of capillary bridge formation on the silica nanoparticle interaction studied by grand canonical Monte Carlo simulations. Langmuir 2013, 29, 12410–12420. [Google Scholar] [CrossRef] [PubMed]
- Ryoo, R.; Joo, S.H.; Kruk, M.; Jaroniec, M. Ordered Mesoporous Carbons. Adv. Mater. 2001, 13, 677–681. [Google Scholar] [CrossRef]
- Wainwright, S.G.; Parlett, C.M.A.; Blackley, R.A.; Zhou, W.; Lee, A.F.; Wilson, K.; Bruce, D.W. True liquid crystal templating of SBA-15 with reduced microporosity. Microporous Mesoporous Mater. 2013, 172, 112–117. [Google Scholar] [CrossRef]
- Pedersen, J.S. Analysis of small-angle scattering data from colloids and polymer solutions: Modeling and least-squares fitting. Adv. Colloid Interface Sci. 1997, 70, 171–210. [Google Scholar] [CrossRef]
- Förster, S.; Timmann, A.; Konrad, M.; Schellbach, C.; Meyer, A.; Funari, S.S.; Mulvaney, P.; Knott, R. Scattering curves of ordered mesoscopic materials. J. Phys. Chem. B 2005, 109, 1347–1360. [Google Scholar] [CrossRef]
- Debye, P.; Anderson, H.R.; Brumberger, H. Scattering by an Inhomogeneous Solid. II. The Correlation Function and Its Application. J. Appl. Phys. 1957, 28, 679–683. [Google Scholar] [CrossRef]

| Abbreviation | Description |
|---|---|
| LCT SBA-15-f | SBA-15 synthesized via LCT, freshly calcined, and stored in a sealed 0.9 mm capillary |
| LCT SBA-15-a | SBA-15-f, stored in a glass vial with a snap-on plastic cap at ambient conditions for one year |
| LCT SBA-15-ad | SBA-15-a is gently dried under vacuum at 323 K for 24 h |
| TLCT SBA-15-f | SBA-15 synthesized via TLCT, freshly calcined, and stored in a sealed 0.9 mm capillary |
| TLCT SBA-15-a | TLCT SBA-15-f stored under the same conditions as LCT SBA-15-a |
| TLCT SBA-15-ad | TLCT SBA-15-a, gently dried under vacuum at 323 K for 24 h |
| Sample | a | r | T | d | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| LCT SBA-15-f | 112.58 (2) | 42.8 (3) | 2.4 (2) | 27.0 (6) | (2) | 13.3 (2) | 0.16 (4) | 45.5 | 618.0 | 4.00 (6) |
| LCT SBA-15-a | 114.33 (3) | 38.7 (5) | 4.7 (2) | 36.9 (10) | (4) | 5.2 (2) | 0.34 (1) | 45.6 | 233.0 | 4.00 (1) |
| LCT SBA-15-ad | 113.75 (5) | 39.7 (7) | 4.6 (2) | 34.4 (14) | (5) | 5.7 (2) | 0.28 (1) | – | – | 4.00 (2) |
| TLCT SBA-15-f | 94.45 (3) | 33.9 (2) | 1.9 (2) | 26.7 (4) | (3) | 9.3 (1) | 0.12 (1) | 31.6 | 239.0 | 3.93 (4) |
| TLCT SBA-15-a | 94.64 (3) | 31.0 (3) | 2.8 (2) | 32.6 (6) | (3) | 8.3 (2) | 0.12 (2) | 30.4 | 241.4 | 3.63 (2) |
| TLCT SBA-15-ad | 94.84 (3) | 33.1 (2) | 2.0 (2) | 28.6 (4) | (3) | 9.1 (1) | 0.12 (1) | – | – | 3.86 (1) |
| Comparison | Peaks Used | Average Strain (%) | Average Strain Uncertainty (%) |
|---|---|---|---|
| TLCT SBA-15 fresh vs. aged | (10), (11), (20), (30) | 0.1 | 0.1 |
| LCT SBA-15 fresh vs. aged | (10), (11), (20), (21), (30) | 1.53 | 0.05 |
| LCT SBA-15 fresh vs. aged degas | (10), (11), (20), (21), (30) | 1.07 | 0.08 |
| LCT SBA-15 aged vs. aged degas | (10), (11), (20), (21), (30) | −0.45 | 0.09 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Tan, X.; Atwi, B.; Bui Duc, H.; Buchmeiser, M.R.; Giesselmann, F. Enhanced Aging Stability of Ordered Mesoporous Silica Materials Synthesized via True Liquid Crystal Templating—A Small-Angle X-Ray Scattering Study. Materials 2026, 19, 1923. https://doi.org/10.3390/ma19101923
Tan X, Atwi B, Bui Duc H, Buchmeiser MR, Giesselmann F. Enhanced Aging Stability of Ordered Mesoporous Silica Materials Synthesized via True Liquid Crystal Templating—A Small-Angle X-Ray Scattering Study. Materials. 2026; 19(10):1923. https://doi.org/10.3390/ma19101923
Chicago/Turabian StyleTan, Xiangyin, Boshra Atwi, Huy Bui Duc, Michael R. Buchmeiser, and Frank Giesselmann. 2026. "Enhanced Aging Stability of Ordered Mesoporous Silica Materials Synthesized via True Liquid Crystal Templating—A Small-Angle X-Ray Scattering Study" Materials 19, no. 10: 1923. https://doi.org/10.3390/ma19101923
APA StyleTan, X., Atwi, B., Bui Duc, H., Buchmeiser, M. R., & Giesselmann, F. (2026). Enhanced Aging Stability of Ordered Mesoporous Silica Materials Synthesized via True Liquid Crystal Templating—A Small-Angle X-Ray Scattering Study. Materials, 19(10), 1923. https://doi.org/10.3390/ma19101923








