Effect of AISI H13 Steel Substrate Nitriding on AlCrN, ZrN, TiSiN, and TiCrN Multilayer PVD Coatings Wear and Friction Behaviors at a Different Temperature Level
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation and Coating Process
2.2. Characterization of the Coatings
2.3. Tribometer Tests and Test Parameters
| Test Parameter | Ambient Temperature, 20 °C | Elevated Temperature, 700 °C |
|---|---|---|
| Load (N) | 5 | |
| Test duration (min.) | 125 | |
| Sliding distance (m) | 235 | 1099 |
| Temperature (°C) | 20 ± 2 | 700 ± 2 |
| Sliding speed (mm/s) | for 20 °C | 30 |
| for 700 °C | 150 | |
| Relative Humidity (%) | 36 | |
| Max contact pressure (GPa) | ||
| Coating A | 1.69 | |
| Coating A(N) | 1.65 | |
| Coating B | 1.80 | |
| Coating B(N) | 1.74 | |
| Coating C | 1.79 | |
| Coating C(N) | 1.75 | |
| Coating D | 1.67 | |
| Coating D(N) | 1.73 | |
| Shear stress (GPa) | ||
| Coating A | 0.56 | |
| Coating A(N) | 0.55 | |
| Coating B | 0.58 | |
| Coating B(N) | 0.56 | |
| Coating C | 0.59 | |
| Coating C(N) | 0.57 | |
| Coating D | 0.54 | |
| Coating D(N) | 0.56 | |
3. Results
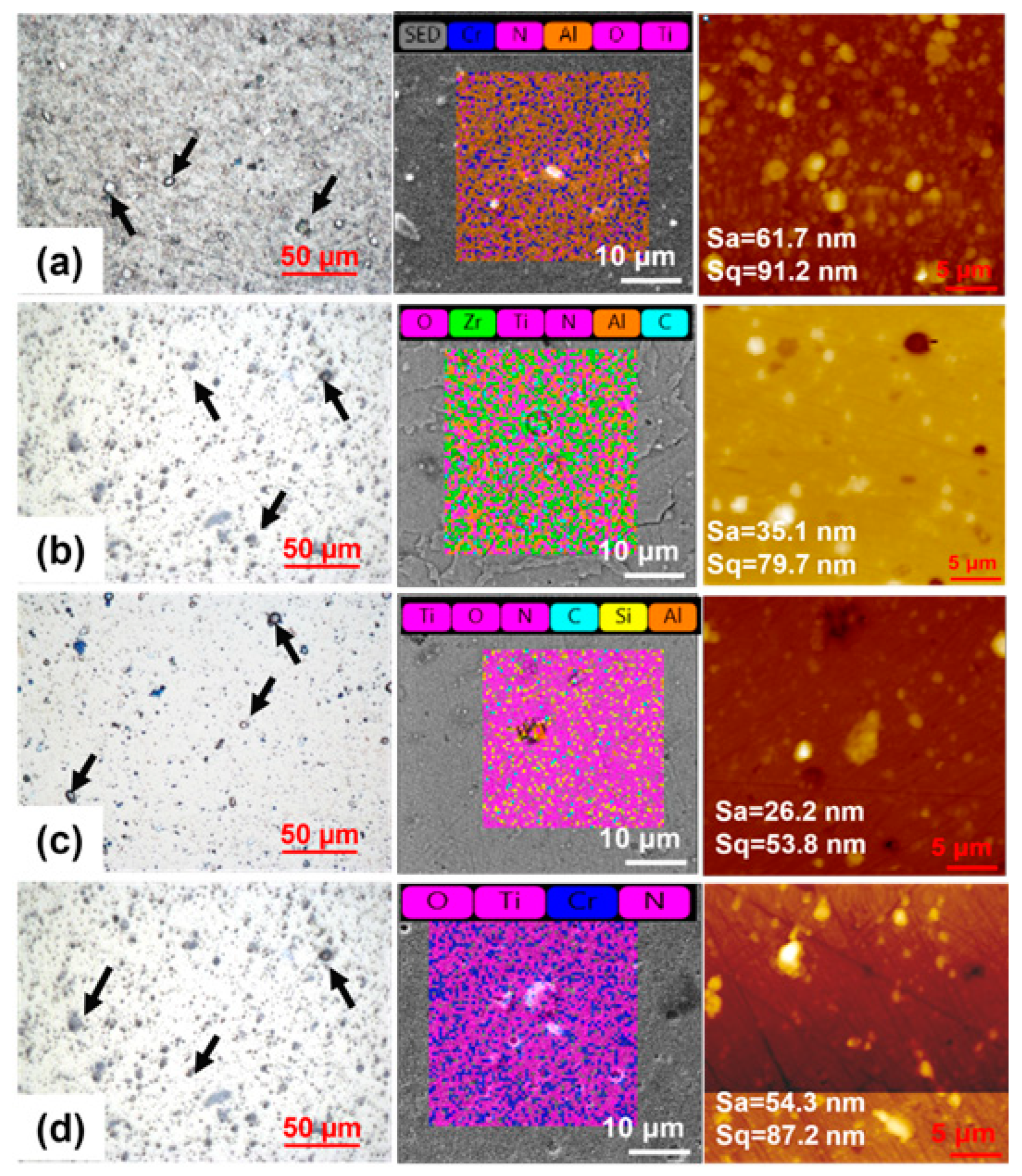
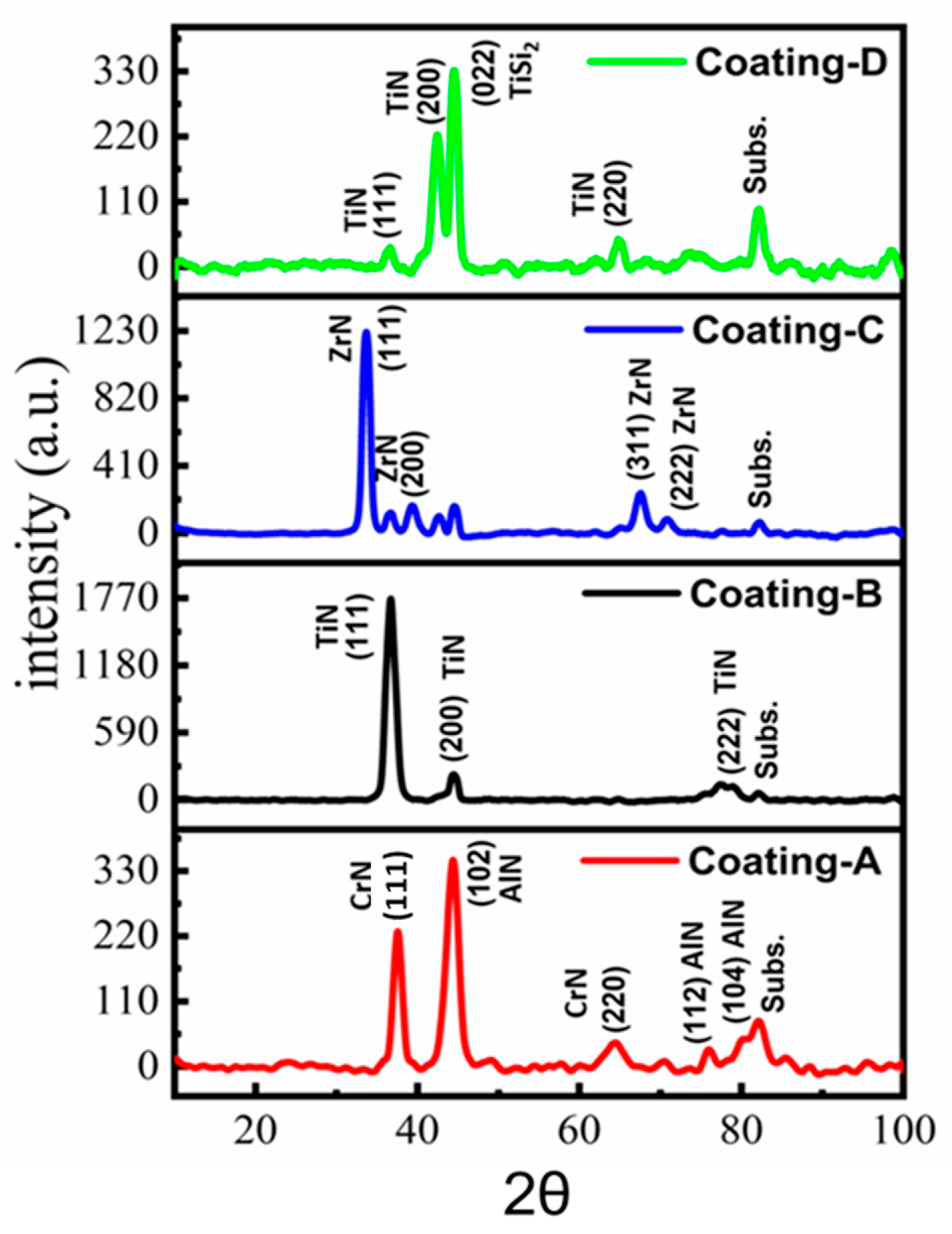
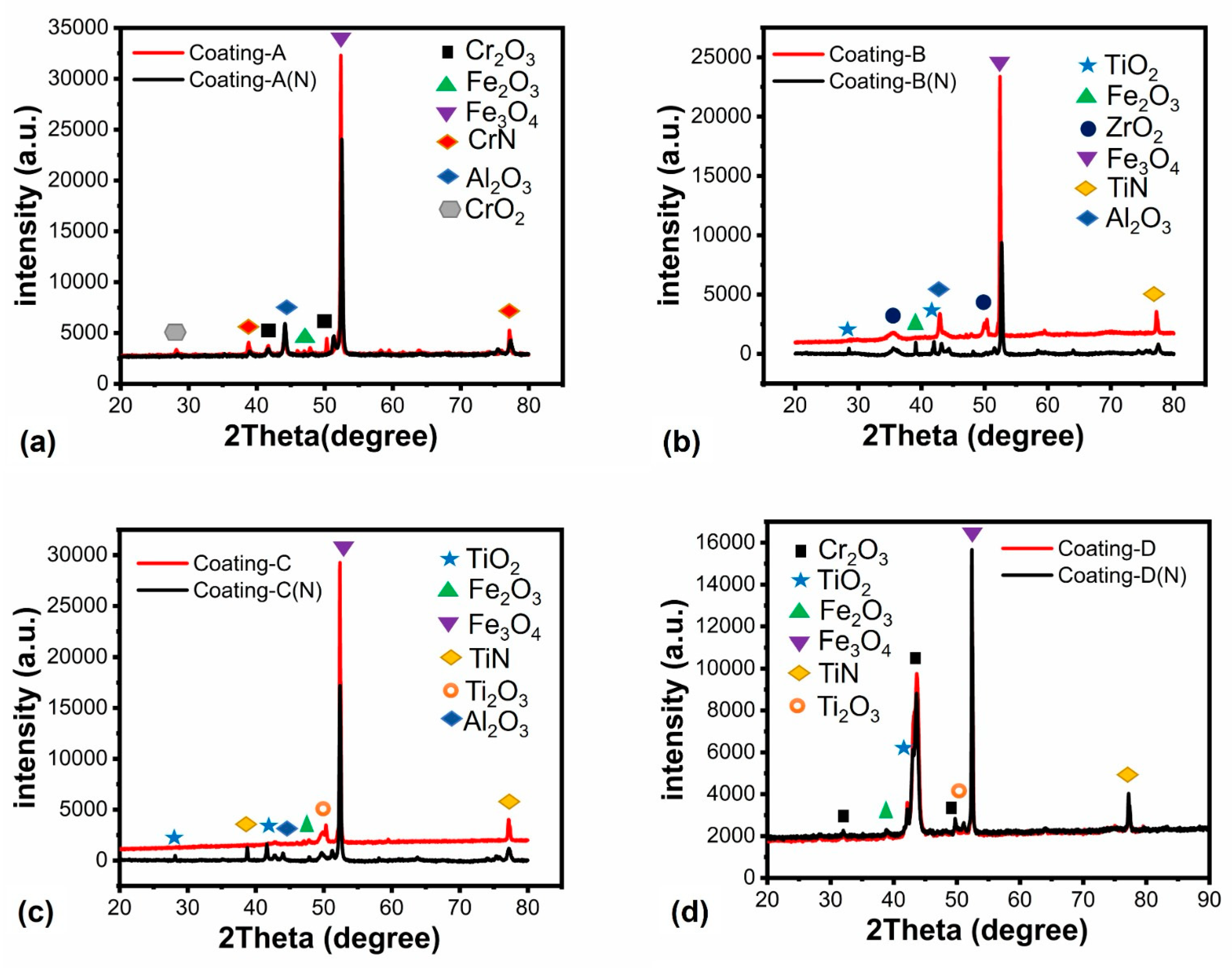
3.1. Morphological Characterization and Mechanical Properties
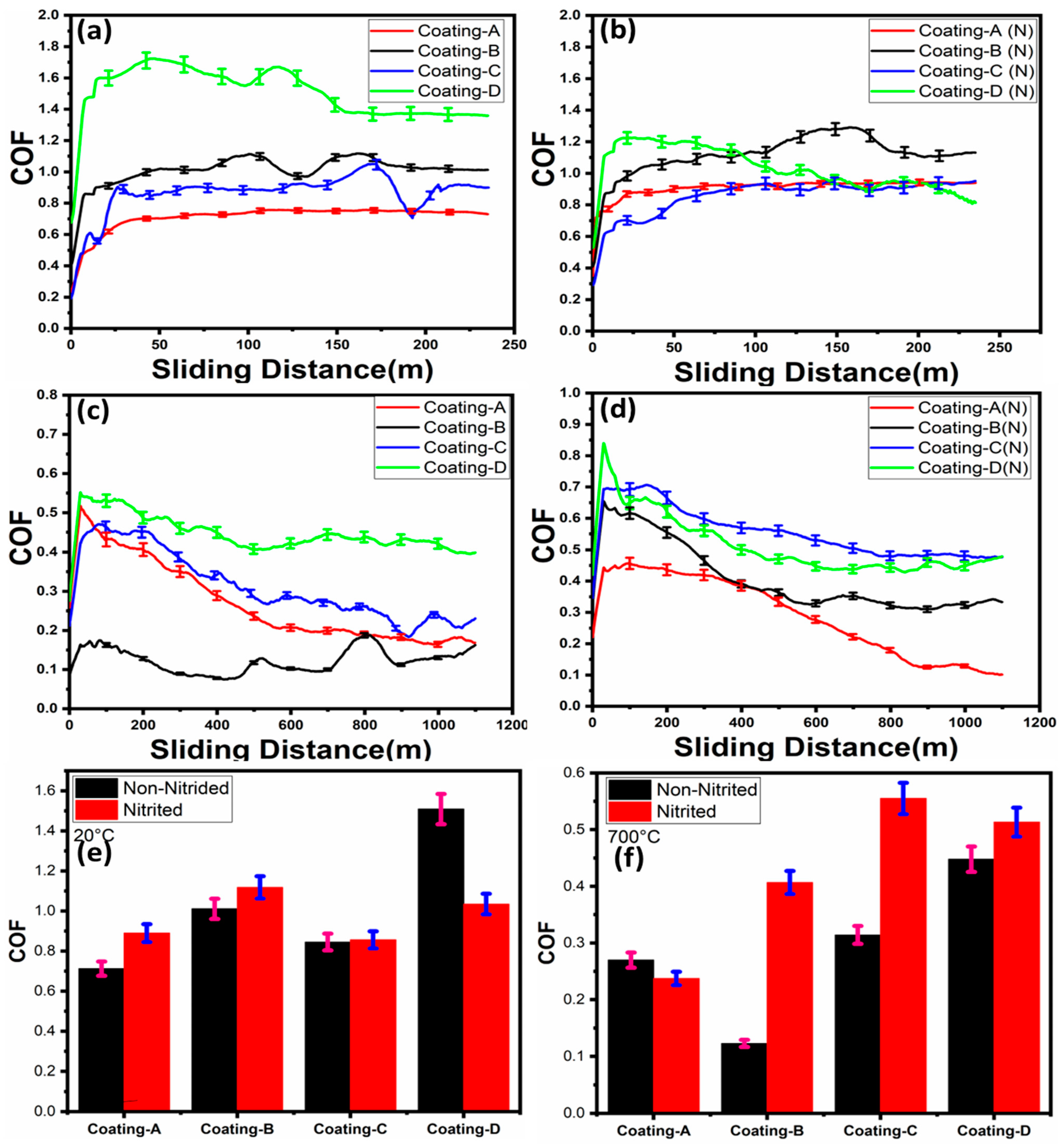
3.2. Friction and Wear Properties
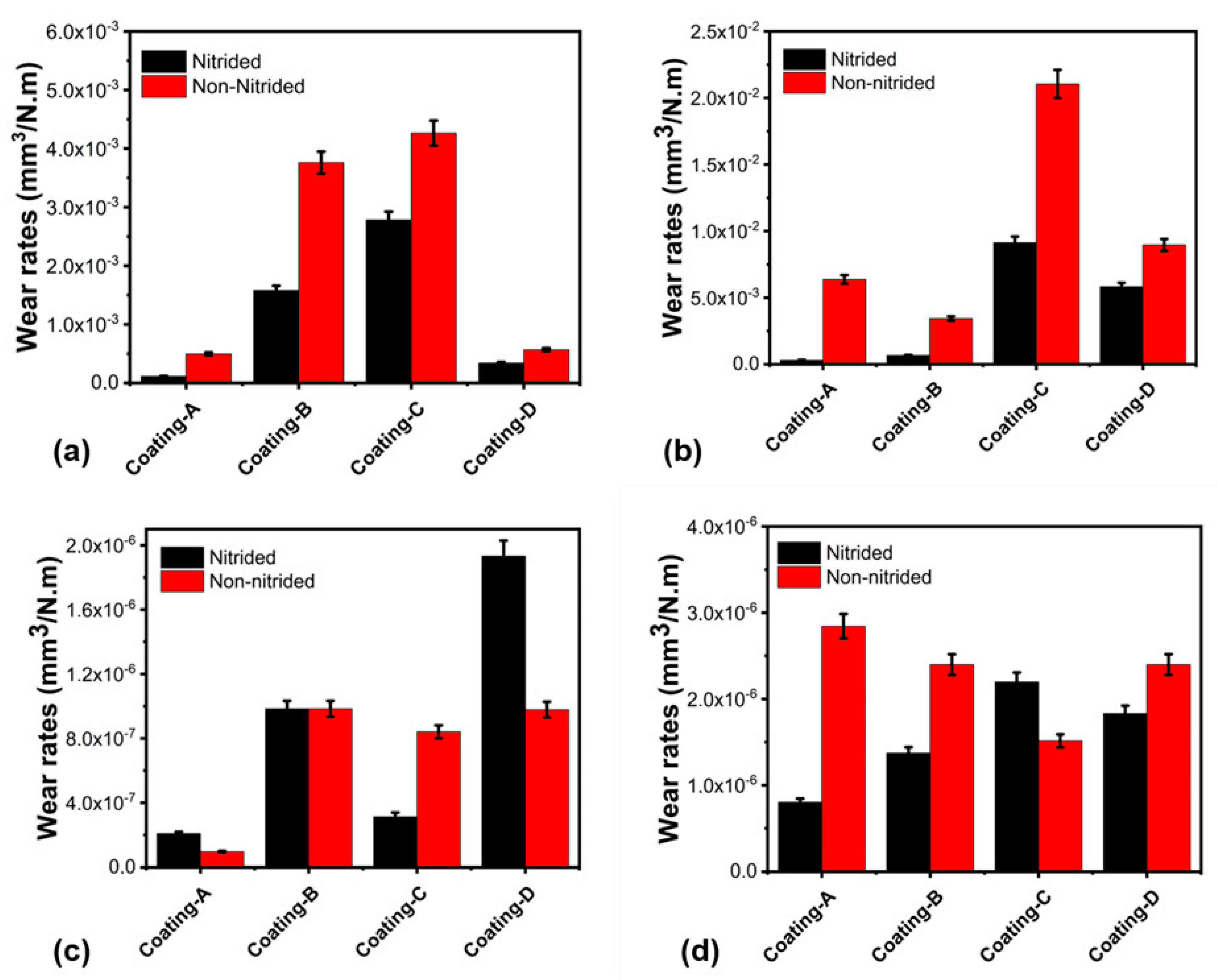
3.3. Wear Rates of the Coatings
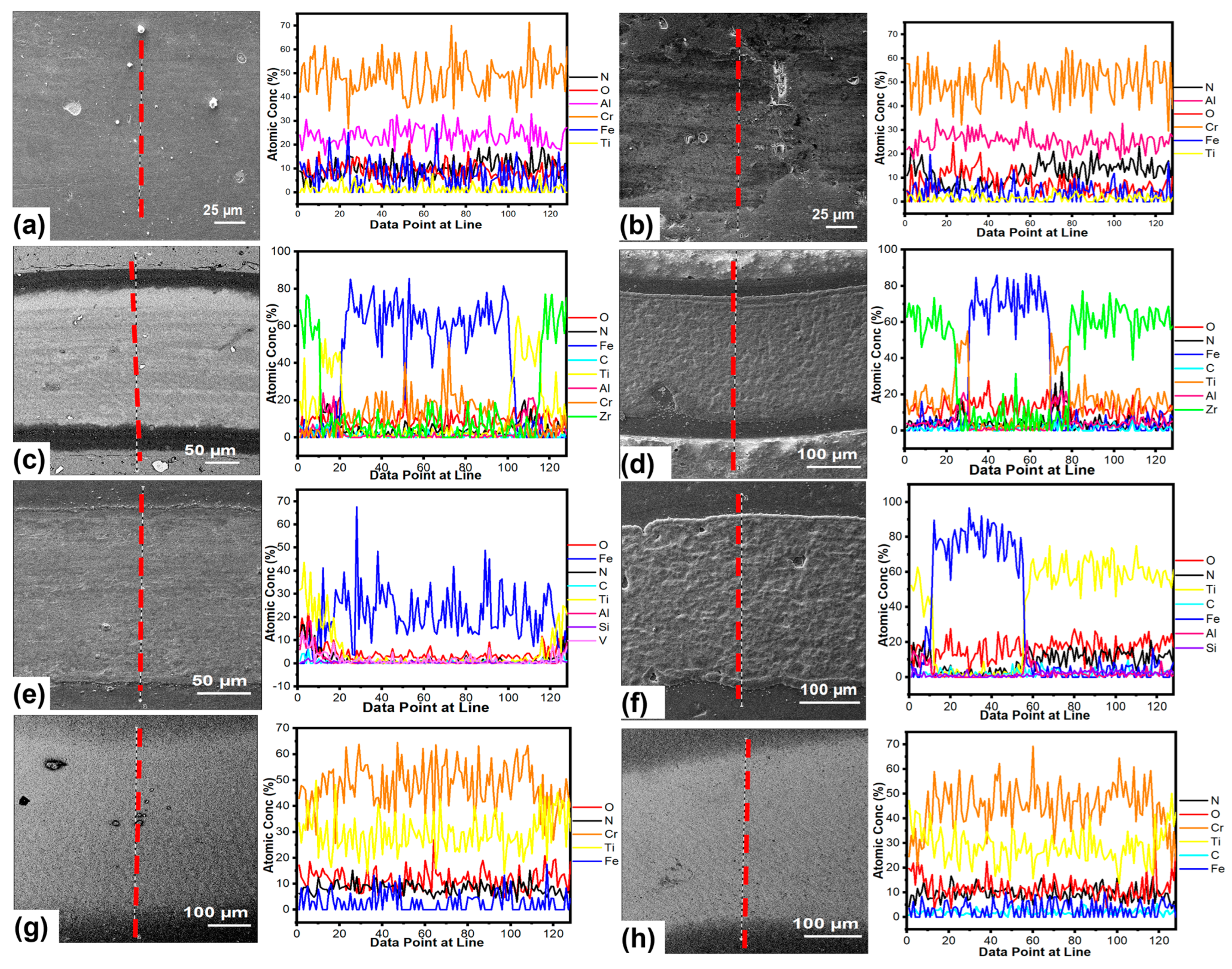
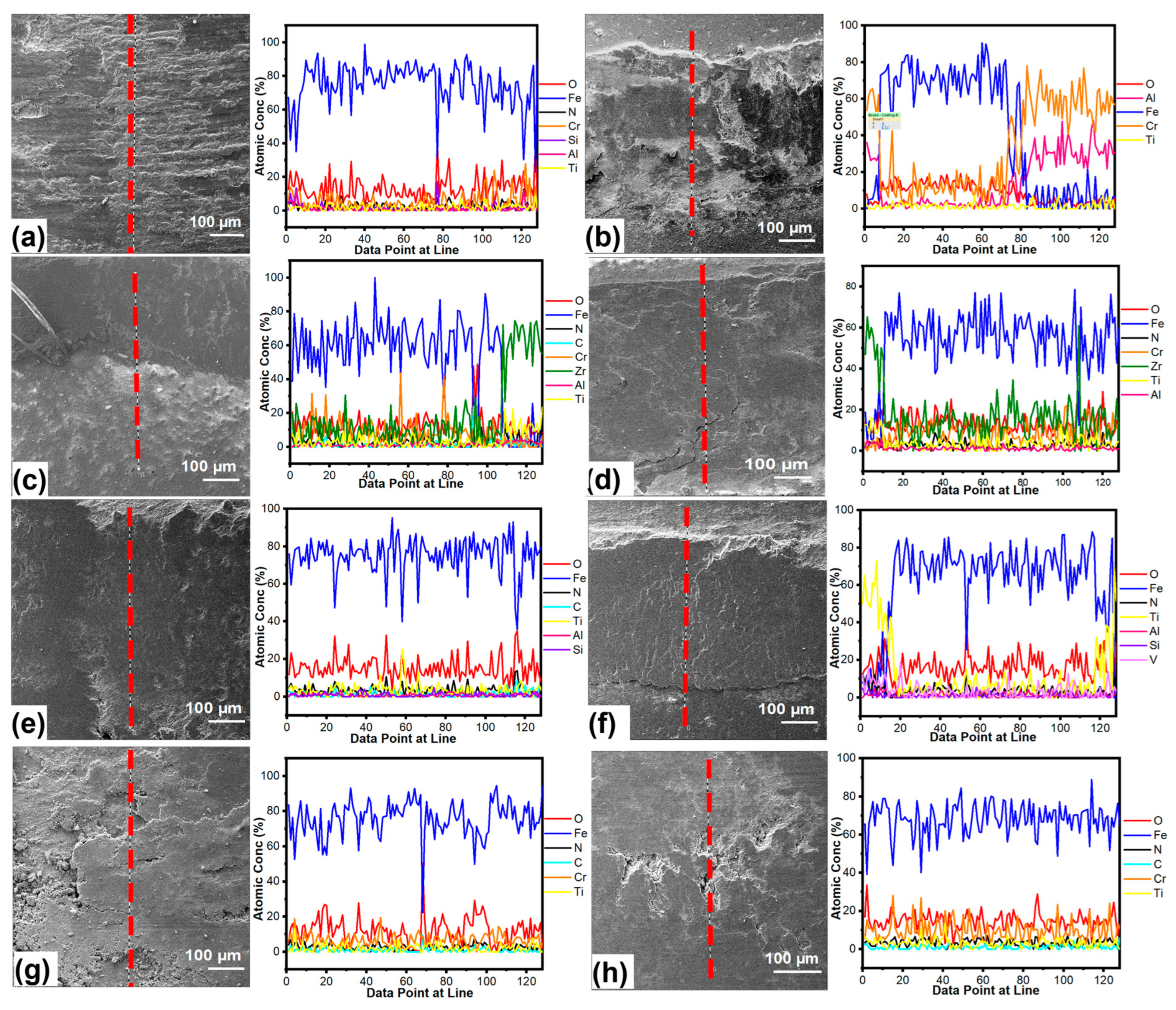
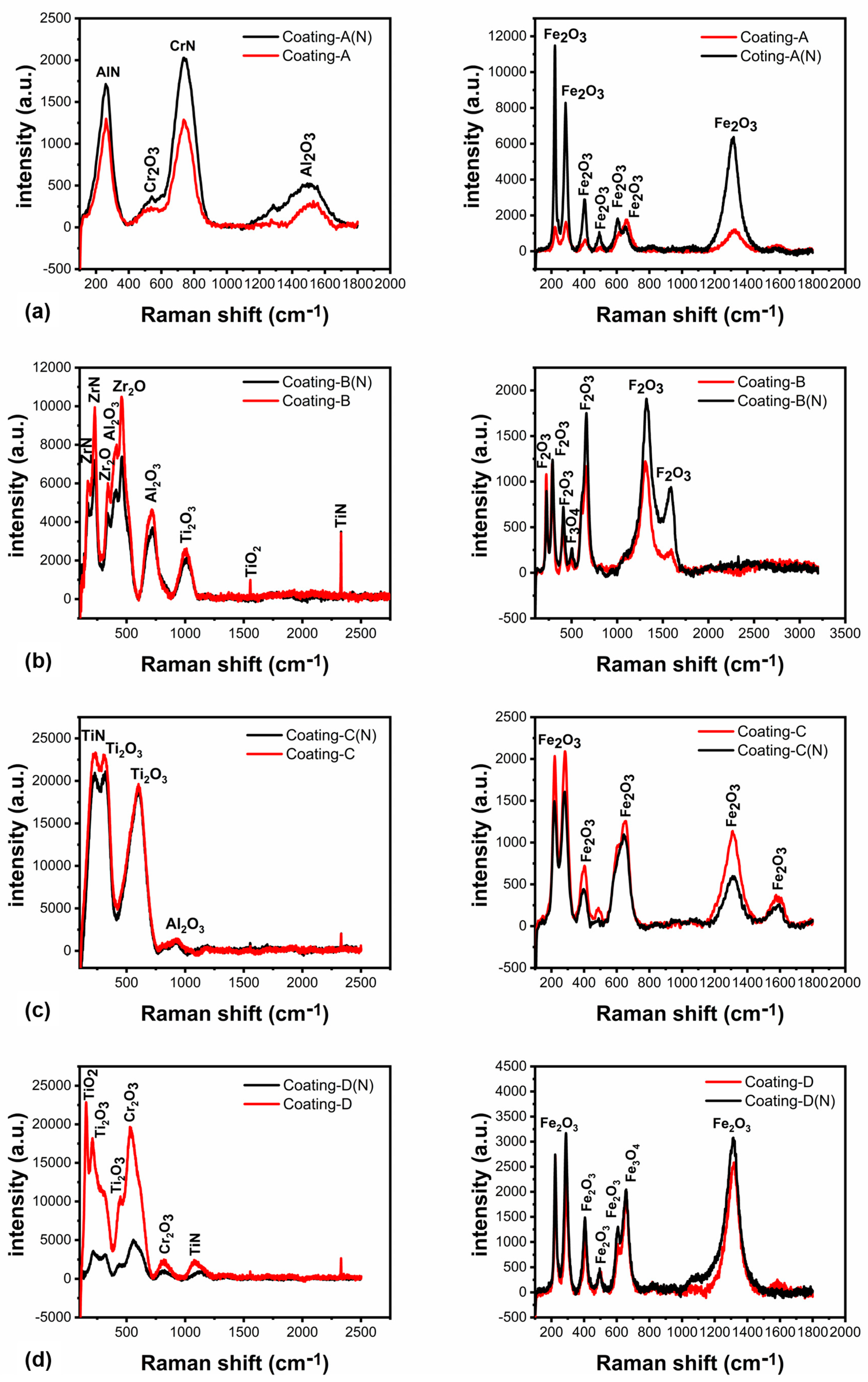
3.4. Chemical Analysis of the Worn Surfaces
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AFM | Atomic force microscope |
| COF | Coefficient of friction |
| EDX | Energy dispersive X-ray |
| PID | Proportional integral derivative |
| PVD | Physical vapor deposition |
| SEM | Scanning electron microscope |
| SPIP | Scanning probe image processor |
| XRD | X-ray diffraction |
| Nomenclature | |
| R | Diameter of the ball (mm) |
| R | Radius of the ball (mm) |
| d | Worn area diameter of the ball (mm) |
| rwt | Wear track radius (mm) |
| wwt | Wear track width (mm) |
| Vb | Volume losses of ball (mm3) |
| Vd | Volume losses of disk (mm3) |
| WR | Wear rate (mm3/N m) |
| H | Hardness (GPa) |
| E | Elastic modulus (GPa) |
| E′ | Effective elastic moduli (GPa) |
| F | Normal load (N) |
| Pmaks | Maximum contact pressure (GPa) |
| Ra | Arithmetic mean roughness of coatings (nm) |
| Sa | Surface average roughness (nm) |
| S | Sliding distance (m) |
| U | Sliding speed (m/s) |
| μm | Length (μm) |
| D | The crystallite sizes (nm) |
| K | Sherrer constant |
| β | Full-width at half maximum of XRD peaks |
| λ | Wavelength of the X-ray source (nm) |
| θ | Angle of diffraction peak (radian) |
| ε | Lattice strain |
| H3/E2 | Modulation ratio |
References
- Mazur, M.; Leary, M.; McMillan, M.; Elambasseril, J.; Brandt, M. SLM Additive Manufacture of H13 Tool Steel with Conformal Cooling and Structural Lattices. Rapid Prototyp. J. 2016, 22, 504–518. [Google Scholar] [CrossRef]
- Barrau, O.; Boher, C.; Gras, R.; Rezai-Aria, F. Analysis of the Friction and Wear Behaviour of Hot Work Tool Steel for Forging. Wear 2003, 255, 1444–1454. [Google Scholar] [CrossRef]
- Rodríguez-Baracaldo, R.; Benito, J.A.; Puchi-Cabrera, E.S.; Staia, M.H. High Temperature Wear Resistance of (TiAl)N PVD Coating on Untreated and Gas Nitrided AISI H13 Steel with Different Heat Treatments. Wear 2007, 262, 380–389. [Google Scholar] [CrossRef]
- Özkan, D.; Alper Yılmaz, M.; Szala, M.; Türküz, C.; Chocyk, D.; Tunç, C.; Göz, O.; Walczak, M.; Pasierbiewicz, K.; Barış Yağcı, M. Effects of Ceramic-Based CrN, TiN, and AlCrN Interlayers on Wear and Friction Behaviors of AlTiSiN+TiSiN PVD Coatings. Ceram. Int. 2021, 47, 20077–20089. [Google Scholar] [CrossRef]
- Tekmen, C.; Toparli, M.; Ozdemir, I.; Kusoglu, I.M.; Onel, K. High Temperature Behaviour of H13 Steel. Int. J. Mater. Res. 2005, 96, 1431–1433. [Google Scholar] [CrossRef]
- Chim, Y.C.; Ding, X.Z.; Zeng, X.T.; Zhang, S. Oxidation Resistance of TiN, CrN, TiAlN and CrAlN Coatings Deposited by Lateral Rotating Cathode Arc. Thin Solid Films 2009, 517, 4845–4849. [Google Scholar] [CrossRef]
- Grzesik, W.; Małecka, J. The Oxidation Behaviour and Notch Wear Formation of TiAlN Coated Tools Using Different Oxidation Techniques. Materials 2021, 14, 1330. [Google Scholar] [CrossRef]
- Perez-Mariano, J.; Lau, K.-H.; Sanjurjo, A.; Caro, J.; Casellas, D.; Colominas, C. TiSiN Nanocomposite Coatings by Chemical Vapor Deposition in a Fluidized Bed Reactor at Atmospheric Pressure (AP/FBR-CVD). Surf. Coat. Technol. 2006, 201, 2217–2225. [Google Scholar] [CrossRef]
- Altaş, E.; Erdogan, A.; Koçyiğit, F. A Comparative Study on the High Temperature Dry Sliding Wear Behavior of TiN and AlTiN/TiSiN Coatings Fabricated by PVD Technique. Ind. Lubr. Tribol. 2019, 71, 861–868. [Google Scholar] [CrossRef]
- Chang, Y.-Y.; Amrutwar, S. Effect of Plasma Nitriding Pretreatment on the Mechanical Properties of AlCrSiN-Coated Tool Steels. Materials 2019, 12, 795. [Google Scholar] [CrossRef]
- Birol, Y. Sliding Wear of CrN, AlCrN and AlTiN Coated AISI H13 Hot Work Tool Steels in Aluminium Extrusion. Tribol. Int. 2013, 57, 101–106. [Google Scholar] [CrossRef]
- Sue, J.A.; Chang, T.P. Friction and Wear Behavior of Titanium Nitride, Zirconium Nitride and Chromium Nitride Coatings at Elevated Temperatures. Surf. Coat. Technol. 1995, 76–77, 61–69. [Google Scholar] [CrossRef]
- López, G.; Staia, M.H. High-Temperature Tribological Characterization of Zirconium Nitride Coatings. Surf. Coat. Technol. 2005, 200, 2092–2099. [Google Scholar] [CrossRef]
- Wu, Z.T.; Sun, P.; Qi, Z.B.; Wei, B.B.; Wang, Z.C. High Temperature Oxidation Behavior and Wear Resistance of Ti0.53Al0.47N Coating by Cathodic Arc Evaporation. Vacuum 2017, 135, 34–43. [Google Scholar] [CrossRef]
- Xia, B.; Zhou, S.; Wang, Y.; Chen, H.; Zhang, J.; Qi, B. Multilayer Architecture Design to Enhance Load-Bearing Capacity and Tribological Behavior of CrAlN Coatings in Seawater. Ceram. Int. 2021, 47, 27430–27440. [Google Scholar] [CrossRef]
- Chang, Y.-Y.; Chang, B.-Y.; Chen, C.-S. Effect of CrN Addition on the Mechanical and Tribological Performances of Multilayered AlTiN/CrN/ZrN Hard Coatings. Surf. Coat. Technol. 2022, 433, 128107. [Google Scholar] [CrossRef]
- Qi, Z.B.; Wu, Z.T.; Liang, H.F.; Zhang, D.F.; Wang, J.H.; Wang, Z.C. In Situ and Ex Situ Studies of Microstructure Evolution during High-Temperature Oxidation of ZrN Hard Coating. Scr. Mater. 2015, 97, 9–12. [Google Scholar] [CrossRef]
- Chen, L.; Holec, D.; Du, Y.; Mayrhofer, P.H. Influence of Zr on Structure, Mechanical and Thermal Properties of Ti–Al–N. Thin Solid Films 2011, 519, 5503–5510. [Google Scholar] [CrossRef]
- Sousa, V.F.C.; Silva, F.J.G.; Alexandre, R.; Fecheira, J.S.; Silva, F.P.N. Study of the Wear Behaviour of TiAlSiN and TiAlN PVD Coated Tools on Milling Operations of Pre-Hardened Tool Steel. Wear 2021, 476, 203695. [Google Scholar] [CrossRef]
- Ezazi, M.A.; Quazi, M.M.; Zalnezhad, E.; Sarhan, A.A.D. Enhancing the Tribo-Mechanical Properties of Aerospace AL7075-T6 by Magnetron-Sputtered Ti/TiN, Cr/CrN & TiCr/TiCrN Thin Film Ceramic Coatings. Ceram. Int. 2014, 10 Pt A, 15603–15615. [Google Scholar] [CrossRef]
- ASTM G99-Standard Test Method for Wear Testing with a Pin-on-Disk Apparatus; ASTM International: West Conshohocken, PA, USA, 2017.
- Nazir, M.H.; Khan, Z.A.; Saeed, A.; Bakolas, V.; Braun, W.; Bajwa, R. Experimental Analysis and Modelling for Reciprocating Wear Behaviour of Nanocomposite Coatings. Wear 2018, 416–417, 89–102. [Google Scholar] [CrossRef]
- Wei, M.X.; Wang, S.Q.; Wang, L.; Cui, X.H.; Chen, K.M. Effect of Tempering Conditions on Wear Resistance in Various Wear Mechanisms of H13 Steel. Tribol. Int. 2011, 44, 898–905. [Google Scholar] [CrossRef]
- Li, S.; Wu, X.; Chen, S.; Li, J. Wear Resistance of H13 and a New Hot-Work Die Steel at High Temperature. J. Mater. Eng. Perform. 2016, 25, 2993–3006. [Google Scholar] [CrossRef]
- Bokuniaeva, A.O.; Vorokh, A.S. Estimation of Particle Size Using the Debye Equation and the Scherrer Formula for Polyphasic TiO2 Powder. J. Phys. Conf. Ser. 2019, 1410, 012057. [Google Scholar] [CrossRef]
- Sampath Kumar, T.; Balasivanandha Prabu, S.; Manivasagam, G. Metallurgical Characteristics of TiAlN/AlCrN Coating Synthesized by the PVD Process on a Cutting Insert. J. Mater. Eng. Perform. 2014, 23, 2877–2884. [Google Scholar] [CrossRef]
- Drozd, K.; Walczak, M.; Szala, M.; Gancarczyk, K. Tribological Behavior of AlCrSiN-Coated Tool Steel K340 Versus Popular Tool Steel Grades. Materials 2020, 13, 4895. [Google Scholar] [CrossRef]
- Popović, M.; Novaković, M.; Bibić, N. Annealing Effects on the Properties of TiN Thin Films. Process. Appl. Ceram. 2015, 9, 67–71. [Google Scholar] [CrossRef]
- Khojier, K.; Savaloni, H.; Shokrai, E.; Dehghani, Z.; Dehnavi, N.Z. Influence of Argon Gas Flow on Mechanical and Electrical Properties of Sputtered Titanium Nitride Thin Films. J. Theor. Appl. Phys. 2013, 7, 37. [Google Scholar] [CrossRef]
- Roman, D.; Bernardi, J.; Amorim, C.L.G.D.; de Souza, F.S.; Spinelli, A.; Giacomelli, C.; Figueroa, C.A.; Baumvol, I.J.R.; Basso, R.L.O. Effect of Deposition Temperature on Microstructure and Corrosion Resistance of ZrN Thin Films Deposited by DC Reactive Magnetron Sputtering. Mater. Chem. Phys. 2011, 130, 147–153. [Google Scholar] [CrossRef]
- Stallard, J.; Poulat, S.; Teer, D.G. The Study of the Adhesion of a TiN Coating on Steel and Titanium Alloy Substrates Using a Multi-Mode Scratch Tester. Tribol. Int. 2006, 39, 159–166. [Google Scholar] [CrossRef]
- Goldbaum, D.; Manimuda, P.; Kamath, G.; Descartes, S.; Klemberg-Sapieha, J.E.; Chromik, R.R. Tribological Behavior of TiN and Ti (Si,C)N Coatings on Cold Sprayed Ti Substrates. Surf. Coat. Technol. 2016, 291, 264–275. [Google Scholar] [CrossRef]
- Walczak, M.; Pasierbiewicz, K.; Szala, M. Effect of Ti6Al4V Substrate Manufacturing Technology on the Properties of PVD Nitride Coatings. Acta Phys. Pol. A 2022, 142, 723. [Google Scholar] [CrossRef]
- Walter, C.; Antretter, T.; Daniel, R.; Mitterer, C. Finite Element Simulation of the Effect of Surface Roughness on Nanoindentation of Thin Films with Spherical Indenters. Surf. Coat. Technol. 2007, 202, 1103–1107. [Google Scholar] [CrossRef]
- Chen, X.; Du, Y.; Chung, Y.-W. Commentary on Using H/E and H3/E2 as Proxies for Fracture Toughness of Hard Coatings. Thin Solid Films 2019, 688, 137265. [Google Scholar] [CrossRef]
- ISO 26443:2008; Rockwell Indentation Test for Evaluation of Adhesion of Ceramic Coatings, Fine Ceramics (Advanced Ceramics, Advanced Technical Ceramics). ISO: Geneva, Switzerland, 2008.
- VDI 3198; Verein-Deutscher-Ingenieure, Daimler-Benz Adhesion Test. VDI-Verlag: Dusseldorf, Duitsland, 1992; p. 7.
- Broitman, E.; Hultman, L. Adhesion Improvement of Carbon-Based Coatings through a High Ionization Deposition Technique. J. Phys. Conf. Ser. 2012, 370, 012009. [Google Scholar] [CrossRef]
- Beake, B.D.; Vishnyakov, V.M.; Harris, A.J. Relationship between Mechanical Properties of Thin Nitride-Based Films and Their Behaviour in Nano-Scratch Tests. Tribol. Int. 2011, 44, 468–475. [Google Scholar] [CrossRef]
- Walczak, M.; Pasierbiewicz, K.; Szala, M. Adhesion and Mechanical Properties of TiAlN and AlTiN Magnetron Sputtered Coatings Deposited on the DMSL Titanium Alloy Substrate. Acta Phys. Pol. A 2019, 136, 294–298. [Google Scholar] [CrossRef]
- Chen, Z.; Zhou, K.; Lu, X.; Lam, Y.C. A Review on the Mechanical Methods for Evaluating Coating Adhesion. Acta Mech. 2014, 225, 431–452. [Google Scholar] [CrossRef]
- Hardell, J.; Hernandez, S.; Mozgovoy, S.; Pelcastre, L.; Courbon, C.; Prakash, B. Effect of Oxide Layers and near Surface Transformations on Friction and Wear during Tool Steel and Boron Steel Interaction at High Temperatures. Wear 2015, 330–331, 223–229. [Google Scholar] [CrossRef]
- Shioga, P.H.T.; Binder, C.; Hammes, G.; Klein, A.N.; Mello, J.D.B.D. Effects of Different Plasma Nitrided Layers on the Tribological Performance of DLC Coatings. Mat. Res. 2016, 19, 1180–1188. [Google Scholar] [CrossRef]
- Ge, P.L.; Bao, M.D.; Zhang, H.J.; You, K.; Liu, X.P. Effect of Plasma Nitriding on Adhesion Strength of CrTiAlN Coatings on H13 Steels by Closed Field Unbalanced Magnetron Sputter Ion Plating. Surf. Coat. Technol. 2013, 229, 146–150. [Google Scholar] [CrossRef]
- Pizzi, A.; Mittal, K. Handbook Af Adhesive Technology; CRC Press: Boca Raton, FL, USA, 2017. [Google Scholar]
- Guo, J.-Y.; Kuo, Y.-L.; Wang, H.-P. A Facile Nitriding Approach for Improved Impact Wear of Martensitic Cold-Work Steel Using H2/N2 Mixture Gas in an AC Pulsed Atmospheric Plasma Jet. Coatings 2021, 11, 1119. [Google Scholar] [CrossRef]
- Ovsyannikov, S.V.; Wu, X.; Shchennikov, V.V.; Karkin, A.E.; Dubrovinskaia, N.; Garbarino, G.; Dubrovinsky, L. Structural Stability of a Golden Semiconducting Orthorhombic Polymorph of Ti2O3 under High Pressures and High Temperatures. J. Phys. Condens. Matter 2010, 22, 375402. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.B.; Kim, M.H.; Lee, Y.C.; Kwon, S.C. High Temperature Oxidation of TiCrN Coatings Deposited on a Steel Substrate by Ion Plating. Surf. Coat. Technol. 2001, 141, 232–239. [Google Scholar] [CrossRef]
- Surmacki, J.; Wroński, P.; Szadkowska-Nicze, M.; Abramczyk, H. Raman Spectroscopy of Visible-Light Photocatalyst–Nitrogen-Doped Titanium Dioxide Generated by Irradiation with Electron Beam. Chem. Phys. Lett. 2013, 566, 54–59. [Google Scholar] [CrossRef]
- Han, R.; Li, W.; Pan, W.; Zhu, M.; Zhou, D.; Li, F. 1D Magnetic Materials of Fe3O4 and Fe with High Performance of Microwave Absorption Fabricated by Electrospinning Method. Sci. Rep. 2014, 4, 7493. [Google Scholar] [CrossRef] [PubMed]
- Mansour, H.; Letifi, H.; Bargougui, R.; De Almeida-Didry, S.; Negulescu, B.; Autret-Lambert, C.; Gadri, A.; Ammar, S. Structural, Optical, Magnetic and Electrical Properties of Hematite (α-Fe2O3) Nanoparticles Synthesized by Two Methods: Polyol and Precipitation. Appl. Phys. A 2017, 123, 787. [Google Scholar] [CrossRef]
- Abdullah, M.M.; Rajab, F.M.; Al-Abbas, S.M. Structural and Optical Characterization of Cr2O3 Nanostructures: Evaluation of Its Dielectric Properties. AIP Adv. 2014, 4, 027121. [Google Scholar] [CrossRef]
- Merkert, P.; Hoffman, M.; Rödel, J. Detection of Prefracture Microcracking in Al2O3 by Acoustic Emission. J. Eur. Ceram. Soc. 1998, 18, 1645–1654. [Google Scholar] [CrossRef]
- Liu, Y.; Cheng, B.; Wang, K.-K.; Ling, G.-P.; Cai, J.; Song, C.-L.; Han, G.-R. Study of Raman Spectra for γ-Al2O3 Models by Using First-Principles Method. Solid State Commun. 2014, 178, 16–22. [Google Scholar] [CrossRef]
- Yan, D.; Zhao, H.; Pei, J.; Wang, X. Metal Ion-Mediated Structure and Properties of α-Fe2O3 Nanoparticles. Mater. Res. Bull. 2018, 101, 100–106. [Google Scholar] [CrossRef]
- Tao, S.; Yin, Z.; Zhou, X.; Ding, C. Sliding Wear Characteristics of Plasma-Sprayed Al2O3 and Cr2O3 Coatings against Copper Alloy under Severe Conditions. Tribol. Int. 2010, 43, 69–75. [Google Scholar] [CrossRef]
- Gomes, A.S.O.; Yaghini, N.; Martinelli, A.; Ahlberg, E. A Micro-Raman Spectroscopic Study of Cr(OH)3 and Cr2O3 Nanoparticles Obtained by the Hydrothermal Method. J. Raman Spectrosc. 2017, 48, 1256–1263. [Google Scholar] [CrossRef]
- Zamani, P.; Valefi, Z. Microstructure, Phase Composition and Mechanical Properties of Plasma Sprayed Al2O3, Cr2O3 and Cr2O3-Al2O3 Composite Coatings. Surf. Coat. Technol. 2017, 316, 138–145. [Google Scholar] [CrossRef]
- Testa-Anta, M.; Ramos-Docampo, M.A.; Comesaña-Hermo, M.; Rivas-Murias, B.; Salgueiriño, V. Raman Spectroscopy to Unravel the Magnetic Properties of Iron Oxide Nanocrystals for Bio-Related Applications. Nanoscale Adv. 2019, 1, 2086–2103. [Google Scholar] [CrossRef]
- Mo, J.L.; Zhu, M.H.; Leyland, A.; Matthews, A. Impact Wear and Abrasion Resistance of CrN, AlCrN and AlTiN PVD Coatings. Surf. Coat. Technol. 2013, 215, 170–177. [Google Scholar] [CrossRef]
- Kuo, C.-C.; Lin, Y.-T.; Chan, A.; Chang, J.-T. High Temperature Wear Behavior of Titanium Nitride Coating Deposited Using High Power Impulse Magnetron Sputtering. Coatings 2019, 9, 555. [Google Scholar] [CrossRef]
- Milošev, I.; Strehblow, H.-H.; Navinšek, B. XPS in the Study of High-Temperature Oxidation of CrN and TiN Hard Coatings. Surf. Coat. Technol. 1995, 74–75, 897–902. [Google Scholar] [CrossRef]
- Otani, Y.; Hofmann, S. High Temperature Oxidation Behaviour of (Ti1−xCrx)N Coatings. Thin Solid Films 1996, 287, 188–192. [Google Scholar] [CrossRef]
- Ahmad, F.; Zhang, L.; Zheng, J.; Sidra, I.; Zhang, S. Characterization of AlCrN and AlCrON Coatings Deposited on Plasma Nitrided AISI H13 Steels Using Ion-Source-Enhanced Arc Ion Plating. Coatings 2020, 10, 306. [Google Scholar] [CrossRef]
- Grützmacher, P.G.; Rammacher, S.; Rathmann, D.; Motz, C.; Mücklich, F.; Suarez, S. Interplay between Microstructural Evolution and Tribo-Chemistry during Dry Sliding of Metals. Friction 2019, 7, 637–650. [Google Scholar] [CrossRef]
- Stott, F.H.; Jiang, J.; Stack, M. Modelling the Beneficial Effects of Oxidation on the Sliding Wear of Metals and Alloys; Datta, P.K., Burnell-Gray, J.S., Eds.; GBR; Royal Society of Chemistry: London, UK, 1997; Volume 1, pp. 143–157. [Google Scholar]
- Kato, H.; Komai, K. Tribofilm Formation and Mild Wear by Tribo-Sintering of Nanometer-Sized Oxide Particles on Rubbing Steel Surfaces. Wear 2007, 262, 36–41. [Google Scholar] [CrossRef]
- Balerio, R.; Kim, H.; Morell-Pacheco, A.; Hawkins, L.; Shiau, C.-H.; Shao, L. ZrN Phase Formation, Hardening and Nitrogen Diffusion Kinetics in Plasma Nitrided Zircaloy-4. Materials 2021, 14, 3572. [Google Scholar] [CrossRef]
- Kundalkar, D.; Mavalankar, M.; Tewari, A. Effect of Gas Nitriding on the Thermal Fatigue Behavior of Martensitic Chromium Hot-Work Tool Steel. Mater. Sci. Eng. A 2016, 651, 391–398. [Google Scholar] [CrossRef]
- Le, N.M.; Schimpf, C.; Biermann, H.; Dalke, A. Effect of Nitriding Potential KN on the Formation and Growth of a “White Layer” on Iron Aluminide Alloy. Met. Mater. Trans. B 2021, 52, 414–424. [Google Scholar] [CrossRef]
- Hacısalihoğlu, İ.; Kaya, G.; Ergüder, T.O.; Mandev, E.; Manay, E.; Yıldız, F. Tribological and Thermal Properties of Plasma Nitrided Ti45Nb Alloy. Surf. Interfaces 2021, 22, 100893. [Google Scholar] [CrossRef]
- Mydłowska, K.; Ratajski, J.; Szparaga, Ł.; Myśliński, P.; Jędrzejewski, R.; Gilewicz, A. Analysis of Thermal Stability of CrCN Coatings Deposited on Nitrided Substrates Using Modulated Temperature Thermodilatometry. Arch. Metall. Mater. 2017, 62, 771–776. [Google Scholar] [CrossRef]

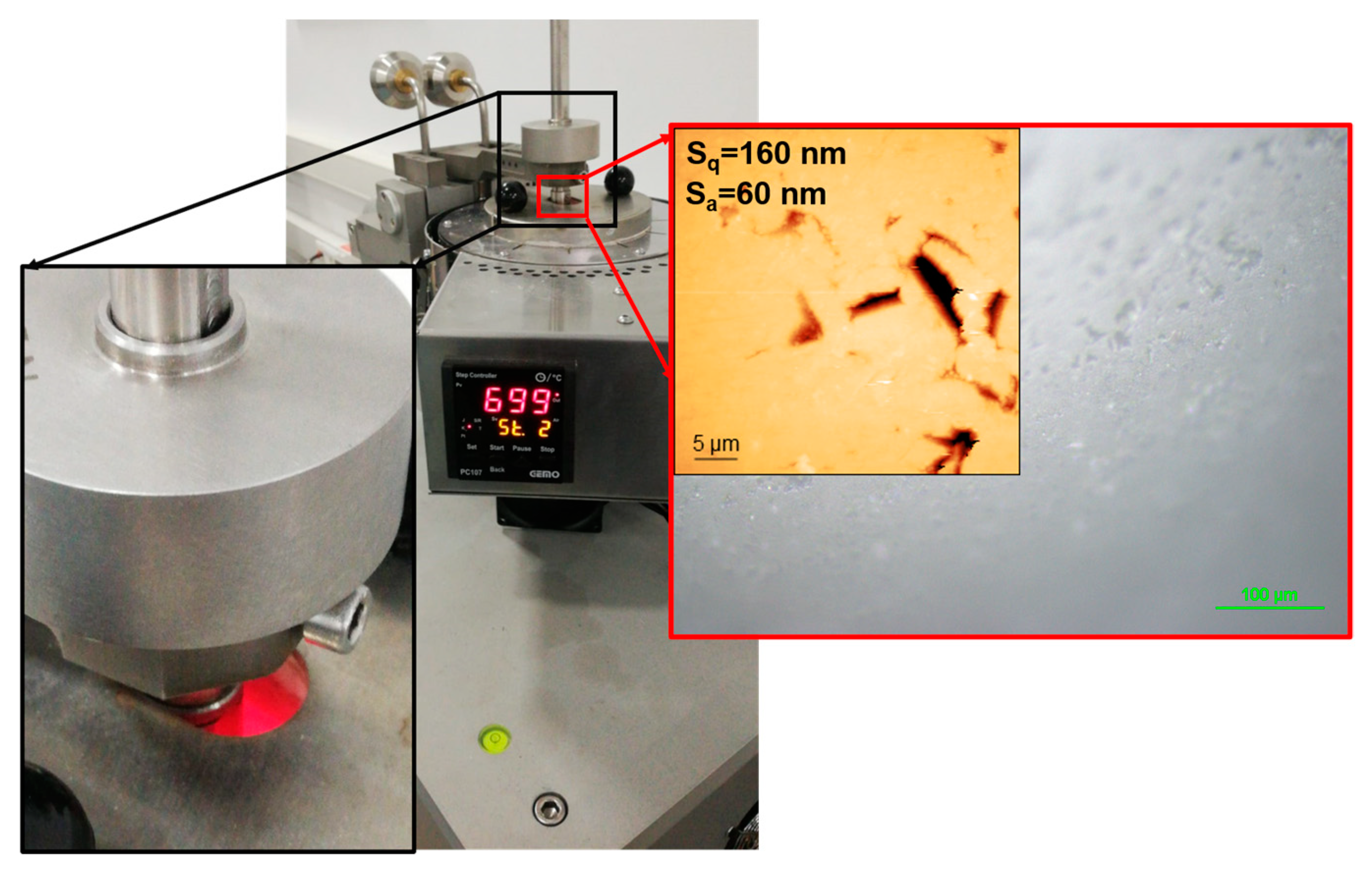




| Coating Array | Duration (Min.) | Coating | Cathode Type and Quantity | Cathode Current (A) | Bias Voltage (V) | Power (W) | Temperature (°C) | Gas Flow sccm | Nitrogen Partial Pressure (mbar) |
|---|---|---|---|---|---|---|---|---|---|
| Coating A | 15 min. | CrN (1 µm) | 2 Cr | 180 | 95 | 17,100 | 450 | 220 | 6 × 10−3 |
| 150 min. | AlCrN (0.6 µm) | 2 AlCr | 120 | 65 | 7800 | 450 | 450 | 3 × 10−2 | |
| Coating B | 45 min. | TiN (0.3 µm) | 1 Ti | 125 | 95 | 11,875 | 450 | 220 | 6 × 10−3 |
| 30 min | TiAlN (0.5 µm) | 1 Ti 1 AlTi | 120 Ti 120 AlTi | 65 | 7800 | 450 | 350 | 2.5 × 10−2 | |
| 25 min. | AlTiN (0.5 µm) | 2 AlTi | 150 | 65 | 14,250 | 450 | 400 | 2.5 × 10−2 | |
| 25 min. | AlTiZrN (1.5 µm) | 2 AlTi 1 Zr | 120 AlTi 180 Zr | 95 | 11,400 17,100 | 450 | 450 | 2.5 × 10−2 | |
| 30 min. | ZrN (0.8 µm) | 1 Zr | 200 | 120 | 2400 | 450 | 400 | 2.5 × 10−2 | |
| Coating C | 15 min. | TiN (0.3 µm) | 1 Ti | 125 | 95 | 11,875 | 450 | 220 | 6 × 10−3 |
| 40 min. | AlTiN (0.5 µm) | 2 AlTi | 150 | 65 | 14,250 | 450 | 400 | 2.5 × 10−2 | |
| 70 min. | AlTiSiN (0.5 µm) | 1 Ti 2 AlTi 1 TiSi | 120 TiSi 150 AlTi | 65 | 7800 14,250 | 450 | 480 | 2.5 × 10−2 | |
| 95 min. | TiSiN (1.2 µm) | 1 TiSi | 150 | 65 | 15,250 | 450 | 480 | 3.2 × 10−2 | |
| Coating D | 45 min. | CrN (1 µm) | 4 Cr | 70 | 120 | 8400 | 380 | 250 | 2 × 10−2 |
| 60 min. | TiCrN (1 µm) | 4 Cr 4 Ti | 70 Cr 60 Ti | 120 | 8400 7200 | 380 | 300 | 2 × 10−2 | |
| 15 min. | TiN (1 µm) | 4 Ti | 60 | 100 | 6000 | 380 | 200 | 1.5 × 10−2 |
| Coating Sample | Hardness (GPa) | Elastic Modulus (GPa) | H/E H3/E2 (GPa) | Sa/Sq (nm) | Coating Thickness (µm) | Preferred Orientation Grain Size (nm) | Strain () |
|---|---|---|---|---|---|---|---|
| Coating A | 42.45 | 438.46 | 0.1 0.4 | 61.7/ 91.2 | 1.6 | 5.8 | 2.2 |
| Coating A(N) | 35.60 | 401.28 | 0.1 0.3 | 1.7 | |||
| Coating B | 49.23 | 490.1 | 0.1 0.5 | 35.1/ 79.7 | 3.4 | 8.4 | 5.6 |
| Coating B(N) | 48.71 | 479.07 | 0.1 0.5 | 3.6 | |||
| Coating C | 42.77 | 460.40 | 0.1 0.3 | 26.2/ 53.8 | 2.7 | 8.6 | 4.7 |
| Coating C(N) | 31.61 | 415.57 | 0.1 0.2 | 2.5 | |||
| Coating D | 43.65 | 534.56 | 0.1 0.3 | 54.3/ 87.2 | 3.0 | 7.5 | 1.1 |
| Coating D(N) | 33.00 | 464.47 | 0.1 0.2 | 2.8 |
| Coating Sample | Wear Rate 20 °C | Wear Rate 700 °C | Ball Wear Rate 20 °C | Ball Wear Rate 700 °C | Wear Depth 20 °C | Wear Depth 700 °C |
|---|---|---|---|---|---|---|
| Coating A | 4.97 × 10−4 | 6.37 × 10−3 | 9.62 × 10−8 | 2.84 × 10−6 | 0.65 | 18.19 |
| Coating A(N) | 4.97 × 10−4 | 3.13 × 10−4 | 2.09 × 10−7 | 8.03 × 10−7 | 0.17 | 7.6 |
| Coating B | 3.95 × 10−3 | 3.43 × 10−3 | 9.83 × 10−7 | 2.39 × 10−6 | 3.20 | 12.51 |
| Coating B(N) | 1.62 × 10−3 | 6.63 × 10−4 | 9.83 × 10−7 | 1.37 × 10−6 | 4.45 | 6.67 |
| Coating C | 4.36 × 10−3 | 2.10 × 10−2 | 8.00 × 10−7 | 1.51 × 10−6 | 5.72 | 24.60 |
| Coating C(N) | 2.92 × 10−3 | 9.13 × 10−3 | 3.13 × 10−7 | 2.19 × 10−6 | 3.12 | 32.27 |
| Coating D | 6.87 × 10−4 | 8.96 × 10−3 | 9.78 × 10−7 | 2.39 × 10−6 | 0.52 | 26.34 |
| Coating D(N) | 1.12 × 10−3 | 5.83 × 10−3 | 1.93 × 10−6 | 1.83 × 10−6 | 0.37 | 18.35 |
| Coating Sample | Hardness (GPa) (Before 700 °C Wear Test) | Hardness (GPa) (After 700 °C Wear Test) | Deviation (%) |
|---|---|---|---|
| Coating A | 42.5 | 26.4 | 43.8 |
| Coating A(N) | 35.6 | 23.9 | 25.8 |
| Coating B | 49.2 | 32.6 | 34.9 |
| Coating B(N) | 48.7 | 36.3 | 25.4 |
| Coating C | 42.8 | 16.9 | 60.5 |
| Coating C(N) | 31.6 | 24.4 | 38.6 |
| Coating D | 43.7 | 19.6 | 55.1 |
| Coating D(N) | 33 | 17.2 | 47.8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Özkan, D.; Yilmaz, M.A.; Karakurt, D.; Szala, M.; Walczak, M.; Bakdemir, S.A.; Türküz, C.; Sulukan, E. Effect of AISI H13 Steel Substrate Nitriding on AlCrN, ZrN, TiSiN, and TiCrN Multilayer PVD Coatings Wear and Friction Behaviors at a Different Temperature Level. Materials 2023, 16, 1594. https://doi.org/10.3390/ma16041594
Özkan D, Yilmaz MA, Karakurt D, Szala M, Walczak M, Bakdemir SA, Türküz C, Sulukan E. Effect of AISI H13 Steel Substrate Nitriding on AlCrN, ZrN, TiSiN, and TiCrN Multilayer PVD Coatings Wear and Friction Behaviors at a Different Temperature Level. Materials. 2023; 16(4):1594. https://doi.org/10.3390/ma16041594
Chicago/Turabian StyleÖzkan, Doğuş, Mustafa Alper Yilmaz, Deniz Karakurt, Mirosław Szala, Mariusz Walczak, Seda Ataş Bakdemir, Cenk Türküz, and Egemen Sulukan. 2023. "Effect of AISI H13 Steel Substrate Nitriding on AlCrN, ZrN, TiSiN, and TiCrN Multilayer PVD Coatings Wear and Friction Behaviors at a Different Temperature Level" Materials 16, no. 4: 1594. https://doi.org/10.3390/ma16041594
APA StyleÖzkan, D., Yilmaz, M. A., Karakurt, D., Szala, M., Walczak, M., Bakdemir, S. A., Türküz, C., & Sulukan, E. (2023). Effect of AISI H13 Steel Substrate Nitriding on AlCrN, ZrN, TiSiN, and TiCrN Multilayer PVD Coatings Wear and Friction Behaviors at a Different Temperature Level. Materials, 16(4), 1594. https://doi.org/10.3390/ma16041594









