Study on Sintering Behavior, Heat and Wear Resistance of Refractory Metal Borides (HfB2, ZrB2) and Al-Ni Modified PDC
Abstract
1. Introduction
- To systematically compare the effects of three additives (HfB2, ZrB2, and Al-Ni) on the heat resistance (e.g., thermal failure temperature, linear thermal expansion coefficient) and wear resistance (e.g., VTL wear cycles, wear area, wear ratio) of PDC under a unified high-temperature and high-pressure (HTHP) sintering process, focusing on clarifying how different additives improve PDC’s mechanical properties;
- To reveal the synergistic strengthening mechanism between the three additives and the PDC matrix, including phase evolution (e.g., formation of HfC/ZrC/AlCoxNi1-X), element interaction, and interface bonding improvement;
- To screen out the additive with the optimal comprehensive performance, thereby providing theoretical support for the development of high-performance PDC suitable for complex downhole environments (where high heat and wear resistance are required).
2. Materials and Methods
2.1. Sample Preparation
2.2. Performance Testing Methods
2.2.1. Heat Resistance Testing Method
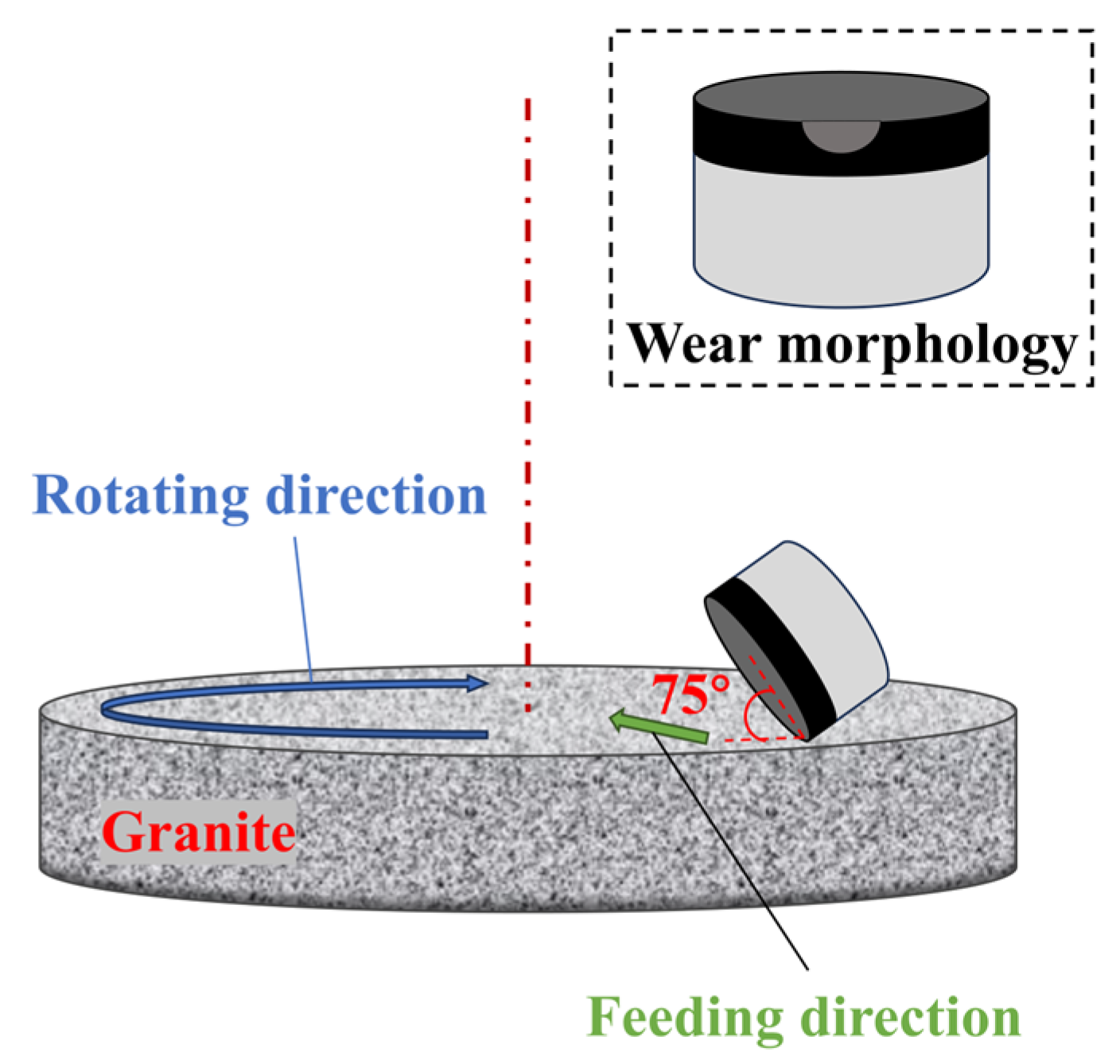
2.2.2. Wear Resistance Testing Method
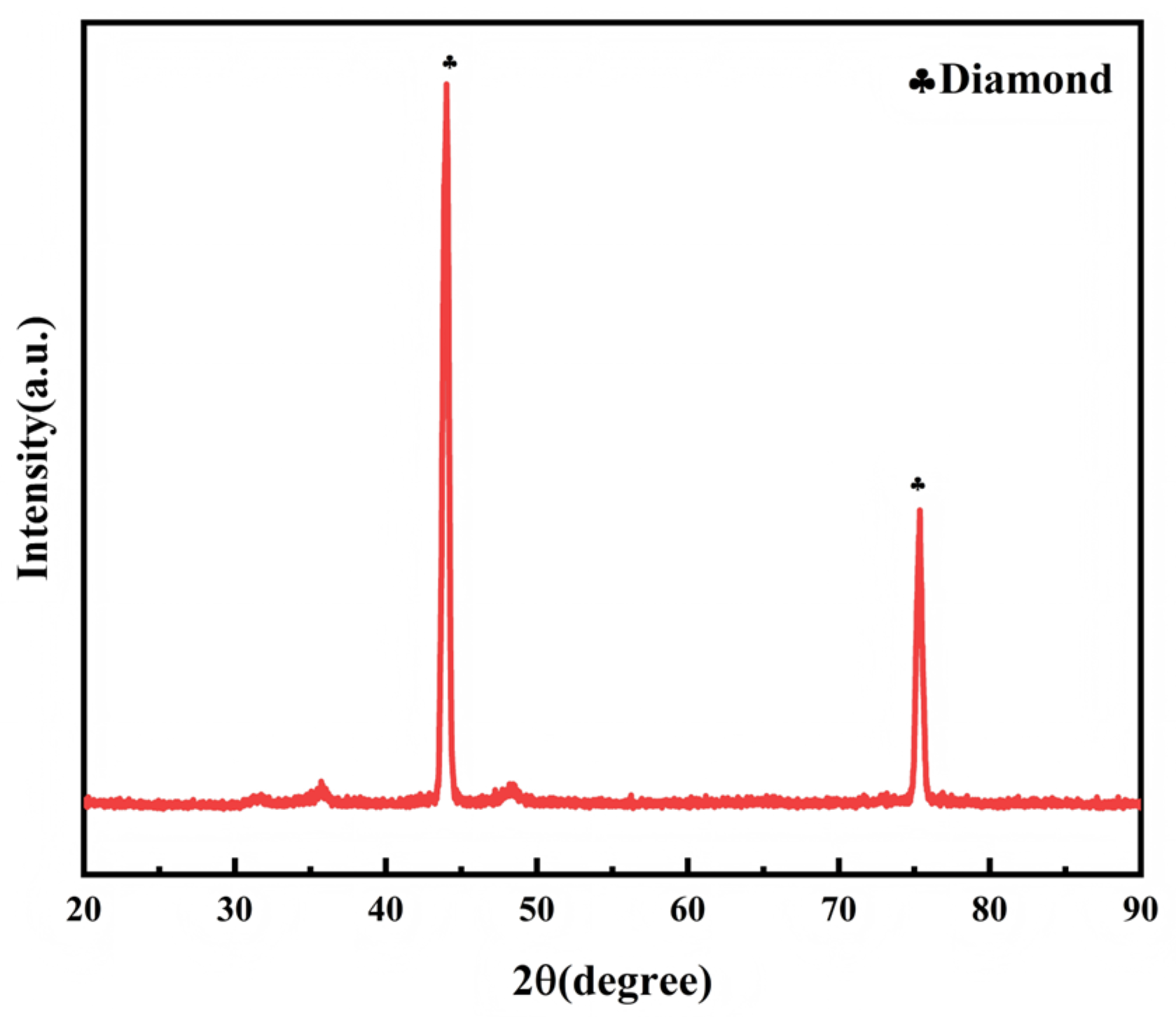
2.3. Characterization Results
3. Results
3.1. Heat Resistance Testing
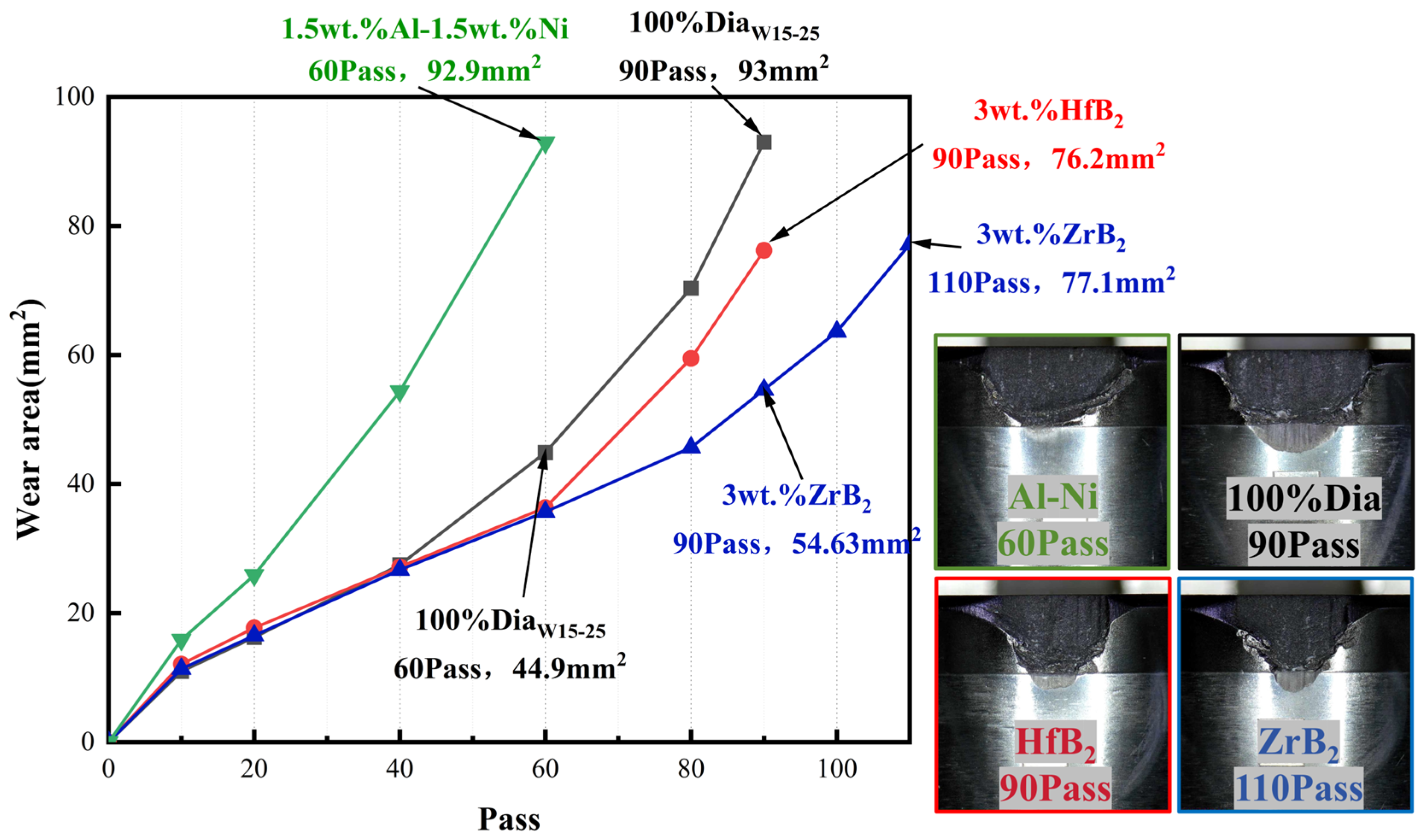
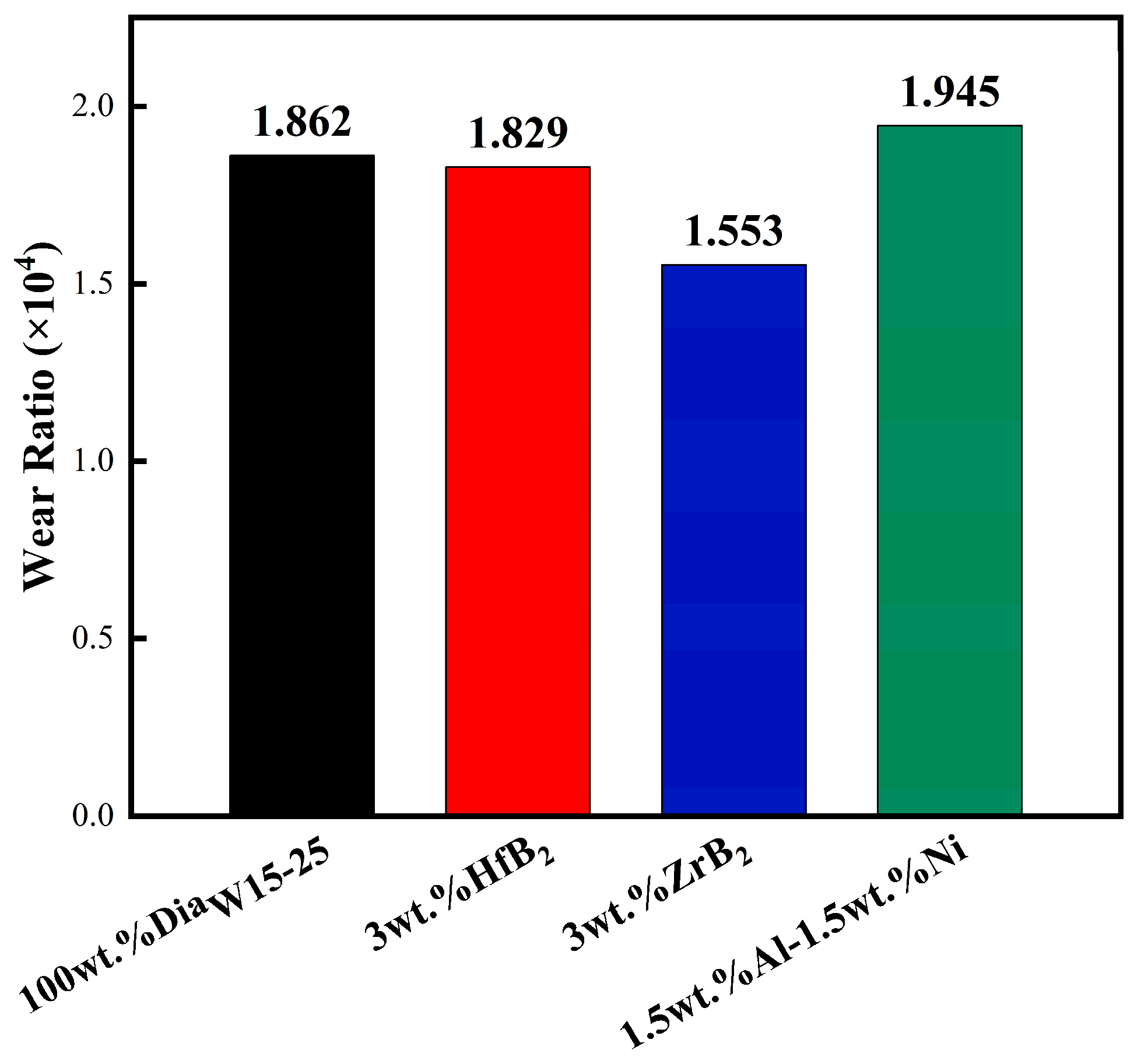
3.2. Wear Resistance Testing
4. Conclusions
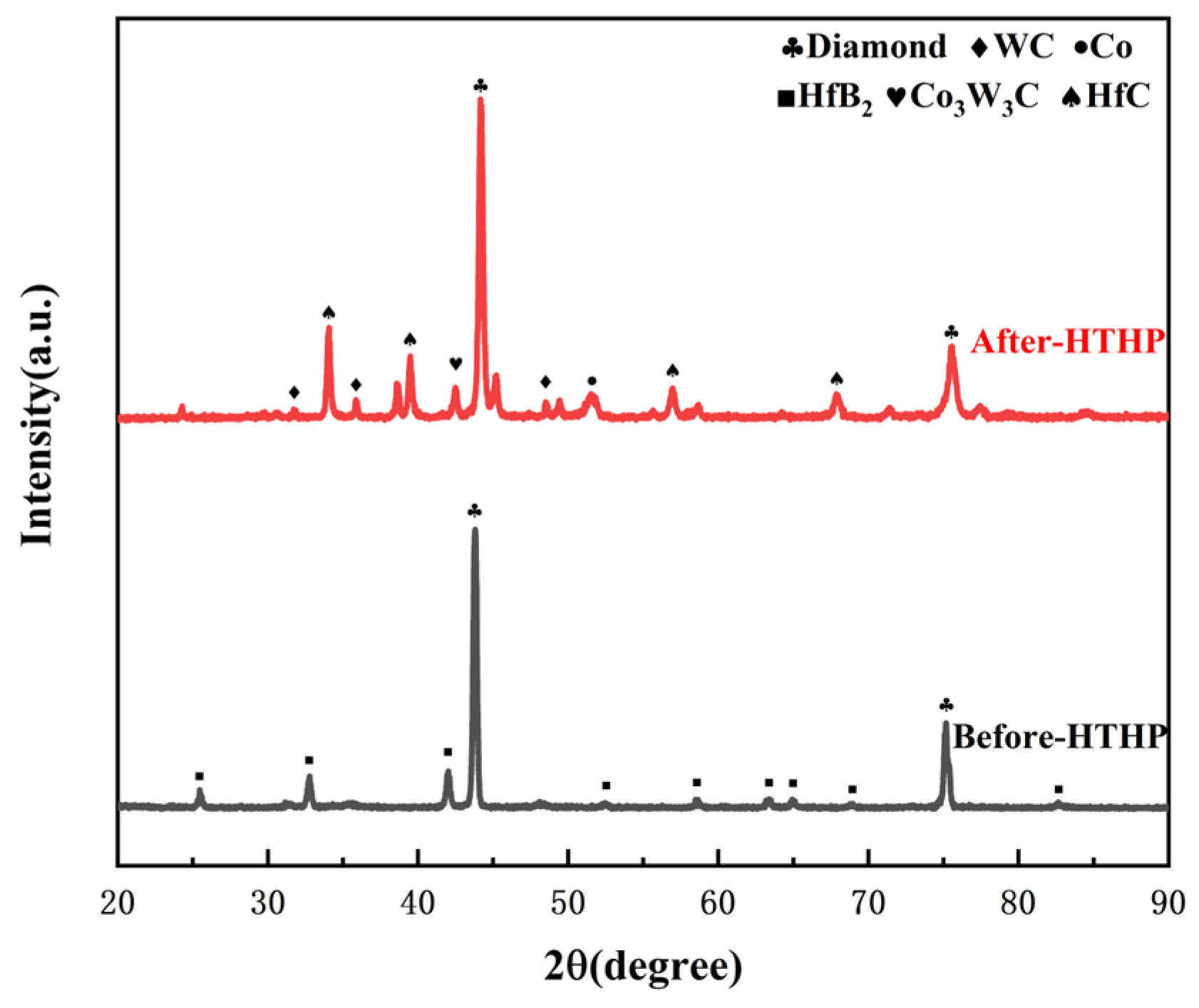
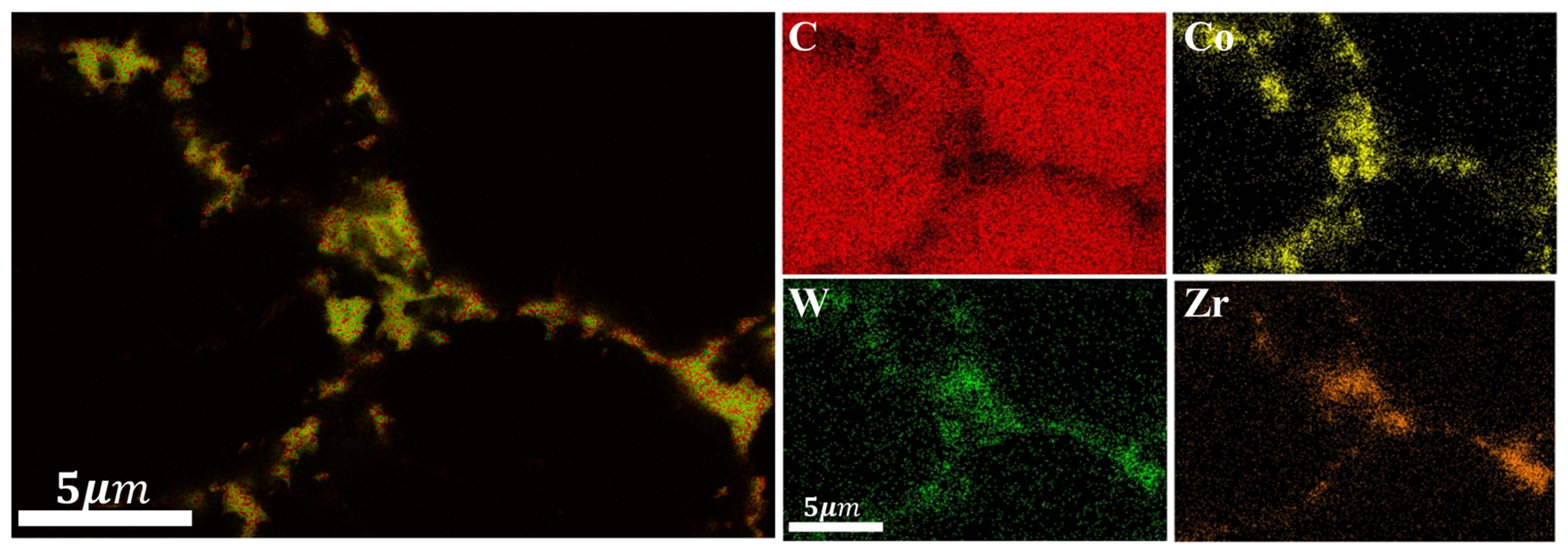
- For PDC with HfB2 addition, in addition to diamond, HfC phases were observed on the surface of the polycrystalline layer after sintering, and the content of HfC increased with the increase in the additive content. Large block-shaped HfC phases and strip-shaped HfC phases were formed between diamond particles on the surface of the polycrystalline layer. Additionally, Co-W-C phases were detected, all of which are phases that enhance mechanical properties. This system exhibited a smaller thermal expansion capacity, which is attributed to the low linear thermal expansion coefficient of the additive, thus providing a certain buffer for the thermal expansion of the system.
- For PDC with ZrB2 addition, Co-W-C, Co-B, and ZrB phases appeared on the surface after sintering. This system could effectively reduce the content of elemental Co in the polycrystalline layer. The experimental group with ZrB2 addition achieved a test result of 110 working cycles, which was a 22.2% increase compared to the blank group. This study also confirmed the rule that reducing the content of elemental Co in the system can improve heat resistance.
- For PDC with Al-Ni addition, intermetallic compounds AlCoxNi1−x and AlNi3C0.5 phases were formed on the surface after sintering, which effectively reduced the content of elemental Co in the polycrystalline layer. PDC in this system had a relatively high wear ratio but a low number of wear cycles.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, Y.G.; Liu, B.; Song, J.Y.; Yan, X.P.; Wu, K.M. Study on the Wear Mechanism of PCD Tools in High-Speed Milling of Al-Si Alloy. Adv. Mater. Res. 2011, 381, 16–19. [Google Scholar] [CrossRef]
- Kim, D.; Beal, A.; Kwon, P. Effect of tool wear on hole quality in drilling of carbon fiber reinforced plastic–titanium alloy stacks using tungsten carbide and polycrystalline diamond tools. J. Manuf. Sci. Eng. 2016, 138, 031006. [Google Scholar] [CrossRef]
- Liang, Y.; Du, B.; Ren, J.; Feng, S. Research on the type and expansion mechanism of internal cracks in rocks under the action of PDC. Sci. Rep. 2025, 15, 25136. [Google Scholar] [CrossRef]
- Guo, Y.; Meng, D.; Cai, Z.; Wang, C.; Yue, W. New insights on impact wear of polycrystalline diamond compact: Effect of interface state on kinetic response and damage behavior. Carbon 2024, 230, 119661. [Google Scholar] [CrossRef]
- Ke, X.; Yang, X.; Prasad, V.; Peng, Q.; Feng, X.; Li, Z. Effect of thermal residual stress on the impact failure of PDC in application. Int. J. Refract. Met. Hard Mater. 2025, 128, 107078. [Google Scholar] [CrossRef]
- Glowka, D.A. Implications of thermal wear phenomena for PDC bit design and operation. In Proceedings of the SPE Annual Technical Conference and Exhibition, Las Vegas, NV, USA, 22–26 September 1985; SPE: Richardson, TX, USA. [Google Scholar] [CrossRef]
- Wentorf, R.; DeVries, R.C.; Bundy, F. Sintered superhard materials. Science 1980, 208, 873–880. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Shao, H.; He, D.; Chen, Y.; Zhang, F. Preparation and Characterization of Diamond-SiC, Nanostructured Diamond-TiC and Diamond-Diamond Direct Bonding Type Polycrystalline. Superhard Mater. Eng. 2015, 27, 6–11. [Google Scholar]
- Qiu, H.; Long, W.; Jianwei, L. Preparation of diamond/vitrified bond composite sintered by sol-gel method and its performance characterization. J. Mater. Eng. 2019, 47, 130–135. [Google Scholar]
- Mingzhi, W.; Yanhui, W.; Jianbing, Z. Study on Interface Stucture and Bond Properties of Polycrystalline Diamond Combined with Titanium as Bonder. J. Synth. Cryst. 1996, 25, 236–239. [Google Scholar]
- Sheng, Y.; Tao, W.; Heyi, L. Properties of Si-Ti-B Doped Diamond/Cemented Carbide Composites. Chin. J. Eng. 1991, 13, 442–446. [Google Scholar] [CrossRef]
- Ueda, F.; Ohashi, T.; Akaishi, M.; Yamaoka, S. Cutting performance of sintered diamond with MgCO3 as a sintering agent. Mater. Sci. Eng. A 1996, 209, 260–263. [Google Scholar] [CrossRef]
- Jaworska, L.; Klimczyk, P.; Szutkowska, M.; Putyra, P.; Sitarz, M.; Cygan, S.; Rutkowski, P. Thermal resistance of PCD materials with borides bonding phase. J. Superhard Mater. 2015, 37, 155–165. [Google Scholar] [CrossRef]
- Jaworska, L. Diamond–ceramic bonding phase composites for application in cutting tools. Mater. Ceram. 2011, 63, 131–137. [Google Scholar]
- Szutkowska, M.; Jaworska, L.; Boniecki, M.; Stobierski, L.; Rozmus, M. Mechanical behavior of diamond matrix composites with ceramic Ti3 (Si, Ge) C2 bonding phase. Int. J. Refract. Met. Hard Mater. 2015, 49, 302–306. [Google Scholar] [CrossRef]
- Jaworska, L.; Olszowka-Myalska, A.; Cygan, S.; Figiel, P.; Karolus, M.; Cyboron, J. The influence of tungsten carbide contamination from the milling process on PCD materials oxidation. Int. J. Refract. Met. Hard Mater. 2017, 64, 60–65. [Google Scholar] [CrossRef]
- Osipov, A.; Klimczyk, P.; Cygan, S.; Melniychuk, Y.A.; Petrusha, I.A.; Jaworska, L.; Bykov, A. IDiamond-CaCO3 and diamond-Li2CO3 materials sintered using the HPHT method. J. Eur. Ceram. Soc. 2017, 37, 2553–2558. [Google Scholar] [CrossRef]
- Osipov, A.; Nauyoks, S.; Zerda, T.W.; Zaporozhets, O. IRapid sintering of nano-diamond compacts. Diam. Relat. Mater. 2009, 18, 1061–1064. [Google Scholar] [CrossRef]
- Ekimov, E.; Ralchenko, V.; Popovich, A. Synthesis of superconducting boron-doped diamond compacts with high elastic moduli and thermal stability. Diam. Relat. Mater. 2014, 50, 15–19. [Google Scholar] [CrossRef]
- Ekimov, E.; Sidorov, V.A.; Sadykov, R.A.; Mel’Nik, N.N.; Gierlotka, S.; Presz, A. Synthesis of carbonado-like polycrystalline diamond in the B4C–graphite system. Diam. Relat. Mater. 2005, 14, 437–440. [Google Scholar] [CrossRef]
- Jaworska, L.; Szutkowska, M.; Klimczyk, P.; Sitarz, M.; Bucko, M.; Rutkowski, P.; Lojewska, J. Oxidation, graphitization and thermal resistance of PCD materials with the various bonding phases of up to 800 C. Int. J. Refract. Met. Hard Mater. 2014, 45, 109–116. [Google Scholar] [CrossRef]
- Lu, Y.-G.; Turner, S.; Ekimov, E.A.; Verbeeck, J.; Van Tendeloo, G. Boron-rich inclusions and boron distribution in HPHT polycrystalline superconducting diamond. Carbon 2015, 86, 156–162. [Google Scholar] [CrossRef]
- Mashhadikarimi, M. Obtaining Triple Layer Polycrystalline Diamond Compact by HPHT Method. 2017. Available online: https://repositorio.ufrn.br/jspui/handle/123456789/23749 (accessed on 5 November 2025).
- Gurgel, D.P.; Mashhadikarimi, M.; Rodrigues, M.A.L.D.O.; Barreto, L.P.D.P.; Filgueira, M.; Gomes, U.U.; Bichler, L. Niobium as a new binder for polycrystalline diamond (PCD) sintered via high pressure-high temperature (HPHT). Int. J. Refract. Met. Hard Mater. 2020, 90, 105234. [Google Scholar] [CrossRef]
- Podborodnikov, I.V.; Shatskiy, A.; Arefiev, A.V.; Bekhtenova, A.; Litasov, K.D. New data on the system Na2CO3–CaCO3–MgCO3 at 6 GPa with implications to the composition and stability of carbonatite melts at the base of continental lithosphere. Chem. Geol. 2019, 515, 50–60. [Google Scholar] [CrossRef]
- Jivanji, M.; Forbes, R.P.; Sithebe, H.; Westraadt, J.E. Effect of ZrB2 additions on the thermal stability of polycrystalline diamond. Int. J. Refract. Met. Hard Mater. 2023, 113, 106202. [Google Scholar] [CrossRef]
- Tkatchenko, V.; Filonenko, V.P.; Bagramov, R.K.; Zibrov, I.P.; Anokhin, A.S.; Bondar, I.V.; Shipkov, A.N. Diamond composites with Al-Co binder: Synthesis, structure, wear resistance. Mater. Lett. 2023, 330, 133317. [Google Scholar] [CrossRef]
- Khabashesku, V.N.; Filonenko, V.; Bagramov, R.; Zibrov, I.; Anokhin, A. Nanoengineered polycrystalline diamond composites with advanced wear resistance and thermal stability. ACS Appl. Mater. Interfaces 2021, 13, 59560–59566. [Google Scholar] [CrossRef]
- Khabashesku, V.N.; Filonenko, V.P.; Bagramov, R.K.; Zibrov, I.P. Diamond Composites Produced from Fluorinated Mixtures of Micron-Sized and Nanodiamonds by Metal Infiltration. Materials 2022, 15, 4936. [Google Scholar] [CrossRef]
- Mishima, Y.; Kato, M.; Kimura, Y.; Hosoda, H.; Miura, S. Improvement in room temperature ductility of intermetallic alloys through microstructural control. Intermetallics 1996, 4, S171–S179. [Google Scholar] [CrossRef]
- Kimura, Y.; Mishima, Y.; Liu, C. Microstructure control and tensile properties of three-phase alloys based on the E21 Co3AlC and B2 CoAl. Intermetallics 2001, 9, 1069–1078. [Google Scholar] [CrossRef]
- Zhao, C.; Liu, B.C. Research on improving the thermal resistance of polyerystalline diamondcompact by adding HfB2. Equip. Geotech. Eng. 2024, 25, 1–6. [Google Scholar]
- Liu, C.; Kou, Z.; He, D.; Chen, Y.; Wang, K.; Hui, B.; Wang, Y. Effect of removing internal residual metallic phases on wear resistance of polycrystalline diamond compacts. Int. J. Refract. Met. Hard Mater. 2012, 31, 187–191. [Google Scholar] [CrossRef]
- Wang, X.; Tu, J.; Liu, B. Effects of initial diamond particle size on the comprehensive mechanical properties of PDC. Ceram. Int. 2025, 51, 10433–10442. [Google Scholar] [CrossRef]







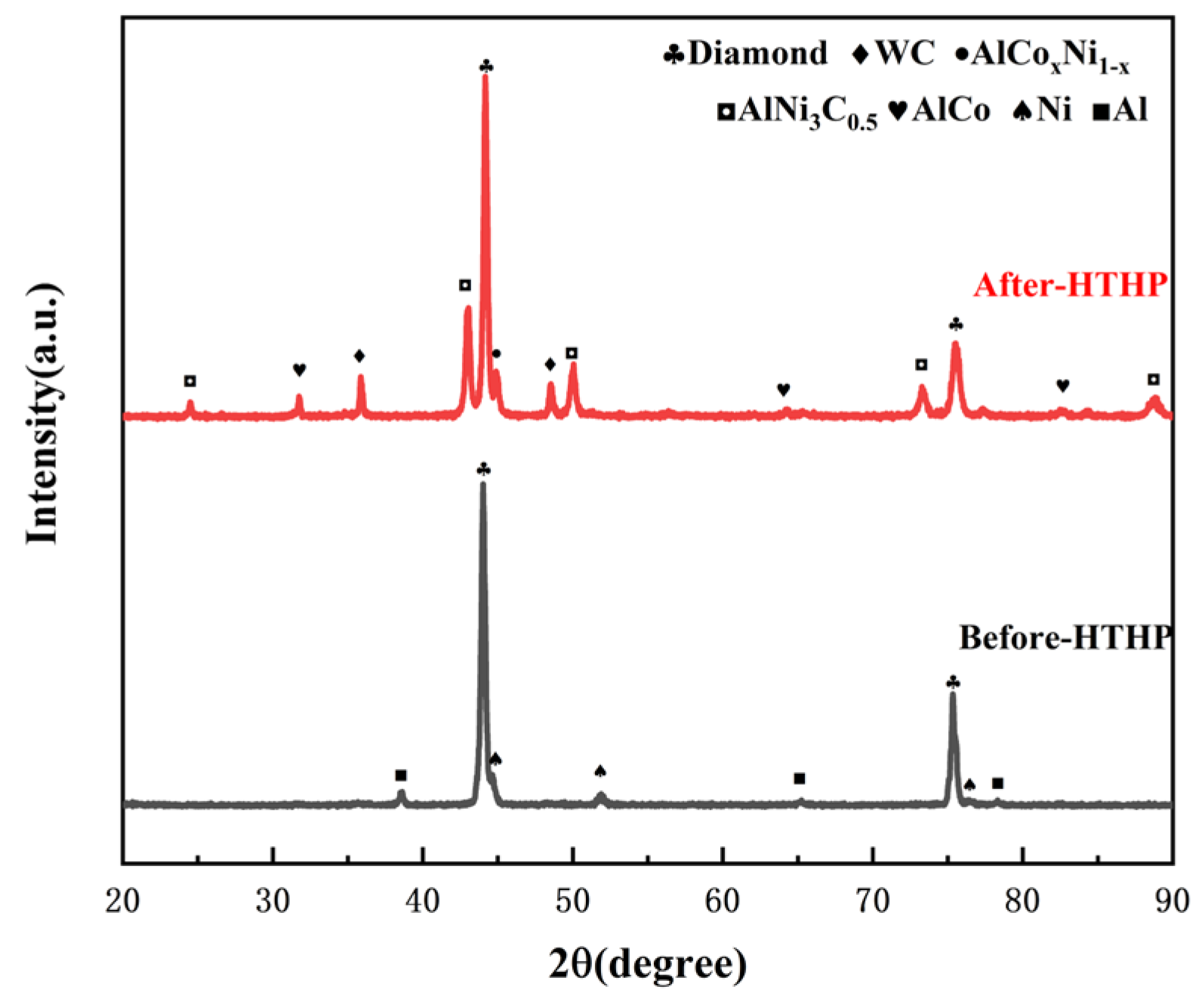
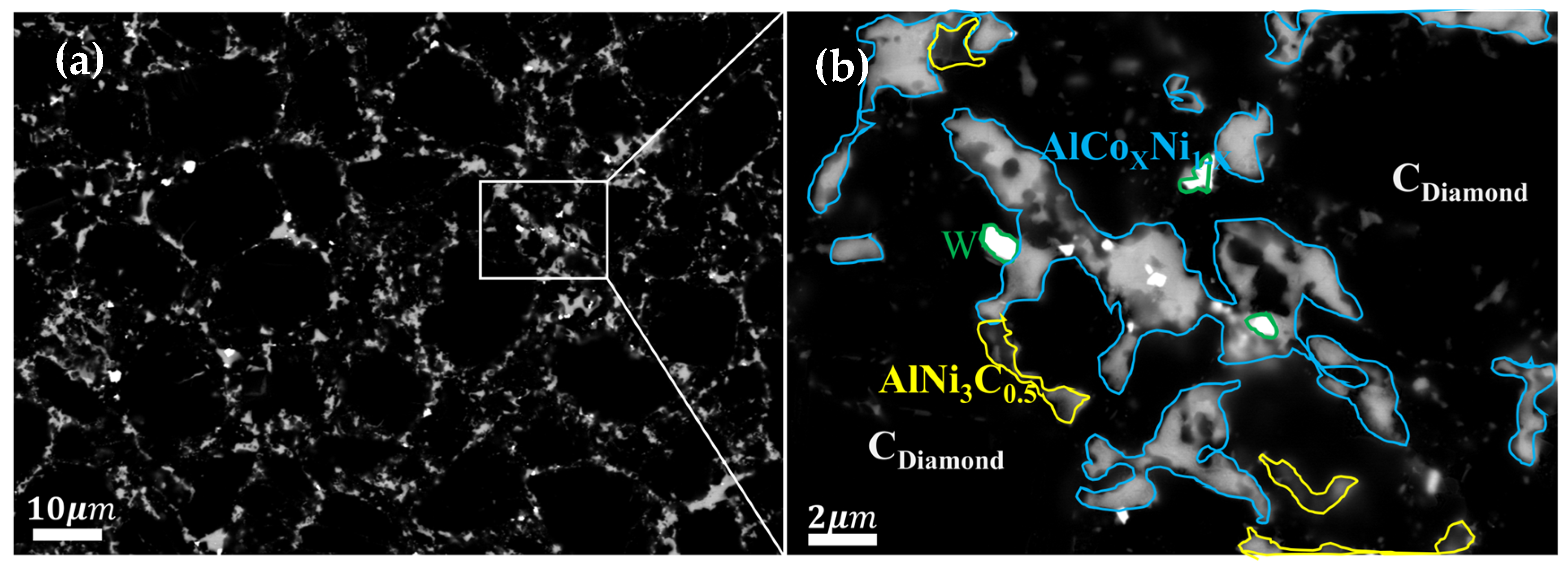
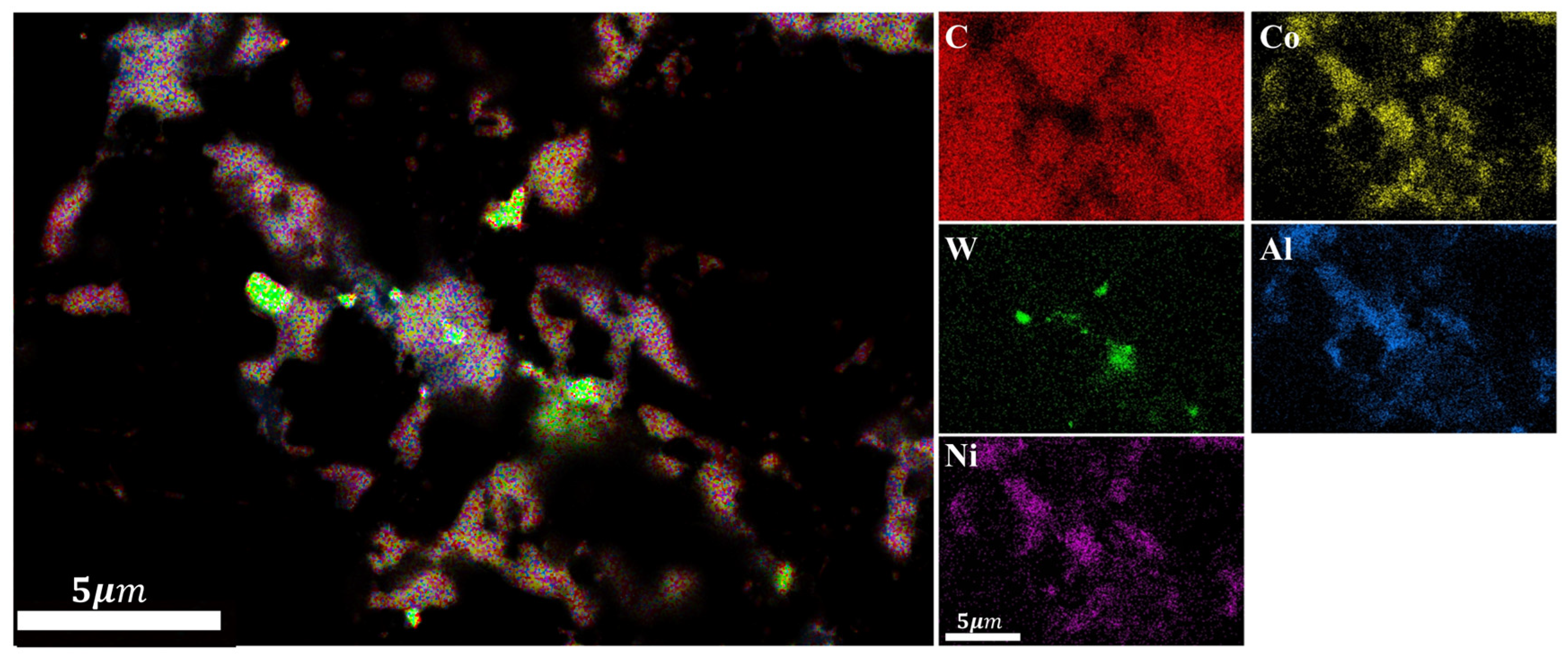
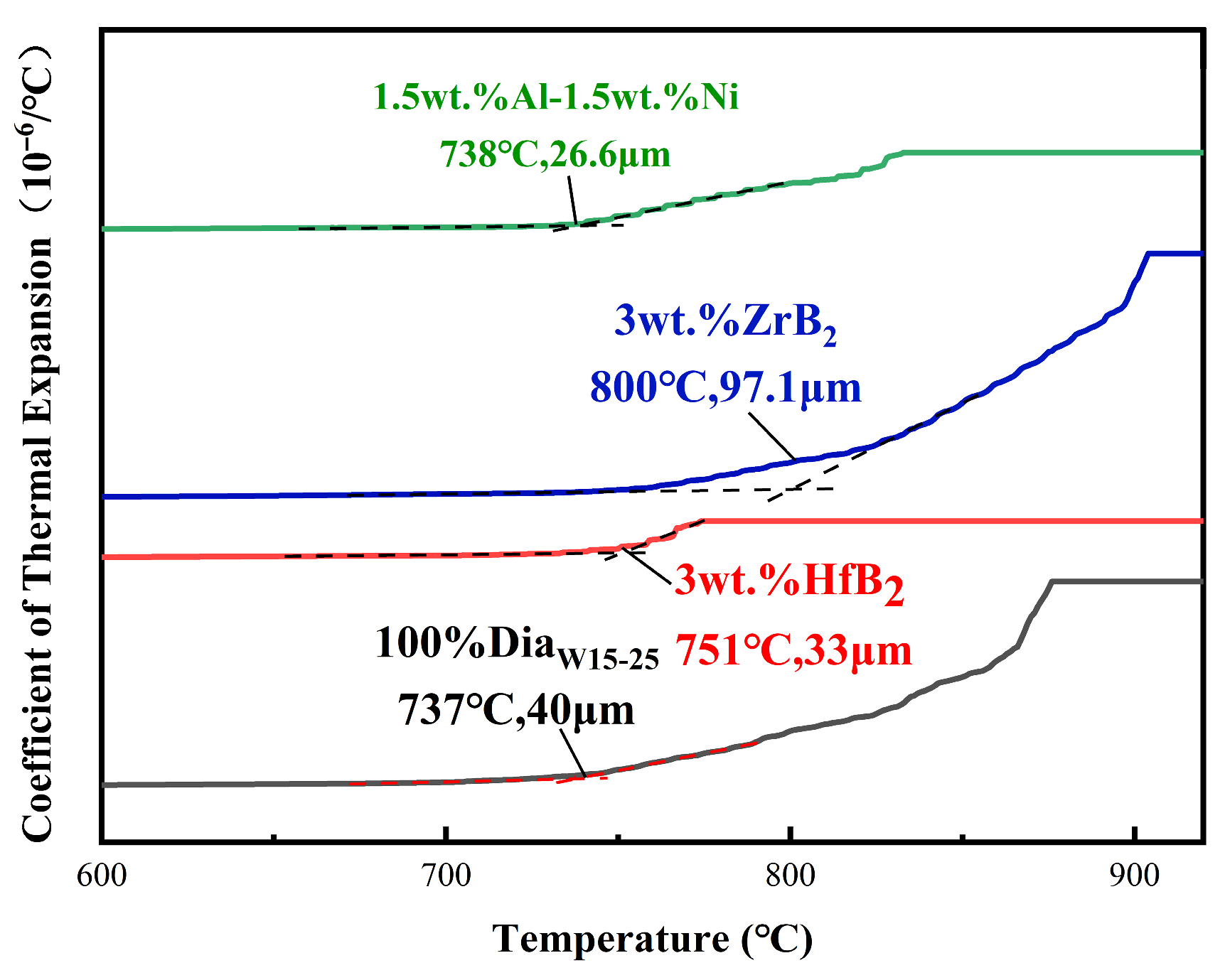

| Experimental Group | Type of Additive | Additive Content | Additive Particle Size |
|---|---|---|---|
| 100wt.%DiaW15-25 | / | 0 | / |
| 3wt.%HfB2-97wt.%DiaW15-25 | HfB2 | 3wt.% | 2 μm |
| 3wt.%ZrB2-97wt.%DiaW15-25 | ZrB2 | 3wt.% | 1 μm |
| 1.5wt.%Al-1.5wt.%Ni-97wt.%DiaW15-25 | Al | 1.5wt.% | 1 μm |
| Ni | 1.5wt.% | 1 μm |
| Element Types | Surface Area |
|---|---|
| C | 90.66 |
| Co | 7.74 |
| W | 1.60 |
| Total Amount | 100 |
| Element Types | Surface Area | Special Area |
|---|---|---|
| C | 89.64 | 81.05 |
| Co | 7.17 | 9.00 |
| W | 1.47 | 3.09 |
| Hf | 1.71 | 6.86 |
| Total Amount | 100 | 100 |
| Element Types | Surface Area | Special Area |
|---|---|---|
| C | 91.66 | 91.62 |
| Co | 5.86 | 4.39 |
| W | 1.02 | 0.92 |
| Zr | 1.46 | 2.08 |
| Total Amount | 100 | 100 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, C.; Dai, W.; Xu, S.; Liu, B. Study on Sintering Behavior, Heat and Wear Resistance of Refractory Metal Borides (HfB2, ZrB2) and Al-Ni Modified PDC. Materials 2025, 18, 5093. https://doi.org/10.3390/ma18225093
Zhao C, Dai W, Xu S, Liu B. Study on Sintering Behavior, Heat and Wear Resistance of Refractory Metal Borides (HfB2, ZrB2) and Al-Ni Modified PDC. Materials. 2025; 18(22):5093. https://doi.org/10.3390/ma18225093
Chicago/Turabian StyleZhao, Chuang, Wenhao Dai, Shaotao Xu, and Baochang Liu. 2025. "Study on Sintering Behavior, Heat and Wear Resistance of Refractory Metal Borides (HfB2, ZrB2) and Al-Ni Modified PDC" Materials 18, no. 22: 5093. https://doi.org/10.3390/ma18225093
APA StyleZhao, C., Dai, W., Xu, S., & Liu, B. (2025). Study on Sintering Behavior, Heat and Wear Resistance of Refractory Metal Borides (HfB2, ZrB2) and Al-Ni Modified PDC. Materials, 18(22), 5093. https://doi.org/10.3390/ma18225093









