Ways to Improve the Efficiency of Devices for Freezing of Small Products
Abstract
:1. Introduction
2. The Basic Part of the Study
2.1. Model Representation of the Problem and Its Solution
2.2. Materials and Methods
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhou, G.; Cui, M.; Wan, J.; Zhang, S.A. Review on Snowmelt Models: Progress and Prospect. Sustainability 2021, 13, 11485. [Google Scholar] [CrossRef]
- Nitsenko, V.; Mardani, A.; Streimikis, J.; Shkrabak, I.; Klopov, I.; Novomlynets, O.; Podolska, O. Criteria for Evaluation of Efficiency of Energy Transformation Based on Renewable Energy Sources. Montenegrin J. Econ. 2018, 14, 237–247. [Google Scholar] [CrossRef]
- Kotenko, S.; Nitsenko, V.; Hanzhurenko, I.; Havrysh, V. The Mathematical Modeling Stages of Combining the Carriage of Goods for Indefinite, Fuzzy and Stochastic Parameters. Int. J. Integr. Eng. 2020, 12, 173–180. [Google Scholar] [CrossRef]
- Karaiev, O.; Bondarenko, L.; Halko, S.; Miroshnyk, O.; Vershkov, O.; Karaieva, T.; Shchur, T.; Findura, P.; Prístavka, M. Mathematical modelling of the fruit-stone culture seeds calibration process using flat sieves. Acta Technol. Agric. 2021, 24, 119–123. [Google Scholar] [CrossRef]
- Xin, Y.; Zhang, M.; Xu, B.; Adhikari, B.; Sun, J. Research Trends in Selected Blanching Pretreatments and Quick Freezing Technologies as Applied in Fruits and Vegetables: A Review. Int. J. Refrig. 2015, 57, 11–25. [Google Scholar] [CrossRef]
- Santarelli, V.; Neri, L.; Sacchetti, G.; Di Mattia, C.D.; Mastrocola, D.; Pittia, P. Response of Organic and Conventional Apples to Freezing and Freezing Pre-Treatments: Focus on Polyphenols Content and Antioxidant Activity. Food Chem. 2020, 308, 125570. [Google Scholar] [CrossRef]
- Tan, X.Y.; Misran, A.; Daim, L.D.J.; Ding, P.; Pak Dek, M.S. Effect of Freezing on Minimally Processed Durian for Long Term Storage. Sci. Hortic. 2020, 264, 109170. [Google Scholar] [CrossRef]
- Alhamdan, A.; Hassan, B.; Alkahtani, H.; Abdelkarim, D.; Younis, M. Cryogenic Freezing of Fresh Date Fruits for Quality Preservation during Frozen Storage. J. Saudi Soc. Agric. Sci. 2018, 17, 9–16. [Google Scholar] [CrossRef] [Green Version]
- Ibarz, A.; Barbosa-Cánovas, G.V. Unit Operations in Food Engineering (Food Preservation Technology Series); CRC Press: Boca Raton, FL, USA, 2003; ISBN 978-1-56676-929-7. [Google Scholar]
- Bondarenko, L.Y. Determination of Optimal Parameters of the Process of Calibration of Seeds of Fruit Stone Crops. Proc. Tavriya State Agrotechnol. Univ. 2010, 5, 162–168. [Google Scholar]
- Blija, A.; Beitane, I.; Aboltins, A.; Boca, S. Heat-Mass Transfer in Layer of Berries during the Freezing Process. In Proceedings of the 16th International Scientific Conference “Engineering for Rural Development”, Jelgava, Latvia, 24–26 May 2017; pp. 700–705. [Google Scholar] [CrossRef]
- Rayman Ergün, A.; Yanat, M.; Baysal, T. The Effects of the Novel Home Freezing System on Microstructure, Color, Antioxidant Activity, and Microbiological Properties of Strawberries. Int. J. Refrig. 2021, 121, 228–234. [Google Scholar] [CrossRef]
- Struchaev, M.; Golub, I.; Palyanychka, N.; Postol, Y. Pat. 129068 Ukraine. Device for Storage of Refrigerated Products; Ukrpatent: Kyiv, Ukraine, 2018. [Google Scholar]
- Struchaev, M.; Zagorko, N.; Bovkun, O.; Palyanychka, N.; Tarasenko, V. Pat. 129278 Ukraine. Device for the Production of Aerated Frozen Products; Ukrpatent: Kyiv, Ukraine, 2018. [Google Scholar]
- Struchaev, M.; Tarasenko, V.; Verkholantseva, V.; Postol, Y.; Shats, V. Pat. 129352: Pat. 129352 Ukraine. Device for Determining the Cryoscopic Temperature of Foodstuffs; Ukrpatent: Kyiv, Ukraine, 2018. [Google Scholar]
- Yalpachik, V.; Struchaev, M.; Tarasenko, V. Experimental Determination of the Coefficient of Thermal Conductivity during Freezing. Proc. Tavriya State Agrotechnol. Univ. 2017, 17, 113–118. [Google Scholar]
- Struchaiev, N.I. Determination of the Amount of Heat during Freezing and Defrosting. Petro Vasylenko Kharkiv Natl. Tech. Univ. Agric. 2015, 2, 130–131. [Google Scholar]
- Bondarenko, L.Y. Research of Dimensional—Mass Parameters of Sowing Material of Fruit Stone Cultures. Proc. Tavriya State Agrotechnol. Univ. 2006, 7, 111–117. [Google Scholar]
- Struchaev, M.; Zagorko, N.; Verkholantseva, V.; Palyanychka, N.; Tarasenko, V.; Malakhov, V. Pat. 131546 Ukraine. Device for Fluidization; Ukrpatent: Kyiv, Ukraine, 2019. [Google Scholar]
- Eglit, A.Y.; Krupenenkov, N.F.; Filatov, A.S.; Kisser, K.V. Methods of Determining the Dynamics of the Temperature Field a Two-Layer Product of Spherical Form during the Freezing. Sci. J. NRU ITMO Ser. Process. Food Prod. Equip. 2015, 24, 187–192. [Google Scholar]
- Tabor, S.; Lezhenkin, A.; Halko, S.; Miroshnyk, A.; Kovalyshyn, S.; Vershkov, A.; Hryhorenko, O. Mathematical Simulation of Separating Work Tool Technological Process. E3S Web Conf. 2019, 132, 01025. [Google Scholar] [CrossRef] [Green Version]
- Lezhenkin, O.; Halko, S.; Miroshnyk, O.; Vershkov, O.; Lezhenkin, I.; Suprun, O.; Shchur, T.; Kruszelnicka, W.; Kasner, R. Investigation of the separation of combed heap of winter wheat. J. Phys. Int. Conf. Appl. Sci. 2021, 1781, 012016. [Google Scholar] [CrossRef]
- Struchaiev, N.I.; Postol, Y. Improving the Energy Efficiency of the Milk Cooler. Proc. Tavriya State Agrotechnol. Univ. 2017, 1, 243–247. [Google Scholar]
- Yalpachik, V.; Struchaiev, N.I.; Verkholantseva, V. Planning of Experimental Researches of Process of Cooling of Grain. Proc. Tavriya State Agrotechnol. Univ. 2015, 1, 3–8. [Google Scholar]
- Fayazbakhsh, M.A.; Bagheri, F.; Bahrami, M. Real-Time Thermal Load Calculation by Automatic Estimation of Convection Coefficients. Int. J. Refrig. 2015, 57, 229–238. [Google Scholar] [CrossRef]
- Li, L.; Pegg, R.B.; Eitenmiller, R.R.; Chun, J.-Y.; Kerrihard, A.L. Selected Nutrient Analyses of Fresh, Fresh-Stored, and Frozen Fruits and Vegetables. J. Food Compos. Anal. 2017, 59, 8–17. [Google Scholar] [CrossRef]
- Struchaev, M.; Oleksienko, V.; Palyanychka, N.; Verkholantseva, V.; Tarasenko, V. Pat. 137137 Ukraine. Cascading Device for Freezing; Ukrpatent: Kyiv, Ukraine, 2019. [Google Scholar]
- Struchaiev, N.; Postol, Y.; Stopin, Y.; Borokhov, I. Determination of the Duration of Spherical-Shaped Berries Freezing Under the Conditions Stationary Heat Flow. In Modern Development Paths of Agricultural Production; Nadykto, V., Ed.; Springer: Cham, Switzerland, 2019; pp. 405–414. [Google Scholar] [CrossRef]
- Bondarenko, L.Y.; Tsymbal, V.I. Investigation of the Shape and Size of the Holes of the Sieves for Calibration of Seed of Fruit Stone Crops. Mech. Electrif. Agric. Interdep. Themat. Sci. Collect. 2008, 4, 176–181. [Google Scholar]
- Karaev, A.; Bondarenko, L.Y. Pat. 48097 Ukraine. Installation for Calibration of Seed Material of Stone Fruit 2010.
- Ergun, S. Fluid Flow through Packed Columns. Chem. Eng. Prog. 1952, 48, 89–94. [Google Scholar]
- Li, L.; Xie, W.; Zhang, Z.; Zhang, S. Pressure Drop in Packed Beds with Horizontally or Vertically Stratified Structure. Nucl. Eng. Technol. 2020, 52, 2491–2498. [Google Scholar] [CrossRef]
- Bazaluk, O.; Postnikova, M.; Halko, S.; Kvitka, S.; Mikhailov, E.; Kovalov, O.; Suprun, O.; Miroshnyk, O.; Nitsenko, V. Energy Saving in Electromechanical Grain Cleaning Systems. Appl. Sci. 2022, 12, 1418. [Google Scholar] [CrossRef]
- Todes, O.M. Thermal Explosion Theory. Zh Fiz Khim 1939, 4, 78–80. [Google Scholar]
- Vargas-Salgado, C.; Hurtado-Pérez, E.; Alfonso-Solar, D.; Malmquist, A. Empirical Design, Construction, and Experimental Test of a Small-Scale Bubbling Fluidized Bed Reactor. Sustainability 2021, 13, 1061. [Google Scholar] [CrossRef]
- Bazaluk, O.; Havrysh, V.; Cherednichenko, O.; Nitsenko, V. Chemically Recuperated Gas Turbines for Offshore Platform: Energy and Environmental Performance. Sustainability 2021, 13, 12566. [Google Scholar] [CrossRef]
- Bazaluk, O.; Havrysh, V.; Nitsenko, V. Energy Efficiency of Inland Waterways Transport for Agriculture: The Ukraine Case Study. Appl. Sci. 2021, 11, 8937. [Google Scholar] [CrossRef]
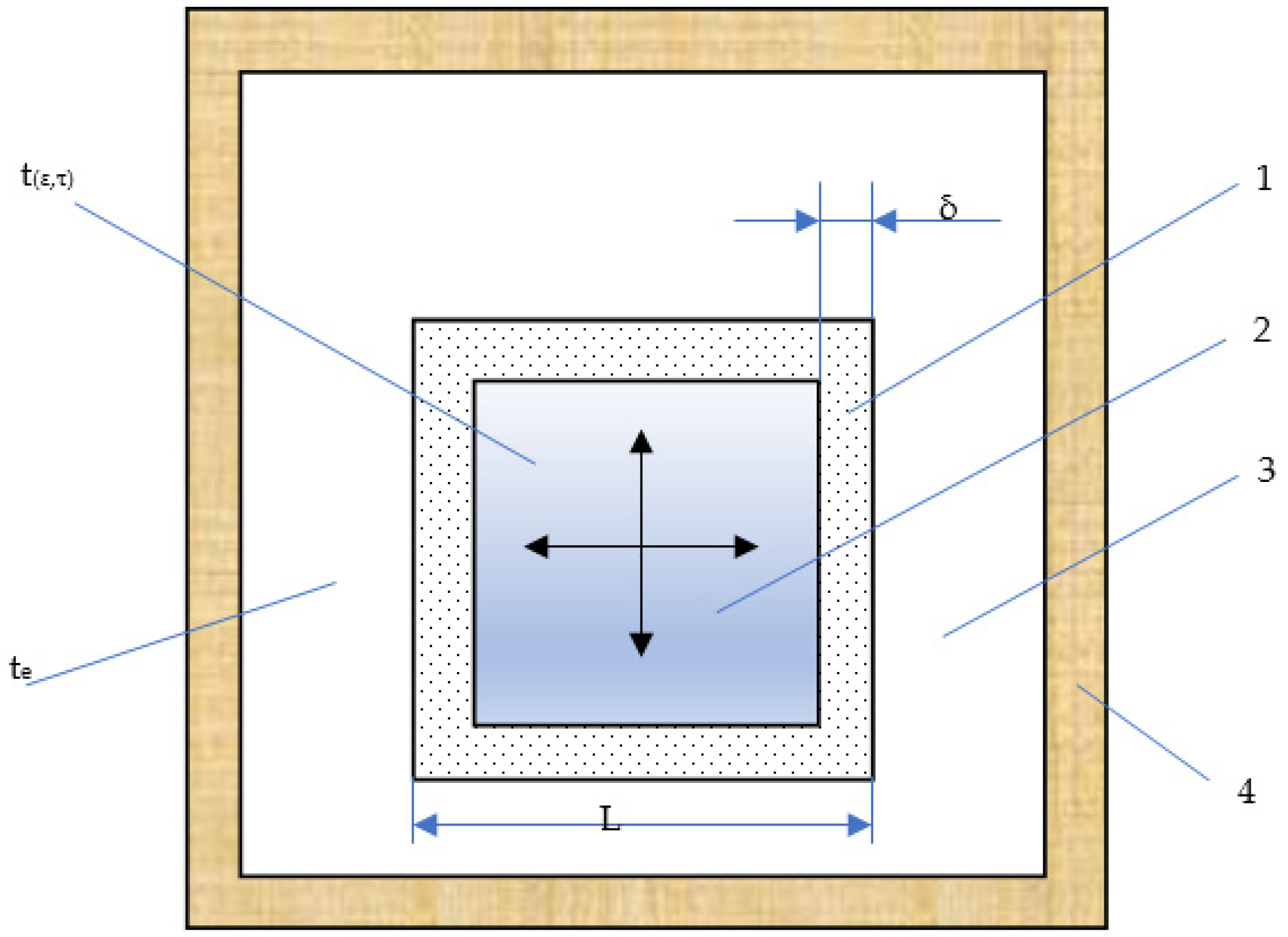
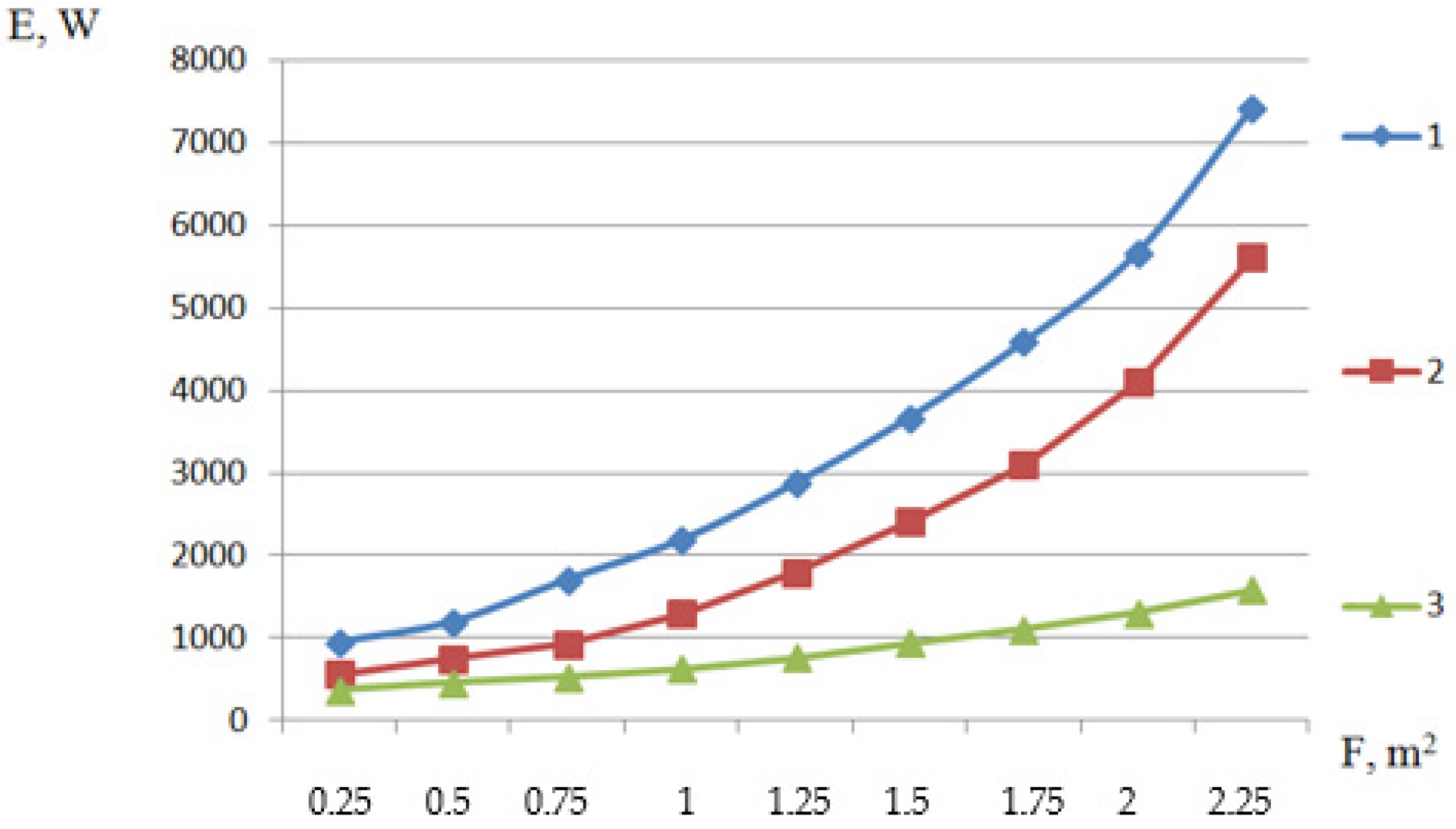
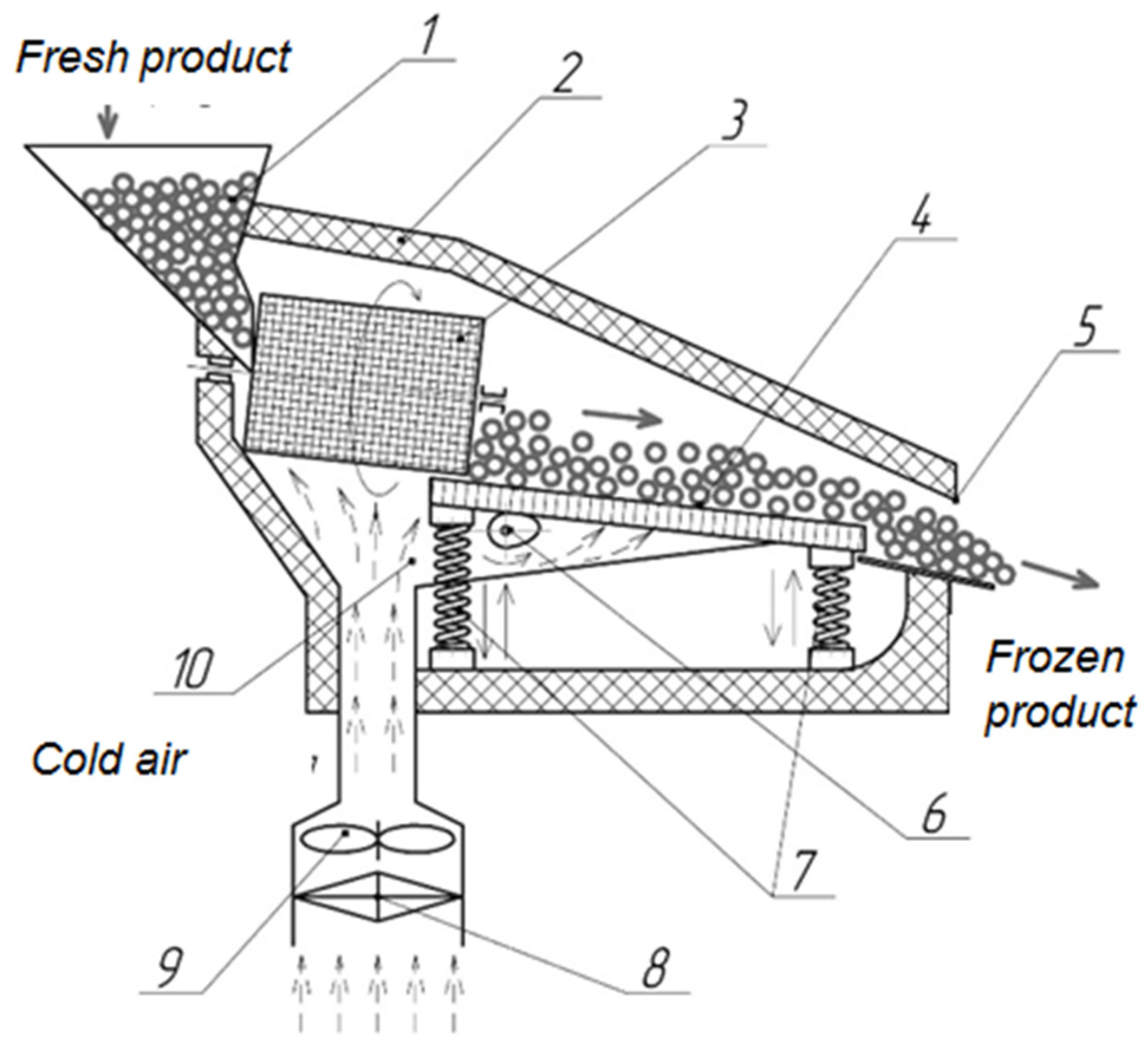
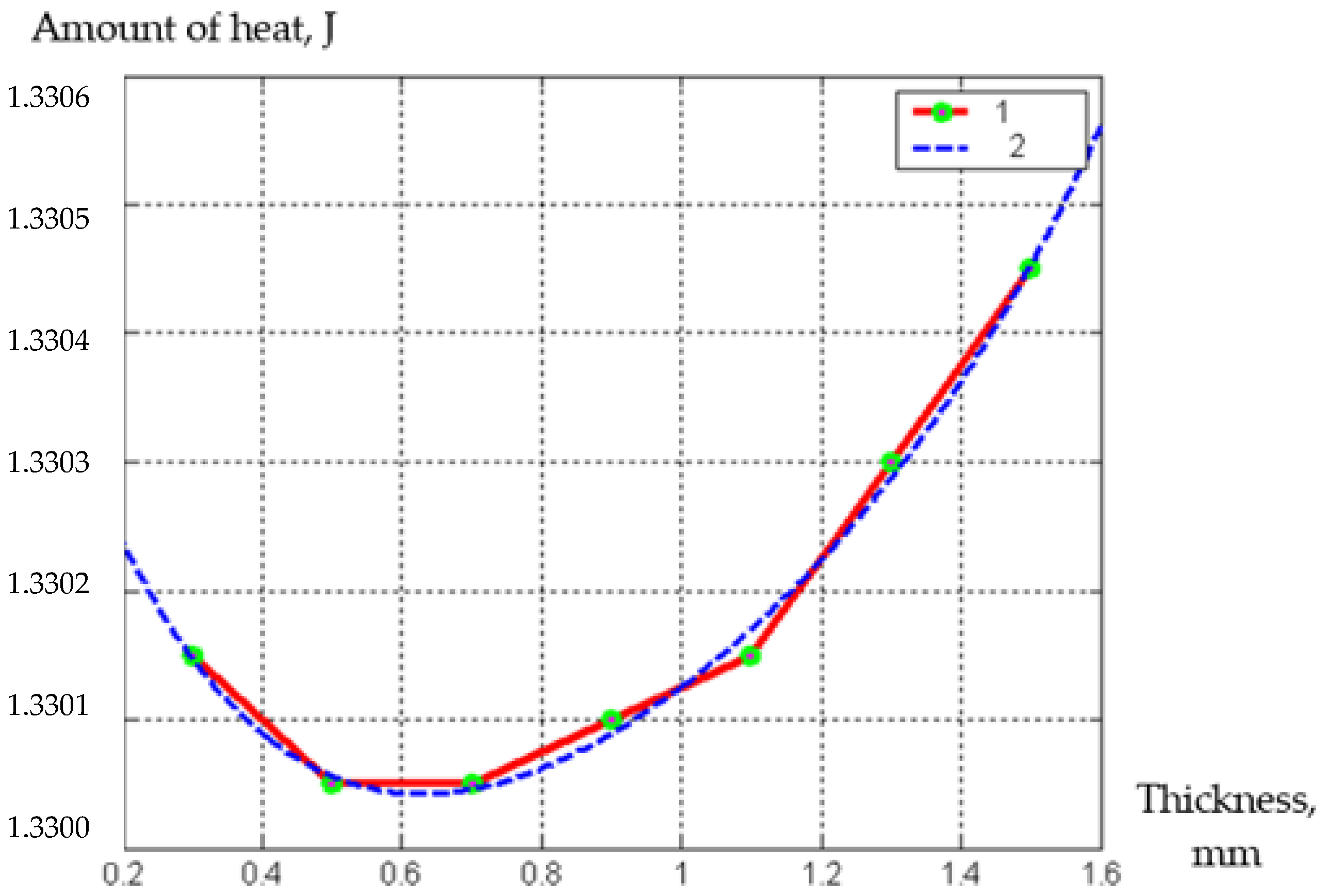
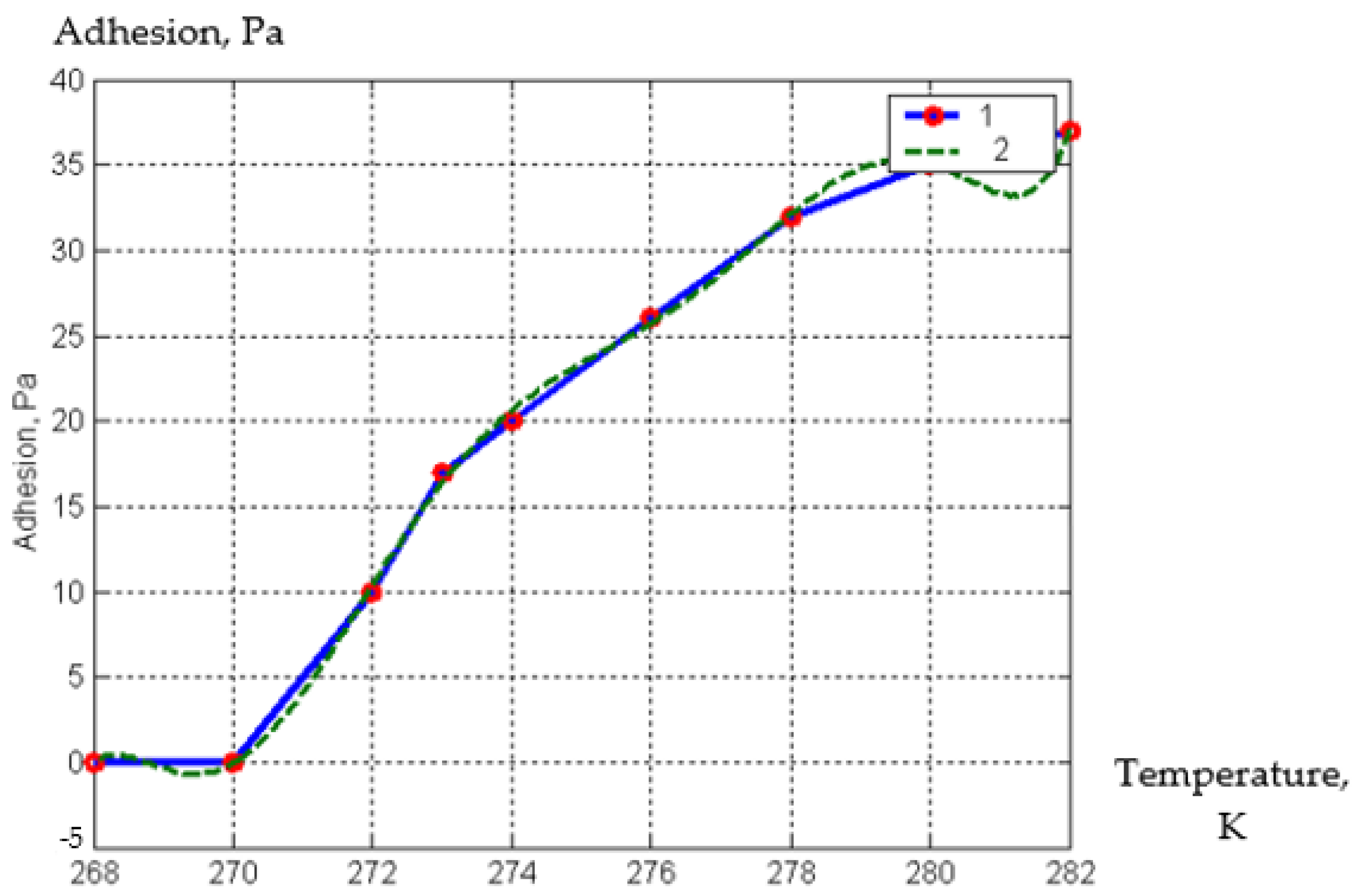
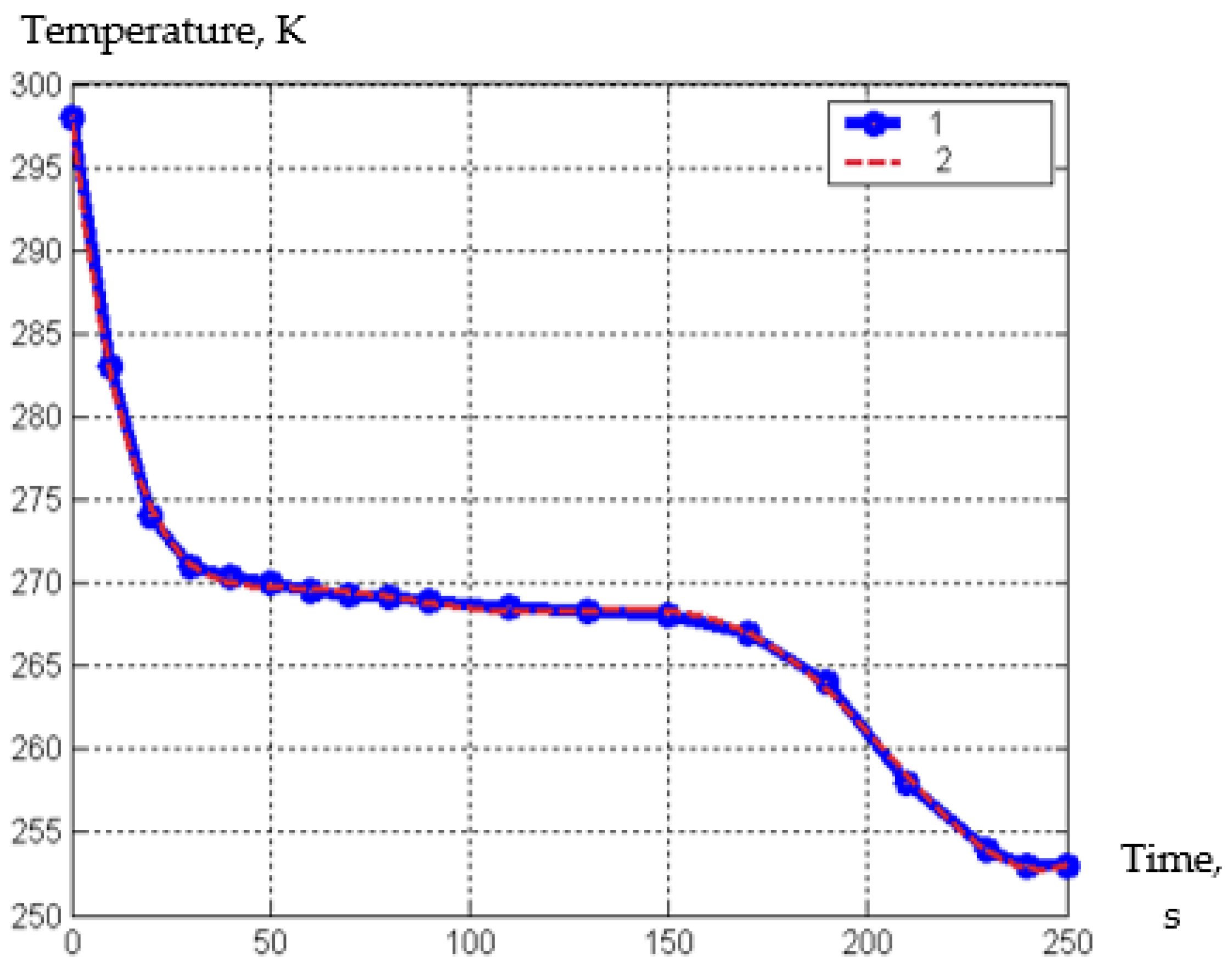
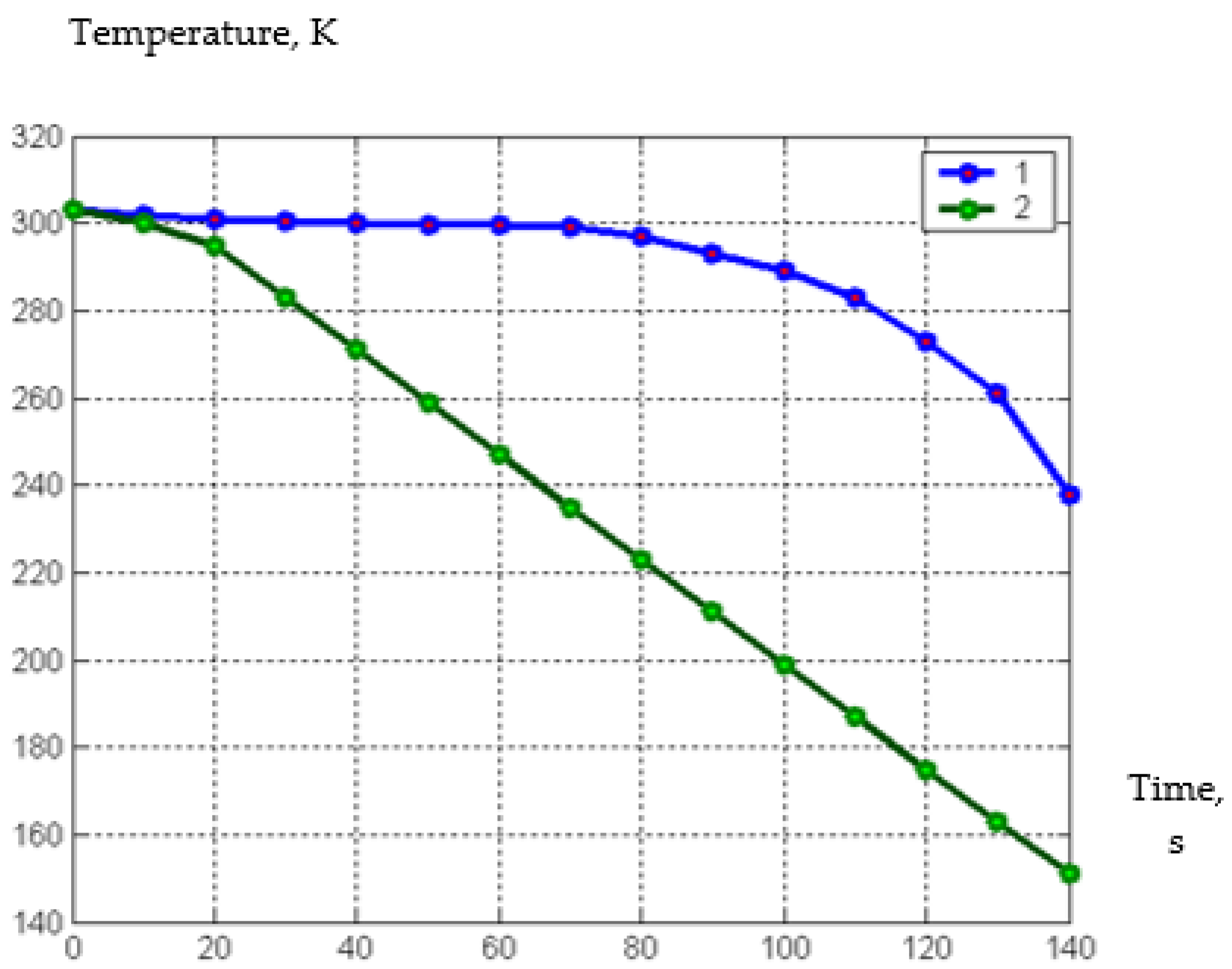
| Parameter | Designation | Meaning |
|---|---|---|
| Temperature, K: | ||
| initial | ti.e., | 298 |
| cryoscopic | tcr | 272 |
| freezing medium | te. | 233 |
| Specific heat capacity, kJ/(m3·K): | ||
| at positive temperatures | cp | 3.8 |
| at low temperatures | cfr | 2.3 |
| Density, kg/m3: | ||
| at positive temperatures | ρ | 940 |
| at low temperatures | ρfr | 700 |
| Specific heat of fusion, kJ/kg | λmlt | 333 |
| Coefficient of thermal conductivity, W/(m·K): | ||
| at positive temperatures | λp | 0.23 |
| at cryoscopic temperature | λcr | 0.2 |
| at low temperatures | λfr | 1.5 |
| Parameter | Temperature, K | |||||
|---|---|---|---|---|---|---|
| 273 | 268 | 263 | 258 | 253 | 248 | |
| The coefficient of thermal conductivity, W/(m·K) | 0.21 | 1.23 | 1.34 | 1.43 | 1.53 | 1.56 |
| Specific heat capacity, kJ/(m3·K) | 3.8 | 2.3 | 2.27 | 2.23 | 2.22 | 2.21 |
| Density, kg/m3 | 940 | 870 | 836 | 821 | 770 | 700 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bazaluk, O.; Struchaiev, N.; Halko, S.; Miroshnyk, O.; Bondarenko, L.; Karaiev, O.; Nitsenko, V. Ways to Improve the Efficiency of Devices for Freezing of Small Products. Materials 2022, 15, 2412. https://doi.org/10.3390/ma15072412
Bazaluk O, Struchaiev N, Halko S, Miroshnyk O, Bondarenko L, Karaiev O, Nitsenko V. Ways to Improve the Efficiency of Devices for Freezing of Small Products. Materials. 2022; 15(7):2412. https://doi.org/10.3390/ma15072412
Chicago/Turabian StyleBazaluk, Oleg, Nikolai Struchaiev, Serhii Halko, Oleksandr Miroshnyk, Larysa Bondarenko, Oleksandr Karaiev, and Vitalii Nitsenko. 2022. "Ways to Improve the Efficiency of Devices for Freezing of Small Products" Materials 15, no. 7: 2412. https://doi.org/10.3390/ma15072412
APA StyleBazaluk, O., Struchaiev, N., Halko, S., Miroshnyk, O., Bondarenko, L., Karaiev, O., & Nitsenko, V. (2022). Ways to Improve the Efficiency of Devices for Freezing of Small Products. Materials, 15(7), 2412. https://doi.org/10.3390/ma15072412









