Unraveling the Phase Transition Behavior of MgMn2O4 Electrodes for Their Use in Rechargeable Magnesium Batteries
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis of MgMn2O4 and MgMn2O4 Doped with Either Co or Ni
2.2. Characterization of MgMn2O4 and MgMn2O4 Doped with Either Co or Ni
2.3. Electrochemical Experiments
3. Results
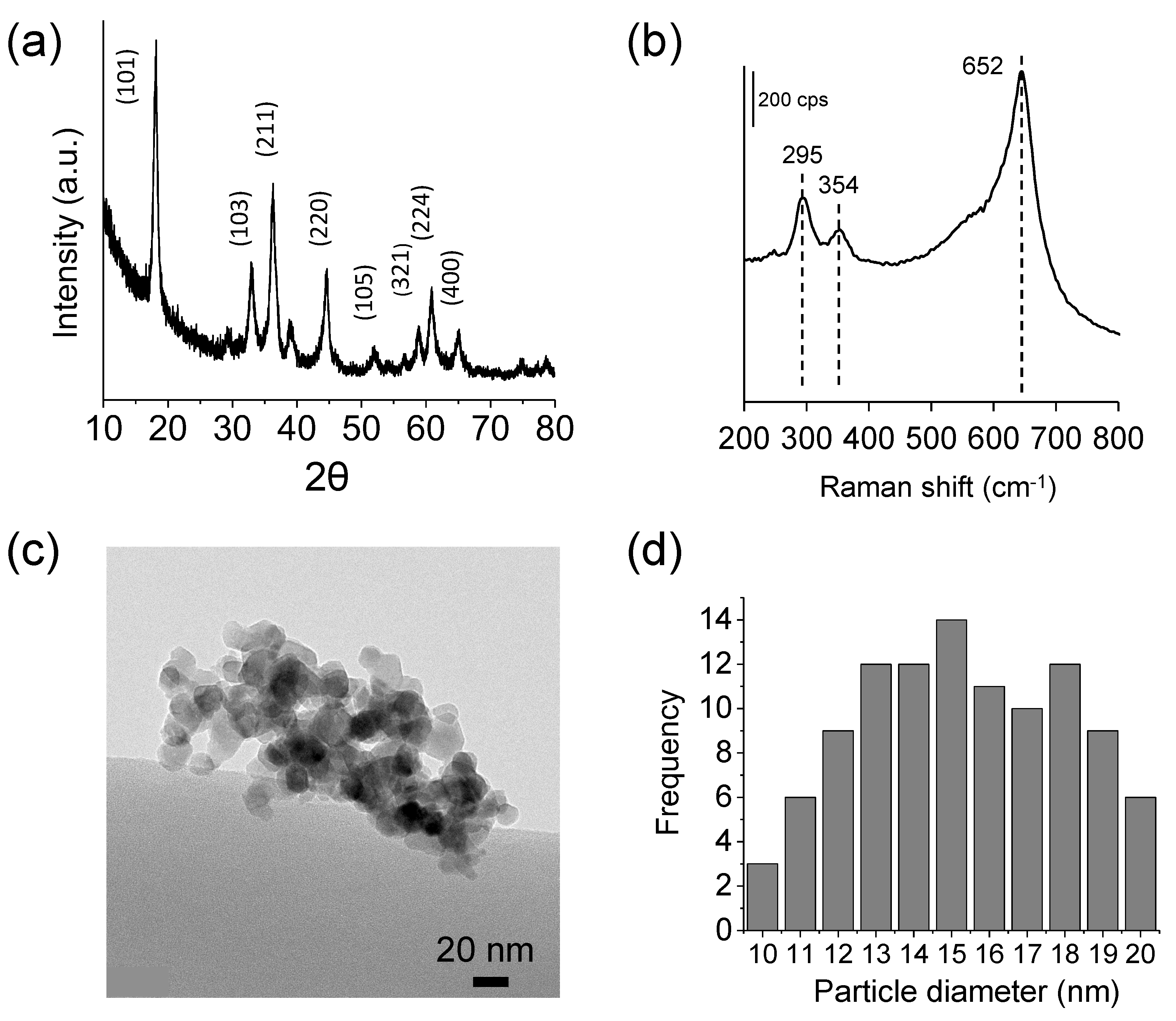
3.1. Physicochemical Characterization of the As-Obtained MgMn2O4
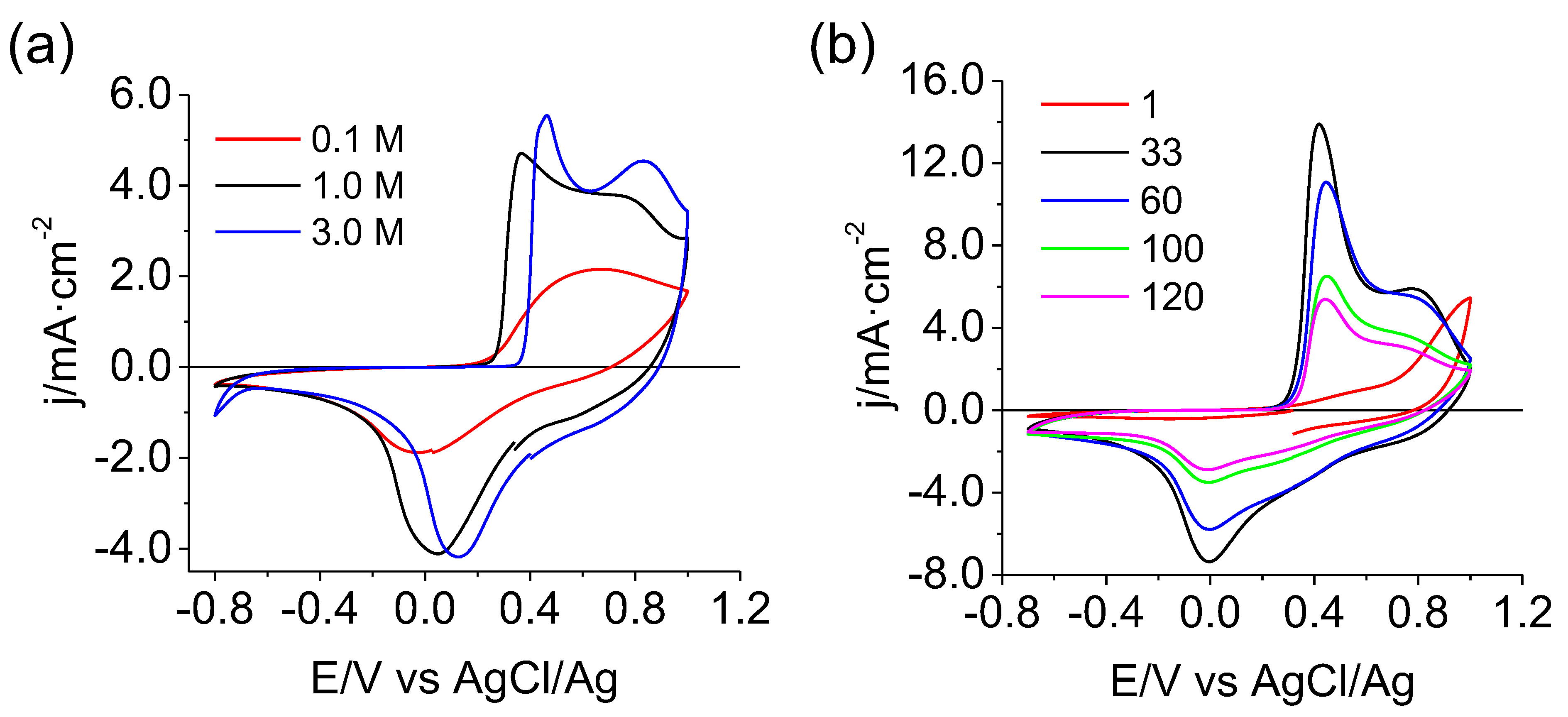
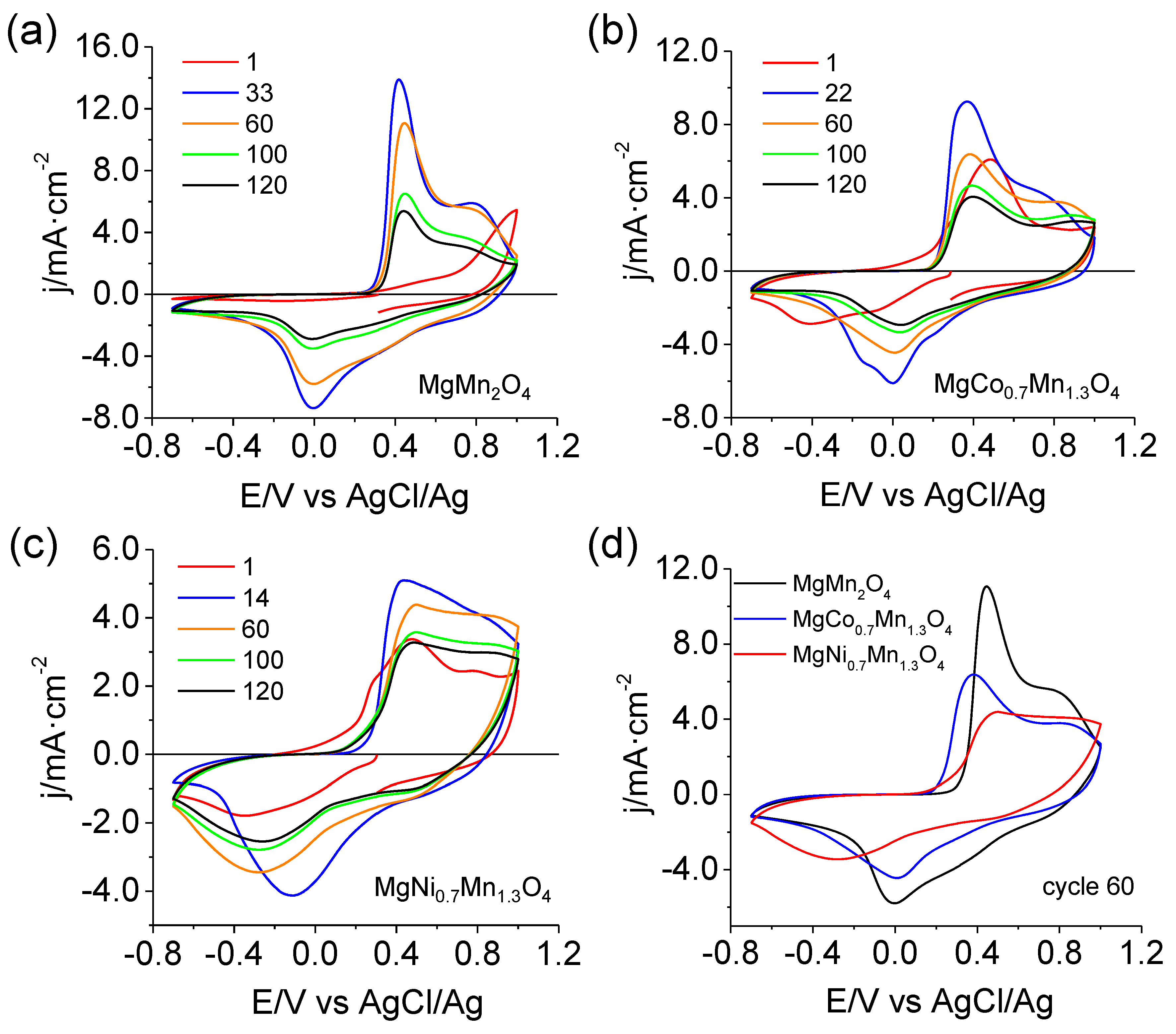
3.2. Electrochemical Behavior of MgMn2O4 in an Aqueous Mg(NO3)2 Electrolyte
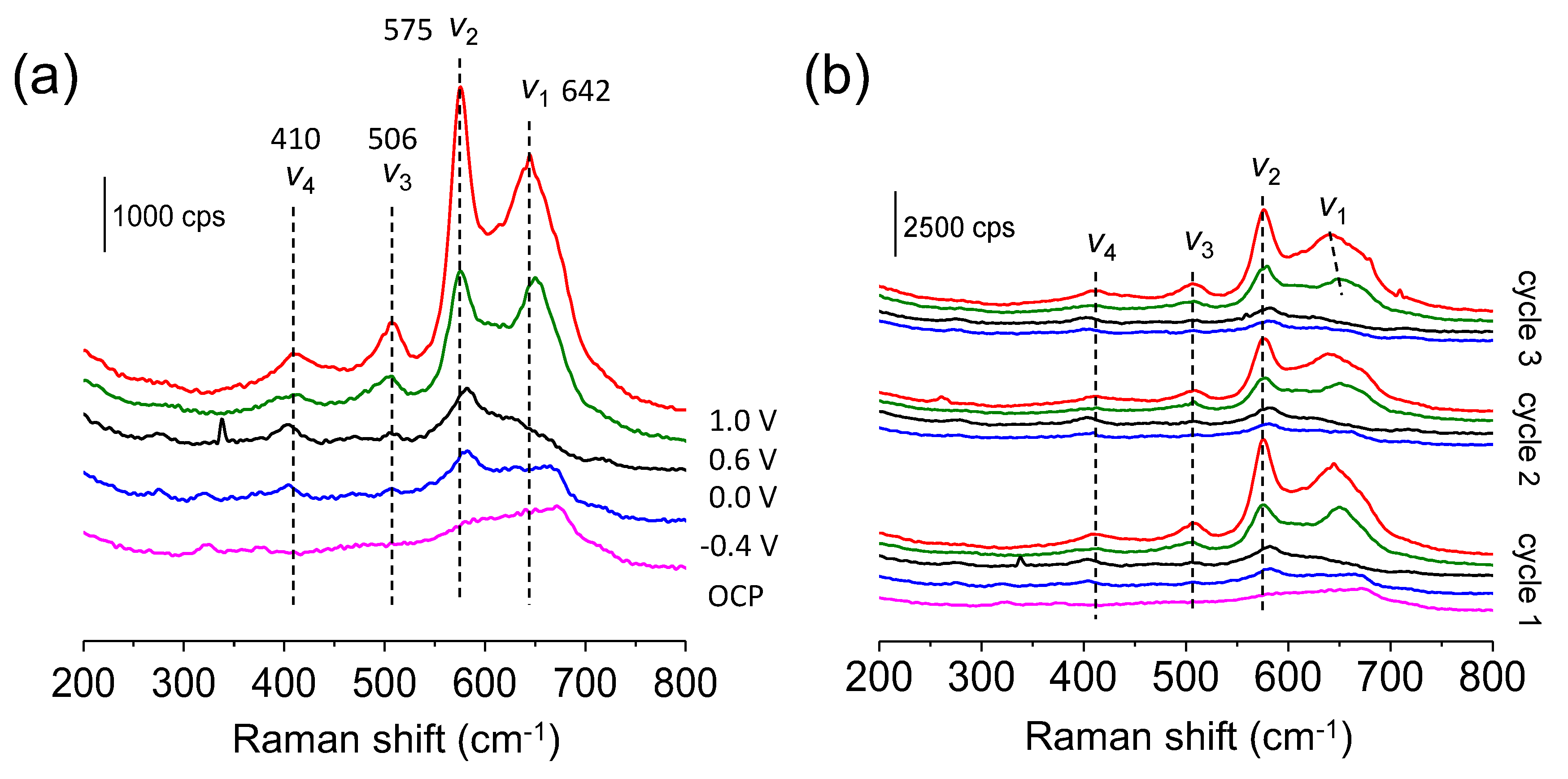
3.3. Electrode/Electrolyte Interfacial Reaction during Cycling
3.4. MgMn2O4 Modified with either Nickel or Cobalt
4. Discussion
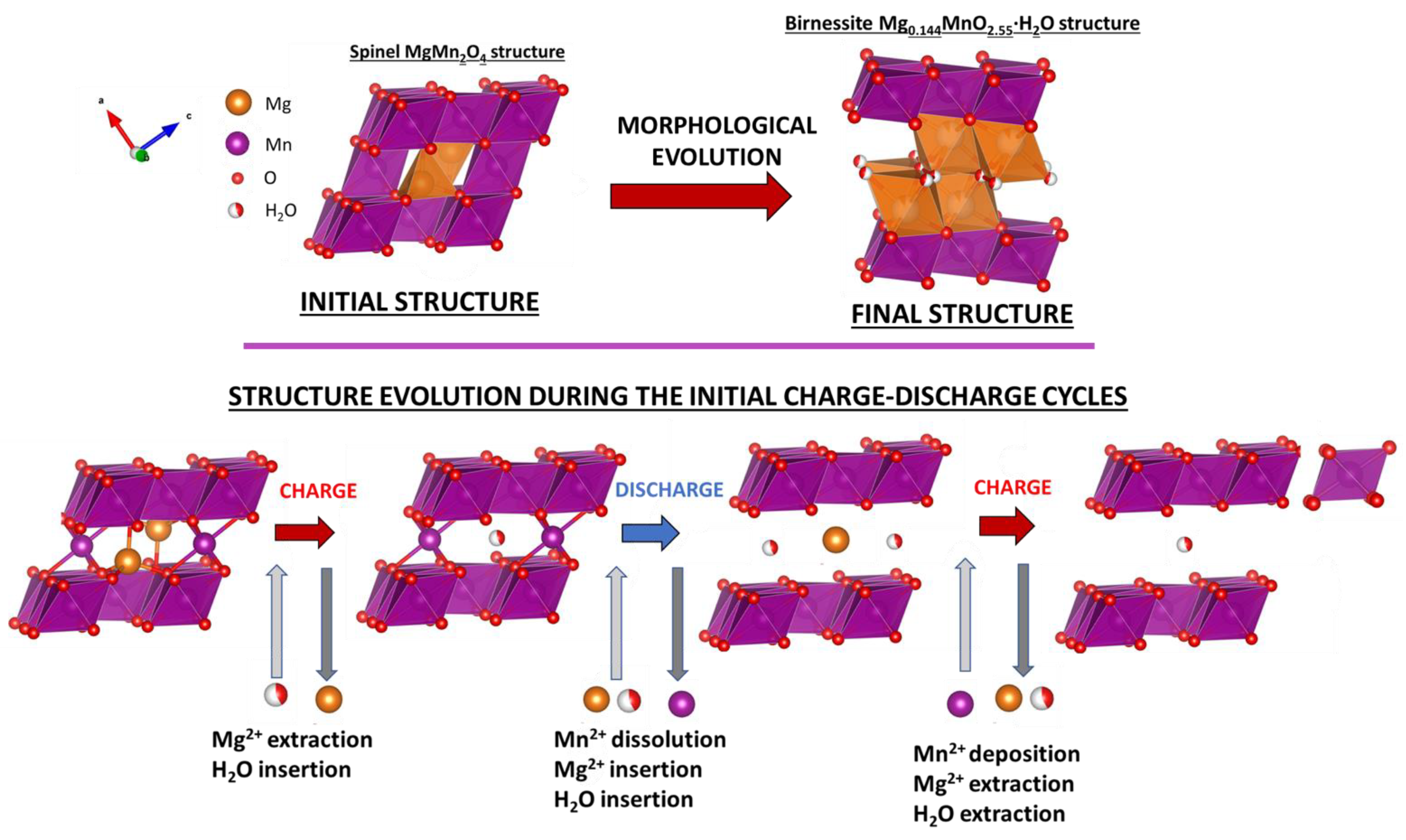
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ma, Z.; MacFarlane, D.R.; Kar, M. Mg Cathode Materials and Electrolytes for Rechargeable Mg Batteries: A Review. Batter. Supercaps 2019, 2, 115–127. [Google Scholar] [CrossRef]
- Levi, E.; Levi, M.D.; Chasid, O.; Aurbach, D. A Review on the Problems of the Solid State Ions Diffusion in Cathodes for Rechargeable Mg Batteries. J. Electroceram. 2009, 22, 13–19. [Google Scholar] [CrossRef]
- Gregory, T.D.; Hoffman, R.J.; Winterton, R.C. Nonaqueous Electrochemistry of Magnesium. J. Electrochem. Soc. 1990, 137, 775–780. [Google Scholar] [CrossRef]
- Mukherjee, A.; Sa, N.; Phillips, P.J.; Burrell, A.; Vaughey, J.; Klie, R.F. Direct Investigation of Mg Intercalation into the Orthorhombic V2O5 Cathode Using Atomic-Resolution Transmission Electron Microscopy. Chem. Mater. 2017, 29, 2218–2226. [Google Scholar] [CrossRef]
- Attias, R.; Salama, M.; Hirsch, B.; Gofer, Y.; Aurbach, D. Solvent Effects on the Reversible Intercalation of Magnesium-Ions into V2O5 Electrodes. ChemElectroChem 2018, 5, 3514–3524. [Google Scholar] [CrossRef]
- Sun, X.; Duffort, V.; Mehdi, B.L.; Browning, N.D.; Nazar, L.F. Investigation of the Mechanism of Mg Insertion in Birnessite in Nonaqueous and Aqueous Rechargeable Mg-Ion Batteries. Chem. Mater. 2016, 28, 534–542. [Google Scholar] [CrossRef]
- Wu, C.; Zhao, G.; Bao, X.; Chen, X.; Sun, K. Hierarchically Porous Delta–Manganese Dioxide Films Prepared by an Electrochemically Assistant Method for Mg Ion Battery Cathodes with High Rate Performance. J. Alloys Compd. 2019, 770, 914–919. [Google Scholar] [CrossRef]
- Aurbach, D.; Lu, Z.; Schechter, A.; Gofer, Y.; Gizbar, H.; Turgeman, R.; Cohen, Y.; Moshkovich, M.; Levi, E. Prototype Systems for Rechargeable Magnesium Batteries. Nature 2000, 407, 724–727. [Google Scholar] [CrossRef]
- Michail, A.; Silván, B.; Tapia-Ruiz, N. Progress in High-Voltage MgMn2O4 Oxyspinel Cathode Materials for Mg Batteries. Curr. Opin. Electrochem. 2022, 31, 100817. [Google Scholar] [CrossRef]
- Kim, C.; Phillips, P.J.; Key, B.; Yi, T.; Nordlund, D.; Yu, Y.S.; Bayliss, R.D.; Han, S.D.; He, M.; Zhang, Z.; et al. Direct Observation of Reversible Magnesium Ion Intercalation into a Spinel Oxide Host. Adv. Mater. 2015, 27, 3377–3384. [Google Scholar] [CrossRef]
- Hannah, D.C.; Sai Gautam, G.; Canepa, P.; Ceder, G. On the Balance of Intercalation and Conversion Reactions in Battery Cathodes. Adv. Energy Mater. 2018, 8, 1800379. [Google Scholar] [CrossRef]
- Truong, Q.D.; Kempaiah Devaraju, M.; Tran, P.D.; Gambe, Y.; Nayuki, K.; Sasaki, Y.; Honma, I. Unravelling the Surface Structure of MgMn2O4 Cathode Materials for Rechargeable Magnesium-Ion Battery. Chem. Mater. 2017, 29, 6245–6251. [Google Scholar] [CrossRef]
- Yin, J.; Brady, A.B.; Takeuchi, E.S.; Marschilok, A.C.; Takeuchi, K.J. Magnesium-Ion Battery-Relevant Electrochemistry of MgMn2O4: Crystallite Size Effects and the Notable Role of Electrolyte Water Content. Chem. Comm. 2017, 53, 3665–3668. [Google Scholar] [CrossRef] [PubMed]
- Cabello, M.; Alcántara, R.; Nacimiento, F.; Ortiz, G.; Lavela, P.; Tirado, J.L. Electrochemical and Chemical Insertion/Deinsertion of Magnesium in Spinel-Type MgMn2O4 and Lambda-MnO2 for Both Aqueous and Non-Aqueous Magnesium-Ion Batteries. CrystEngComm 2015, 17, 8728–8735. [Google Scholar] [CrossRef]
- Tao, S.; Huang, W.; Liu, Y.; Chen, S.; Qian, B.; Song, L. Three-Dimensional Hollow Spheres of the Tetragonal-Spinel MgMn2O4 Cathode for High-Performance Magnesium Ion Batteries. J. Mater. Chem. A Mater. 2018, 6, 8210–8214. [Google Scholar] [CrossRef]
- Zhang, H.; Ye, K.; Shao, S.; Wang, X.; Cheng, K.; Xiao, X.; Wang, G.; Cao, D. Octahedral Magnesium Manganese Oxide Molecular Sieves as the Cathode Material of Aqueous Rechargeable Magnesium-Ion Battery. Electrochim. Acta 2017, 229, 371–379. [Google Scholar] [CrossRef]
- Kim, S.; Nam, K.W.; Lee, S.; Cho, W.; Kim, J.S.; Kim, B.G.; Oshima, Y.; Kim, J.S.; Doo, S.G.; Chang, H.; et al. Observation of an Anomalous Spinel-to-Layered Phase Transition Mediated by Crystal Water Intercalation. Angew. Chem. 2015, 54, 15094–15099. [Google Scholar] [CrossRef]
- Kim, S.; Lee, S.; Nam, K.W.; Shin, J.; Lim, S.Y.; Cho, W.; Suzuki, K.; Oshima, Y.; Hirayama, M.; Kanno, R.; et al. On the Mechanism of Crystal Water Insertion during Anomalous Spinel-to-Birnessite Phase Transition. Chem. Mater. 2016, 28, 5488–5494. [Google Scholar] [CrossRef]
- Yang, E.; Kim, H.; Kim, S.; Kim, I.; Kim, J.; Ji, H.; Choi, J.W.; Jung, Y. Origin of Unusual Spinel-to-Layered Phase Transformation by Crystal Water. Chem. Sci. 2018, 9, 433–438. [Google Scholar] [CrossRef]
- Hatakeyama, T.; Okamoto, N.L.; Shimokawa, K.; Li, H.; Nakao, A.; Uchimoto, Y.; Tanimura, H.; Kawaguchi, T.; Ichitsubo, T. Electrochemical Phase Transformation Accompanied with Mg Extraction and Insertion in a Spinel MgMn2O4 Cathode Material. Phys. Chem. Chem. Phys. 2019, 21, 23749–23757. [Google Scholar] [CrossRef]
- Truong, Q.D.; Kobayashi, H.; Nayuki, K.; Sasaki, Y.; Honma, I. Atomic-Scale Observation of Phase Transition of MgMn2O4 Cubic Spinel upon the Charging in Mg-Ion Battery. Solid State Ion. 2020, 344, 115136. [Google Scholar] [CrossRef]
- Liu, T.; Dai, A.; Lu, J.; Yuan, Y.; Xiao, Y.; Yu, L.; Li, M.; Gim, J.; Ma, L.; Liu, J.; et al. Correlation between Manganese Dissolution and Dynamic Phase Stability in Spinel-Based Lithium-Ion Battery. Nat. Commun. 2019, 10, 4721. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, H.; Samukawa, K.; Nakayama, M.; Mandai, T.; Honma, I. Promoting Reversible Cathode Reactions in Magnesium Rechargeable Batteries Using Metastable Cubic MgMn2O4 Spinel Nanoparticles. ACS Appl. Nano Mater. 2021, 4, 8328–8333. [Google Scholar] [CrossRef]
- Yokozaki, R.; Kobayashi, H.; Mandai, T.; Honma, I. Effect of Al Substitution on Structure and Cathode Performance of MgMn2O4 Spinel for Magnesium Rechargeable Battery. J. Alloys Compd. 2021, 872, 159723. [Google Scholar] [CrossRef]
- Sone, K.; Hayashi, Y.; Mandai, T.; Yagi, S.; Oaki, Y.; Imai, H. Effective 3D Open-Channel Nanostructures of a MgMn2O4 positive Electrode for Rechargeable Mg Batteries Operated at Room Temperature. J. Mater. Chem. A Mater. 2021, 9, 6851–6860. [Google Scholar] [CrossRef]
- Zhang, D.; Chen, Q.; Zhang, J.; Sun, T. MgMn2O4/Multiwalled Carbon Nanotubes Composite Fabricated by Electrochemical Conversion as a High-Performance Cathode Material for Aqueous Rechargeable Magnesium Ion Battery. J. Alloys Compd. 2021, 873, 159872. [Google Scholar] [CrossRef]
- Liu, G.; Chi, Q.; Zhang, Y.; Chen, Q.; Zhang, C.; Zhu, K.; Cao, D. Superior High Rate Capability of MgMn2O4/RGO Nanocomposites as Cathode Materials for Aqueous Rechargeable Magnesium Ion Batteries. Chem. Comm. 2018, 54, 9474–9477. [Google Scholar] [CrossRef]
- Yu, Y.; Guo, J.; Xiang, M.; Su, C.; Liu, X.; Bai, H.; Bai, W.; Duan, K. Enhancing the Durable Performance of LiMn2O4 at High-Rate and Elevated Temperature by Nickel-Magnesium Dual Doping. Sci. Rep. 2019, 9, 16864. [Google Scholar] [CrossRef]
- Taniguchi, I.; Song, D.; Wakihara, M. Electrochemical Properties of LiM1/6Mn11/6O4 (M=Mn, Co, Al and Ni) as Cathode Materials for Li-Ion Batteries Prepared by Ultrasonic Spray Pyrolysis Metho. J. Power Sources 2002, 109, 333–339. [Google Scholar] [CrossRef]
- Han, J.; Yagi, S.; Ichitsubo, T. Suppressive Effect of Fe Cations in Mg(Mn1−xFex)2O4 Positive Electrodes on Oxidative Electrolyte Decomposition for Mg Rechargeable Batteries. J. Power Sources 2019, 435, 226822. [Google Scholar] [CrossRef]
- Medina, A.; Rodríguez, A.I.; Pérez-Vicente, C.; Alcántara, R. Magnesium Deintercalation from the Spinel-Type MgMn2-YFeyO4 (0.4≤y≤2.0) by Acid-Treatment and Electrochemistry. Chem. Eur. 2021, 27, 12599–12609. [Google Scholar] [CrossRef]
- Banu, A.; Sakunthala, A.; Thamilselvan, M.; Kumar, P.S.; Suresh, K.; Ashwini, S. Preparation, Characterization and Comparative Electrochemical Studies of MgMXMn2-XO4 (X = 0, 0.5; M = Ni/Co). Ceram. Int. 2019, 45, 13072–13085. [Google Scholar] [CrossRef]
- Malavasi, L.; Galinetto, P.; Mozzati, M.C.; Azzoni, C.B.; Flor, G. Raman Spectroscopy of AMn2O4 (A = Mn, Mg and Zn) Spinels. Phys. Chem. Chem. Phys. 2002, 4, 3876–3880. [Google Scholar] [CrossRef]
- Malavasi, L.; Ghigna, P.; Chiodelli, G.; Maggi, G.; Flor, G. Structural and Transport Properties of Mg1-XMnxMn2O4±δ Spinels. J. Solid. State Chem. 2002, 166, 171–176. [Google Scholar] [CrossRef]
- Malavasi, L.; Mozzati, M.C.; Flor, G. RF Sputtering Deposition of MgMn2O4 Spinel Thin Films. J. Mater. Science 2004, 39, 1671–1675. [Google Scholar] [CrossRef]
- Julien, C.M.; Massot, M. Lattice Vibrations of Materials for Lithium Rechargeable Batteries I. Lithium Manganese Oxide Spinel. Mater. Sci. Eng. B 2003, 97, 217–230. [Google Scholar] [CrossRef]
- Taddesse, P.; Gebrekiros, H.; Semu, G.; Duressa, M.; Chemeda, Y.C.; Murali, N.; Babu, K.V. Investigation of Structural, Vibrational Spectroscopic and Properties Study of LiMn2O4 and LiMn1.9Cu0.05Fe0.05O4 Cathode Materials. Results Mater. 2021, 12, 100224. [Google Scholar] [CrossRef]
- Tortosa, M.; Manjón, F.J.; Mollar, M.; Marí, B. ZnO-Based Spinels Grown by Electrodeposition. J. Phys. Chem. Solids 2012, 73, 1111–1115. [Google Scholar] [CrossRef]
- Bernardini, S.; Bellatreccia, F.; Casanova Municchia, A.; della Ventura, G.; Sodo, A. Raman Spectra of Natural Manganese Oxides. J. Raman Spectrosc. 2019, 50, 873–888. [Google Scholar] [CrossRef]
- Julien, C.; Massot, M.; Baddour-Hadjean, R.; Franger, S.; Bach, S.; Pereira-Ramos, J.P. Raman Spectra of Birnessite Manganese Dioxides. Solid. State Ion. 2003, 159, 345–356. [Google Scholar] [CrossRef]
- Lu, J.; Zhan, C.; Wu, T.; Wen, J.; Lei, Y.; Kropf, A.J.; Wu, H.; Miller, D.J.; Elam, J.W.; Sun, Y.K.; et al. Effectively Suppressing Dissolution of Manganese from Spinel Lithium Manganate via a Nanoscale Surface-Doping Approach. Nat. Commun. 2014, 5, 5693. [Google Scholar] [CrossRef] [PubMed]
- Hou, Z.; Dong, M.; Xiong, Y.; Zhang, X.; Ao, H.; Liu, M.; Zhu, Y.; Qian, Y. A High-Energy and Long-Life Aqueous Zn/Birnessite Battery via Reversible Water and Zn2+ Coinsertion. Small 2020, 16, 2001228. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.H.; Lin, Y.Q.; Althouse, Z.D.; Liu, N. Dissolution-Redeposition Mechanism of the MnO2 Cathode in Aqueous Zinc-Ion Batteries. ACS Appl. Energy Mater. 2021, 4, 12267–12274. [Google Scholar] [CrossRef]
- Zhao, H.; Liu, L.; Xiao, L.; Hu, Z.; Han, S.; Liu, Y.; Chen, D.; Liu, X. The Effects of Co Doping on the Crystal Structure and Electrochemical Performance of Mg(Mn2-X Cox)O4 Negative Materials for Lithium Ion Battery. Solid State Sci. 2015, 39, 23–28. [Google Scholar] [CrossRef]
- Jin, W.; Yin, G.; Wang, Z.; Fu, Y.Q. Surface Stability of Spinel MgNi0.5Mn1.5O4 and MgMn2O4 as Cathode Materials for Magnesium Ion Batteries. Appl. Surf. Sci. 2016, 385, 72–79. [Google Scholar] [CrossRef]
- Persson, I. Structures of Hydrated Metal Ions in Solid State and Aqueous Solution. Liquids 2022, 2, 210–242. [Google Scholar] [CrossRef]
- Lee, B.; Lee, H.R.; Kim, H.; Chung, K.Y.; Cho, B.W.; Oh, S.H. Elucidating the Intercalation Mechanism of Zinc Ions into α-MnO2 for Rechargeable Zinc Batteries. Chem. Comm. 2015, 51, 9265–9268. [Google Scholar] [CrossRef]
- Momma, K.; Izumi, F. VESTA 3 for Three-Dimensional Visualization of Crystal, Volumetric and Morphology Data. J. Appl. Crystallogr. 2011, 44, 1272–1276. [Google Scholar] [CrossRef]

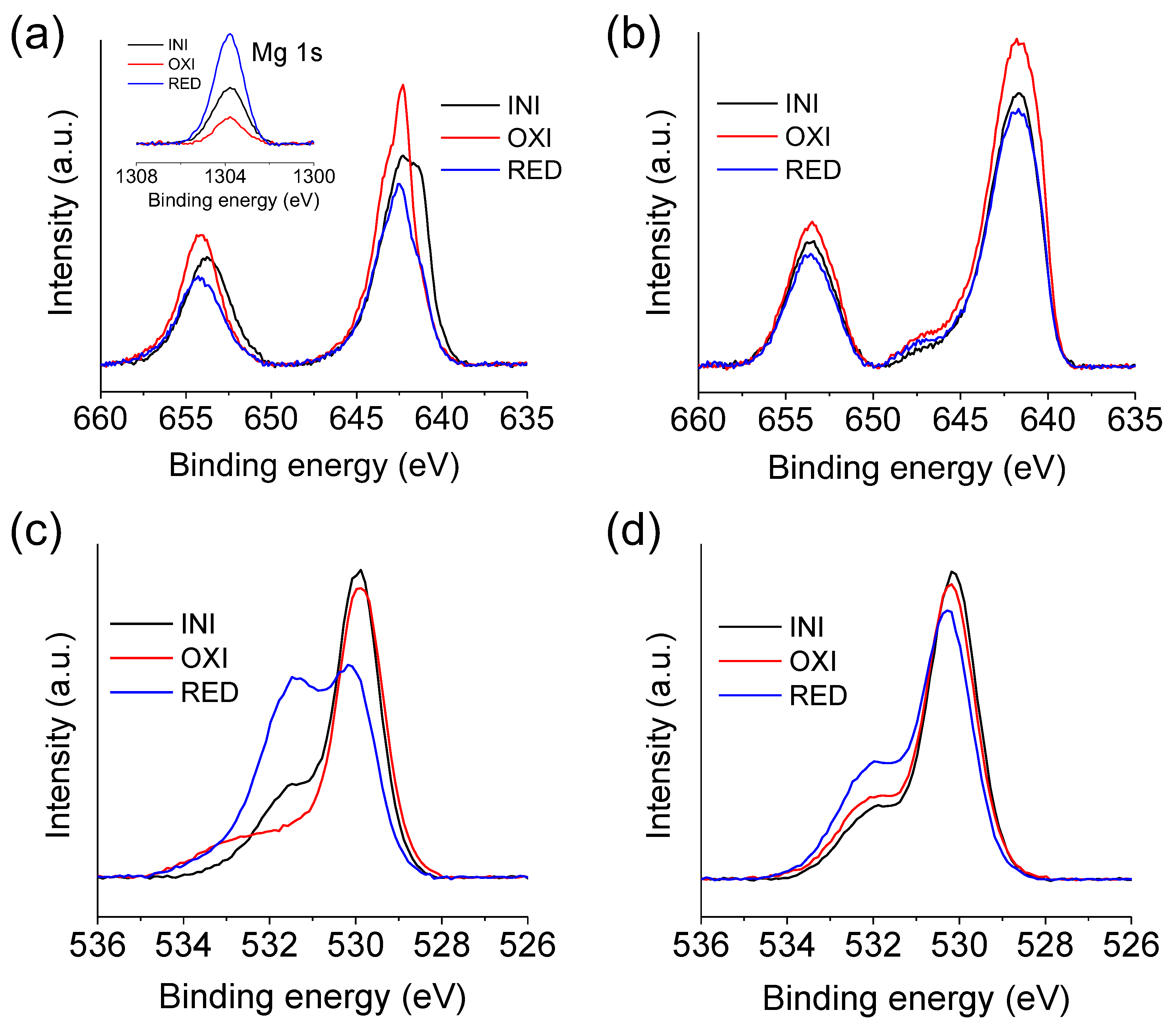

| Ratio Mg/Mn Surface | Ratio Mg/Mn after Etching | |
|---|---|---|
| Pristine MgMn2O4 | 0.42 | 0.54 |
| Oxidized MgMn2O4 | 0.16 | 0.27 |
| Reduced MgMn2O4 | 0.96 | 0.78 |
| Mn(III) | Mn(IV) | ||||
|---|---|---|---|---|---|
| Pristine MgMn2O4 | Mn 2p peak (eV) | 641.1 | 642.5 | 644.4 | 646.5 |
| Content (%) | 83 | 17 | |||
| Oxidized MgMn2O4 | Mn 2p peak (eV) | 641.2 | 642.2 | 643.2 | 644.9 |
| Content (%) | 44 | 56 | |||
| Reduced MgMn2O4 | Mn 2p peak (eV) | 641.4 | 642.4 | 643.5 | 644.8 |
| Content (%) | 56 | 44 | |||
| Mn-O-Mn | Mn-OH | H-O-H | ||
|---|---|---|---|---|
| Pristine MgMn2O4 | O 1s peak (eV) | 529.9 | 531.5 | 532.9 |
| Content (%) | 70 | 28 | 2 | |
| Oxidized MgMn2O4 | O 1s peak (eV) | 529.9 | 531.4 | 533.0 |
| Content (%) | 73 | 16 | 11 | |
| Reduced MgMn2O4 | O 1s peak (eV) | 530.0 | 531.4 | 532.7 |
| Content (%) | 40 | 50 | 10 |
| Max. Capacity (mAh g−1) | Capacity for Scan 120 (mAh g−1) | Retention Capacity for Scan 120 (%) | |
|---|---|---|---|
| MgMn2O4 | 144 | 71 | 49 |
| MgCo0.7Mn1.3O4 | 118 | 71 | 60 |
| MgNi0.7Mn1.3O4 | 94 | 72 | 77 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miralles, C.; Lana-Villarreal, T.; Gómez, R. Unraveling the Phase Transition Behavior of MgMn2O4 Electrodes for Their Use in Rechargeable Magnesium Batteries. Materials 2023, 16, 5402. https://doi.org/10.3390/ma16155402
Miralles C, Lana-Villarreal T, Gómez R. Unraveling the Phase Transition Behavior of MgMn2O4 Electrodes for Their Use in Rechargeable Magnesium Batteries. Materials. 2023; 16(15):5402. https://doi.org/10.3390/ma16155402
Chicago/Turabian StyleMiralles, Carmen, Teresa Lana-Villarreal, and Roberto Gómez. 2023. "Unraveling the Phase Transition Behavior of MgMn2O4 Electrodes for Their Use in Rechargeable Magnesium Batteries" Materials 16, no. 15: 5402. https://doi.org/10.3390/ma16155402
APA StyleMiralles, C., Lana-Villarreal, T., & Gómez, R. (2023). Unraveling the Phase Transition Behavior of MgMn2O4 Electrodes for Their Use in Rechargeable Magnesium Batteries. Materials, 16(15), 5402. https://doi.org/10.3390/ma16155402






